Abstract
Objectives:
Articular cartilage lesions are painful and debilitating conditions. A novel aragonite-based scaffold has been developed for the treatment of chondral and osteochondral lesions of the knee. A randomized controlled trial comparing the scaffold to surgical standard of care (SSOC: debridement or microfracture) showed superior clinical outcomes at two years post-operatively. The objective of this analysis is to compare the clinical and safety outcomes of the aragonite-based scaffold to SSOC at five years post-operatively. The hypothesis was that the aragonite-based scaffold would be superior to SSOC in subjective scores, treatment failure rates and conversion to arthroplasty or osteotomy.
Methods:
A total of 251 patients were included in 26 medical centres across the world and were randomly assigned to receive either the aragonite-based scaffold or SSOC in a 2:1 ratio. Patients aged 21 to 75 years with one to three cartilage defects ICRS Grade 3a or above located on the femoral condyles and/or trochlea with a symptomatic total treatable area 1-7cm2 were enrolled. Exclusion criteria included: KOOS Pain subscale score at baseline <20 or >65 (maximum pain=0, pain-free=100); bony defect depth >8mm, articular cartilage lesions in the tibia or the patella ICRS grade 4a or above, severe OA of the index knee (Kellgren-Lawrence Grade 4), BMI > 35kg/m2, and previous surgical cartilage treatment within six months of enrolment. Patients underwent arthroscopy to confirm study eligibility followed immediately by randomization and treatment according to group assignment. The SSOC (debridement or microfracture) for each patient was determined by a prespecified algorithm based on age, lesion size, and level of osteoarthritis. Patients randomized to the investigational arm received the cell-free, off-the-shelf, porous, biocompatible, and biodegradable aragonite-based implant via a mini-open or open arthrotomy procedure. All patients underwent the same rehabilitation program including progressive partial weightbearing to reach full weightbearing after six weeks. Most patients regained full active range of motion after approximately two months. Interim follow-up evaluations occurred at 6, 12, 18, 24, 36, and 48 months after treatment. The primary safety