Abstract
Background:
Cartilage lesions are frequently encountered during anterior cruciate ligament reconstruction (ACLR) and may influence long-term outcomes. Surgical options include debridement, microfracture, osteochondral autograft transfer (OAT), osteochondral allograft transplantation (OCA), and autologous chondrocyte implantation (ACI), yet the optimal approach remains controversial.
Purpose:
To systematically review clinical outcomes of cartilage procedures performed during ACLR and compare them with cases where cartilage lesions were left untreated.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
Following PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, PubMed, Ovid MEDLINE, and Scopus were searched through July 15, 2025. Inclusion criteria comprised studies reporting outcomes of primary ACLR with concomitant cartilage lesions treated with debridement, microfracture, OAT, OCA, ACI, or no treatment. Outcomes assessed included patient-reported outcome measures, return to sport, osteoarthritis (OA) progression, and reoperation rates. Methodological quality was evaluated using the Modified Coleman Methodology Score, and the certainty of evidence for each outcome was further assessed using the Grading of Recommendations Assessment, Development and Evaluation framework.
Results:
A total of 14 studies (1003 patients; mean age range, 28-39 years; mean follow-up range, 2.1-95 months) met the inclusion criteria. Microfracture and debridement resulted in significant postoperative improvement but did not consistently outperform no treatment, with some studies reporting worse Knee injury and Osteoarthritis Outcome Score (KOOS) Sport and Recreation and Quality of Life subscale scores after microfracture. OAT, OCA, and ACI produced greater improvements in patient-reported outcomes, with OAT showing higher return-to-sport rates (63.6% vs 38.5% for microfracture) and ACI demonstrating substantial pain reduction and functional gains. OA progression was highest after microfracture (up to 48%) and lowest with OAT and ACI. OCA improved KOOS domains but had the highest reoperation rate (32%). The overall certainty of evidence was very low due to heterogeneity, small sample sizes, and methodological limitations.
Conclusion:
Microfracture and debridement performed during primary ACLR did not consistently improve outcomes compared with no treatment. In contrast, OAT, OCA, and ACI demonstrated greater functional improvements. Limited evidence from a single study suggested a higher return-to-sport rate with OAT than with microfracture, though further research is required to confirm this finding. OA progression appeared more frequent after microfracture, whereas ACI and OAT were associated with lower rates of progression and reoperation. Substantial limitations in available data and study design prevent definitive conclusions related to the effect of articular cartilage treatment at the time of primary ACLR.
Registration:
CRD420251137854 (PROSPERO identifier).
Keywords
Anterior cruciate ligament (ACL) injuries are among the most common knee pathologies in young, active individuals and athletes. Their incidence has increased in recent decades due to greater participation in sports and recreational activities. 16 Up to 46% of patients undergoing ACL reconstruction (ACLR) have associated chondral defects that may result from the initial trauma or develop progressively due to joint instability and altered load distribution.13,20,44 These cartilage lesions have limited intrinsic healing capacity due to the avascular nature of hyaline cartilage, predisposing the joint to progressive degeneration and early onset osteoarthritis (OA), and may compromise long-term outcomes after ACLR when not addressed surgically.21,32
A variety of surgical techniques are available to treat focal articular cartilage defects, aiming to restore the integrity of the joint surface and prevent further degeneration. Options include debridement and chondroplasty (indicated for small or unstable lesions to alleviate symptoms by smoothing lesion borders 3 ), marrow stimulating techniques such as microfracture and subchondral drilling (promote the formation of reparative fibrocartilaginous tissue by accessing the subchondral bone marrow8,37), and cartilage replacement techniques including osteochondral autograft transfer (OAT) and osteochondral allograft transplantation (OCA) (the former is suitable for small defects in young patients, and the latter is used for larger, more complex lesions enabling restoration of both cartilage and subchondral bone with viable donor tissue). 22 Other advanced options are autologous chondrocyte implantation (ACI) or second- or third-generation ACI (also known a matrix-assisted ACI [MACI]).15,22 These are 2-stage procedures involving harvesting and culturing of autologous chondrocytes followed by reimplantation, with the aim of regenerating hyaline-like cartilage.5,17,39 Clinical outcomes vary by technique: while microfracture demonstrates good short-term results, durability is often limited 19 ; in contrast, OAT and OCA have improved long-term outcomes in well-selected patients.9,25 ACI and its advanced forms have produced promising results demonstrating tissue regeneration and durable clinical improvement, particularly for young patients with isolated lesions and minimal joint degeneration. 27
In patients presenting with combined ACL and cartilage injuries, a comprehensive treatment approach addressing both ligamentous instability and the chondral defect is essential to optimize clinical outcomes. Several studies have demonstrated that by combining ACL and cartilage treatment, joint degeneration can be delayed and significant improvements can be seen in knee function and symptoms compared with isolated treatment of either condition. 36 On the contrary, some studies have reported favorable outcomes after ACLR when full-thickness chondral defects were left untreated, although cartilage lesions are often considered a negative prognostic factor. 31 This controversy suggests that a nuanced approach should be taken for each patient, as outcomes are influenced by multiple factors, including patient age, lesion size and location, degree of joint degeneration, timing of intervention, and the selected surgical technique.2,6,30
The primary objective of this systematic review was to evaluate the clinical outcomes of various cartilage treatment techniques performed in conjunction with primary ACLR. An additional aim was to compare these outcomes with cases where chondral lesions were left untreated. The authors hypothesized that appropriately addressing cartilage lesions at the time of primary ACLR would lead to improved patient-reported outcomes (PROs) compared with leaving such lesions untreated.
