Abstract
Background:
Hip osteoarthritis is a debilitating condition that leads to progressive joint pain and stiffness. While total hip arthroplasty provides definitive treatment, intra-articular injections offer a less invasive alternative for patients. Several injection options are available, including corticosteroids (CS), hyaluronic acid (HA), and platelet-rich plasma (PRP). Previous reviews and network meta-analyses have compared the short-term efficacy of these injections, but it remains unclear if a particular injection provides superior symptom relief for up to 12 months.
Purpose:
To provide an updated summary of the current hip intra-articular injection literature and compare the efficacy of all injection types at 3 months, 6 months, and 12 months.
Study Design:
Systematic review; Level of evidence, 1.
Methods:
Four databases were queried: Web of Science, Embase, MEDLINE, and Cochrane Central Register of Controlled Trials. The primary outcome measures were the Western Ontario and McMaster Universities Osteoarthritis Index Total Score (WOMAC–Total) and the visual analog scale (VAS) at 3, 6, and 12 months. The Cochrane risk-of-bias tool was used to assess study quality. Treatment effects were expressed as mean differences for the WOMAC–Total and standardized mean differences for the VAS.
Results:
A total of 14 studies were included in the final analysis with 1254 participants. Eight unique intra-articular injection types were identified: CS, HA of varying molecular weights (low, high, and ultra-high), PRP, CS + high molecular weight HA, PRP + HA, and standard of care/placebo (SOC/PBO) group. When compared with SOC/PBO, no statistically significant differences in WOMAC–Total and VAS outcomes were observed between any injections at 3, 6, or 12 months.
Conclusion:
There were no statistically significant differences in WOMAC–Total and VAS outcomes at any time point between all injection types to baseline. Future studies should compare the long-term efficacy of various intra-articular injections with a control and examine the efficacy of combined injections.
Registration:
CRD42024574937 (PROSPERO identifier).
Hip osteoarthritis (OA) is a debilitating condition that causes pain and stiffness, which can substantially affect patients’ physical function, mental health, and overall quality of life.29,46 Many patients suffer from hip OA, as it is estimated to affect 7.95% of the adult population in North America and 12.59% in Europe. 18 While severe hip OA can be definitively treated with total hip arthroplasty, the procedure may not be suitable for all patients based on the associated risks of the surgery and patients’ own preferences. An alternative to total hip arthroplasty is intra-articular injections, which provide temporary symptom alleviation for patients. Several injection options are available on the market, including corticosteroids (CS), hyaluronic acid (HA), and platelet-rich plasma (PRP).19,21 Different types of intra-articular injections act in different ways to relieve hip OA symptoms. For example, CS inhibits the activity of proinflammatory cytokines and helps reduce inflammation by attenuating the immune system. 31 PRP is derived from autologous blood and contains a high concentration of platelets, growth factors (eg, vascular endothelial growth factor and platelet-derived growth factor), and immunomodulatory cytokines (eg, interleukin 1–beta, IL-1Ra). 52 It has been suggested that immunomodulatory cytokines reduce inflammation in the joint space, ultimately alleviating hip OA symptoms. 1 Last, HA alleviates OA by providing chondroprotective, anti-inflammatory, and lubrication effects in the joint space.17,23 Given the diverse intra-articular injection options, it can be challenging for clinicians and patients to choose the best option. Hence, it is clinically important to determine the relative efficacy of injections. Elucidating the relative efficacy of these injections will improve outcomes, guide protocols, and optimize the use of health care resources.
Prior reviews and network meta-analyses (NMAs) offer important contributions to the field, but existing results have been based on short-term follow-up durations (eg, up to 6 months for Gazendam et al 21 ) or outdated database searches (eg, Zhao et al 51 ). Zhao et al also did not differentiate between different formulations of HA, such as high molecular weight (HMW-HA) versus low molecular weight (LMW-HA), and some evidence suggests they may vary in efficacy.7,37,49,50 Results from existing NMAs are also inconsistent. Gazendam et al 21 is the most recent synthesis of the literature and compared the efficacy of HA, CS, and PRP with up to 6 months of follow-up. The authors reported no difference in symptom alleviation between HA, CS, PRP, and PRP + HA compared with controls. Zhao et al compared HA, PRP, CS, and PRP + HA with controls. They reported significant advantages of HA and CS in reducing pain at 3 months compared with controls (saline or local anesthetic injections). The conflicting results in the literature leave the comparative efficacy of available injections unclear.
