Abstract
Background:
The role of magnetic resonance imaging (MRI) in the treatment protocol of lateral epicondylitis is controversial.
Purpose:
To explore the relationship between the radiohumeral synovial plica (RHSP) and surrounding anatomic landmarks through cadaveric studies, perform a precise and detailed morphometric parametric analysis of RHSP using MRI in patients with lateral epicondylitis, and assess the predictive power of RHSP syndrome on the failure of nonoperative treatment.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
There were a total of 8 elbow specimens in this study. The MRI findings were compared with those in anatomic sections and histological preparations. A total of 459 patients with lateral epicondylitis from 3 hospitals were included and divided into a nonoperative treatment failure group (n = 138) and a nonoperative treatment success group (n = 321). The prognostic prediction model for nonoperative treatment was developed using the logistic regression method. Model 1 was based on logistic regression results, and model 2 was model 1 with the parameter of RHSP syndrome removed.
Results:
The RHSP is contiguous with the radiohumeral articular capsule and subtly mingles with the lateral collateral ligament (LCL) complex and common extensor tendon (CET), forming an enthesis. Furthermore, the authors found that patients in the nonoperative treatment failure group had greater RHSP dimensions and a higher prevalence of RHSP syndrome (all P < .05). In addition, sex, RHSP syndrome, size of CET tear, grade of LCL complex abnormality, and muscle edema were independent predictors of failure for nonoperative treatment. The AUC of model 1 (0.839; 95% CI, 0.796-0.881) was significantly higher than that of model 2 (0.805; 95% CI, 0.760-0.851) (P = .024).
Conclusion:
The authors developed a model to predict the failure of nonoperative treatment in patients with lateral epicondylitis. Additionally, RHSP syndrome significantly improves the model's predictive capacity.
Lateral epicondylitis (LE) is a painful disorder of the lateral elbow associated with the origin of the common extensor tendon (CET) and is one of the most common pain syndromes in the upper extremity.1,36 Most patients with LE will experience significant improvement in pain after 6 to 12 months of nonsurgical treatment. 40 However, there are some patients who require surgical treatment because long-term nonoperative treatment is ineffective.7,23 It has been proposed that this ineffectiveness may be because the pain source in LE involves not only CET lesions but also coexisting radiohumeral synovial plica (RHSP) syndrome.34,38 A comprehensive diagnosis of the source of lateral elbow pain is essential for the timely selection of appropriate treatment, pain relief, and restoration of function.
There is limited knowledge about RHSP and controversy about the reported symptoms and examination methods in patients with RHSP syndrome.2,9,26,29 It has been reported that only 25% of patients with posterolateral elbow pain exhibit typical painful snapping during the clinical physical examination. 26 Therefore, when LE is combined with RHSP syndrome, it is difficult for physicians to differentiate between different causes of pain based on clinical presentation and physical examination alone. In this condition, magnetic resonance imaging (MRI) can be helpful to the clinicians in diagnosing and determining treatment plans as a reliable tool for ruling out other possible causes and assessing the severity of the disease.5,16,29,35 Therefore, we first aimed to explore the relationship between RHSP and surrounding anatomic landmarks through cadaveric studies. In addition, we performed precise and detailed morphometric parametric analysis of RHSP in patients with LE by MRI. Finally, the predictive ability of RHSP syndrome to predict the failure of nonoperative treatment was assessed.
Methods
Cadaveric Study
The study was conducted following the principles of the Helsinki Declaration and received approval from the institutional review board. Informed consent was obtained from the deceased donor's family.
Cadaveric Elbow Specimens and MRI
Elbow joints from 4 fresh human cadavers (including 2 females and 2 males; mean age at death, 68 years) were obtained, and permissions were obtained for use. All cadaveric elbow specimens were rapidly deep-frozen at −80°C (Haier Biomedical; DW-40W100). Before further analysis, each elbow specimen was evaluated by anteroposterior and lateral radiographs to rule out articular disorders or osseous abnormalities. Detailed MRI scans were performed after the specimens had been thawed gradually for 24 hours at room temperature. A 3.0-T MRI device (Philips Achieva) and appropriate coil were used to perform coronal, axial, and sagittal proton density–fat suppression (PD-FS) scans. Detailed parameters are given in Table S1.