Methods
Search Process
This systematic review adhered to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 guidelines 24 and was prospectively registered with PROSPERO. Two independent reviewers (N.T. and L.D.M.) systematically searched PubMed, Ovid MEDLINE, and Scopus to identify studies evaluating postoperative outcomes of cartilage repair techniques performed in conjunction with ACLR. The literature search encompassed all relevant publications available in these databases through July 15, 2025. In all 3 databases, search terms included combinations of keywords such as ("ACL reconstruction" OR "anterior cruciate ligament surgery") AND ("cartilage lesion" OR "chondral defect" OR "osteochondral") AND ("treatment" OR "repair" OR "microfracture" OR "debridement" OR "no treatment" OR "ignored") AND ("outcome" OR "return to sport" OR "functional score" OR "progression of osteoarthritis").
Eligibility Criteria
The inclusion criteria for this review were (1) studies involving patients who underwent primary ACLR with intraoperatively visualized cartilage lesions; (2) studies reporting on cartilage lesions identified during ACLR in patients undergoing treatment including no intervention, debridement, microfracture, OATS, OCA, ACI, or other cartilage stabilization technique; (3) studies that reported postoperative outcomes such as PRO measures, return to sport, reoperation rate, or progression of OA; (4) studies classified as level 1 to 4 evidence; and (5) articles published in English. For studies that included both ACL-intact and ACLR cohorts, only the ACLR cohort arms were included in our analysis to ensure that results reflected outcomes of true concomitant ACLR with cartilage lesions. Exclusion criteria included (1) studies involving multiligamentous knee injuries or revision ACLR; (2) cadaveric, animal, or biomechanical studies; (3) studies lacking relevant clinical outcome data, such as those focused solely on surgical technique or intraoperative findings; and (4) study designs including editorials, letters to the editor, systematic reviews, and non–peer reviewed sources such as conference abstracts without full-text availability.
Data Extraction
Two authors (N.T. and L.D.M.) independently assessed the eligibility of the identified studies by screening titles, abstracts, and full texts. Any discrepancies were resolved through discussion or, when necessary, with input from a third author (J.T.M.). Data were systematically extracted from the included studies, including study design, patient demographics, characteristics of the cartilage lesions, details of cartilage treatment techniques, associated injuries, and outcomes such as patient-reported outcome measures, OA progression, and reoperation rates.
Methodology Quality Assessment
Study quality was appraised using the Modified Coleman Methodology Score (MCMS), 4 a widely recognized tool developed to evaluate the methodological quality of clinical studies. Two reviewers (J.T.M. and F.C.) independently conducted the evaluations, and any disagreements were settled through consensus or, when needed, by involving a third reviewer (T.F.V.). The MCMS assesses 10 specific aspects of study design, including sample size, follow-up length, prospective versus retrospective design, clarity in the surgical technique description, rehabilitation protocols, outcome definitions, assessment methodology, and selection criteria. Each component receives a score ranging from 0 to 10, culminating in a total score out of 100. A higher total score reflects stronger methodological integrity, emphasizing elements such as validated outcomes, appropriate blinding procedures, and comprehensive follow-up. Based on the total score, studies were rated as fair (50-69), good (70-84), or excellent (≥85), providing a structured and consistent approach for evaluating methodological quality.
Additionally, risk of bias for randomized controlled trials was assessed using the Cochrane risk-of-bias 2 (RoB 2) tool, which evaluates potential bias across categories including randomization, deviations from intended interventions, missing outcome data, outcome measurement, and selective reporting. 35 For nonrandomized comparative studies, we applied the Risk of Bias in Non-Randomized Studies–of Interventions (ROBINS-I) tool, which is specifically designed to account for the unique challenges of observational study designs, with structured evaluation of bias due to confounding, selection of participants, classification of interventions, deviations from intended interventions, missing data, outcome measurement, and selection of reported results. 34
The certainty of evidence for each outcome was evaluated using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach, considering factors for downgrading (risk of bias, inconsistency, indirectness, imprecision, publication bias) and upgrading (large effect, dose-response, confounding). Certainty was rated as high, moderate, low, or very low. 12
Statistical Analysis
Given the substantial methodological and clinical differences among the included studies such as variability in outcome measures, follow-up periods, and patient demographics, quantitative meta-analysis was not appropriate. Therefore, a descriptive qualitative synthesis was undertaken. The extracted information encompassed mean values, standard deviations, and reported changes in both objective physical examination results and PROs from baseline to final follow-up, along with the incidence of OA progression and reoperation.
Results
Study Selection
A total of 219 records were identified through database searches, including Scopus (n = 102), PubMed (n = 64), and Ovid MEDLINE (n = 53). After removing 100 duplicates—4 manually and 96 via Covidence—119 studies remained for title and abstract screening. Of these, 80 were excluded based on irrelevance to the review question. The remaining 39 full-text articles were assessed for eligibility, and no studies were excluded because of retrieval issues. A total of 25 studies were excluded at the full-text stage for various reasons: not in English (n = 1), wrong setting (n = 2), wrong outcomes (n = 6), wrong comparator (n = 3), wrong indication (n = 1), wrong study design (n = 3), lack of full-text availability (n = 4), or lack of PROs (n = 5). Ultimately, 14 studies ¶ met the inclusion criteria and were included in the final review (Figure 1).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram for study selection.
