Objectives:
Bone marrow aspirate concentrate (BMAC) is a commonly clinically used cell source for muscular skeletal injury repair. However, it is often required multiple injections with multiple harvests which caused pain for patient. Therefore, one harvest and multiple injections will be desired, but this process requires frozen (banking) stem cells. The objective of this study is to determine whether the articular cartilage regenerative potential of BMAC will be affected by the cryopreservation technique using destabilized medial meniscus (DMM) model.
Methods:
Preparation of fresh and frozen BMAC. Bone marrow was drawn from patients’ iliac crest. BMAC was prepared according to the standard clinical protocol. BMAC was then split into fresh and frozen portions. For the fresh-BMAC group, 100ul of BMAC was injected into the injured knee at 4 weeks after DMM. For frozen-BMAC group, BMAC was frozen by separating cells and plasma. Briefly, BMAC was spun down, plasma was aspirated in separate tubes and frozen directly. Cells were frozen in a freezing medium with 10% DMSO for 4 weeks. When it’s time for injection, we thawed the cells, removed DMSO, combined with frozen plasma, and injected same living cell number and volume as fresh BMAC based on the cell viability after cryopreservation. In vivo osteoarthritis (OA)induction using DMM model and BMAC treatment. 12 weeks female RNU nude rats were divided into 3 groups (N=10/group), group 1, PBS; group 2, Fresh-BMAC; group 3, Frozen-BMAC. This study was approved by IACUC. All rats were subjected to DMM surgery by following the previous published method. When OA was developed at 4 weeks after surgery, PBS, Fresh BMAC and frozen BMAC were injected into the knee joints. Rats were sacrificed at 8 weeks after injection and rat knee diameter were measured before dissection. 5 rats from each group were dissected to separate femur and tibia and observed grossly and imaged. After imaging, articular cartilage was dissected for RNA extraction from both tibia plateau and femur condyle, cDNA synthesis and Q-PCR were subsequently performed. Micro-CT scan and histology: Another 5 rats from each group were fixed in formalin for 5 days and subjected to micro-CT scan for proximal tibia. After Micro-CT scanning, rat knees were decalcified using formic acid and processed and sagittal section were cut for histology. Histology was conducted according to previously described protocol [1-3].Statistical analysis: All data were analyzed using GraphPad Prism 10 software. P<0.05 was considered statistically significant.
Results:
