Abstract
Background:
Insufficient tendon-bone healing remains a major cause of rotator cuff repair failure. The inability to reestablish a native-like enthesis results in scar formation and inferior mechanics. From a biomechanical standpoint, maximizing footprint contact and minimizing micromotion are critical determinants of early repair stability. Current greater tuberosity preparation techniques are primarily directed toward smoothing the footprint surface and exposing marrow elements to enhance biologic integration; however, the biomechanical consequences of deliberately controlled surface geometry on tendon fixation strength remain undefined.
Hypothesis:
Controlled decortication channels, termed biogrooves, would reduce supraspinatus (SSP) tendon displacement and improve footprint contact area compared with standard double-row repair with a smooth surface.
Study Design:
Controlled laboratory study.
Methods:
Six fresh-frozen cadaveric shoulders were acquired. The SSP of all specimens was sharply dissected from the humeral origin. Each specimen underwent a standard double-row repair on PCF-25 Sawbone blocks. Biogrooves that were 6-mm in width and isosceles cross-section were created with a high-speed conical rotary tool and were sequentially tested across 5 conditions: (1) no biogrooves, (2) one 2 mm–deep biogroove, (3) two 2 mm–deep biogrooves, (4) two 3 mm–deep biogrooves, and (5) one 3 mm–deep biogroove. Constructs were mounted on a servohydraulic testing machine and ramped to a single load of 70 N over 30 seconds. Tendon displacement was measured under tension, and ultrasound imaging assessed footprint infill. Repeated-measures analysis of variance with Bonferroni correction was used for comparisons.
Results:
All biogroove conditions significantly reduced tendon displacement compared with the no-biogroove condition (P < .05). Mean displacement decreased by 19.5% with one 2-mm biogroove (P = .010), 25.4% with two 2-mm biogrooves (P = .021), 31.1% with two 3-mm biogrooves (P = .001), and 31.8% with one 3-mm biogroove (P = .005). The single 3-mm biogroove demonstrated a 14.3% reduction in displacement compared with the 2-mm biogroove (P = .0459). Ultrasound confirmed complete tendon infill across all groove conditions.
Conclusion:
Biogrooves reduced SSP tendon displacement and improved footprint conformity compared with standard double-row repair in this laboratory model. Groove depth was more influential than groove number, with a single 3-mm biogroove providing as much improvement as two 3-mm biogrooves.
Clinical Relevance:
Biogrooves enhance the mechanical environment for biologic healing.
Rotator cuff repair remains a technically demanding procedure with failure rates reported as high as 20% to 60%, largely attributable to inadequate tendon-to-bone healing at the repair interface.26,29 After repair, the biologic healing process progresses through inflammation, proliferation, and remodeling; however, this cascade does not reproduce the native enthesis, instead forming a fibrovascular scar tissue interface that is structurally and biomechanically inferior to the native insertion.11,12,14,36 Because early tendon-bone healing is highly sensitive to the local mechanical environment, excessive micromotion and limited interface contact can disrupt interface stability and promote disorganized-scar mediated attachment.6,9 Maximizing footprint contact and minimizing micromotion at the tendon interface are therefore critical technical goals that directly address this limitation. Consequently, considerable effort has been directed toward improving fixation constructs that maximize initial mechanical stability and create favorable conditions for biologic healing.1,18
Biomechanical investigations of rotator cuff repair have prioritized fixation-focused strategies that increase footprint contact and reduced interface motion as they have been described as critical for repair durability.1,5,25,30 In response to these mechanical demands, Millett et al 22 first described the linked repair concept, which subsequently evolved into the modern transosseous-equivalent and double-row configurations. These techniques established key construct-level principles that have been shown to biomechanically to improve load sharing, increase footprint coverage, and reduce gap formation at the tendon-bone interface.21,23,31,32 These advances have markedly improved the mechanical environment for healing, yet clinical retear rates remain high, suggesting that further mechanical refinements alone may yield diminishing returns.13,20 Although modern configurations achieve superior time-zero biomechanics, the increased repair tension and tendon compression may theoretically compromise vascularity and healing potential. Consequently, recent discussions have emphasized the need to balance mechanical stability with biologic viability during repair. 8