The current study updates prior work by extending follow-up to 12 months, incorporating new randomized controlled trials (RCTs) published since 2021, and differentiating molecular weight formulations of HA. Compared with a conventional meta-analysis, which only synthesizes outcomes from direct comparisons, NMA facilitates indirect comparisons among treatments within the same network. 20 These indirect comparisons are advantageous in the presence of literature gaps or limited sample sizes. 11 The current NMA will evaluate the efficacy of intra-articular injections for managing hip OA at 3, 6, and 12 months.
Methods
This systematic review and NMA was prospectively registered on PROSPERO. We adhered to The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-analyses of Health Care Interventions (see complete checklist in Appendix Table A1). 25
Study Identification
Four databases were systematically searched for relevant studies from inception to November 20, 2023: Web of Science (Core Collection), Embase, MEDLINE, and Cochrane Central Register of Controlled Trials (CENTRAL). No language or date limits were applied. The search strategy included key terms covering hip OA, injection options, and delivery routes. The complete search strategy is available in Supplemental Material Tables S2 to S5 (available separately). The reference section of previous reviews was also searched for relevant articles.
Inclusion criteria for this review included RCTs investigating adult patients (≥18 years of age) with a diagnosis of hip OA. To be eligible, studies were required to assess the use of intra-articular injections, have a minimum follow-up period of ≥3 months, and report the mean and standard deviation of the Western Ontario and McMaster Universities Osteoarthritis Index Total Score (WOMAC–Total) and the visual analog scale (VAS). The exclusion criteria included studies not available in English, unpublished studies, and cadaveric studies. Additionally, we excluded crossover RCTs, pseudo-randomized RCTs, case studies, case series, retrospective observational studies, and any trial that did not report the mean and standard deviation for WOMAC–Total and VAS.
Two reviewers (C.L. and J.D.) independently screened the articles in duplicate. Eligible studies compared any intra-articular injection therapy with another injection. Studies deemed eligible by both reviewers then underwent subsequent independent full-text screening. Disagreements between the 2 reviewers were resolved through discussion, with 2 authors (D.L.L. and A.A.) providing additional consultation if needed.
Data Extraction
Two reviewers (C.L. and J.D.) performed data extraction independently and in duplicate using a standardized data extraction form established a priori. Discrepancies during extraction were resolved through discussions between the 2 reviewers. From each included article, the following information was extracted: publication year, number of participants, number of hip joints, mean age, mean body mass index, adverse events (AEs), severe adverse events (SAE), and the Kellgren-Lawrence (KL) grade. We extracted the WOMAC–Total score and the VAS score at baseline, 3 months, 6 months, and 12 months.
Risk of Bias
The revised Cochrane risk-of-bias tool for randomized trials (RoB2) was used to evaluate the risk of bias for all articles. 45 The 2 reviewers independently assessed the risk of bias for all articles, and conflicts were resolved with consultation with 2 other authors of the team (D.L.L. and A.A.).
Statistical Analysis
All statistical analyses were completed on R Statistical Software (Version 1.4.1717) using the netmeta package.3,36 For direct comparisons, pairwise meta-analyses were conducted to estimate pooled mean differences and 95% CIs for WOMAC–Total outcomes. For VAS, we calculated the standardized mean differences because of heterogeneity in scale formats across studies. For example, some studies used a 0 to 10 scale (eg, Di Sante et al 16 ), while others used a 0 to 100 scale (eg, Rezende et al 38 ).
