Objectives:
Fresh osteochondral allograft transplantation (OCA) is commonly used to treat large articular chondral or osteochondral defects, especially in younger patients, because it allows for delivery of mature, viable hyaline cartilage at the site of the defect. Incorporation into the adjacent native host tissue is pivotal for surgical success, and recent long-term outcome studies have demonstrated failure rates of 10-46% post transplantation, for poorly understood reasons.[1] The immunologic response may play a critical role in OCA transplantation outcomes; however, the mechanisms and biological consequences of the immune response in humans are poorly understood.[2] The purpose of this study was to evaluate the local and systemic host immune response and clinical outcomes following fresh OCA transplantation at up to one-year follow-up.
Methods:
After IRB approval and patient consent, a total of 10 patients (mean age 30.5 years; 60% male) with a focal chondral defect >2cm2 of total area in the femoral condyle underwent OCA transplantation surgery. All osteochondral allografts (mean donor age 20.5 ± 5.0 years; 70% male) were obtained from the same tissue bank to minimize variability. Fresh cylindrical plugs of the unused portion of each graft were immediately processed for evaluation of cell viability (live/dead cell staining and caspase 3 immunostaining) and proteoglycan content (Safranin-O/Fast Green staining)u. Peripheral blood, knee joint synovial fluid, MRI and patient-reported knee pain (Numeric Rating Scale [NRS]) and function scores (International Knee Documentation Committee [IKDC]), were obtained both