Anatomic Specimen Preparation
After imaging, all cadaveric specimens were frozen again at −80°C for >24 hours. Frozen specimens were then sectioned into 2 mm–thick slices in the axial (n = 3), coronal (n = 3), and sagittal (n = 2) planes based on MRI sections using a stainless steel band saw (American Meat Equipment Corp). Two physicians (R.B. and J.C.) compared MRI scans and cadaveric sections to determine the presence of RHSP in the elbow specimen and the anatomic features of RHSP (Figures 1 and 2).

Sagittal images of the radiohumeral synovial plica (RHSP) seen in the elbow specimen obtained in a 56-year-old man. (A) Midsagittal schematic of the elbow. (B) Sagittal elbow specimen section. (C) Sagittal proton density–fat suppression image of the elbow specimen. Panels A, B, and C are the same levels of the elbow specimen. The RHSP (arrow) was found to be a triangular thickening of the synovium that extended into the elbow joint cavity between the capitellum (star) and the radial head (triangle). (D and F) Hematoxylin-eosin and Masson trichrome staining of the elbow specimen (original magnification, ×4). The RHSP (arrow) is located at the proximal edge of the annular ligament (AL). (E and G) Local magnification of RHSP (rectangular part of D and F; original magnification, ×100). RHSP consists of a synovial epithelium on the outside and a thick fibrous shaft (including fat and vascular tissue) on the inside.

Coronal images of the radiohumeral synovial plica (RHSP) seen in the elbow specimen obtained in a 71-year-old woman. (A) Midcoronal schematic of the elbow. The lateral epicondyle of the humerus is divided into 3 layers: the inner layer is the RHSP (layer 1, black), the middle layer is the lateral collateral ligament complex (layer 2, dark gray), and the outer layer is the common extensor tendon (CET) (layer 3, light gray). (B) Coronal elbow specimen section. (C) Coronal proton density–fat suppression image of the elbow specimen. The star indicates the capitellum and the triangle the radial head. Panels A, B, and C are the same levels of the elbow specimen. (D and F) Hematoxylin-eosin and Masson trichrome staining of the elbow specimen (original magnification, ×4). The RHSP (arrow) was found in the elbow joint cavity between the capitellum (star) and the radial head (triangle). (E and G) Local magnification (rectangular part of D and F) of the RHSP (original magnification. × 400). The RHSP (star) and lateral collateral ligament complex (rhombus) are mingled without distinct boundaries.
Histological Examination
The histological sections were fixed in 10% neutral phosphate-buffered formalin, dehydrated in ascending ethanol, decalcified with ethylenediaminetetraacetic acid and formic acid, and then embedded in paraffin. Subsequently, 4 μm–thick sections were cut. Histological sections were stained with hematoxylin-eosin and Masson trichrome and observed under the light microscope.
Clinical Study
Study Design and Participants
The study was approved by the institutional review board, and the requirement of informed consent was waived. This retrospective study included consecutive patients with LE treated at 3 hospitals between January 2017 and June 2023. All patients had typical symptoms of lateral elbow pain, which was diagnosed with LE by physical examination, and underwent MRI of the elbow within a week of diagnosis. The exclusion criteria were as follows: (1) patient had systemic comorbidities, (2) history of elbow surgery or arthroscopic procedure, (3) insufficient clinical information, (4) loss of follow-up, or (5) poor MRI quality. Initially, all patients underwent nonoperative treatment. Failure of nonoperative treatment was defined as the absence of improvement in pain or dysfunction after 6 to 12 months of such management. This time frame is widely recognized in the literature as the point at which surgical intervention needs to be considered when symptoms persist despite nonoperative treatment, as most people experience pain relief and functional recovery within this duration.7,15,23 Based on the outcome of nonoperative treatment, patients were divided into 2 groups: the nonoperative treatment success group and the nonoperative treatment failure group.
