Abstract
Background:
Pediatric patellofemoral instability is common, and medial patellofemoral ligament (MPFL) reconstruction is a highly effective treatment option. Femoral graft fixation methods include suture anchors (SAs) and interference screws (ISs), although comparative biomechanical studies in pediatric bone are lacking.
Purpose:
To compare the biomechanical properties of SA versus IS femoral graft fixation in pediatric MPFL reconstruction.
Study Design:
Controlled laboratory study.
Methods:
Ten fresh-frozen pediatric knees underwent femoral MPFL graft fixation using SA and IS (mean age, 9.5 years; range, 8-10 years). Posterior tibialis tendon allograft was used in both groups. For the IS group, 8-mm tunnels were drilled and 8 × 25–mm biocomposite IS was used for graft fixation. For the SA group, a 1.8-mm knotless all-suture SA was placed and secured to the graft. Specimens were loaded onto an Instron 5944 and subjected to cyclic preconditioning, followed by an ultimate failure load at 6 mm/s. The load displacement curve was then used to calculate stiffness (N/mm). Data were analyzed using repeated-measures analysis of variance to account for multiple measurements on the same specimens.
Results:
IS graft fixation demonstrated significantly higher ultimate failure load than SA (246.4 ± 79.7 N vs 90.4 ± 20.6 N, P < .001). Similarly, stiffness was significantly greater for IS compared to SA (17.8 ± 4.1 N/mm vs 13.4 ± 2.2 N/mm, P = .009). All but 1 IS reconstruction failed by graft pullout from the femoral tunnel, whereas all 10 SA reconstructions failed by anchor pullout.
Conclusion:
IS offers significantly higher ultimate failure load and stiffness values compared to SA femoral MPFL graft fixation in human pediatric knees. Mean ultimate failure load values for femoral MPFL fixation in pediatric bone may be lower than in adult bone, based on comparative adult studies. These results provide initial biomechanical data comparing 2 commonly used femoral fixation methods in skeletally immature patients.
Clinical Relevance:
This study characterized the biomechanical strength of SA and IS femoral MPFL fixation in pediatric bone, revealing differences in ultimate failure load and stiffness between constructs and compared to adult bone. While SA may pose less risk for growth alteration from physeal injury in younger patients, greater “time-zero” MPFL fixation strength with IS may allow for accelerated rehabilitation and earlier return to sport, which is a key patient and family satisfaction variable. In some circumstances, SA fixation may be preferred due to physeal proximity, and postoperative therapy regimens may be modified to account for lower fixation strength. These findings can help surgeons optimize implant selection, tailor rehabilitation protocols, and minimize the risk of graft failure in skeletally immature patients.
Keywords
Patellar instability is a common knee disorder that primarily affects young, active individuals.7,15,25 It encompasses a range of conditions, from intermittent subluxation to recurrent dislocation, and is associated with significant risk for patellofemoral osteoarthritis and long-term disability. 19 The medial patellofemoral ligament (MPFL) is the major ligamentous restraint to lateral patellar displacement and is ruptured in most patients with patellar dislocation.2,4,18
Multiple surgical options exist to treat recurrent patellar instability, and most involve reconstruction of the MPFL or repair/plication of the MPFL and medial soft tissues.11,17 Graft positioning is a major factor in complications reported after MPFL reconstruction, with the femoral fixation point being the most critical to avoid undesired anisometry.1,31 In skeletally immature patients, the MPFL femoral origin is in close proximity to the distal femoral physis,24,26,27 presenting a unique challenge for implant selection in children. To avoid direct physeal injury, numerous surgical techniques have been described, such as all-epiphyseal blind-socket femoral tunnel drilling, adductor magnus tendon transfer, and soft tissue pulley methods at the adductor or medial collateral ligament. 29 Pediatric bone may further complicate fixation because of its higher proportion of cancellous bone compared with the denser cortical bone found in adults, resulting in decreased resistance to tensile and compressive forces.5,8,30 Additionally, immature bone's greater plasticity and lower stiffness may allow for greater deformation under load, potentially reducing the effectiveness of fixation constructs. 30
Various studies have characterized the biomechanical properties of MPFL fixation techniques in reconstruction procedures using animal or adult cadaveric tissue, with suture anchor (SA) and interference screw (IS) fixation being the most common methods.3,9,12,14,16,23 IS fixation, which requires a larger socket and reaming of bone tunnels, is anatomically constrained, particularly in pediatric patients with open physes. 16 In contrast, SA fixation involves a smaller footprint and poses less risk of clinically significant physeal violation, which can lead to growth arrest and angular deformity in the skeletally immature.3,13
To our knowledge, no studies have compared the biomechanics of SA versus IS graft fixation techniques in pediatric specimens. Given the proximity of the MPFL origin to the femoral physis and unique bone characteristics in the skeletally immature, who have relatively high rates of patellar instability, there is a need to understand the biomechanical performance of commonly employed fixation methods. Thus, the aim of this study was to compare the biomechanical properties of SA versus IS for femoral fixation of a posterior tibialis tendon allograft in MPFL reconstruction in skeletally immature pediatric cadaveric specimens.