Statistical significance was determined based on the 95% CIs. A comparison was considered statistically significant if the confidence interval did not include the null value. A random-effects model was used to account for potential heterogeneity across studies, with between-study variance (τ2) estimated using the DerSimonian-Laird method. 15 Heterogeneity was further quantified using the I2 statistic. An I2 value of 0% indicated no observed heterogeneity, while higher values reflected increasing inconsistency among study results. 24 In contrast to traditional direct comparisons, NMA evaluated both direct and indirect evidence across a network of treatments. Probability scores (P-scores) were derived from networks and were used to compare the relative efficacy between injections. P-scores are frequentist analogs to the Surface Under the Cumulative Ranking (SUCRA) scores in Bayesian NMAs. 40 P-scores are derived from point estimates and standard errors of treatment effects. Similar to SUCRA scores, P-scores measure the mean extent of certainty that one injection type is better than another. Unlike SUCRA, P-scores do not reflect Bayesian probability-based ranking and do not require resampling methods. 40 P-scores range from 0 to 1. A P-score close to 1 indicates that a particular injection is likely the most effective compared with all other injections. In contrast, a P-score of 0 indicates that the treatment is likely the least effective.
A network plot was created to illustrate the relationship between injections among included studies. The size of the nodes was proportional to the number of studies evaluating each injection, and the thickness of the edges represented the relative number of direct connections between the connected nodes. Isolated nodes indicated a lack of direct comparison, which could limit the precision of indirect estimates.
Results
After removing duplicates, a total of 1909 studies were identified. After title and abstract review, 63 studies progressed to full-text review and were assessed for eligibility. Ultimately, 14 studies met the inclusion criteria and were included in the final analysis (Figure 1).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram. The PRISMA flow diagram is adapted from the template outlined in Page et al. 34
Study Characteristics
Across all injections, the 14 included studies covered a total of 1254 participants. Table 1 summarizes all included articles. Eight different intra-articular injections were included in the current NMA: (1) CS, (2) ultra–high molecular weight hyaluronic acid (UHMW-HA), (3) HMW-HA, (4) LMW-HA, and (5) PRP. Additionally, combination therapies including (6) CS plus HMW-HA (CS + HMW-HA), (7) PRP plus HMW-HA (PRP + HMW-HA) and standard of care/placebo (SOC/PBO) were included. The (SOC/PBO) group served as a comparator when possible, and included participants who received saline or local anesthetic injections (eg, Migliore et al 32 used mepivacaine, Richette et al 39 used saline water). Some studies included a noninjection arm. For example, Paskins et al 35 had a noninjection arm, which educated patients on noninjection ways to alleviate symptoms, such as exercise and weight loss. These noninjection groups were excluded from the final analysis. The network diagram for WOMAC–Total and VAS at 3 months, 6 months, and 12 months can be found in the Supplemental Material (Figures S1 and S2, available separately).
Summary of Participant Demographics a

Levels of hip osteoarthritis severity are reported as KL grade. Overall, HA was the most studied treatment. Note that in Rezende et al, 38 the 4-mL hylan G-F20 arm showed an inconsistency in the KL grade reported: 36 hips were included, but only 24 KL 2 and 10 KL 3 hips were accounted for. Villanova-Lopez 48 combined KL 3 and 4. We reported the data the authors presented. BMI, body mass index; CS, corticosteroids; HA, hyaluronic acid; HMW, high molecular weight; KL, Kellgren-Lawrence; LMW, low molecular weight; NR, not reported by the authors or not applicable to the study; PRP, platelet-rich plasma; UHMW, ultra–high molecular weight.
Risk of Bias
Based on the RoB2 assessment, 8 studies were rated as having a low risk of bias, and 6 studies were rated as having some concerns regarding risk of bias (Figure 2). Most of the bias arose from the randomization process, missing data, and reported results.