MRI Examinations
MRI examinations of the elbow were conducted at 3 hospitals using a 3.0-T MRI scanner (Philips Achieva) with a dedicated coil. The MRI protocol consisted of T1-weighted imaging (T1WI) in the coronal plane and PD-FS sequences in the axial, coronal, and sagittal planes. Detailed parameters are given in Table S1.
MRI Analysis
All elbow MRI scans were reviewed by an experienced musculoskeletal radiologist (R.B.) who was blinded to the clinical data. The RHSP is a triangular synovial thickening located at the proximal border of the annular ligament, which is attached to the joint capsule and extends medially along the elbow joint cavity between the capitellum and the radial head. In PD-FS and T1WI sequences, the RHSP appears as low signal intensity. The presence of RHSP in patients with LE was assessed by signal characteristics and anatomic location on MRI. Following established methodologies, quantitative parameters of the RHSP, including length, thickness, and cross-sectional area, were measured on the image slice that displayed its largest dimensions.4,29,35 RHSP dimensional parameters were assessed from the coronal and sagittal planes. RHSP length was measured from the inner tip of the RHSP to the outer capsule at the maximal width plane (Figure 3, B and F). The RHSP thickness was measured at a distance of 4 mm from the annular ligament–capsular complex (Figure 3, C and G). The area was evaluated by drawing a triangle or polygon around the RHSP (Figures 3, D and H). RHSP length, thickness, and area were measured similarly on coronal and sagittal images. If the area in the sagittal images was >20 mm2, the RHSP was considered pathological (RHSP syndrome).4,35 In the PD-FS sequence, RHSP with higher signal intensity than muscle was considered to have abnormally increased signal. 4
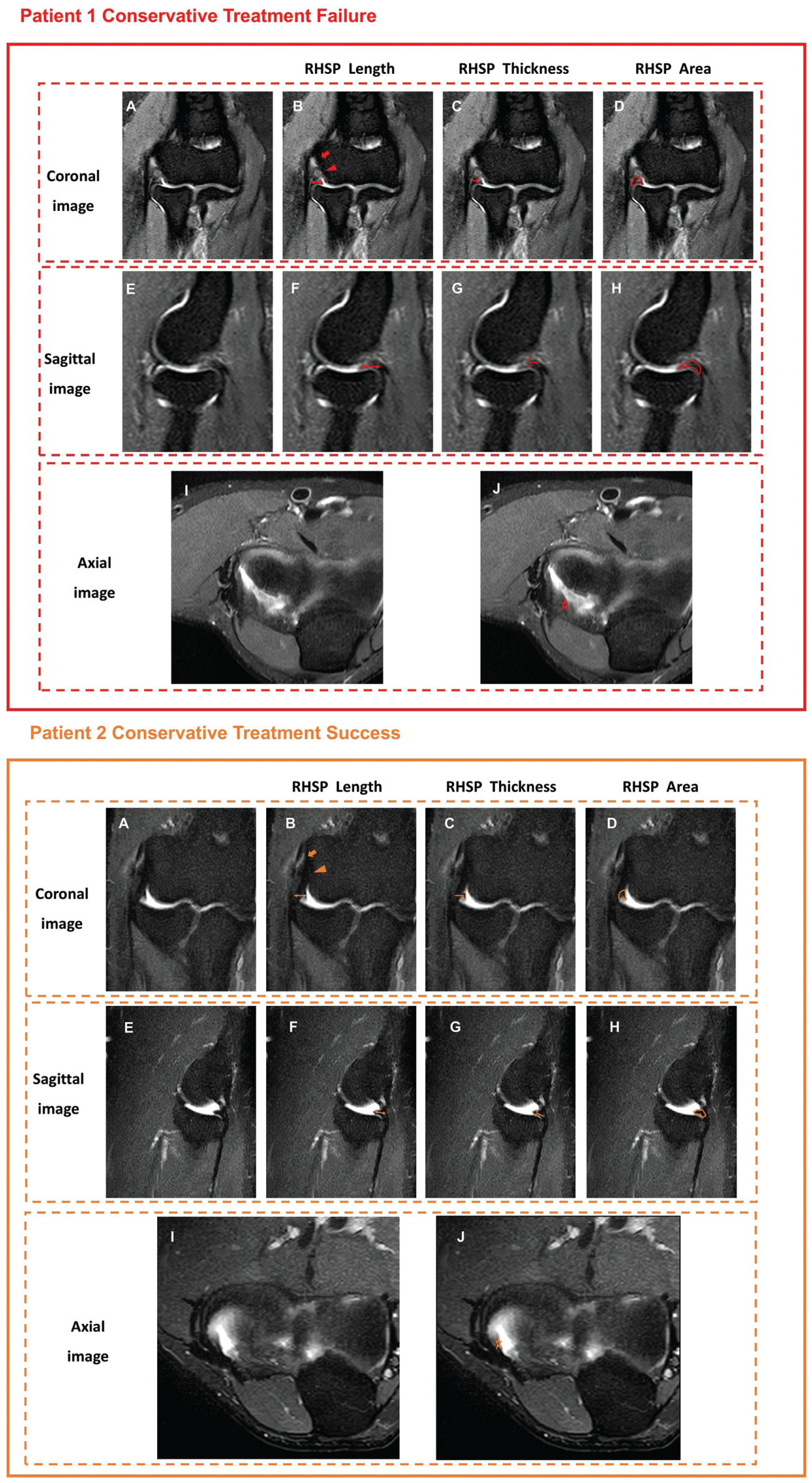