In contrast to fixation-focused strategies, some biologic approaches to rotator cuff repair have targeted the footprint itself. Pioneered by Steadman's early work in microfracture, the concept of mechanically modifying bone to release marrow elements during repair represented a paradigm shift in orthopaedic biologics. The microfracture or “healing response” technique demonstrated that controlled perforation of subchondral bone could release mesenchymal stem cells, cytokines, and growth factors that enhance fibrocartilaginous repair tissue formation, thereby improving the biologic environment for tendon or cartilage healing.27,28 Subsequent studies expanded this concept to the tendon-bone interface, showing that decortication of the greater tuberosity enhances tendon-to-bone contact, increases vascularity, and promotes biologic integration. 7 The principle that controlled surface modification can influence the healing environment continues to shape the modern understanding of decortication in orthopaedic surgery.3,17
Although biologically motivated, decortication is typically performed by creating a uniformly smooth footprint surface with limited consideration of how footprint surface geometry may contribute to load sharing and resistance to slip at the tendon-bone interface. By contrast, in many applied disciplines, surface geometry is deliberately engineered to control interfacial motion under load. In materials science, surface scoring enhances adhesion and load transfer between interfaces. Similarly, grooved surfaces improve resistance to shear in mechanical systems such as tire treads that increase grip or scored brake pads that enhance friction by introducing geometric barriers that disrupt slip planes and oppose sliding. Analogous principles are observed in nonbiologic systems outside of engineering, including agriculture, where harrow plowing is used to break up smooth soil crusts and reduce shallow surface sliding. Although computational and experimental models have examined enthesis-inspired geometric features such as angulation, flaring, and interdigitation at the microscale to mitigate stress concentrations, their application at the macroscale, particularly through controlled surface modification of the native bone footprint, has not been explored.10,33 Applying these engineering principles directly, controlled grooving of the greater tuberosity footprint could improve fixation mechanics while remaining compatible with modern repair techniques.
Therefore, this study introduces a novel approach of controlled decortication channels, which we have named biogrooves, created within the rotator cuff footprint and evaluates their effect on displacement and tendon infill using a repeated-measures cadaveric model. We hypothesized that the addition of biogrooves would decrease displacement and increase tendon infill compared with standard double-row repairs without decortication.
Methods
Specimen Preparation
Six fresh-frozen human cadaveric shoulders (4 unpaired, 1 paired) with intact supraspinatus (SSP) tendons were obtained (mean age, 59 years; range, 52-64 years; 6 females). Under donor consent, specimens were previously donated to a tissue bank (Science Care) designated for medical research and purchased by the research team for biomechanical research usage. Institutional review board (IRB) approval was not required as studies using decedent cadavers are IRB exempt. Shoulders with evidence of prior surgery, rotator cuff pathology, cystic changes, or poor tendon quality were excluded. After screening, the SSP tendon was sharply dissected free from its humeral origin for preparation.
Experimental Design
A repeated-measures design was used, with each specimen tested sequentially across 5 conditions (states) in the following order: (1) no biogrooves; (2) 2-mm depth, 1 biogroove; (3) 2-mm depth, 2 biogrooves; (4) 3-mm depth, 2 biogrooves; and (5) 3-mm depth, 1 biogroove (Figure 1). Transition from state 3 to state 4 was achieved by increasing the depth of the 2 existing grooves by 1 mm using the same preparation method. To transition from state 4 to state 5, the medial biogroove was filled with bone wax (Ethicon; Johnson & Johnson), leaving the lateral biogroove intact. A cross-sectional schematic of the groove configurations is shown in Figure 2.

Schematic of the 5 sequential testing states: (1) no biogroove, (2) 2-mm depth, 1 biogroove, (3) 2-mm depth, 2 biogrooves, (4) 3-mm depth, 2 biogrooves, and (5) 3-mm depth, 1 biogroove. In state 5, the second biogroove from state 4 was filled with bone paste to restore a smooth surface. Lateral row anchors (green circles) are positioned superiorly within a 45° sloped cut (shaded rectangle) and medial row anchors (black circles) inferiorly.