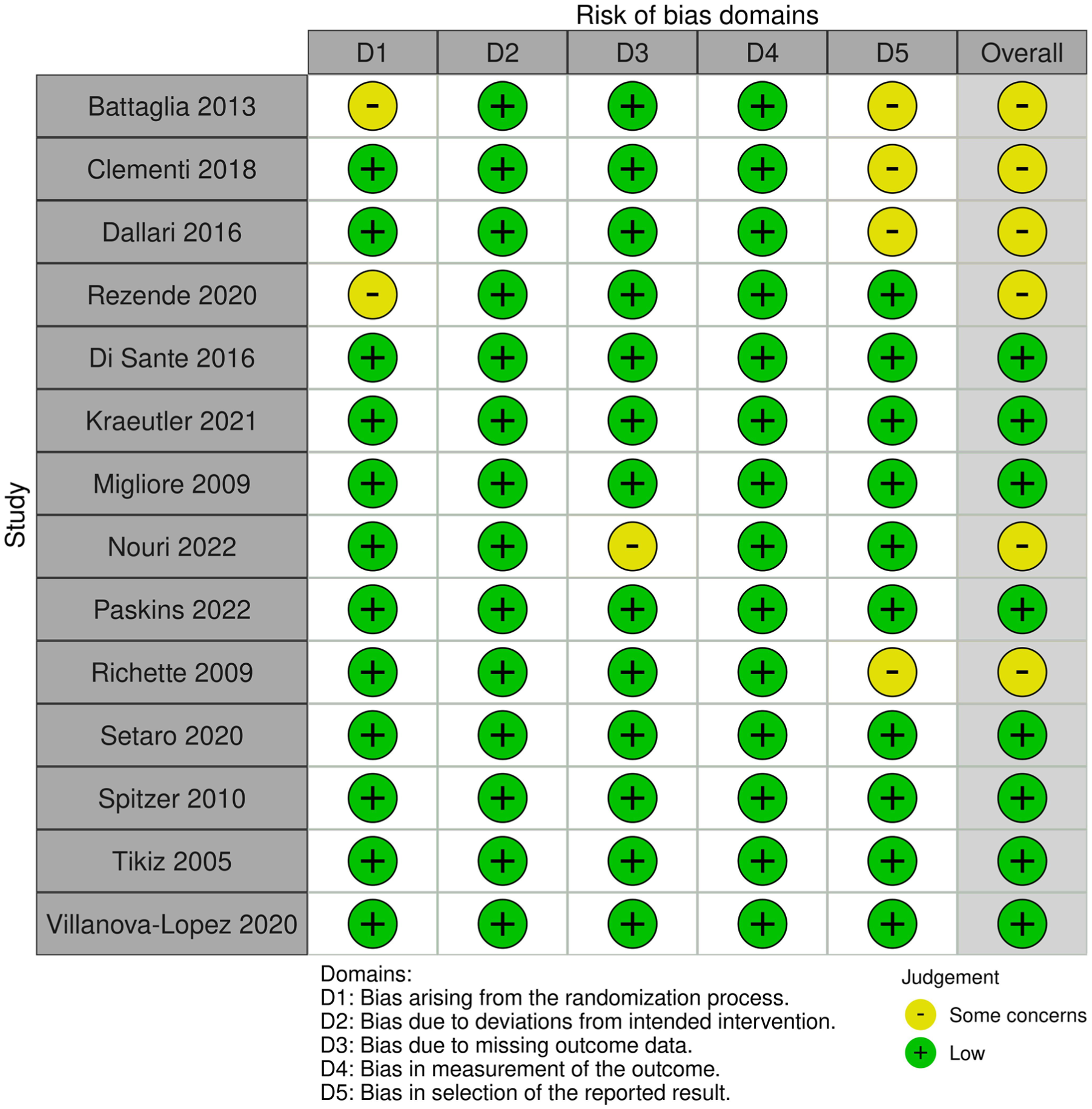
Summary plot for risk of bias. Six studies had some concerns for risk of bias. Bias arose from domain 1 (bias arising from the randomization process), domain 3 (bias due to missing outcome data), and domain 5 (bias in selection of the reported result).
WOMAC–Total
At 3 months, 6 articles reported the WOMAC–Total score for 5 injections involving 305 participants.10,27,38,39,41,47 Results from these 6 articles showed no difference in WOMAC–Total between all injections and SOC/PBO (Figure 3) (τ2 = 0.0; I2 = 0.0%). At 6 months, 9 articles reported 8 injections, covering 941 participants. ** These 9 studies also indicated no difference in WOMAC–Total score between all injection types and placebo (Figure 4) (τ2 = 24.42; I2 = 59% [95% CI, 0.0%-83.4%]). At 12 months, 5 articles reported the WOMAC–Total score for 4 injections involving 301 participants.10,27,38,41,48 Since no articles reported WOMAC–Total outcomes with SOC/PBO, PRP was set as the baseline, and there was no significant difference between other injections with PRP (Figure 5) (τ2 = 0.0; I2 = 0.0%).

