Abstract
Background:
Studies on posterior ankle impingement syndrome (PAIS) that focus on clinical features, characteristics, and surgical outcomes are limited.
Purpose:
To investigate postoperative outcomes of PAIS and identify the factors affecting it.
Study Design:
Case series; Level of evidence, 4.
Methods:
Demographic data, participating sports, the competing level of the sport, the source of pain, and concomitant pathologies were reviewed in 267 ankle samples from 240 athletes. For surgical cases, the following data were reviewed: preoperative pain duration, pre- and postoperative scores on the Japanese Society for Surgery of the Foot (JSSF) ankle-hindfoot scale, and time of return to training (RTT) and return to sports (RTS). Comparisons were statistically analyzed (1) between pre- and postoperative scores on the JSSF scale, (2) for the difference in RTT/RTS between sources of pain (bony vs soft tissue) and among sports and competition levels, and (3) for the relationship between preoperative pain duration and RTT/RTS.
Results:
The most common sports were ballet, followed by soccer, rugby, baseball, basketball, and swimming/water polo. Ankles of student athletes (mean ± SD age, 17.2 ± 2.6 years; range, 12-22) were the most commonly affected. The source of pain was bony impingement in 221 ankles (82.8%). The most common concomitant pathology was tenosynovitis of the flexor hallucis longus. A total of 147 ankles (55.1%) in 125 athletes were treated surgically. The preoperative JSSF scale score of 83.9 points significantly improved postoperatively to 99.4 points (P < .00001). RTT and RTS did not differ significantly between sources of pain and among sports and competition levels. The duration of preoperative pain was significantly correlated positively with RTT and RTS (P = .009 and P < .001, respectively).
Conclusion:
PAIS has been observed in many sports and is often associated with flexor hallucis longus–related pathologies, caused mainly by bony impingement. The preoperative pain duration was positively related to RTT and RTS. Surgical treatment of PAIS improves symptoms in most patients.
Posterior ankle impingement syndrome (PAIS) is a clinical disorder characterized by posterior ankle pain during plantarflexion of the ankle.10,11 The pain is usually aggravated by repetitive plantarflexion or hyperplantarflexion of the ankle. Therefore, PAIS is often observed in athletes who are required to forcefully or repeatedly flex their ankles. Previous studies have reported that PAIS is commonly observed in athletes such as ballet dancers and soccer players,2,5,16 runners, and gymnasts. 8 PAIS has also been reported in swimmers since they perform repetitive kicking motions that require forced ankle plantarflexion. 18
The main cause of the syndrome is the presence of osseous and soft tissue lesions.6,16,21 The former includes pathologies represented by the os trigonum, an enlarged Stieda process (hypertrophic posterior talar process), or loose bodies, whereas the latter includes flexor hallucis longus (FHL) tenosynovitis, synovitis, or anomalous muscles. Although the prevalence is low, pathologies related to joint origin, such as joint capsule issues, have also been reported. 16 Soft tissue–related pathologies are less common in men, 10 and pathologies related to FHL are commonly observed among ballet dancers. 16 The initial approach to PAIS is nonsurgical treatment, which includes resting or reduction of activity load, avoiding provocative activities, use of nonsteroidal anti-inflammatory drugs, and injection of steroid to the posterior ankle or subtalar joint.8,9 Although nonoperative treatment has been suggested, previous literature has reported that approximately 40% of the cases eventually require surgery.7,21 Currently, surgical interventions for PAIS are mostly conducted endoscopically, owing to its low complication rates and early return to sports (RTS).1,3,4,5,8,10,16,21,22 However, RTS varies by sport and competition level. Furthermore, the length of preoperative symptoms is said to relate to the duration of return to training (RTT) and RTS. 1 Taking everything into account, there seems to be a lack of information on the clinical features and characteristics, such as the level of athletes and competing sports, and the surgical outcomes and affecting factors. In addition, postoperative results such as time to RTS can differ by participating sports. Thus, a large-scale study is required to better understand this syndrome and its features, along with the postoperative results and affecting factors.
Therefore, this study aimed to investigate the postoperative results of PAIS in athletes and establish its clinical characteristics, focusing on concomitant pathologies, competing level of athletes, and participating sports, with factors related to RTS and RTT. We hypothesized that the clinical characteristics and surgical results would differ among sports and athlete participation levels. Specifically, we suspected that those with bony impingement would show earlier RTS than those with soft tissue impingement and that professional athletes would RTS earlier than athletes from other levels.