Magnetic resonance imaging (MRI) proton density–fat suppression sequences obtained in patients with lateral epicondylitis. Patient 1 underwent surgery after nonoperative treatment failed. Patient 2 underwent successful nonoperative treatment. Panels A, E, and I are MRI scans of the coronal, sagittal, and axial planes of patients 1 and 2. (B and F) The length of the radiohumeral synovial plica (RHSP) (double arrow). (C and G) The craniocaudal thickness of the RHSP 4 mm from the capsule (double arrow). (D and H) The area of the RHSP (polygon outlined by solid line). (J) Axial position of the RHSP (low-signal polygon marked by star). Note the enhanced signal within the RHSP and increased RHSP area in patient 1 compared with patient 2. Patient 1 has a common extensor tendon (CET) tear (MRI grade 2, arrow), and the lateral collateral ligament (LCL) complex shows thickened edema (MRI grade 1, triangle). Patient 2 has CET tendinosis (MRI grade 1, arrow) and normal LCL complex (MRI grade 0, triangle).
On PD-FS images, CET tendinosis was defined as signal intensity higher than muscle signal intensity but lower than fluid-like signal intensity, with or without an increased tendon thickness. The tear was defined as the presence of a fluid-like signal intensity within the tendon. The tear size was the maximum length of the fluid-like signal within the tendon. CET abnormalities were graded as follows: grade 1: tendinosis or tear <20% of the tendon thickness; grade 2: tear between 20% and 80% of the tendon thickness; and grade 3: tear >80% of the tendon thickness. Lateral collateral ligament (LCL) complex abnormalities were graded as follows: grade 0: normal; grade 1: thickening, thinning, partial tear, or periligamentous edema; and grade 2: complete tear. Muscle edema, joint effusion, cartilaginous defect, subchondral cyst, and subchondral bone marrow edema were classified as present or absent.
Statistical Analysis
All statistical analyses were performed using the SPSS statistical package (Version 26.0; IBM). Continuous variables are presented as median and interquartile range (IQR). Categorical variables are expressed as numbers and percentages. Differences between the 2 groups were analyzed using the Student t test, Mann-Whitney U test, or chi-square test, as appropriate.