Study Characteristics
A total of 14 studies ¶ published between 2003 and 2025 were included in this review, comprising 1003 patients who underwent ACLR with or without concomitant cartilage procedures. The level of evidence ranged from 2 to 4. Sample sizes varied from 10 to 269 patients. The mean age of participants across studies ranged from 28 to 39 years. The follow-up duration varied widely, ranging from 2.1 ± 0.2 months to 95 ± 66 months. Various cartilage treatment strategies were reported, including microfracture,23,40 OAT,10,28 debridement, 38 or no cartilage treatment.31,42 Several studies also included comparator groups with intact cartilage.33,38,42
Most ACLRs were performed arthroscopically using either hamstring tendon autograft or bone–patellar tendon–bone autograft. Some studies used a mix of graft types,29,31,42 while others did not report graft details 41 (Table 1).
Characteristics of Included Studies a

Data are presented as mean, mean ± SD, or median (range). ACI, autologous chondrocyte implantation; ACLR, anterior cruciate ligament reconstruction; BPTB, bone–patellar tendon–bone; LOE, level of evidence; MCMS, Modified Coleman Methodology Score; OAT, osteochondral autograft transfer; OCA, osteochondral allograft transplantation.
Methodology Quality and Risk of Bias
Methodological quality, as assessed by the MCMS, was excellent in 4 studies (scores ≥85), good in 7 studies (scores 70-84), and fair in 3 studies (scores 60-69). No studies were rated as poor (Table 1). For the single randomized controlled trial, overall risk of bias was judged as having some concerns with the Cochrane RoB 2 tool, primarily because of issues with randomization and outcome measurement (Table 2). For nonrandomized comparative studies, the overall risk of bias was consistently rated moderate with the ROBINS-I tool, with confounding variables and participant selection contributing most to the downgrading (Table 3).
Cochrane RoB 2 for Randomized Controlled Trial a

RoB 2, risk-of-bias 2.
ROBINS-I Tool a

ROBINS-I, Risk of Bias in Non-Randomized Studies–of Interventions.
Application of the GRADE framework demonstrated that the certainty of evidence across all outcomes and interventions was predominantly rated as very low, with only select PRO comparisons reaching low certainty. Downgrades were consistent across domains because of the observational design of included studies, small and heterogeneous cohorts, risk of bias, inconsistency between results, and imprecision in estimates (Table 4).
Grading of Recommendations Assessment, Development and Evaluation of Findings (GRADE) a

Certainty of evidence is presented as ●●●● high, ●●●○ moderate, ●●○○ low, ●○○○ very low. ACI, autologous chondrocyte implantation; ACLR, anterior cruciate ligament reconstruction; ADL, Activities of Daily Living; IKDC, International Knee Documentation Committee; KL, Kellgren-Lawrence; KOOS, Knee injury and Osteoarthritis Outcome Score; NR, not reported; OA, osteoarthritis; OAT, osteochondral autograft transfer; OCA, osteochondral allograft transplantation; PRO, patient-reported outcome; QOL, Quality of Life; RTS, return to sport; Sport, Sport and Recreation; TKA, total knee arthroplasty; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index; VAS, visual analog scale.
Cartilage Lesion Characteristics and Treatments
All 14 studies reported detailed characteristics and management of cartilage lesions. Cartilage lesion size varied widely among studies. When reported, lesion size ranged from as small as 1 cm2 to as large as 6.6 cm2. The most commonly affected location was the medial femoral condyle, followed by the lateral femoral condyle, trochlea, and patella. Less frequently involved sites included the tibial plateau, medial tibial plateau, and lateral tibial plateau.
Various cartilage treatment modalities were employed. Microfracture was the most frequently utilized technique, either alone or in combination with other procedures, and was reported in studies such as Osti et al, 23 Gudas et al, 10 Imade et al, 14 and Røtterud et al. 31 Debridement was another common approach, used either as a standalone treatment or alongside microfracture.10,23,31,38,42 ACI was employed in several studies,1,11,29 with some incorporating periosteal membrane or collagen membranes. OAT was reported in 4 studies.10,14,18,28 One study 41 utilized OCA. Associated injuries were frequently reported. Meniscal tears were the most common concomitant pathology (Table 5).