pre-transplantation (baseline) and at 2 and 6 weeks, and 3, 6 and 12 months post-transplantation. Peripheral blood mononuclear cells (PBMCs) were analyzed by flow cytometry to characterize the immune cell types using the following surface markers: CD235a, CD45, CD19, CD3, CD4, CD8, CD14, CD16, CD161, CD1c, HLA-DR. Multiplex ELISA was utilized to analyze synovial fluid protein content. Spearman correlation analyses were performed to evaluate associations between the biological variables and clinical outcome measures. Statistical analyses were performed using GraphPad Prism 6. P<0.05 was considered significant.
Results:
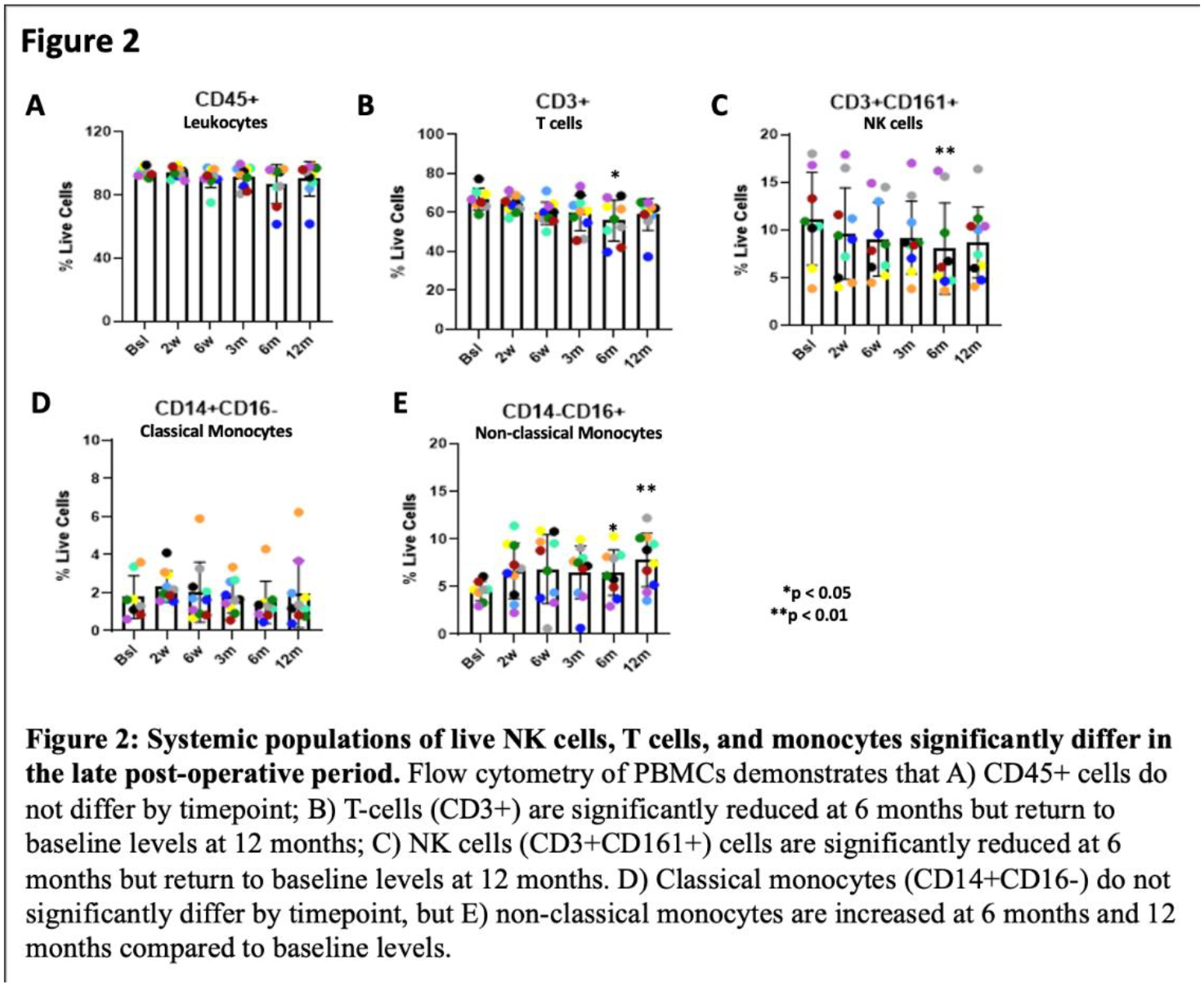
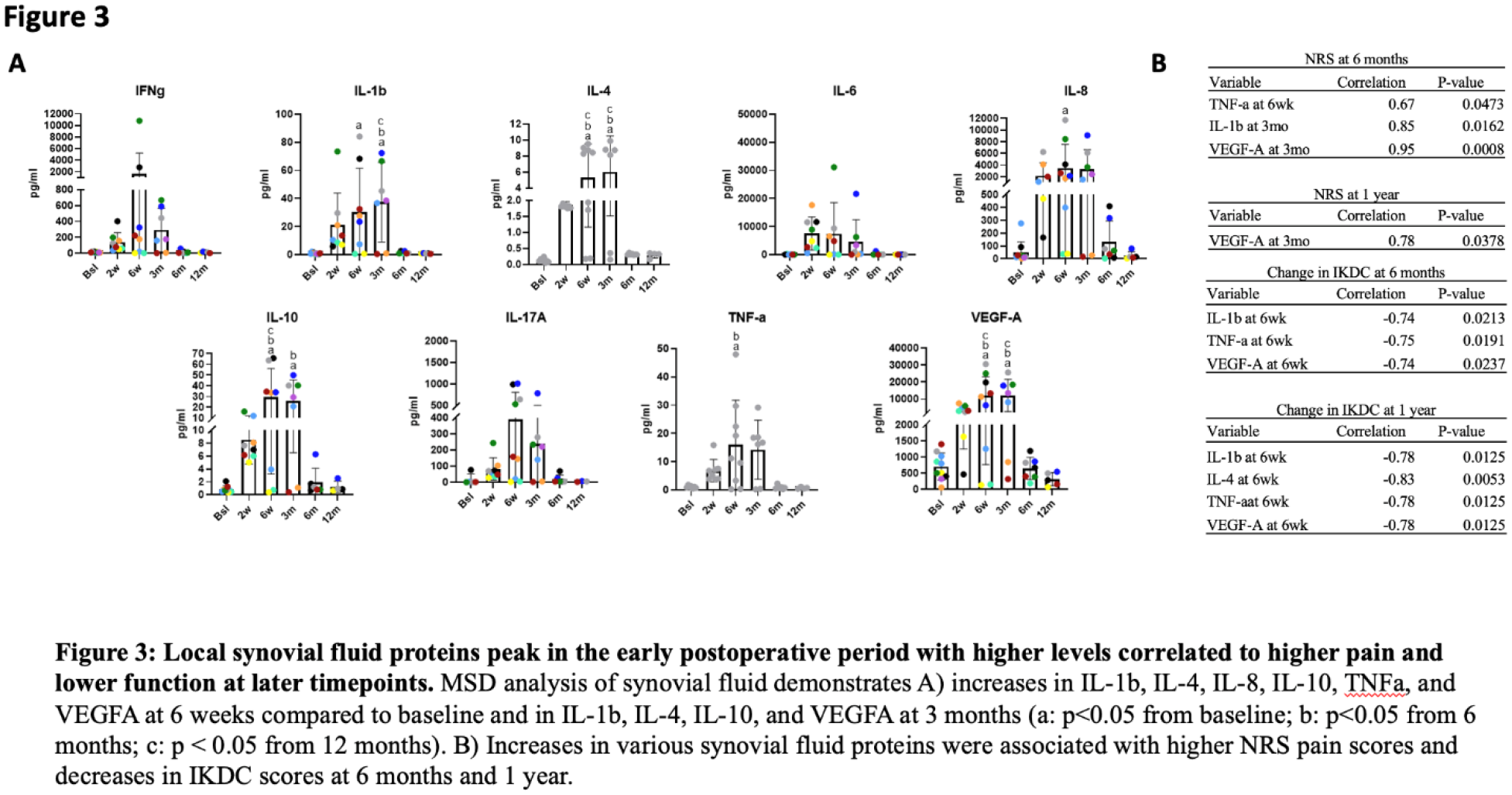
The mean time from graft procurement to implantation was 24.7 ± 3.3 days. The majority of the cells in the unused OCA plugs were live with a higher number of dead cells and decreased proteoglycan content concentrated in the superficial zone of the hyaline cartilage (Fig 1A). Mean proteoglycan loss was 10.0% (range 0.3% to 33.1%) (Fig 1B). Decreased proteoglycan content was significantly correlated with increased donor age (R=-0.70, p=0.023) (Fig 1C). Analyses of PBMCs demonstrated reduced NK and T cells at 6-month after OCA vs. baseline, but these levels returned back to baseline at 12 months (Fig 2A). Additionally, there was an increase in non-classical monocytes (CD14-CD16+) at 6 and 12 months post-operatively relative to baseline numbers (Fig 2B). However, we did not find statistically significant correlations between these flow cytometry findings and clinical outcomes scores, Similarly, we did not identify statistically