Methods
This retrospective observational study was approved by the institutional review board of the affiliated clinic (No. 24-01). This study was conducted in accordance with the Declaration of Helsinki and its later amendments or comparable ethical standards. 4 Clinical records were reviewed for patients who visited our 2 affiliated clinics or hospitals and were diagnosed with PAIS between April 2014 and March 2023. Data were extracted from the clinical records in each facility, and the extraction was performed by the senior author (H.N.).
Diagnosis of PAIS was made by a single experienced sports orthopaedic surgeon (T.K.) who specializes in foot and ankle surgery, and it was based on the medical history (posterior ankle or hindfoot pain) and physical findings of reproducible posterior ankle pain induced by forced hyperplantarflexion of the ankle (posterior impingement hyperflexion test 13 ). Participants were included if they were diagnosed with PAIS in accordance to the aforementioned diagnostic physical examination. Participants were excluded if they were diagnosed with PAIS but were nonathletes.
All athletes underwent imaging to include plain radiographs of the anteroposterior and lateral views along with computed tomography (CT) scan and magnetic resonance imaging. Bony impingement was diagnosed on the basis of the presence of the os trigonum, hypertrophic posterior talar process, or loose body on plain radiographs and CT scans. Soft tissue impingement was diagnosed by confirming the absence of previously mentioned bony structures with CT scans and the presence of soft tissue pathologies such as FHL tenosynovitis, synovitis, or anomalous muscles around the posterior talar process with magnetic resonance imaging (Figure 1).

Typical images of (a) bony impingement with os trigonum and (b) soft tissue impingement with tenosynovitis around the flexor hallucis tendon. Arrow, os trigonum; arrowhead, synovium; asterisk, swelling of around the flexor hallucis tendon.
Those who were not treated at other medical facilities underwent nonsurgical rehabilitation. Nonoperative treatment was performed for at least 3 months, and the exercise load was controlled by ceasing or alleviating the activity load based on the pain levels. Rehabilitation was conducted by physical therapists, and injection of local anesthesia and corticosteroid was performed in certain cases for pain relief, if requested by the athletes. Surgical treatment was suggested if 3-month nonoperative treatment was unsuccessful. For athletes who visited our medical facilities, after having a consultation with the athletic trainer of the team with which they were affiliated regarding possible surgical indication after failed nonsurgical treatment and for those who decided to undergo surgical treatment, the affected ankle was evaluated in detail, including a thorough physical examination combined with previous multimodal imaging to confirm concomitant pathologies, such as FHL-related pathologies (FHL stenosis, tenosynovitis, tendinosis, and tendon tears), synovitis, anterior talofibular ligament (ATFL) injury, and anterior ankle impingement syndrome (AAIS). AAIS was diagnosed on the basis of complaints of anterior ankle pain and confirmation of bony spurs along the anterior tibia border and/or talar neck on plain radiographs and CT scans.
All surgical procedures were conducted endoscopically in the prone position by creating 2 portals, posterolateral and posteromedial.17,20 Patients with concomitant pathologies who required surgical treatment underwent both treatments simultaneously. For bony impingement cases, the os trigonum was removed by using a bur or an arthroscopic forceps. After the removal, the posterior contour of the talus was confirmed by fluoroscopy, and the posterior process of the talus was resected until the contour became rounded and smooth, without impinging against the tibia or the calcaneus. For soft tissue impingement, synovectomy of the ankle was performed, and if the patient had FHL-related pathologies, the tendon sheath of the FHL tendon was resected, along with synovitis around the tendon. The presence of the tear was confirmed, and since there were no cases that required suturing or repairing of the tendon, debridement was performed accordingly. For AAIS cases, the patient was turned to the supine position. By using the ankle traction system, anteromedial and anterolateral portals were created. Through the portals, bony spurs of the tibia and talus were resected by a bur or an arthroscopic forceps. Removal of the spur was confirmed by fluoroscopy. Postoperatively, the athletes were permitted to bear weight based on their tolerance without any splints or braces, except for the application of compressive bandages around the ankle to protect the surgical site. Intrinsic foot muscle training was performed immediately after surgery. Two weeks after postoperative suture removal, formal physical therapy was initiated, which focused on aggressive range of motion exercises, muscle strengthening, and proprioceptive exercises. RTT was allowed after achieving full range of motion, recovering muscle strength, and completing sports-specific movements. RTS was defined when the coach or athletic trainer permitted the athlete to return to a scrimmage or game. RTT and RTS definitions were based on a previous report regarding recovery from sports injury. 12 For athletes who had concomitant surgery for ATFL injury via the arthroscopic Brostrom technique, the ankle was immobilized with a below-knee cast for 3 weeks postoperatively. After cast removal, weightbearing was allowed to tolerate pain, and range of motion exercises were initiated. Subsequent rehabilitation was modified accordingly.