In WOMAC–Total at 3 months, there was no statistically significant difference between all injections and SOC/PBO. HMW-HA ranked highest on the P score. HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; MD, mean difference; PRP, platelet-rich plasma; UHMW, ultra–high molecular weight; SOC/PBO, standard of care/placebo; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.

In WOMAC–Total at 6 months, there was no statistically significant difference between all injections and SOC/PBO. PRP+HMW-HA ranked highest on the P score. CS, corticosteroids; HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; MD, mean difference; PRP, platelet-rich plasma; UHMW, ultra–high molecular weight; SOC/PBO, standard of care/placebo; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.

In WOMAC–Total at 12 months, no studies reported results for a standard of care/placebo, so different injection options were compared against PRP. There was no statistically significant difference between all injections against PRP, and PRP ranked highest on the P score. HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; MD, mean difference; PRP, platelet-rich plasma; UHMW, ultra–high molecular weight; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Based on P-score rankings, HMW-HA was the highest-ranking injection at 3 months (P-score, .65). At 6 months, PRP + HMW-HA was the highest-ranking injection (P-score, .86). At 12 months, PRP was the highest-ranking injection (P-score, .71).
Visual Analog Scale
At 3 months, 6 articles reported pain outcomes covering 381 participants.4,10,32,38,41,47 There was no difference between 4 different types of injection treatments against SOC/PBO (Figure 6) (τ2 = 0.24; I2 = 74.2% [95% CI, 28.0%-90.8%]). At 6 months, 7 articles reported 7 injections, involving 497 participants, with no difference across all injection types to SOC/PBO (Figure 7) (τ2 = 0.18; I2 = 74.1% [95% CI, 44.7%-87.9%]).4,32,33,38,41,47,48 Finally, at 12 months, 6 articles reported VAS, comparing 5 different injections and including 481 participants.4,10,13,38,41,48 However, no article reported outcomes for SOC/PBO at 12 months, so PRP was used as the baseline. All injections had no significant difference compared with PRP at 12 months (Figure 8) (τ2 = 0.16; I2 = 73.6% [95% CI, 26.1%-90.6%]).

In the visual analog scale at 3 months, there was no statistically significant difference between all injections and SOC/PBO. LMW-HA ranked highest on the P score. HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; SMD, standardized mean difference; PRP, platelet-rich plasma; SOC/PBO, standard of care/placebo; UHMW, ultra–high molecular weight.

In the visual analog scale at 6 months, there was no statistically significant difference between all injections and SOC/PBO. PRP ranked highest on the P score. HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; SMD, standardized mean difference; PRP, platelet-rich plasma; SOC/PBO, standard of care/placebo; UHMW, ultra–high molecular weight.

In the visual analog scale at 12 months, no studies reported results for SOC/PBO, so different injection options were compared against PRP. There was no statistically significant difference between all treatments against PRP, and LMW-HA ranked highest on the P score. HA, hyaluronic acid; HMW, high molecular weight; LMW, low molecular weight; SMD, standardized mean difference; PRP, platelet-rich plasma; SOC/PBO, standard of care/placebo; UHMW, ultra–high molecular weight.
At 6 months, PRP was likely the highest-ranking injection (P-score, .77). At 12 months, LMW-HA was the highest-ranking injection (P-score, .84)
Safety Profile
Nine studies reported AEs related to the injection (Table 2). †† The most commonly reported AE was injection-site pain, which was reported across 7 studies affecting 38 participants.4,13,32,35,39,44,47 Three studies reported 4 SAEs.35,39,44 The SAEs included 1 case of rapidly destructive OA requiring hip replacement, 1 case of subacute bacterial endocarditis, 1 case of spontaneous abortion, and 1 case of choroidal dystrophy (Table 3).
Adverse Reactions a

AE, adverse event; n, number of participants who reported the specified AE; N, total number of participants in that arm; CS, corticosteroids; HA, hyaluronic acid; HMW, high molecular weight; KL, Kellgren-Lawrence; LMW, low molecular weight; PRP, platelet-rich plasma.