Methods
An institutional review board deemed that approval was not required for this study, as it was based on cadaveric tissue and did not include patient identifiers, the use of genetic information, or contact with the patient's family, as per guidelines by the US Department of Health and Human Services (www.hhs.gov). A tissue-harvesting facility (Allosource) sourced the cadaveric tissue, which had family consent for use in research before conducting this study.
Ten fresh-frozen pediatric cadaveric knee specimens were used (mean age, 9.5 years; range, 8-10 years). The femur was isolated and stripped of all soft tissue attachments. Each specimen underwent femoral graft fixation with SA, followed by IS. The MPFL femoral origin was identified using fluoroscopy, as described by Schöttle et al 20 in both fixation techniques. Moisture of the cadaveric specimens and allograft tendons was maintained with saline sprays during preparation and testing. Mean dimensions for the posterior tibial allograft measured 7 to 8 mm in diameter and 22 to 26 cm in length.
In the SA group, a 1.8-mm all-suture SA (ASA) with an embedded No. 2 FiberWire suture (Arthrex) was placed. Appropriate purchase was confirmed by applying tension to both suture ends. A posterior tibial tendon allograft was folded to form a looped construct, positioning the 2 free limbs side by side (Figure 1A). Each graft limb was secured to the ASA using 1 of the 2 embedded suture ends. For each limb, 6 locking Krackow stitches were placed, 3 along each side of the tendon (Figure 1B).