Descriptive data were reviewed on sex, age at initial diagnosis, participating sports, level of competition (professional, amateur, recreational, or student), dominant hand/foot (according to the participating sports or daily life), source of impingement (bony or soft tissue), concomitant pathologies, and the affected side. Bilateral ankle cases were treated as independent cases. Athletes who were treated surgically were evaluated for preoperative pain duration (weeks), pre- and postoperative Japanese Society for Surgery of the Foot (JSSF) ankle-hindfoot scale,14,15 concomitant surgery, postoperative complications, and weeks of RTT and RTS. Postoperative complications were evaluated by complaints from athletes and physical examination during postoperative outpatient follow-up. RTT and RTS were confirmed via interviews with the athletes during periodic follow-up visits and consultation after surgery; if the athlete could not visit, then confirmation was done via phone calls or text messaging to the trainer/coach. The JSSF scale is a clinically valid and reliable objective and subjective clinical rating system for evaluation of foot and ankle disorders. 15 It is a modified scale and combines the American Orthopaedic Foot and Ankle Society scale and foot rating scale from the Japanese Orthopaedic Association. The JSSF ankle-hindfoot scale score is based on 100 points, with 40 assigned to pain, 50 to function, and 10 to alignment. The JSSF scale scores were evaluated preoperatively and at the last postoperative follow-up by a single experienced sports orthopaedic surgeon (T.K.).
Statistics
Categorical data are described as absolute numbers and percentages and continuous data as mean and standard deviation. After confirmation of normality, comparisons between the pre- and postoperative JSSF scale scores were statistically analyzed with a paired t test. The relationship between surgical cases and competing levels was analyzed by a chi-square test. Differences in RTT/RTS between sources of pain were analyzed with an unpaired t test. Differences in RTT/RTS for participating sports and level of competition were analyzed by analysis of variance. The groups were compared individually by a post hoc Tukey-Kramer honestly significant difference test. The relationship between preoperative pain duration and RTT/RTS was analyzed by the Spearman rank correlation coefficient. In addition, the relationship between preoperative pain duration and the presence of FHL-related pathologies was analyzed by the Mann-Whitney U test. Postoperative JSSF scale scores, RTT, and RTS were also compared between athletes with and without FHL-related pathologies, using a Mann-Whitney U test. All statistical analyses were performed in JMP Pro 15 software (SAS Institute). Results were considered statistically significant at P < .05.
Results
During the review period, 267 ankles (132 male, 135 female; mean ± SD age, 21.4 ± 7.2 years; range, 12-55 years) from 240 athletes were diagnosed with PAIS. The most common sports involved were ballet (100 ankles; 37.4%), soccer (51 ankles; 19.1%), rugby (15 ankles; 5.2%), baseball (13 ankles; 4.9%), basketball (11 ankles; 4.1%), and swimming/water polo (10 ankles; 3.7%). The source of pain was bony impingement (including the os trigonum, hypertrophic posterior talar process, and loose body) in 221 ankles (82.8%) and soft tissue impingement in the rest. The most common concomitant pathology was FHL (125 ankles), followed by synovitis (79 ankles), AAIS (21 ankles), and ATFL injury (19 ankles).
A total of 55% of athletes were diagnosed in the right ankle, 41.6% in the left, and 3.4% in both, and 164 (61.7%) complained about the dominant side. The percentage of PAIS on the dominant side for each sport was as follows: 58% for ballet, 71% for soccer, 64% for rugby, 92% for baseball, 64% for basketball, and 60% for swimming. The percentages of bony impingement for each sport were as follows: 80% for ballet, 82% for soccer, 79% for rugby, 69% for baseball, 82% for basketball, and 100% for swimming. There were 152 affected ankles among student athletes (56.8%; mean age, 17.2 ± 2.6 years), followed by 74 among professional athletes (27.8%; 25.3 ± 5.8 years), 26 among amateur athletes (9.7%; 28.3 ± 8.4 years), and 15 among recreational athletes (5.6%; 33.1 ± 10.1 years). Details of the competition levels in the participating sports are presented in Table 1.