Independent predictors of failure for nonoperative treatment were assessed using logistic regression. To avoid multicollinearity among the highly correlated RHSP parameters, the representative variable “RHSP syndrome” was selected for inclusion in the regression analyses. Demographic and MRI characteristics were included in univariate analysis, and then all significant parameters in the univariate analyses were analyzed using backward stepwise multivariate logistic regression (P < .05). To isolate the predictive contribution of RHSP syndrome, we constructed 2 predictive models: model 1 included all significant parameters in the multivariate logistic regression analysis (P < .05), and model 2 was model 1 with “RHSP syndrome” removed. The ability of the parameters to predict the failure of nonoperative treatment was verified using the area under the curve (AUC) of the receiver operating characteristic curve. The DeLong test was used to compare the AUCs of the 2 models. In addition, we constructed a nomogram model to visualize the probability of operative treatment and evaluated the performance of model 1. The covariance of the parameters in the model was assessed through the variance inflation factor. Correlations were assessed using the Spearman or Pearson method, as appropriate. To ensure the reliability of the MRI analysis, images of 30 patients were randomly selected and reassessed by the same musculoskeletal radiologist (R.B.) at 3-month intervals and independently interpreted by another experienced musculoskeletal radiologist (P.W.). The intraobserver and interobserver reliability of MRI parameters were assessed using intraclass correlation coefficients. A 2-tailed P value <.05 was considered statistically significant.
Results
Cadaveric Study
Anatomic sections, MRI, and corresponding histological slides are shown in Figures 1 and 2. All elbow specimens in our study had clear RHSP. On MRI scans, the RHSPs were evaluated with the same parameters observed on cadaveric elbow specimens. On coronal images, the mean length, thickness, and area of RHSP were 5.18 ± 1.06 mm, 1.02 ± 0.65 mm, and 9.98 ± 3.48 mm2. On sagittal images, the mean length, thickness, and area of RHSP were 5.83 ± 0.51 mm, 1.62 ± 0.38 mm, and 10.42 ± 2.40 mm2. Histologically, the RHSP is externally composed of synovial epithelium and internally composed of a fibrous shaft, including fat and vascular tissue. The lateral epicondyle can be divided into 3 layers from medial to lateral, according to the anatomic positioning of the RHSP, LCL complex, and CET relative to the lateral epicondyle (Figure 2). We further observed that the RHSP attaches to the LCL complex and, in continuity with the CET, to the lateral epicondyle, with no clear anatomic boundary demarcating these 3 structures.
Clinical Study
Demographic and MRI Characteristics
Table 1 presents a comparison of demographic and MRI characteristics between the operative group and the nonoperative group. There were 527 patients with LE, and 68 were excluded according to the exclusion criteria. Therefore, 459 patients were included in the final study: 321 patients with successful nonoperative treatment and 138 patients with nonoperative treatment failure. The study population consisted of 186 males and 273 females, with a median age of 47 years (IQR, 41-53 years). The nonoperative treatment failure group consisted of significantly fewer males than the successful treatment group (P = .002). Regarding MRI findings, an RHSP was identified in 358 patients (78.0%). The thickness and area of the RHSP were significantly greater in the failure group than in the successful group (all P < .05). The prevalence of RHSP syndrome was significantly higher in the nonoperative treatment failure group (P < .001). Furthermore, CET tears were significantly larger in the nonoperative treatment failure group (P < .001). The nonoperative treatment failure group had significantly higher CET and LCL complex abnormality grades (both P < .001). In addition, muscle edema and subchondral cyst were more common in the nonoperative treatment failure group (P < .001 and P = .039, respectively). The intra- and interobserver reliability of MRI parameters was good (Table S2).