Cartilage Lesion Characteristics, Treatments, and Concomitant Procedures a
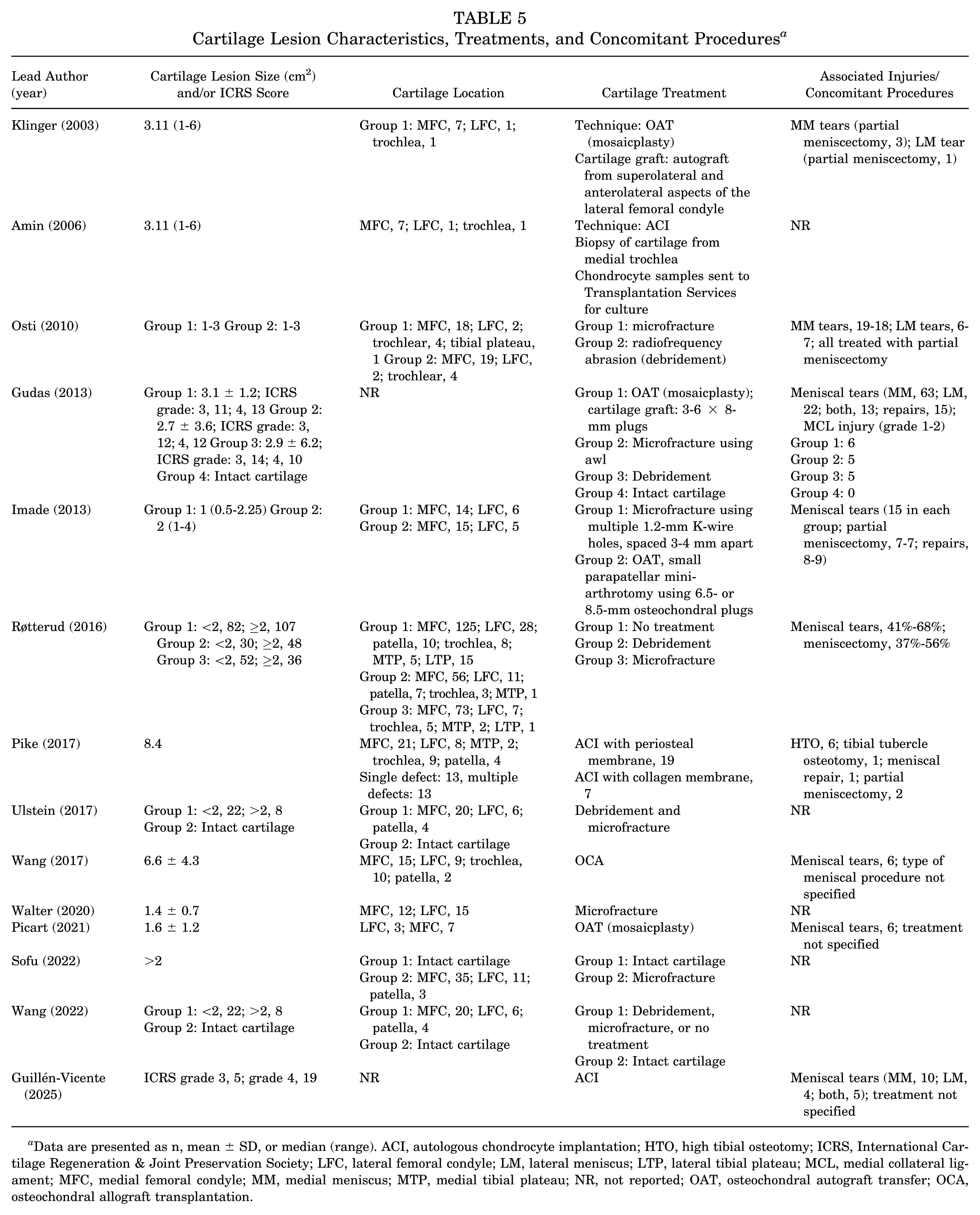
Data are presented as n, mean ± SD, or median (range). ACI, autologous chondrocyte implantation; HTO, high tibial osteotomy; ICRS, International Cartilage Regeneration & Joint Preservation Society; LFC, lateral femoral condyle; LM, lateral meniscus; LTP, lateral tibial plateau; MCL, medial collateral ligament; MFC, medial femoral condyle; MM, medial meniscus; MTP, medial tibial plateau; NR, not reported; OAT, osteochondral autograft transfer; OCA, osteochondral allograft transplantation.
Outcomes After No Treatment, Debridement, or Microfracture of Cartilage Lesions During ACLR Compared With Other Treatment Strategies
Four studies10,23,31,42 reported PROs comparing no treatment, debridement, and other cartilage treatment strategies during ACLR (Table 6).
PROs After No Treatment, Debridement, or Microfracture of Cartilage Lesions During ACLR Compared With Other Treatment Strategies a
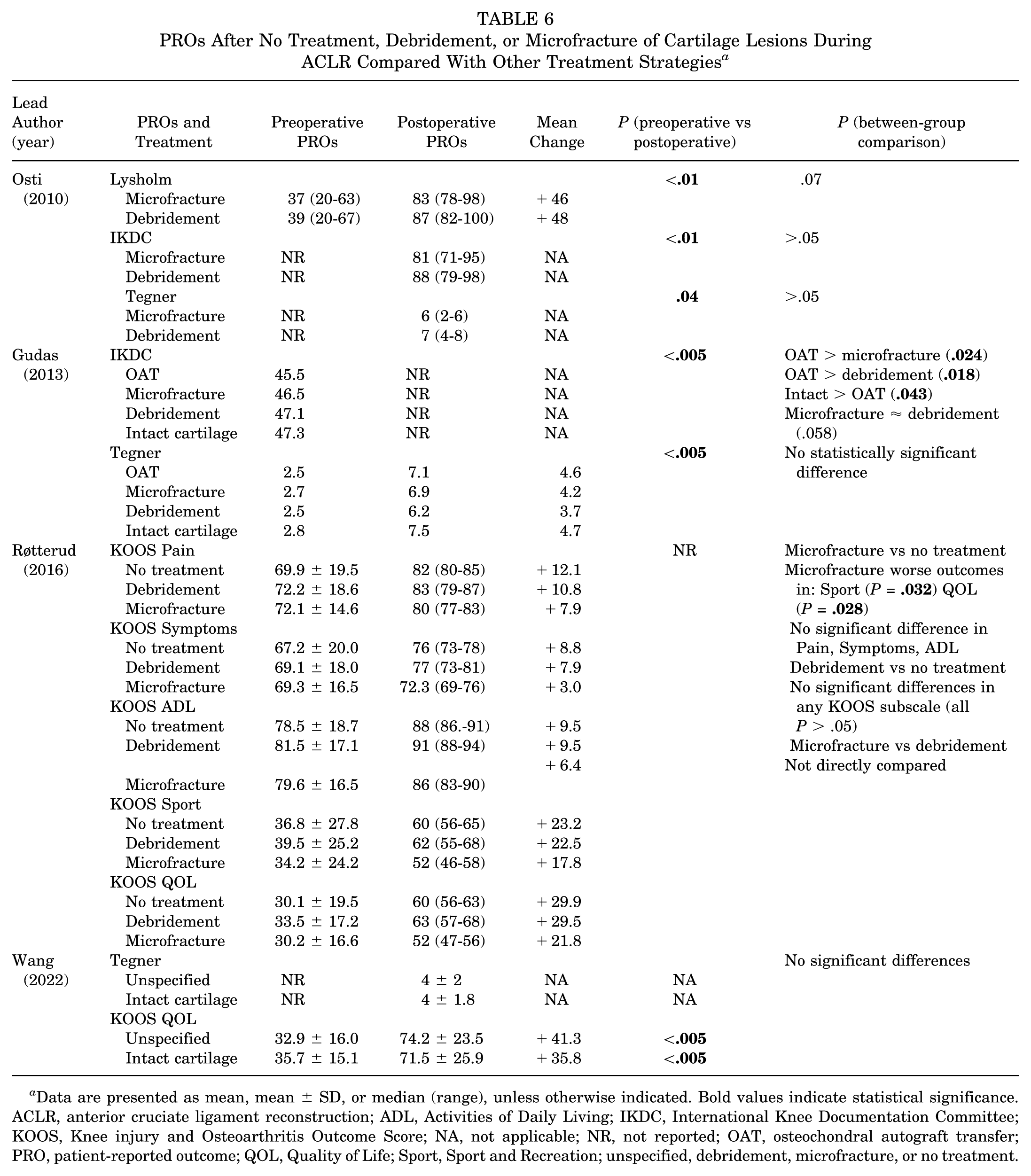
Data are presented as mean, mean ± SD, or median (range), unless otherwise indicated. Bold values indicate statistical significance. ACLR, anterior cruciate ligament reconstruction; ADL, Activities of Daily Living; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; NA, not applicable; NR, not reported; OAT, osteochondral autograft transfer; PRO, patient-reported outcome; QOL, Quality of Life; Sport, Sport and Recreation; unspecified, debridement, microfracture, or no treatment.
Osti et al 23 compared microfracture and debridement. Both groups showed significant improvement in Lysholm scores postoperatively (microfracture: from 37 to 83; debridement: from 39 to 87; both P < .01). Improvements in International Knee Documentation Committee (IKDC) and Tegner scores were also statistically significant (P < .01 and P = .04, respectively), but no significant differences were found between treatment groups (P > .05) (Table 6).