Data of Each Sport and Competition Level a

Data are presented as No. (%).
A total of 147 ankles (55.1%) in 125 athletes were surgically treated. Among surgical cases, the ankles of athletes participating in ballet (56 ankles; 56% of all ballet dancers) were the most commonly affected, followed by soccer (33 ankles; 64.7%), rugby (12 ankles; 80%), basketball (7 ankles; 63.6%), and swimming/water polo (7 ankles; 77.8%). Regarding the competition level, the ankles of student athletes accounted for the most, with 85 (57.8% of overall surgical cases; 55.9% of all student athletes), followed by professional athletes with 45 (30.6% and 60.8%), amateur athletes with 13 (8.8% and 50%), and recreational athletes with 4 (2.7% and 26.7%) (Table 1). Professional athletes tended to show a higher ratio of surgery than amateur athletes, although the small number of surgical cases among amateur athletes hindered us from statistically analyzing those accurately.
The preoperative JSSF scale score of 83.9 ± 7.2 points (range, 44-87) significantly improved postoperatively to 99.4 ± 2.1 points (range, 80-100; P < .00001). The most common concomitant surgery was resection of the FHL tendon pulley (96 ankles; 65.7%), followed by synovectomy around the FHL tendon and tendon sheath (53 ankles; 36.1%). There were 6 cases of postoperative complications (4.1%), with 1 mild infection that healed with oral antibiotics and the rest with surgical site pain, all of which were relieved after postoperative rehabilitation. Mean preoperative pain duration was 51.9 ± 53.7 weeks (median, 36 weeks), and the mean postoperative follow-up period was 8.9 ± 9.2 months. There were no recurrent cases among any of the athletes who were treated surgically and returned to their previous competition levels.
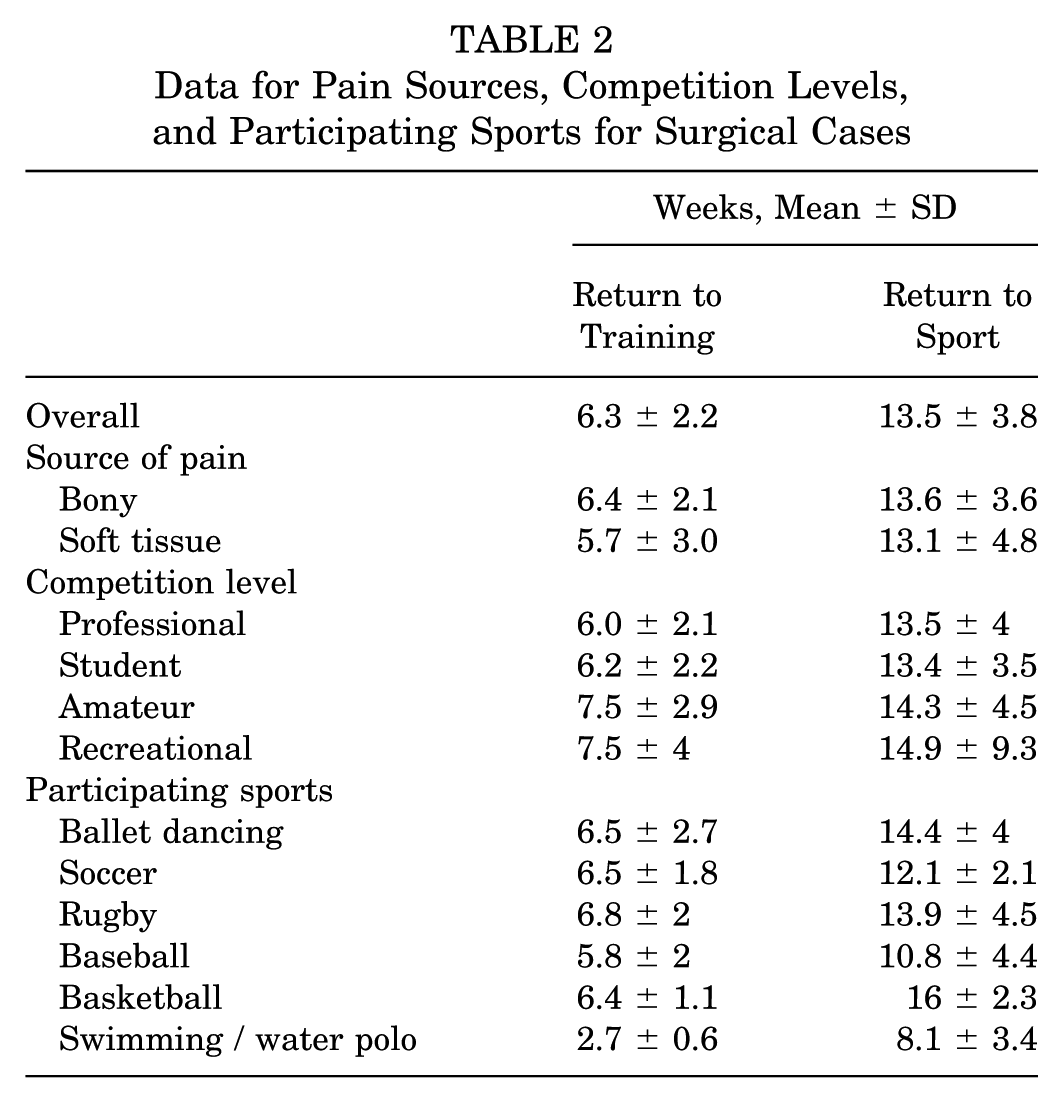
The mean numbers of weeks for RTT and RTS were 6.3 ± 2.2 and 13.5 ± 3.8, respectively. The mean numbers of weeks for RTT and RTS after bony impingement were 6.4 ± 2.1 and 13.6 ± 3.6, and the corresponding mean numbers after soft tissue impingement were 5.7 ± 3.0 and 13.1 ± 4.8. There were no significant differences in the mean RTT and RTS values based on pain sources. Mean RTT and RTS for each competition level and participating sport were not significantly different and are presented in Table 2.
Data for Pain Sources, Competition Levels, and Participating Sports for Surgical Cases
The duration of preoperative pain was significantly correlated with the duration of RTT (r = 0.24; P = .009) and RTS (r = 0.43; P < .001) (Figure 2).

Relationship between preoperative pain duration and return to training/sport.
The mean value for preoperative pain duration in athletes with FHL-related pathologies was 52.5 ± 50.2 weeks (median, 40 weeks), and that in athletes without FHL-related pathologies was 46.7 ± 51.5 weeks (median, 28 weeks), with no significant statistical difference. Postoperative JSSF scale scoring of those with FHL-related pathologies was 99.3 ± 2.4 points (range, 80-100), as compared with 99.6 ± 1.1 points (range, 96-100) for those without FHL-related pathologies, with no significant statistical difference. The mean numbers of weeks for RTT and RTS of those with FHL-related pathologies were 6.5 ± 2.4 and 13.6 ± 3.7, respectively, with corresponding values of 5.8 ± 1.9 and 13.7 ± 4.3 weeks for those without FHL-related pathologies. There were no significant differences in mean RTT and RTS values based on the presence of FHL-related pathologies (Table 3).