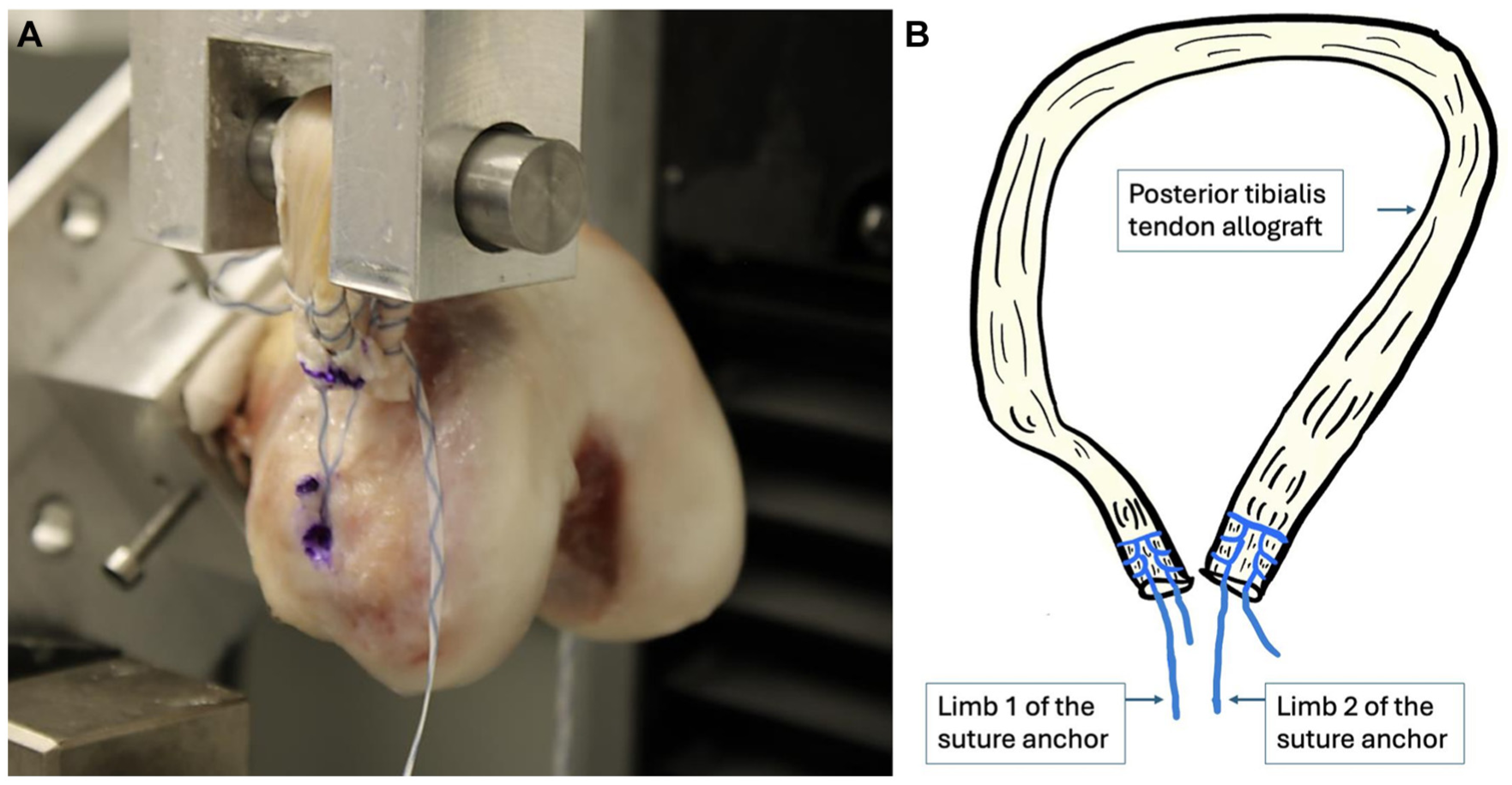
(A) Image captured during testing demonstrating the looped graft configuration, with each free limb of the allograft secured to opposite ends of the suture anchor for attachment to the Instron testing system. (B) Schematic illustration of graft preparation for the suture anchor (SA) group. The graft was folded to create a looped construct, allowing for secure attachment to the Instron system. Each limb of the posterior tibialis tendon allograft was secured to one of the embedded FiberWire sutures from the all-suture anchor using running locked Krackow stitches.
In the IS group, the anatomic femoral origin of the MPFL was again identified. A 2-mm Kirschner wire was introduced across the femur, parallel to the joint line and exiting at the lateral epicondyle. An 8-mm reamer was then reamed over the K-wire to create a bone tunnel extending to the lateral cortex. The 2 free limbs of a posterior tibial tendon allograft were each whipstitched with 3 suture throws using a FiberLoop (Arthrex) and a straight needle. The free ends of the suture were passed through the femoral tunnel, and each limb of the graft was pulled 2 cm into the tunnel. The graft was fixed into the femoral tunnel using an 8 × 25–mm biocomposite interference screw (Arthrex) (Figure 2A).