For Tikiz 2005, n is the number of hips, and N is the total number of hips.
Severe Adverse Reactions a

AE, adverse effect; CS, corticosteroids; HA, hyaluronic acide; HMW, high molecular weight; OA, osteoarthritis.
Discussion
This study provides an updated review and comparison of intra-articular injections for hip OA. The current NMA included 8 different types of intra-articular injections for hip OA: CS, PRP, LMW-HA, HMW-HA, UHMW-HA, PRP + HMW-HA, CS + HMW-HA, and SOC/PBO. The efficacy of these injections was assessed on WOMAC–Total and VAS. We found no statistically significant differences between all injections at 3 months, 6 months, and 12 months on WOMAC–Total and VAS.
The NMA-derived P-scores suggest that some injections may be superior at specific time points, even though direct comparisons revealed no statistically significant differences between them. It is important to acknowledge that P-scores do not directly provide a measure of statistical significance. Instead, P-scores rank injections based on their probability of being superior, considering the point estimates and standard errors across the network. 40 P-scores should always be interpreted in the context of confidence intervals. While an injection with the highest P-score may appear superior, a confidence interval that includes zero indicates that the difference compared with SOC/PBO is statistically insignificant and the evidence may be inconclusive. 8
The current NMA shows no statistically significant difference between UHMW-HA, HMW-HA, and LMW-HA. The current evidence adds to the existing literature, which has been inconsistent regarding whether molecular weights affect efficacy. Some evidence suggests that HMW-HA is better than LMW-HA for managing OA symptoms.37,49,50 Others have argued that they are comparable.2,12,22,28,42 While we report variations in P-score rankings between UHMW-HA, HMW-HA, and LMW-HA, the P-scores alone are insufficient to conclude if one HA formulation is better than another. The rapid degradation of materials in the joint space could account for the lack of significant long-term differences between HA formulations and across all studied injections. For instance, the terminal half-life of corticosteroids in the joint space is less than a week. 14 Their relatively short half-life could contribute to their short period of efficacy. It has been suggested that HA with a higher molecular weight is better at resisting degradation in the joint space, but results of our NMA do not support this theory.7,17,30 The lack of significant long-term differences across all injections reflects the complex pathophysiology of hip OA, and no single treatment can address all aspects of the disease process.
Results from the current NMA have important implications for both clinical practice and future research. Our results show that no injection is superior. Practically, this means clinicians do not need to favor one type over another based solely on efficacy. Instead, factors such as patient preference, cost, and availability can play a larger role in the shared decision-making process. For patients, knowing that all injections perform similarly could have important implications for the cost of care, as some health care systems or insurance plans may only cover certain injections but not others. 5 Our review of the literature also reveals a gap: no studies have compared an injection with a SOC/PBO group at 12 months of follow-up. This gap makes it difficult to infer if any injection provides benefits beyond SOC/PBO in the long term.
Another gap in the literature is the limited evidence on combination therapies, such as PRP + HMW-HA and CS + HMW-HA. Only 3 studies in our review used a combination therapy. Theoretically, combination therapies provide better symptom alleviation by combining the complementary benefits of different injections. For example, PRP offers regenerative and immunomodulatory effects, while HA contributes mechanical and anti-inflammatory benefits.9,26 Although our findings indicate no statistically significant advantage for combination therapies, the lack of significant differences may be due to the limited evidence in the literature and the small sample sizes of the 3 included studies. Meta-analyses in knee OA suggest that combination therapy is more advantageous than single therapy.26,43 However, these findings should be interpreted with caution since current evidence relies on a limited number of primary studies.
Limitations
This study is not without its limitations. We were unable to stratify outcomes by KL grade because the included studies did not report results stratified by KL grade. None of the included studies had an SOC/PBO group at 12 months, which limited our ability to compare the efficacy of injections against a baseline. Finally, heterogeneity is an inherent limitation of NMAs, as variability across included studies introduces bias. 11 The P value does not adjust for the uncertainty or heterogeneity in the network. Injections with wide confidence intervals or high variability may still receive high P values. 11 Given that the current NMA only included 14 trials, we acknowledge that the P value should be interpreted with caution, as the P value ranking may fluctuate with the addition of future studies.