Data Regarding the Presence of Flexor Hallucis Longus–Related Pathologies a

FHL, flexor hallucis longus; JSSF, Japanese Society for Surgery of the Foot ankle-hindfoot scale.
Discussion
This study, with >200 athletes with PAIS, is the only large-scale study besides the report by Zwiers et al 22 published in 2018. Similar to previous studies, the results showed that ballet dancers and soccer players most commonly presented with PAIS.2,16 The results also aligned with a recently published report that showed that PAIS is often observed in swimmers. 18 Furthermore, our study showed that PAIS can occur in players of rugby, basketball, and baseball, which has not yet been reported in the literature. It is possible that rugby and basketball players present with PAIS owing to their quick movements, such as cutting, quick turns, and jumping with high intensity. A distinctive point for baseball players was that >90% of athletes developed PAIS on the dominant side, which equals the throwing arm and the stance leg. As the pitcher strides toward home plate, ankle plantarflexion of the stance leg increases to shift the center of gravity forward. 19 Repetitive plantarflexion of the stance leg during the pitching motion can cause inflammation of the hindfoot, resulting in PAIS of the ankle on the dominant side.
Several pathologies have been reported to be associated with PAIS,6,16,21 and this study showed that pathologies related to the FHL were most frequent and most associated with surgery. These findings are in line with those of previous studies, which reported that FHL-related pathology was one of the most commonly observed pathologies among patients with PAIS.1,16,22 Zwiers et al 22 showed that the presence of FHL tendinopathy is negatively correlated with postoperative satisfaction and positively correlated with recurrence rate. Furthermore, our results revealed a significant correlation between preoperative pain duration and returning to the field. A previous study also revealed a significant correlation between the duration of symptoms preoperatively and RTT and RTS. 1 These facts highlight the importance of preoperative diagnosis, especially in relation to FHL, management of FHL-related pathologies during surgery, and determination of surgical treatment for failed nonoperative cases, as it may affect early RTT and RTS. Even though our results showed that preoperative pain duration was not associated with the presence of FHL-related pathologies, the median preoperative pain duration in athletes with and without FHL-related pathologies was 40 and 28 weeks, respectively. Future studies are needed to determine factors associated with FHL-related pathologies to enhance the treatment of PAIS, especially when determining the type of surgery.