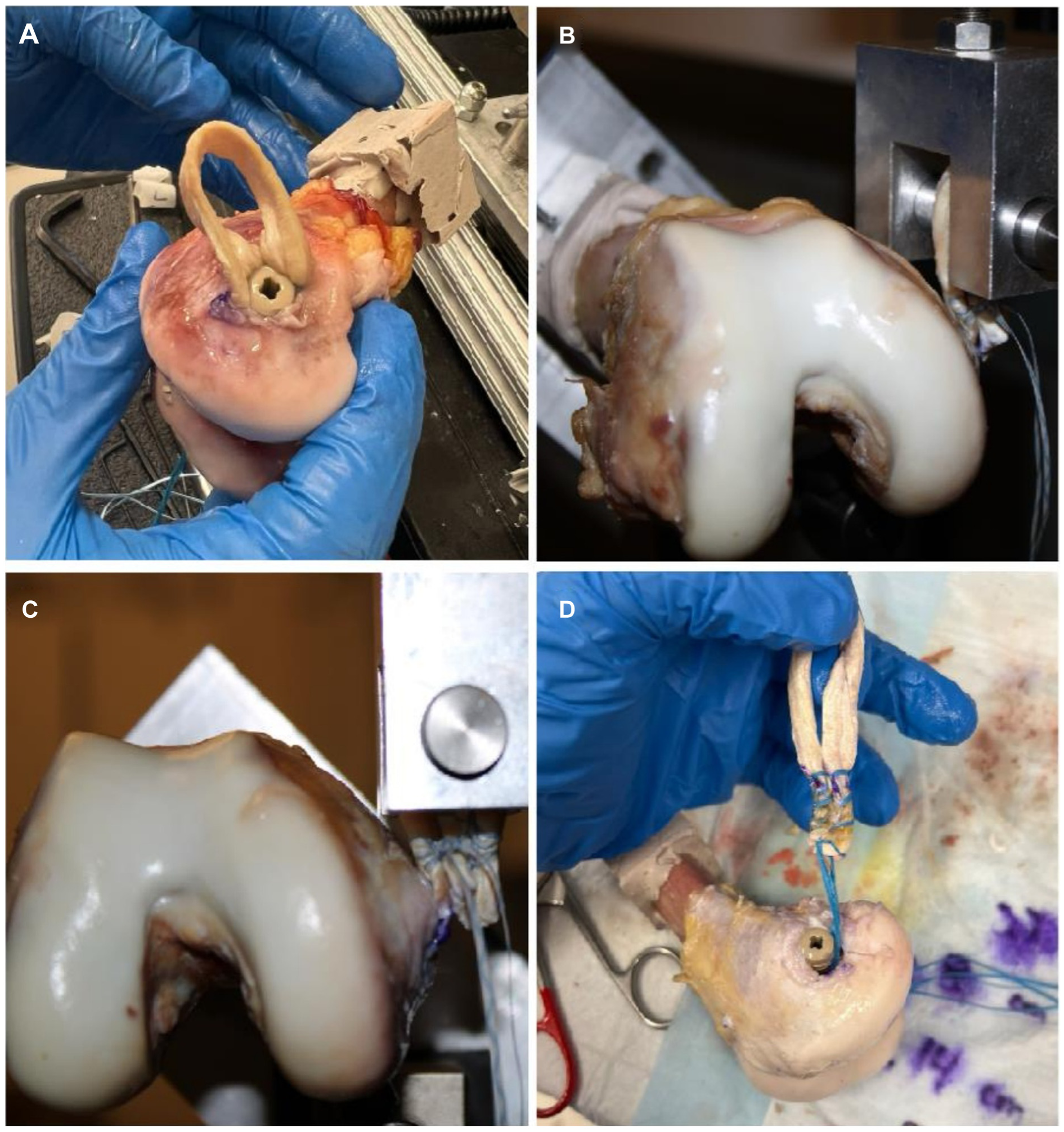
(A) Medial condyle of the right knee, with femoral fixation of posterior tibial tendon autograft with a 9-mm biocomposite interference screw. (B, C) Biomechanical testing setup of the right knee in the Instron 5944, with femoral fixation with an all-suture anchor. (D) Medial condyle of the right knee, with interference screw fixation demonstrating graft pullout after biomechanical testing.