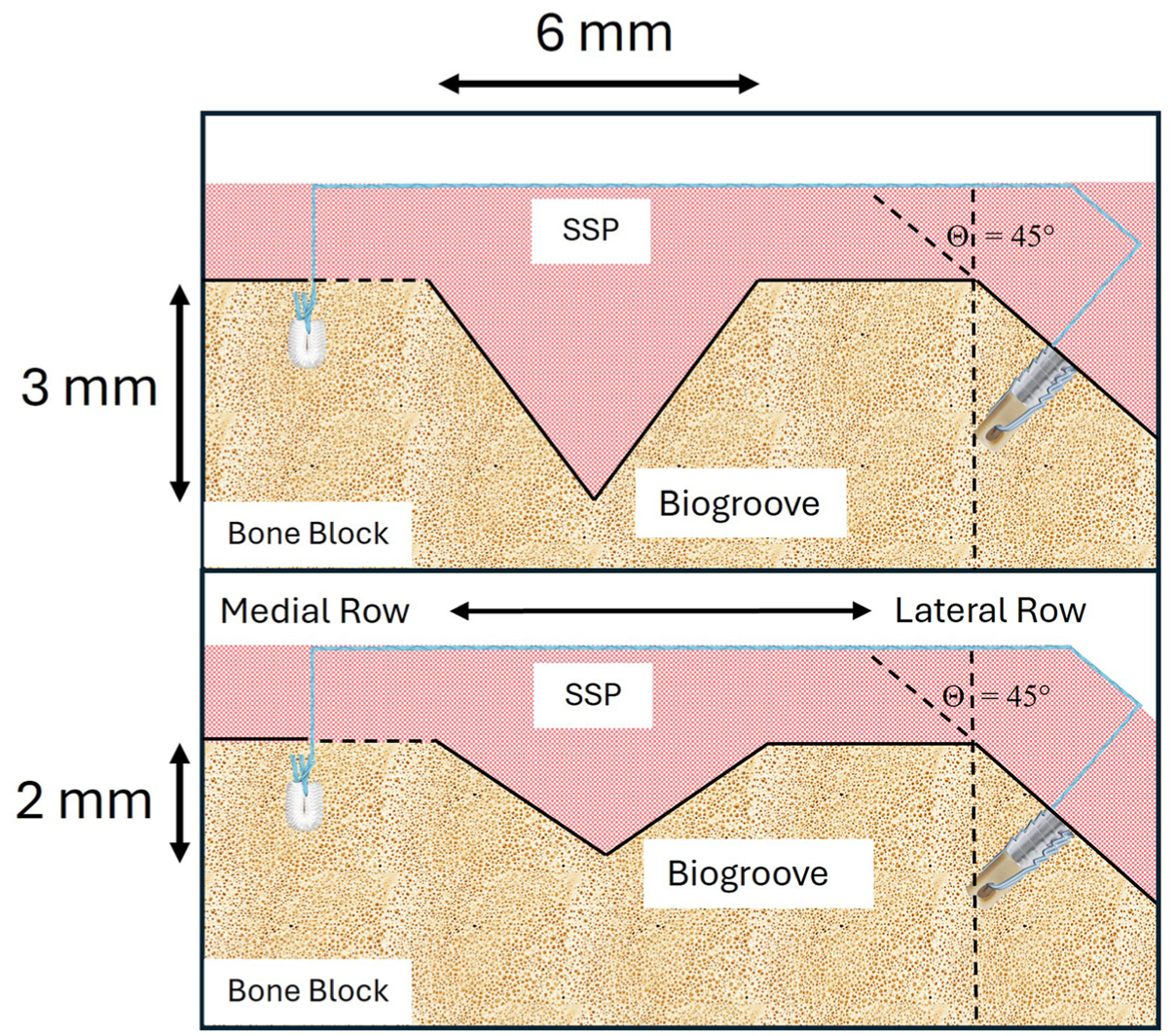
Cross-sectional schematic of biogroove dimension profiles for the biogroove configurations. The superior layer (pink) represents the supraspinatus tendon, and the underlying layer (tan) represents the prepared bone surface. A soft suture anchor is positioned in the medial row, whereas a knotless lateral-row anchor is placed along the 45° plane.
Surgical Technique of Supraspinatus Repair
A PCF-25 bone block (rigid polyurethane foam cancellous bone analog with uniform density and closed cell porosity; Pacific Research Laboratories) was prepared with a 45° sloped cut to replicate the greater tuberosity footprint. The block was premarked to standardize anchor positioning: 3 medial row anchors were marked 10 mm apart and 25 mm from the 45° slope, whereas the 2 lateral row anchors were marked 25 mm apart and centered within the 45° slope. The biogrooves were centered within the prepared footprint and positioned parallel to one another with an intergroove spacing of approximately 3 to 5 mm. The medial row sites were drilled using a 2.6-mm drill bit. The lateral row sites were drilled using a 3.4-mm drill bit and tapped with a 4.75-mm punch (SwiveLock; Arthrex).
A knotless 2.6-mm soft anchor (FiberTak RC Blue; Arthrex) was placed into each of the 3 medial row anchor sites, the suture retention ring and inserter were removed, and the remaining suture limbs were pulled slowly to set the anchor. After complete anchor seating, the sliding suture was removed. For each medial row anchor, the single end of the suture (FiberTape; Arthrex) was loaded onto a Scorpion suture passer, and the 2 limbs were brought through the SSP tendon in a single pass. Each suture limb was then tensioned independently to remove any potential slack.