significant correlations between MRI scores and IKDC scores (p=0.909) or NRS scores (p=0.904) using linear regression analysis. There were significantly increased levels of IFNg, IL-1β, IL-4, IL-8, IL-10, TNFα, and VEGFA in the post-operative synovial fluid analysis, particularly at the 6-week and 3-month timepoints (Fig 3A). Some of these proteins, particularly IL-1β, TNFα, VEGFA, and IL-4 were negatively correlated with NRS and IKDC scores at 6 and 12 months post-operatively (Fig 3B).
Conclusions:
We identified modest time-dependent changes in systemic populations of CD3+ T cells, NK cells, and non-classical monocytes after OCA, but these changes were not correlated with clinical outcomes. Using histopathologic analyses, we found that there is a wide variability in the cell viability and that proteoglycan content of allografts is likely age-dependent and higher in OCA from younger donors. We uncovered increased levels of selected synovial fluid proteins, and a negative correlation between IL-1β, TNFα, VEGFA, and IL4 levels and NRS and IKDC scores. Interestingly, the synovial fluid protein changes preceded changes in functional outcomes. Limitations of this study include the small number of patients and the lack of long-term clinical outcomes. While further investigation into the role of immunogenicity in OCA surgery is warranted, our findings suggest changes in PBMCs are modest and not significantly correlated with clinical outcome measures. However, changes in synovial fluid proteins could potentially be used to predict outcomes following OCA surgery, and may provide potential targets for early-stage intervention.