endpoint was the rate of adverse events, reoperations and revisions up to 60 months. Treatment failures were defined as any secondary intervention, including intra-articular injection or any surgery in the treated joint. Patients who were categorized as treatment failures due to intra-articular injection continued to be followed for safety outcomes. Data were analysed using Fisher’s exact test for categorical variables because of the expected low rate of treatment failure and serious adverse events.
Results:
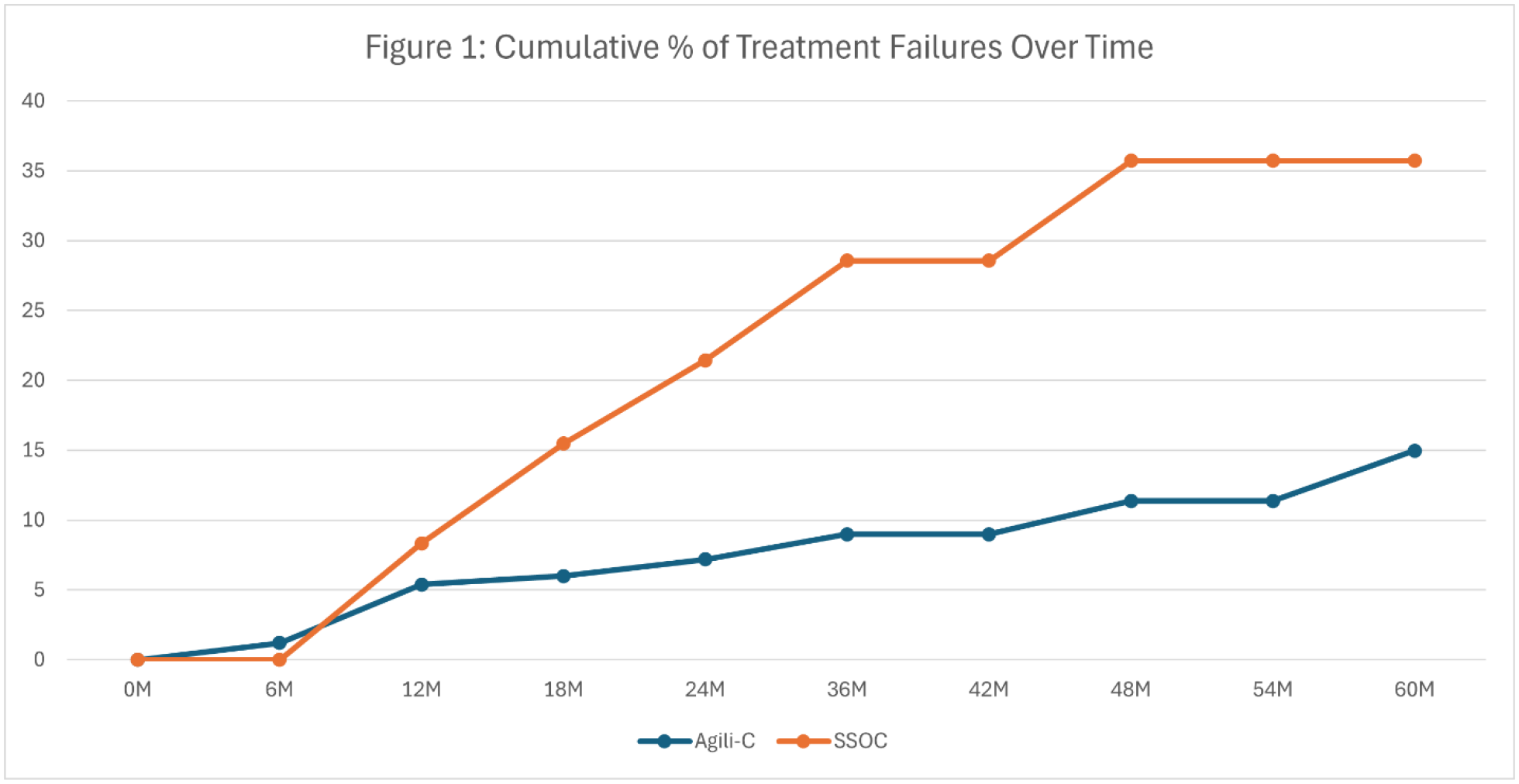
At five years’ follow-up, the implant group showed a significantly lower rate of treatment failure as compared to controls (15.0% vs 35.7%, p<0.001; Fig. 1). Twelve patients in the scaffold group (7.2%) underwent a secondary surgical intervention involving implant removal prior to the five-year follow-up. Four of these patients (2.4%) had their implants removed following a traumatic fall that damaged the implant. At five years’ follow-up, 1.8% (n=3) of patients in the scaffold group had received a knee replacement or osteotomy as compared to 9.5% (n=8, including n=2 osteotomies) in the SSOC group (p=0.008; Tables 1 & 2). For patients with mild to moderate OA, the scaffold significantly outperformed SSOC with respect to treatment failure rates (13.2% vs 40.7%, p<0.001). The scaffold group showed lower treatment failure rates than the SSOC group for patients with no to minimal OA (16.5% vs 26.7%, p=0.282) though the difference was not statistically significant. The scaffold also showed significant superiority over SSOC for lesions larger than 3cm2, showing a treatment failure rate of 14.3% in the scaffold group vs 39.0% in the control (p=0.003). The scaffold outperformed SSOC for patients with lesions smaller than 3cm2 (15.9% vs 32.6%, p=0.061), but the difference in treatment failure rates was not statistically significant (Table 3).
Conclusions:
The aragonite-based scaffold outperformed the control group at five years’ follow-up with respect to clinical scores and rates of treatment failure and conversion to arthroplasty or osteotomy. The analysis suggests that the novel device offers a safe and effective treatment for cartilage degeneration in patients with chondral and osteochondral lesions in the knee joint.