One suture tail from each medial row anchor was preloaded through the eyelet of a 4.75-mm knotless anchor (SwiveLock) and advanced into 1 of the 2 lateral row anchor sites. The anchor body was then inserted by rotating the driver handle clockwise until flush with the bone block. This process was repeated for the second lateral anchor and remaining suture tails, completing the double-row construct. After initial lateral row fixation, a metallic suture clamp was crimped onto the suture limbs at a standardized reference point to record the initial tensioned position. During subsequent reattachment after groove creation, the lateral row anchors were advanced until this reference distance was reproduced, thereby standardizing lateral row suture tension across all test states.
Biogroove Creation
To create the biogrooves, the repair surface was first exposed by removing the lateral row anchors and elevating the tendon. All biogrooves were standardized to 6 mm in width with an isosceles cross-sectional geometry. During preliminary pilot testing, biogroove widths of 1 mm, 2 mm, and 6 mm were trialed in both isosceles and right triangular configurations. A 6 mm–wide isosceles geometry provided the most reliable ultrasound visualization of tendon infill in this benchtop model. Based on these observations, groove depths of 2 mm and 3 mm were selected to represent shallow and moderate decortication levels that would be practically achievable with arthroscopic burrs.
All biogrooves were manually created using a high-speed rotatory tool (Dremel Lite 7760) fitted with a 20° conical cutting bit. A conical cutting bit was used in this benchtop model to create a reproducible isosceles groove geometry with well-defined shoulders across sequential testing states. After biogroove creation, the footprint was irrigated with saline and cleared of debris. The tendon was then replaced back onto the footprint, and the lateral row anchors were reinserted into the pretapped holes such that the anchor site, suture paths, and suture tension of the lateral row were standardized across all test states. After the tendon was resecured to the Sawbone block, biomechanical testing resumed.
Biomechanical Testing Protocol
The Sawbone block and cadaveric tendon were mounted to a materials tensile testing machine (Bionix Tabletop System; MTS Systems). The block was clamped to the end effector of the testing machine, and the musculotendinous portion of the SSP tendon was fixed within a custom clamp secured to the base of the testing machine such that the repair surface was loaded in medial-lateral shear (Figure 3).
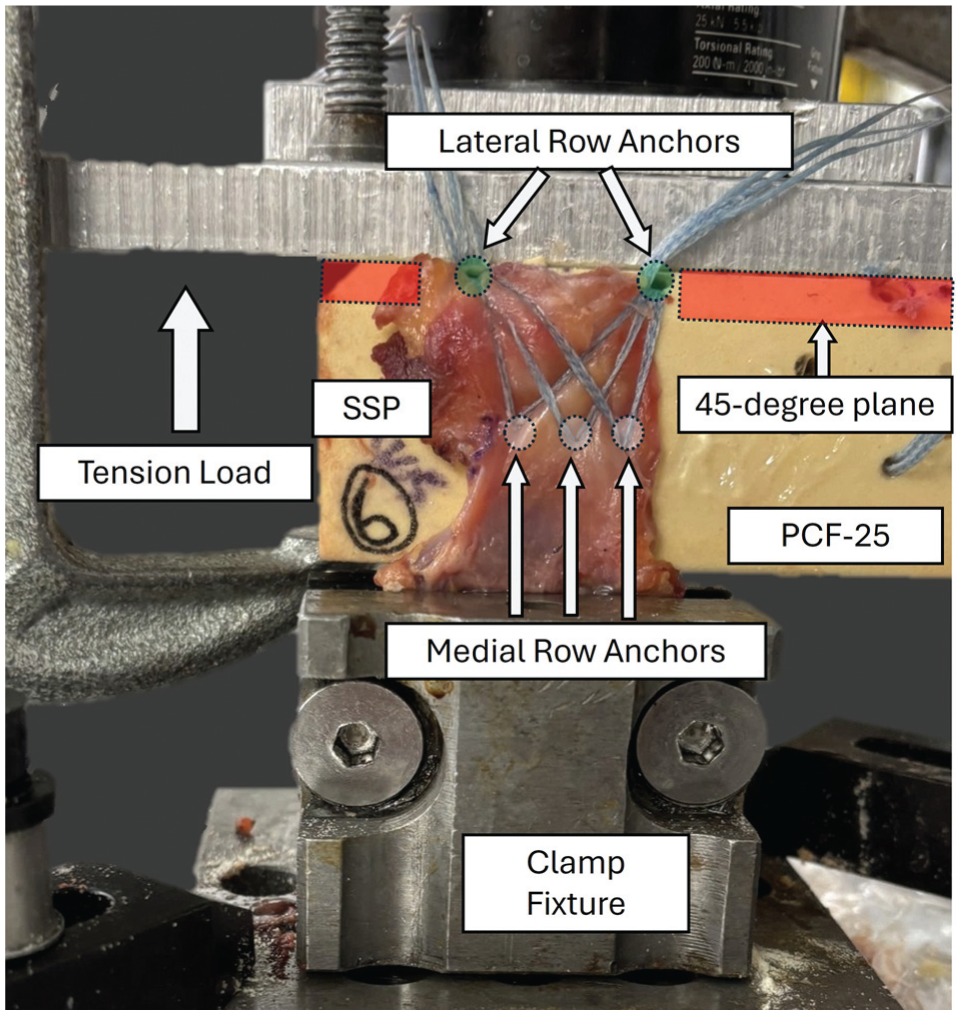
Biomechanical testing setup of the supraspinatus (SSP) repair construct on the Sawbone block. Soft suture medial row anchors are positioned inferiorly, while the knotless lateral-row anchor is placed along the 45° plane superiorly.