Conclusion
This systematic review and NMA evaluated the efficacy of intra-articular injections for reducing hip OA symptoms. We compared 8 types of injections at 3, 6, and 12 months and found no significant differences among all injection types at any time point. There is a lack of placebo-controlled trials with longer-term follow-up, which reflects a gap in the literature. Further research on biologic treatments and combination therapies is needed to clarify their comparative efficacy for hip OA.
Footnotes
Appendix
Cochrane Central Register of Controlled Trials (CENTRAL) Search Strategy a

| Line # | Search statement | Results |
|---|---|---|
| #1 | (platelet-rich or platelet-derived or (platelet NEAR/3 (concentrate or plasma or gel or fibrin or growth factor or lysate)) or "PRP" or "PRGF" or "PRF" or (autologous NEAR/3 (plasma or protein)) or "APS" or "ACP"):ti,ab | 10,120 |
| #2 | MeSH descriptor: [Platelet-Rich Plasma] explode all trees | 967 |
| #3 | (steroid* or cortico* or glucocortico* or dexamethason* or methylprednisolon* or betamethason* or triamcinolon* or hydroxycortico* or hydrocort* or cortison* or prednison* or prednisolon*):ti,ab | 81,282 |
| #4 | MeSH descriptor: [Steroids] explode all trees | 68,923 |
| #5 | MeSH descriptor: [Dexamethasone] explode all trees | 5687 |
| #6 | MeSH descriptor: [Methylprednisolone] explode all trees | 3109 |
| #7 | MeSH descriptor: [Prednisolone] explode all trees | 5586 |
| #8 | MeSH descriptor: [Prednisone] explode all trees | 4497 |
| #9 | MeSH descriptor: [Hydrocortisone] explode all trees | 6812 |
| #10 | MeSH descriptor: [Cortisone] explode all trees | 178 |
| #11 | MeSH descriptor: [Betamethasone] explode all trees | 1655 |
| #12 | MeSH descriptor: [Triamcinolone] explode all trees | 1648 |
| #13 | (orthobiolog* or prolotherap* or ((proliferat* or regenerat* or biolog*) NEAR/5 (therap* or inject*)) or "MSC" or "MSCs" or allogen* or stem cell* or mesenchymal or stromal or progenitor or (marrow NEAR/3 (concentrat* or aspirat*)) or "BMAC" or (adipo* NEAR/3 (microfrag* or micro-frag*)) or lipoaspirat* or "SVF" or "ASA" or placent* or umbilic* or amnio*):ti,ab | 68,957 |
| #14 | MeSH descriptor: [Prolotherapy] explode all trees | 42 |
| #15 | MeSH descriptor: [Cell- and Tissue-Based Therapy] explode all trees | 8748 |
| #16 | MeSH descriptor: [Stem Cells] explode all trees | 1152 |
| #17 | MeSH descriptor: [Biological Products] explode all trees | 34,103 |
| #18 | (hyaluron* or hylan* or viscosup* or synvisc* or orthovisc* or monovisc* or durolane):ti,ab | 5451 |
| #19 | MeSH descriptor: [Hyaluronic Acid] explode all trees | 2139 |
| #20 | MeSH descriptor: [Viscosupplementation] explode all trees | 62 |
| #21 | #1 or #2 or #3 or #4 or #5 or #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20 | 238,487 |
| #22 | (intra-articular or inject* or intraarticular):ti,ab | 109,409 |
| #23 | MeSH descriptor: [Injections] explode all trees | 25,077 |
| #24 | #22 or #23 | 121,597 |
| #25 | (osteoarthr* or arthr* or degenerat*):ti,ab | 68,198 |
| #26 | hip:ti,ab,kw | 29,415 |
| #27 | #25 and #26 | 8684 |
| #28 | MeSH descriptor: [Osteoarthritis, Hip] explode all trees | 1235 |
| #29 | #27 or #28 | 8833 |
| #30 | #21 and #24 and #29 | 407 |
| #31 | #30 in Trials | 402 |
MeSH, Medical Subject Headings.
Final revision submitted November 17, 2025; accepted December 22, 2025.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