Gudas et al 10 compared 4 groups: OAT, microfracture, debridement, and intact cartilage. Significant pre- to postoperative improvements were observed in Tegner scores across all groups (P < .005), though the between-group difference was not statistically significant. Based on IKDC scores, OAT had significantly better outcomes than microfracture (P = .024) and debridement (P = .018), while intact cartilage had significantly better outcomes than OAT (P = .043). Microfracture and debridement outcomes were comparable (P = .058) (Table 6).
Røtterud et al 31 evaluated Knee injury and Osteoarthritis Outcome Score (KOOS) subscores across 3 groups: no treatment, debridement, and microfracture. All groups showed postoperative improvements across KOOS Pain, Symptoms, Activities of Daily Living (ADL), Sport and Recreation (Sport), and Quality of Life (QOL). However, microfracture was associated with significantly worse outcomes than no treatment in the KOOS Sport (P = .032) and KOOS QOL (P = .028) domains. No significant differences were found between no treatment and debridement in any KOOS subscale (P > .05). Microfracture and debridement were not directly compared in this study (Table 6).
Wang et al 42 assessed KOOS QOL and Tegner scores. Both the debridement/microfracture/no treatment group and the intact cartilage group showed statistically significant improvement in KOOS QOL from preoperative to postoperative assessment (P < .005), but no statistically significant differences were found between the groups (Table 6).
Outcomes After ACI, OAT, and OCA of Cartilage Lesions During ACLR
PROs improved after cartilage restoration procedures performed during ACLR, including ACI, OAT, and OCA. Specifically, 3 studies1,11,29 reported outcomes after ACI, 4 studies10,14,18,28 evaluated OAT, and 1 study 41 reported outcomes after the OCA procedure. Pike et al 29 reported significant improvement in the Modified Cincinnati score (mean change, +1.92; P < .0001) and a significant reduction in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores (mean change, –18.77; P < .0001), indicating improved function. Guillén-Vicente et al 11 demonstrated a significant increase in IKDC scores (mean change, +39.0; P < .05) and reduction in pain VAS (mean change, –5.3; P < .05). Similarly, Amin et al 1 reported numerical improvements in PROs, although no statistical values were provided (Table 7).