The specimen was preconditioned by applying 30 N of tension to the specimen for 2 minutes. An initial 5 N of tension was then applied to the specimen, and the position of the end effector was recorded. Tension was then increased to 70 N over 30 seconds, the tension was held for up to 30 seconds, and the final position of the end effector was recorded. The specimen was then unloaded for transition to the next state. Displacement was calculated as the difference between the initial and final position of the end effector. The loading conditions were chosen to be large enough to adequately detect differences in displacement between states while low enough to avoid specimen damage and minimize order bias during repeated-measures testing. During initial repeatability testing, it was determined that 70 N was the maximum tension at which no measurable change in tendon displacement occurred when all other testing conditions were held constant. Thus, 70 N was chosen as the final load point for repeatability testing.
These loading conditions were applied to the specimen across all test states. For the initial and final loading points for all biogroove test states, an ultrasound machine (Canon Aplio i800; 15-MHz probe) was used to capture an image of the repair site and to assess whether the tendon had filled the biogroove cavity (Figure 4). The initial loading point was defined as the state in which the tendon was positioned on the bone block but not yet secured with the double-row construct. Tendon infill was initially considered a quantitative variable; pilot testing demonstrated nearly complete infill (100%) across all biogroove depths and loading states tested. Accordingly, ultrasound was used qualitatively to verify tendon seating rather than as a variable for statistical comparison.
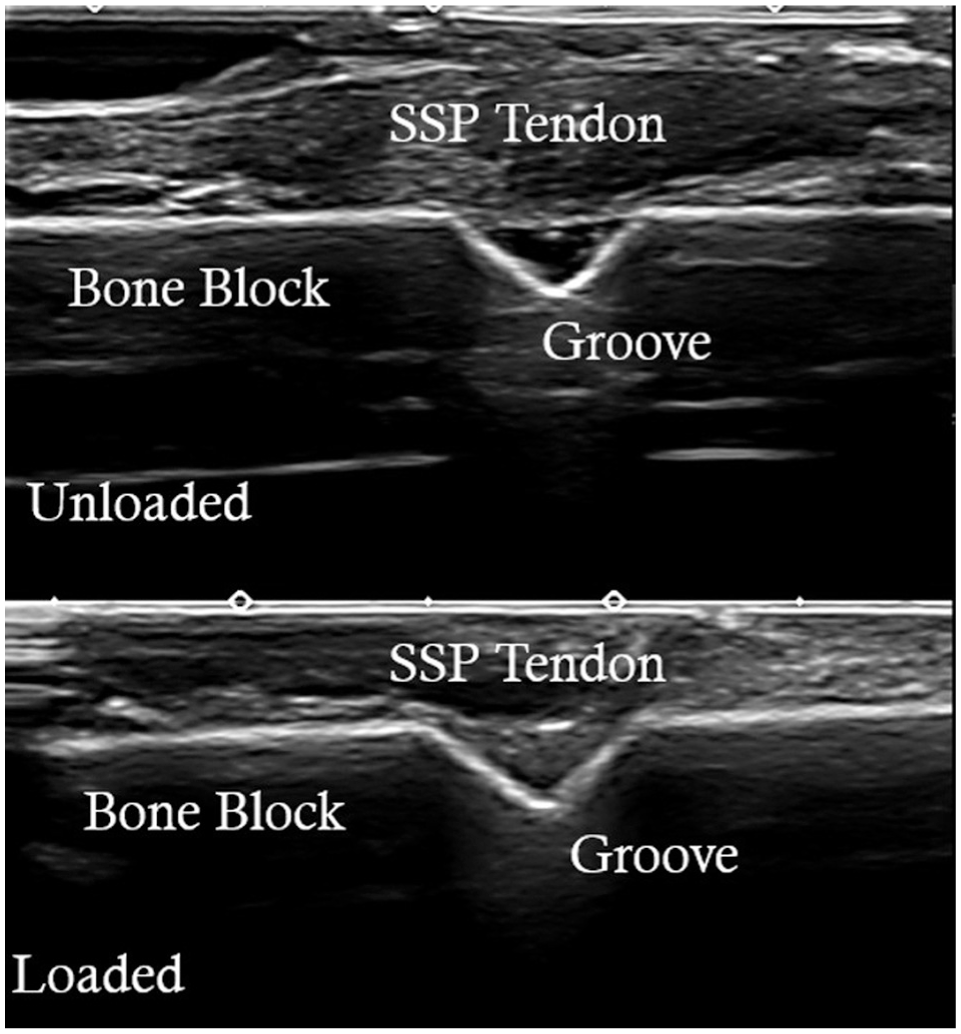
Ultrasound image of a single 3-mm biogroove in initial (unloaded) and final (loaded) states. Complete tendon infill was observed in the loaded state, demonstrating footprint contact under applied tension. SSP, supraspinatus.
Statistical Analysis
Statistical analysis and graphs were performed using MATLAB (Version R2023; The MathWorks) script. P values <.05 were considered statistically significant. The primary dependent variable of this study was tendon displacement (in millimeters). A 1-way repeated-measures analysis of variance was performed to evaluate whether the test state influenced tendon displacement. If the P value was <.05, a pairwise comparison of the means was determined, corrected for multiple comparisons using the Bonferroni method.