The femurs were potted and loaded into an Instron 5944. The looped end of the graft was secured to the load cell in a configuration that ensured a lateral force vector to simulate a patellar dislocation (Figure 2B). Each graft was preconditioned from 0 to 30 N for 20 cycles to reduce tissue hysteresis. The constructs were then pulled at a rate of 6 mm/s until a 50% decrease in load was observed, which was defined as failure. Stiffness (N/mm) was calculated in a post hoc fashion using the best-fit slope of the load displacement curve. Ultimate failure load (N) was measured for each specimen.
Statistical Analysis
Based on a power analysis, a sample size of 10 specimens, undergoing 2 test conditions each, provided at least 80% power to detect a 30% difference in maximum slope and a 50% difference in maximum load between SA and IS fixation methods. Data were analyzed using repeated-measures analysis of variance to account for multiple measurements on the same specimens. Normality was assessed visually using quantile-quantile plots. All analyses were run in RStudio version 2023.12.1+402 using a 2-sided level of significance of .05.
Results
The IS graft fixation construct demonstrated significantly higher ultimate failure load compared to ASA (mean ± SD = 246.4 ± 79.7 N vs 90.4 ± 20.6 N, P < .001) (Figure 3A). Similarly, stiffness was significantly greater for IS compared to ASA (mean ± SD = 17.8 ± 4.1 N/mm vs 13.4 ± 2.2 N/mm, P = .009) (Figure 3B). All but 1 IS reconstruction failed by graft pullout from the femoral tunnel (Figure 2C, D), whereas all 10 ASA reconstructions failed by anchor pullout.

(A) Boxplot distribution of ultimate failure load in interference screw (IS) and all-suture suture anchor (ASA) femoral fixation. (B) Box plot distribution of stiffness in IS and ASA femoral fixation. The mean value is marked with a red diamond.
Discussion
This study demonstrates significantly higher ultimate failure load and stiffness values for IS compared to ASA femoral MPFL graft fixation in pediatric cadaveric specimens. Prior work in adult and animal models has shown mixed results when comparing femoral fixation methods in MPFL reconstruction. A recent meta-analysis of biomechanical studies in adults found that femoral graft fixation with IS led to higher ultimate failure load compared to SA, although stiffness was not different. 22 However, individual biomechanical studies have reported conflicting findings. In a cadaveric study of MPFL femoral fixation in adult knees, Gould et al 9 found no significant difference in ultimate failure load between biocomposite SA and IS, although IS fixation showed greater stiffness. In contrast, Smith et al 28 showed significantly greater ultimate stiffness and ultimate failure load with IS compared to ASA for femoral fixation in a porcine model. In our pediatric cohort, IS fixation outperformed ASA across both biomechanical metrics. While a higher cancellous-to-cortical bone ratio in pediatric femurs may reduce resistance to pullout, particularly in ASA constructs, the heterogeneity of fixation methods across all studies used across studies limits direct comparison and broader inferences.
The proximity of the MPFL femoral origin to the distal femoral physis is a critical consideration in pediatric patients with significant remaining growth. Numerous cadaveric studies have demonstrated the location of the MPFL femoral attachment close to the physis, near the femoral attachment of the medial collateral ligament and the adductor tubercle, underscoring the challenges of safely placing fixation constructs in this region.6,26,27 Physeal injury about the knee has been reported to induce growth alterations after MPFL reconstruction procedures, leading to significant deformity and the need for corrective surgery.21,27,32 Although IS fixation demonstrated superior biomechanical performance in our study, it requires larger bone tunnels that may increase the risk disrupting the physis and surrounding perichondral ring. In contrast, ASA, with a smaller footprint, poses less risk of physeal injury, making it appealing in skeletally immature populations. These findings highlight the need for careful surgical planning and technique in pediatric MPFL reconstruction, balancing fixation strength with the preservation of physeal integrity and avoidance of growth disturbance.