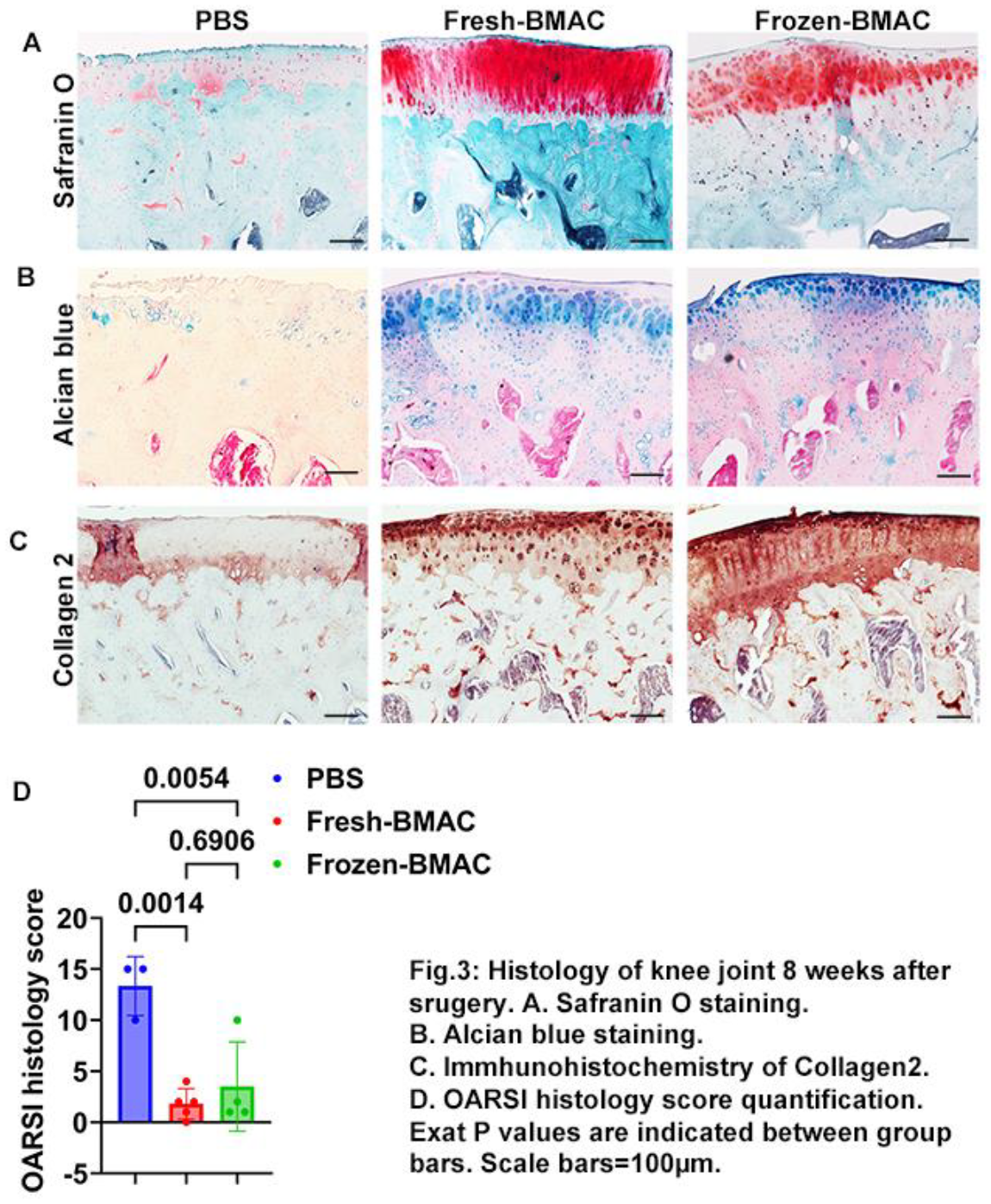
Both fresh and frozen BMAC improved macroscopic core of tibia plateau cartilage. 8 weeks after BMAC treatment, we measured knee diameter. We found both fresh-BMAC and frozen-BMAC treatment decreased knee diameter of nude rats compared to PBS treated group (Fig.1A). Fresh-BMAC and frozen BMAC treatment also improved femur condyle macroscopic score, but there is no significant difference compared to PBS group (Fig.1B, C). However, both fresh and frozen BMAC significantly improved tibia plateau cartilage macroscopic score compared to PBS group, but no significant was found between Fresh-BMAC and Frozen-BMAC (Fig.1D, E). Q-PCR results showed both fresh-BMAC and frozen-BMAC treatment groups significantly decreased MMP9 mRNA expression in the injured articular cartilage (Fig.1F). Fresh-BMAC and Frozen-BMAC group also showed a trend of decreasing MMP13 expression (Fig.1G). Fresh-BMAC and frozen-BMAC treatment did not significantly affect epiphysis and subchondral bone. We performed Micro-CT scanning and quantified the epiphysis and subchondral bone microarchitecture of both lateral and medial side of tibia plateau. We found no significant difference of all bone microarchitecture parameters of lateral epiphysis, subchondral bone and medial epiphysis trabecular bone. We only found PBS group showed trend of decreasing trabecular number in the medial subchondral bone (Fig.2A-F). BMAC treatment improved histology score of tibia plateau cartilage, no difference was found between fresh and frozen BMAC. We further performed Safranin O staining to detect cartilage matrix glycosaminoglycan (GAG). We found that fresh-BMAC and frozen-BMAC groups had majority of the cartilage repaired with GAG positive cartilage when compared to PBS group. OARSI histology score quantification indicated fresh-BMAC and frozen-BMAC groups had significantly lower OARSI score than PBS group, but no significant difference between fresh and frozen BMAC treatment (Fig.3A, D). Furthermore, we performed alcian blue staining to detect hyaluronic acid and acid mucin, the result was similar as we observed in Safranin O staining (Fig.3B). Immunohistochemistry staining of collagen 2 demonstrated significant loss of collagen 2 in PBS group, while strong collagen 2 staining in was found in both chondrocytes and extracellular matrix in BMAC treated groups (Fig.3C). Importantly, no significant difference was found between fresh and frozen BMAC treatment.
Conclusions:
In summary, we found frozen BMAC can maintain similar function as fresh BMAC for cartilage repair after being frozen for one month.