Our study showed a relatively long duration of preoperative pain in some patients, including some outliers. There may be several reasons for this. First, although athletes experienced symptoms caused by PAIS, we did not recommend surgery if they did not feel as though the symptoms would hinder their participation or affect their performance, regardless of the competing level. Second, a visit to our clinic was not always the first visit to seek medical attention. Therefore, reporting of pain occurrence in some athletes was based on memory, which could have led to a longer duration of pain due to its ambiguity.
Several previous studies have shown that full RTS ranged from 7.1 to 14.8 weeks,4,5,10,16 which varied depending on the details of the participants or the number of players included in the study. Ribbans et al 16 concluded that soccer players tended to develop RTS earlier than ballet dancers. Another report showed that the time to RTS between professional and amateur athletes was similar, and this was investigated in several types of sports with small sample sizes. 3 Yet, one study reported recovery time delays for soft tissue–related PAIS 17 and that dancers required a longer time for recovery as compared with soccer players. 8 Our study did not show any significant differences in RTT and RTS between bony and soft tissue impingement. For example, baseball required a longer duration to fully recover (mean RTS, 16 weeks) and had the highest percentage of soft tissue impingement among sport categories. In contrast, swimming/water polo athletes recovered within a short period after surgery in all surgical cases diagnosed with bony impingement (mean RTS, 8 weeks). Because different sport categories have different demands, sport characteristics can influence the time to RTS, 3 which requires further investigation.
The only large-scale study in the past reported that 20% of the patients were professional athletes, mostly (33%) soccer players. 22 A study that focused on PAIS among swimmers revealed that the prevalence of PAIS significantly increased in those who conducted high-intensity training as compared with those who conducted low- and moderate-intensity training. 18 Our study showed that PAIS was highly prevalent in student athletes. This indicated that PAIS could also be present in high-level student athletes, suggesting that more attention should be paid to PAIS in these athletes.
Our study had several limitations. First, patient-reported outcome measures were not evaluated. As the study was conducted retrospectively by reviewing clinical records, we were unable to obtain these measures. In addition, only the JSSF score was obtained as the outcome measure. Future studies should be conducted using patient-reported outcome measures, along with other globally validated scales. In addition, establishing the minimal clinically important differences on the JSSF scale is required in future studies. Also, the JSSF score was obtained by a single surgeon, which may have induced a bias. Second, the mean follow-up period of patients who underwent surgical treatment was relatively short. As most professional athletes and high-level student athletes returned to their training facilities that were moderated by the athletic trainers of their teams, some athletes did not return to our medical facilities for routine check-ups, which hindered long-term follow-up. This could have led to missed recurrence cases in athletes. Third, the number of athletes differed among sports. This limited our statistical analysis of the participating sports, along with multivariate analysis. Fourth, recall bias might have affected the duration of preoperative pain recollection, and accounting for bilateral cases as independent cases could have introduced bias. Fifth, we did not collect objective data such as range of motion or strength, and performance was not evaluated. Although objective data are important, we focused on returning to previous level of competition, rather than regaining range of motion or strength. To further analyze multivariately, objective data and performance should be evaluated in the future study. The importance of this study is that it reveals the details and characteristics of PAIS among athletes from different sports, especially surgical outcomes and affecting factors. To get deeper insights on PAIS in athletes, future studies that address the aforementioned limitations are warranted.
Conclusion
We conducted this study to investigate characteristics of athletes with PAIS. PAIS is observed in athletes in many sports, is caused mainly by bony impingement, and is often associated with FHL-related pathology. Surgery was performed in relatively high-level athletes, and preoperative pain duration was positively related to the time of RTT and RTS. Surgical treatment of PAIS improves symptoms in most patients.
Footnotes
Acknowledgements
All authors thank the staffs of our institutions for their assistance and support in this study. They are also grateful to Editage for English-language editing.
Final revision submitted January 13, 2026; accepted January 18, 2026.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