Results
A graphical display of all tendon displacement data can be found in Figure 5. All states with biogrooves exhibited a reduction in tendon displacement compared with the nonbiogroove state. Compared with the nonbiogroove state, 1 biogroove of 2 mm deep resulted in a 19.5% reduction in tendon displacement, 2 biogrooves of 2 mm deep resulted in a 25.4% reduction in tendon displacement, 2 biogrooves of 3 mm deep resulted in a 31.1% reduction in tendon displacement, and 1 biogroove of 3 mm deep resulted in a 31.8% reduction in tendon displacement. Furthermore, 1 biogroove of 3 mm deep resulted in 18.2% less tendon displacement compared with 1 biogroove with 2 mm displacement.
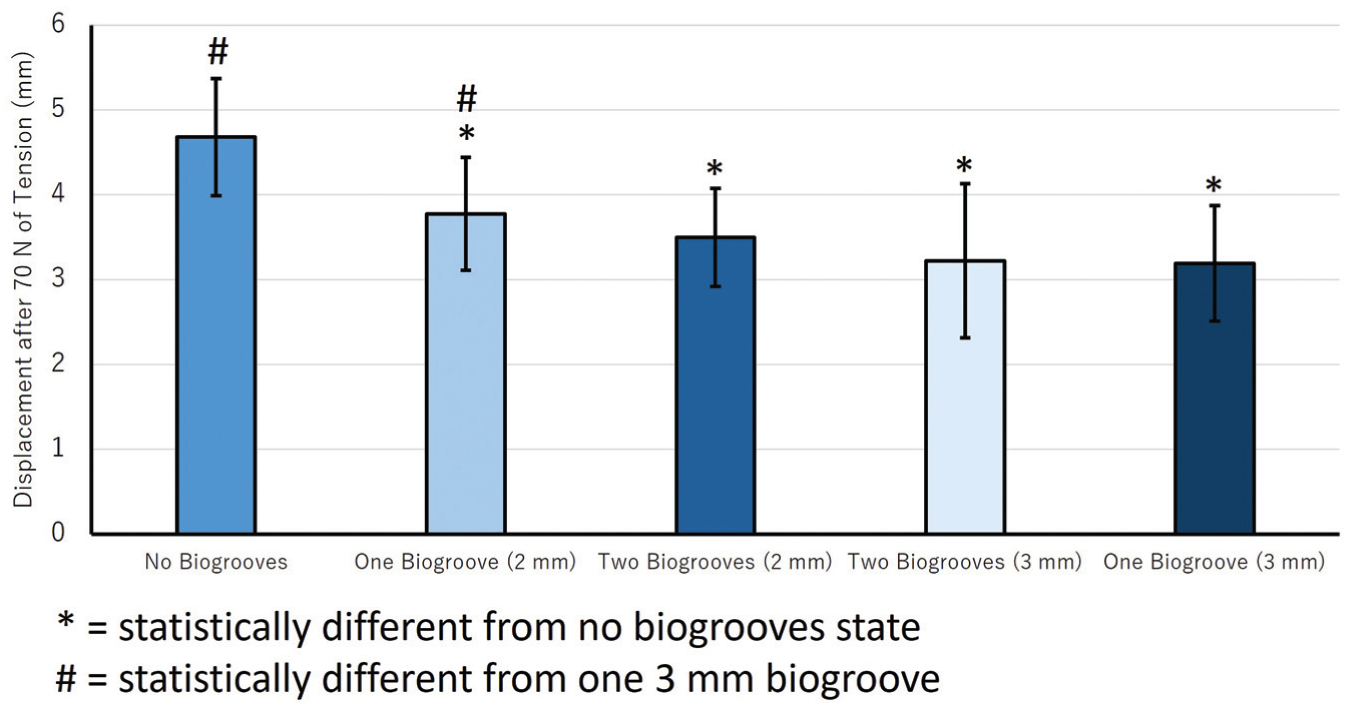
Tendon displacement after 70 N of tension across all 5 testing states. *Statistically significant difference from the no-biogroove state (P < .05). #Statistically significant difference from 1 biogroove at 3 mm depth (P < .05).
Ultrasound confirmed complete tendon infill in all biogroove loaded states (Figure 4). Infill was not observed at the initial state, when the tendon was placed on the bone block but not secured with the double-row construct.
Discussion
This study evaluated the biomechanical effect of controlled decortication channels, or biogrooves, on SSP tendon fixation during rotator cuff repair. The primary finding was that deliberate modification of footprint surface geometry using biogrooves altered tendon displacement behavior compared with a standard double-row repair performed on a uniformly smooth footprint. Among the configurations tested, a single 3-mm biogroove demonstrated the lowest tendon displacement. Additionally, lower displacement values were observed in the deeper biogroove conditions compared with the shallower ones. Multiple biogrooves did not significantly reduce displacement compared with single biogroove configurations. Ultrasound confirmed complete tendon infill across all biogroove states.
The reduction in tendon displacement observed with biogroove preparation can be attributed to mechanical changes in footprint geometry. Compared with shallow configurations, a single deep biogroove is likely to increase shear resistance by creating a physical barrier that the tendon must overcome to translate along the bone surface. As the tendon is tensioned, the biogroove shoulders generate greater normal forces that oppose sliding and stabilize the interface. In contrast, multiple