PROs After ACI, OAT, or OCA of Cartilage Lesions During ACLR a

Data are presented as mean, mean ± SD, or median (range) unless otherwise indicated. Bold values indicate statistical significance. Lower WOMAC scores indicate improved function. ACI, autologous chondrocyte implantation; ACLR, anterior cruciate ligament reconstruction; ADL, Activities of Daily Living; IKDC, International Knee Documentation Committee; KOOS, Knee injury and Osteoarthritis Outcome Score; NA, not applicable; NR, not reported; OAT, osteochondral autograft transfer; OCA, osteochondral allograft transplantation; PRO, patient-reported outcome; QOL, Quality of Life; Sport, Sport and Recreation; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
For OAT, multiple studies showed significant benefits. Klinger et al 18 observed improvements in Tegner (mean change, +2.2; P < .05) and Lysholm scores (mean change, +28; P < .05). Gudas et al 10 and Imade et al 14 also reported significant improvement in Tegner and IKDC scores, respectively. Picart et al 28 demonstrated significant improvements across all KOOS domains and Lysholm scores, indicating comprehensive patient-perceived benefit postoperatively (Table 7).
OCA yielded improvements across all KOOS domains in the study by Wang et al, 41 with significant gains observed in KOOS Pain (mean change, +11.9; P < .001), KOOS Symptoms (mean change, +10.4; P = .01), KOOS ADL (mean change, +10.7; P = .001), KOOS Sport (mean change, +25.6; P < .001), and KOOS QOL (mean change, +40.0; P < .001) (Table 7).
Return to Sports
Return-to-sport outcomes were reported in 1 study, by Imade et al, 14 comparing microfracture and OAT procedures. Among patients who participated in sports before injury, 38.5% (5 of 13) in the microfracture group returned to their preinjury level of sports, compared with 63.6% (7 of 11) in the OAT group. These findings suggest a higher rate of return to the same sports level after OAT compared with microfracture (Table 8).
Return to Sports Details a

OAT, osteochondral autograft transfer.
Osteoarthritis Progression and Reoperation
Among studies evaluating OA progression and reoperation after cartilage restoration during ACLR, OA progression was variably reported. Klinger et al 18 observed progression of 1 KL grade in 4 cases without any reoperation or conversions to TKA at a mean follow-up of 38 months. Osti et al 23 reported significantly greater radiographic OA progression in the microfracture group (48%) compared with the debridement group (20%) based on Fairbank grading (P < .05). Pike et al 29 found OA progression in 2 of 26 patients, with 1 case occurring alongside graft failure. Ulstein et al 38 noted KL grade ≥2 in 12 of 29 patients at 6.8 years (Table 9).
Osteoarthritis Progression and Reoperation After Cartilage Restoration Procedures During Anterior Cruciate Ligament Reconstruction a

ACI, autologous chondrocyte implantation; ACL, anterior cruciate ligament; ACLR, ACL reconstruction; KL, Kellgren-Lawrence; NR, not reported; OAT, osteochondral autograft transfer; OCA, osteochondral allograft transplantation; TKA, total knee arthroplasty.
Reoperation rates were highest in the Wang cohort, 41 with 8 of 25 patients (32%) undergoing reoperation after OCA procedures, although the specific types of reoperation were not reported. In contrast, no reoperations were reported by Klinger et al, 18 Picart et al, 28 or Guillén-Vicente et al 11 at their respective follow-up periods. Amin et al 1 reported that 2 of 9 patients (22.2%) required manipulation under anesthesia due to postoperative stiffness after ACI (Table 9).
Discussion
The most important finding of this study was that cartilage repair procedures—such as microfracture or debridement—did not consistently result in superior clinical outcomes compared with leaving cartilage lesions untreated during ACLR. Notably, some large cohort studies demonstrated that untreated full-thickness cartilage lesions produced comparable, or in some cases even better, PROs than those treated with microfracture. For example, Røtterud et al 31 reported that patients who underwent microfracture had significantly worse KOOS Sport and KOOS QOL subscores compared with those who received no treatment, while debridement and no treatment yielded similar results across all KOOS subscales. Similarly, studies by Osti et al 23 and Gudas et al 10 found no statistically significant differences in PROs between microfracture and debridement. However, Gudas et al 10 also reported that OAT resulted in significantly better outcomes than both microfracture and debridement. Notably, debridement of articular cartilage defects may provide no clinical benefit as compared with neglecting incidental lesions identified at the time of ACLR and reported outcomes likely lack sensitivity to determine any significant effect on patient outcomes.
These findings challenge the assumption that all cartilage defects encountered during ACLR require surgical treatment. Wang et al 42 also reported no statistically significant differences in KOOS QOL and Tegner scores between patients who underwent debridement, microfracture, or no treatment for cartilage lesions and those with intact cartilage. This suggests that the mere presence of a cartilage lesion does not necessarily predict worse clinical outcomes. While techniques such as microfracture and debridement are commonly used because of their technical simplicity and low cost, our findings raise concerns about their long-term efficacy. Microfracture may stimulate fibrocartilaginous repair tissue, which lacks the biomechanical properties of native hyaline cartilage and may deteriorate over time, particularly in active individuals or those with larger defects. 7 Furthermore, outcomes after marrow stimulation techniques are known to decline with increasing age, as subchondral marrow response and fibrocartilage formation are less robust in older individuals. The inclusion of patients aged >35 years in several studies may therefore have contributed to the inconsistent results observed. Lesion size also appears to be an important prognostic factor. Previous studies have shown that defects >2 cm2 tend to respond less favorably to microfracture or other marrow stimulation procedures, which may partially explain the variability in outcomes among the included studies. Similarly, although debridement can alleviate mechanical symptoms by smoothing unstable cartilage margins, available evidence indicates that its long-term outcomes are comparable with no treatment in many cases. Therefore, the choice between microfracture, debridement, or no treatment should be individualized—particularly for small, contained defects—where the expected benefits of intervention may not outweigh the risks, costs, or potential for later deterioration. Many cartilage defects identified during ACLR are discovered incidentally and are often small or asymptomatic. Because advanced techniques such as ACI or OCA require staged preparation or graft matching, they are not feasible in the same operation. As a result, available options are typically limited to debridement, microfracture, or osteochondral autograft, which are generally appropriate for small, contained lesions.