Ultimate failure load for ASA graft fixation in our study using pediatric cadaveric specimens did not exceed published values for native MPFL strength, 10 raising concerns about the stability of the construct in the immediate postoperative period. Although fixation constructs are not expected to resist forces equivalent to those seen during traumatic dislocation, they should provide sufficient strength to withstand normal physiologic loads encountered during daily activities and early rehabilitation. In the absence of published biomechanical data on native MPFL properties in pediatric specimens, these comparisons are challenging. Nonetheless, surgeons should be aware that the time-zero femoral MPFL fixation strength of ASA constructs may be lower, potentially providing a limited safety margin during early postoperative loading, particularly in the setting of aggressive rehabilitation, which could increase the risk of graft failure. In contrast, IS fixation, with a higher initial fixation strength, may allow for earlier loading and accelerated rehabilitation protocols, potentially reducing the time to return to activity. Ultimately, the long-term clinical impact of these fixation techniques remains unclear. While concerns have been raised about potential graft overconstraint with IS constructs, this risk is likely more influenced by nonanatomic or nonisometric graft placement than by implant strength alone. 22
Limitations
This study has limitations. It is possible that the unidirectional force vector imparted on these constructs did not fully capture the constellation of forces that can stress these fixation constructs in vivo. Additionally, as a cadaveric laboratory study, our findings represent a picture of time-zero MPFL fixation strength with testing that simulates ultimate failure rather than the progressive healing and stress that patients undergo during healing and rehabilitation. Given the limited availability of pediatric-sized allografts, posterior tibialis tendons were used in all constructs. All posterior tibialis tendon allografts were provided by the tissue bank, preprocessed and selected based on similar size and gross appearance to minimize variability between constructs. While these grafts may be larger than the native MPFL in pediatric patients, they allowed for standardized comparison of fixation techniques. Similarly, 8-mm femoral tunnels were drilled to the far cortex to permit passage and consistent seating of the grafts for fixation with the available 8-mm interference screws. Although this method may not replicate physeal-sparing, all-epiphyseal techniques used clinically in skeletally immature patients, it enabled consistent graft fixation depth and construct configuration for uniform biomechanical comparison across all specimens. Importantly, the fixation constructs used in this study were not direct replications of those employed in prior adult or animal biomechanical studies. Comparisons of absolute strength and stiffness values across studies should be interpreted with caution. Despite these limitations, to our knowledge, this is the first biomechanical study to evaluate femoral fixation methods in MPFL reconstruction in skeletally immature human cadaveric specimens. Further studies are necessary to evaluate the in vivo failure rates and complications associated with these techniques in pediatric patients to determine their feasibility and efficacy beyond time-zero biomechanical research.
Conclusion
In this biomechanical study of 2 femoral fixation techniques in a pediatric cadaveric model of MPFL reconstruction, IS demonstrated higher ultimate failure load and stiffness than ASA. Femoral fixation with IS may allow for earlier loading and expedited rehabilitation protocols than ASA. While ASA poses less risk of physeal injury, clinicians should be aware of construct stability in the immediate postoperative period. This study highlights the need for tailored approaches to rehabilitation protocols in children undergoing surgical MPFL reconstruction.
Footnotes
Acknowledgements
The authors thank Todd Huft, Tom Cycota (former CEO), and Dean Elliott (CEO) of AlloSource. Their generous donation of cadaveric specimens allowed for the findings of this study. Their support continues to propel forward the field of biomechanical research and enhances our understanding and treatment of children. The authors also honor and acknowledge the families who made the gifts of donation to this study group. Their donations have made this research possible and will contribute to the lives of other families and their children.
Final revision submitted January 19, 2026; accepted January 24, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: I.H. and K.M. have received support for education from Evolution Surgical. H.B.E. has received consulting fees from OrthoPediatrics and support for education from Pylant Medical. M.R.S. has received consulting fees from WishBone Medical and support for education from MedInc of Texas. M.T. has received consulting fees from OrthoPediatrics. Y.-M.Y. has received consulting fees from Smith & Nephew and OrthoPediatrics. T.G. has received support for education from Paladin Technology Solutions and is an associate editor for AJSM. K.G.S. has received consulting fees from OrthoPediatrics and support for education from Evolution Surgical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