biogrooves did not provide incremental stability. This finding suggests a threshold effect: Once sufficient depth is achieved to engage the tendon, additional biogrooves yield diminishing returns. According to contact mechanics theory, subdividing a continuous contact interface into multiple smaller regions can disperse local forces and reduce the normal pressure at each biogroove shoulder, thereby diminishing the degree of mechanical interlock.4,15 The ordered pattern of mean displacement observed across testing states supports this interpretation. Taken together, these results point toward depth as the most critical design parameter for footprint surface preparation.
To our knowledge, this is the first study to evaluate the effect of controlled footprint decortication geometry on the biomechanics of rotator cuff repair. Previous investigations have largely prioritized improving time-zero fixation strength of repair constructs. These, however, are constrained by the competing effect of increased tension on biologic healing.8,34 Modern double-row and transosseous-equivalent repairs already restore much of the native footprint and improve load-to-failure characteristics compared with earlier single-row techniques.21,31 Despite these advances, retear and nonhealing rates remain substantial,26,29 suggesting that construct-level improvements alone may not fully address the local mechanical environment governing early tendon-bone interface behavior.1,5,26,29,30
Prior work supports the biologic rationale for footprint preparation: Decortication and marrow exposure can improve tendon-to-bone contact and create a more favorable healing milieu.3,14,17,18,28 Animal biomechanics studies further reinforce that surface perforation can enhance interfacial biology and mechanics. In a recent rat rotator cuff model, drilling of the footprint while preserving native fibrocartilage increased marrow-derived chondrogenic cells and produced a higher ultimate load to failure at 8 weeks. 24 Similarly, in a rabbit model, combining bone marrow stimulation with losartan improved tendon matrix organization and significantly increased load to failure, demonstrating that biologic modulation can potentiate the mechanical benefits of marrow stimulation. 16 Collectively, these findings suggest that in addition to continued refinement of anchors and suture constructs, footprint surface geometry itself may represent an additional, underexplored contributor to the mechanical environment of tendon-bone healing.