In contrast, more advanced cartilage restoration techniques, such as OAT, OCA, and ACI, have been reported to yield favorable outcomes in smaller, well-selected cohorts. Notably, previous literature5,25 indicates that cartilage restoration techniques such as OCA and ACI tend to provide superior outcomes for larger (>2 cm2) or full-thickness lesions compared with microfracture or debridement, regardless of concomitant ACLR status. For example, studies using ACI reported significant improvements in PROs, and a low rate of OA progression at midterm follow-up. 5 Similarly, OAT yielded superior outcomes in several comparative studies and a higher return to preinjury sports levels compared with microfracture.25,26,45 Multiple studies showed that OAT resulted in significant improvements in both function and activity scores. For instance, Gudas et al 10 and Klinger et al 18 reported significantly higher Tegner and Lysholm scores at 3-year follow-up in patients undergoing OAT. Picart et al 28 also found meaningful improvements across all KOOS subscales at >6 years of follow-up, further supporting the durability of OAT in this setting. These findings suggest that OAT may offer more robust restoration of knee function, especially in younger patients with small to medium-sized lesions.
Similarly, ACI yielded statistically significant improvements in IKDC, VAS, WOMAC, and Modified Cincinnati scores across several studies.1,11,29 Pike et al 29 reported a reduction of −18.77 points in WOMAC and an increase of +1.92 in the Modified Cincinnati score (both P < .0001), indicating improvements in both pain and function. Guillén-Vicente et al 11 observed a 39-point increase in IKDC and a 5.3-point reduction in VAS pain, further emphasizing ACI's potential in symptom relief and functional restoration. Amin et al 1 also reported improvements in pain and function scores, although specific P values were not provided. OCA, though only reported in a single study by Wang et al, 41 demonstrated the largest gains in KOOS domains, with improvements up to +40 points in KOOS QOL and +25.6 in KOOS Sport. These improvements suggest a meaningful enhancement in quality of life and athletic performance, even in patients with larger or more complex lesions not amenable to autografts or ACI. However, the use of OCA is constrained by graft availability, cost, and a higher observed reoperation rate.9,39 Advanced procedures such as ACI/MACI and OCA often require a delay for cell culture or graft matching, during which cartilage damage may progress and instability can increase the risk of meniscal injury. These factors should be considered when planning cartilage treatment with ACLR.
When evaluating return to sports, evidence remains very limited. Only a single study 14 directly compared OAT and microfracture, reporting higher return-to-sport rates with OAT. Given this narrow evidence base, conclusions should be considered preliminary. In their study, 63.6% of patients in the OAT group returned to the same level of sport, compared with only 38.5% in the microfracture group. While the sample size was small, this difference is clinically meaningful and aligns with the understanding that OAT restores more biomechanically competent tissue than fibrocartilage produced by microfracture.7,25,26
With respect to OA progression and reoperation, the data were heterogeneous but revealed important trends. Reported reoperation rates varied widely across studies, ranging from 0% to 32%. However, these values were observed at markedly different follow-up durations, making direct comparisons between procedures inappropriate. Therefore, findings should be interpreted descriptively rather than as a ranked comparison. Osti et al 23 reported significantly higher rates of OA progression in patients treated with microfracture (48%) compared with debridement (20%), raising concerns about the long-term durability of fibrocartilaginous tissue. Conversely, studies evaluating OAT and ACI generally reported low rates of OA progression and minimal need for reoperation. Notably, no OA progression or reoperations were observed in patients treated with OAT by Picart et al 28 and in ACI-treated patients by Guillén-Vicente et al, 11 reinforcing their long-term benefits. In contrast, OCA was associated with a reoperation rate of 32% in the study by Wang et al, 41 the highest among the evaluated procedures. While this may reflect the more challenging lesion profiles in patients receiving OCA, it also highlights the importance of careful patient selection and long-term monitoring.
Collectively, these findings suggest that while OAT, ACI, and OCA are technically demanding and resource-intensive, they may yield better functional outcomes, higher return to sport, and less OA progression in the right clinical context. Such contexts may include younger, high-demand patients with large (>2-4 cm2), symptomatic full-thickness lesions; lesions located on weightbearing zones of the femoral condyles; or cases where previous marrow stimulation techniques have failed. In contrast, patients with smaller (<2 cm2), asymptomatic, or incidentally detected lesions—particularly in nonweightbearing areas—may be better served with a conservative or minimal intervention approach. However, the variability in follow-up length, outcome reporting, and patient selection across studies limits the ability to draw definitive conclusions. Importantly, some untreated lesions showed no significant long-term detriment, supporting a more selective approach to cartilage intervention.42,43
This systematic review highlights that not all cartilage lesions encountered during ACLR require surgical intervention. While simple techniques such as microfracture and debridement are commonly employed, they do not consistently yield superior outcomes compared with leaving lesions untreated. In contrast, advanced cartilage restoration procedures like OAT, OCA, and ACI demonstrated favorable PROs, higher return-to-sport rates, and lower risk of osteoarthritis progression in appropriately selected patients. These findings support a more individualized approach to cartilage treatment during ACLR, reserving complex procedures for symptomatic, high-demand patients with larger or more advanced lesions, while avoiding unnecessary intervention in small or asymptomatic defects.
Limitations
This study has several limitations inherent to the available literature. Most importantly, the decision of when and how to treat a cartilage defect by surgery is highly variable and may or may not be due to a clinical contribution of the co-existing defect at the time of ACL reconstruction. Not uncommonly, a decision to treat an identified defect at the time of ACLR may not necessarily occur because of symptoms associated with that defect, but rather because of concerns that the defect might or could become symptomatic in the future. Separating the disability of an acute ACL injury from any clinically relevant symptoms contributed to by a cartilage defect is difficult and imprecise at best. There are several other important limitations that temper the ability to make conclusions about the effect of cartilage treatment performed at the time of ACLR. First, there was substantial heterogeneity among the included studies in terms of study design, patient demographics, cartilage lesion characteristics (size, location, and grade), surgical techniques, and rehabilitation protocols, which limited our ability to perform a quantitative meta-analysis with patient-level data. It should be noted that 1 included study (Klinger et al 18 ) utilized an open ACLR technique, which differs from the arthroscopic approach in other studies and may introduce methodological and clinical bias when comparing results across cohorts. Second, most included studies were level 3 or 4 evidence, with only a few prospective comparative studies, thereby introducing potential selection and reporting biases including the inability to capture and report upon all cartilage defects with specificity at the time of ACLR. Third, follow-up durations varied widely, ranging from <2 years to over a decade, which may affect the comparability of outcomes, particularly regarding OA progression and long-term function. Fourth, the lack of standardized outcome measures across studies hindered direct comparisons with several PROs lacking sensitivity for cartilage-specific impairment. While some studies used KOOS, others used Lysholm, IKDC, Tegner, or WOMAC, complicating interpretation of the overall clinical effect. Fifth, many studies included major concomitant procedures such as meniscal repair or partial meniscectomy, high tibial osteotomy, and tibial tubercle osteotomy. These procedures are potential confounders, as they independently influence pain, function, and progression of osteoarthritis, minimizing the ability to quantify the clinical effect of an articular cartilage lesion or its response to treatment. Sixth, return-to-sport outcomes were reported in only 1 study, and reoperation rates were inconsistently documented, limiting the strength of conclusions in these domains. Future high-quality, prospective studies with standardized reporting are needed to better define optimal treatment strategies. Last, from a methodological standpoint, the application of the GRADE framework highlights the limitations of the current evidence base for concomitant cartilage procedures at the time of ACLR. Across all outcomes, the certainty of evidence was judged to be predominantly very low, with only select PROs reaching low certainty. The primary drivers of downgrading were the observational design of nearly all included studies, small sample sizes, heterogeneity in interventions and outcome measures, and imprecision in reporting. Inconsistencies between studies further limited confidence in pooled interpretation. These limitations underscore that existing literature cannot support strong conclusions or definitive recommendations regarding the optimal cartilage treatment strategy in this setting. Instead, the findings should be interpreted cautiously, and future high-quality, prospective comparative studies with standardized outcome reporting are needed to clarify the relative benefits and risks of microfracture, debridement, OAT, ACI, and OCA when performed alongside ACLR. Overall, this review underscores that the management of cartilage lesions during ACLR remains insufficiently studied. The current evidence is largely limited to small, heterogeneous, nonrandomized studies, preventing clear treatment recommendations. Future well-designed, prospective comparative studies are needed to better define the role of various cartilage procedures in this setting.
Conclusion
Microfracture and debridement performed during primary ACLR did not consistently improve outcomes compared with no treatment. In contrast, OAT, OCA, and ACI demonstrated greater functional improvements. Limited evidence from a single study suggested a higher return-to-sport rate with OAT than with microfracture, though further research is required to confirm this finding. OA progression appeared more frequent after microfracture, whereas ACI and OAT were associated with lower rates of progression and reoperation. Substantial limitations in available data and study design prevent definitive conclusions related to the impact of articular cartilage treatment at the time of primary ACLR.
Footnotes
Final revision submitted November 23, 2025; accepted November 30, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.B.Y. has received consulting fees from Stryker, JRF Ortho, and Olympus America; nonconsulting fees from Arthrex; education payments from Medwest Associates; and honoraria from JRF Ortho. B.J.C. has received consulting fees from Aesculap Biologics, Arthrex, Bioventus, DJO, Vericel Corporation, Acumed, Anika Therapeutics, OSSIO, and Pacira Pharmaceuticals Incorporated; nonconsulting fees from Terumo BCT; royalties from Arthrex; hospitality payments from GE Healthcare; and honoraria from Vericel Corporation. J.C. has received consulting fees from Smith & Nephew, RTI Surgical, Vericel, Arthrex, Depuy Synthes Products, and Linvatec; education payments from Medwest Associates, Arthrex, and Smith & Nephew; hospitality payments from Stryker and Medical Device Business Services; and a grant from Arthrex.