This study identifies footprint geometry as a modifiable parameter that can augment contemporary fixation techniques. We found that under the conditions tested, tendon engagement depended more on biogroove depth than number, suggesting that depth is a primary determinant of shear resistance. Beyond depth, groove geometry, placement, and number are likely to interact to influence interfacial mechanics. The isosceles profile used in this study was selected primarily for its reproducibility using a conical cutting bit while allowing clear ultrasound visualization of tendon infill. Pilot testing of unidirectional configurations right triangle yielded comparable mechanical results but lacked consistent visualization of tendon engagement within the groove. Alternative configurations such as directional or overlapping patterns may enhance anisotropic shear resistance or improve tendon interdigitation. However, more complex or aggressive geometries may also remove greater volumes of cortical bone and introduce stress concentrations that increase fracture risk, particularly when grooves are placed near anchor sites or in osteoporotic bone.2,35 Additionally, repetitive cyclic loading may introduce tendon wear or abrasion at the groove edges, particularly if sharp transitions are present. Future studies incorporating cyclic loading and modified groove edges will be necessary to evaluate potential wear mechanisms and optimize groove design or durability. The 6-mm groove width was selected after pilot testing to ensure consistent ultrasound visualization and tendon seating but occupies a substantial portion of the usable footprint. 19 Translation to practice will require scaling to footprint-specific anatomy, defining safe spacing relative to anchor tracts, and developing standardized instrumentation. Future work should compare cross-sectional profiles and orientations, assess cyclic loading behavior, and evaluate how suture configuration interacts with groove placement.
If validated through cyclic loading and biologic studies, controlled footprint engineering could represent a simple, workflow-compatible method to further reduce micromotion and strengthen the tendon-bone interface. Biogroove preparation may augment the time-zero mechanical conditions that support biologic integration during the early healing. This approach complements advances in double-row and transosseous equivalent fixation by focusing on the footprint itself. Ultimately, precise mechanical surface preparation may help bridge the gap between mechanical stability and biologic healing, offering a practical and scalable method to improve clinical outcomes in rotator cuff repair.
Limitations
This study has several limitations. First, it was performed at time-zero in cadaveric specimens and therefore cannot account for biologic healing or long-term durability. Second, an a priori power analysis was not performed given the repeated-measures design, with each specimen serving as its own control. Although statistically significant differences between testing states were observed, the modest sample size remains a limitation. Third, to accommodate the repeated-measures protocol, biomechanical loading was restricted to shear forces up to 70 N, which does not replicate the multidirectional and cyclic forces experienced by the rotator cuff in vivo. Fourth, groove width was standardized to 6 mm to facilitate reliable ultrasound visualization, which exceeds typical clinical dimensions and may limit direct generalizability. Fifth, only 1 groove geometry was tested, and the effects of alternative configurations, groove spacing, and placement relative to anchors remain unknown. We also acknowledge that surface modification inherently reduces cortical thickness and could alter local stress distribution, particularly in osteoporotic bone, but fracture risk and anchor fixation strength were not directly measured. Sixth, testing was performed on standardized bone blocks rather than native humeri, limiting generalizability to clinical bone quality and anatomy.
Conclusion
Biogroove reduced SSP tendon displacement and improved footprint conformity compared with standard double-row repair in this laboratory model. Groove depth was more influential than groove number, with a single 3-mm biogroove providing as much improvement as two 3-mm biogrooves.
Footnotes
Final revision submitted December 30, 2025; accepted January 4, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.T.P. reports funding grants and travel reimbursement from Arthrex; consulting or advisory for Arthrex, Arthrosurface, Anika Therapeutics, and JRF Ortho; funding grants from the US Department of Defense and the National Institutes of Health; and board membership with Arthroscopy Association of North America, American Academy of Orthopaedic Surgeons, American Orthopaedic Society for Sports Medicine, American Shoulder and Elbow Surgeons, San Diego Shoulder Institute, Society of Military Orthopaedic Surgeons, and Arthroscopy. P.J.M. has received IP royalties, consulting fees, and research support from Arthrex; research support from Ossur, Siemens, and Smith & Nephew; and publishing royalties and financial or material support from Springer; and he holds stock or stock options in VuMedi. T.D. and M.H. report employment with Arthrex.
Ethical approval was not sought for the present study.