Demographic and MRI Characteristics of Patients With Nonoperative Treatment Success and Failure a
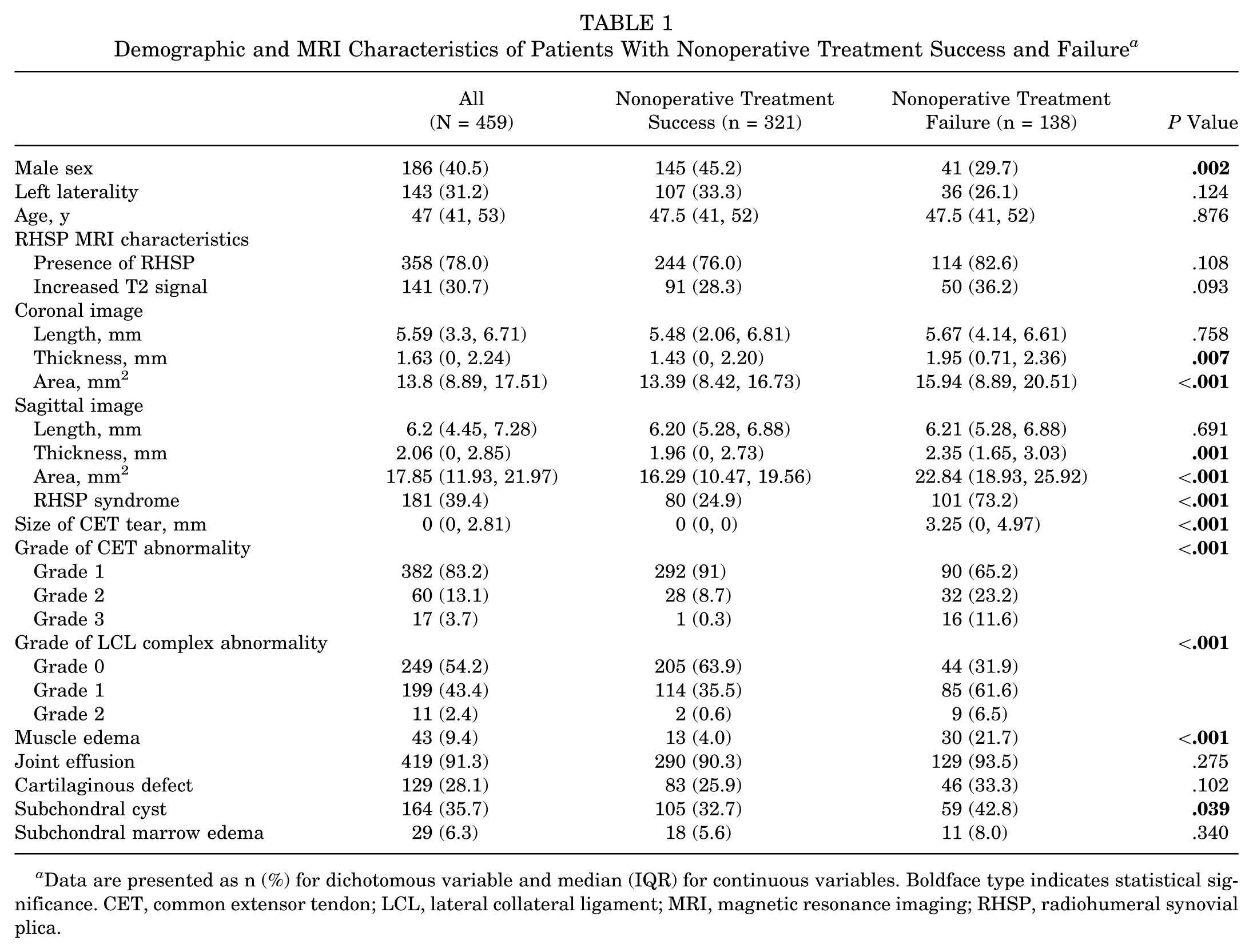
Data are presented as n (%) for dichotomous variable and median (IQR) for continuous variables. Boldface type indicates statistical significance. CET, common extensor tendon; LCL, lateral collateral ligament; MRI, magnetic resonance imaging; RHSP, radiohumeral synovial plica.
Correlation of MRI Features
As shown in Table 2, the length, thickness, and area of RHSP were positively correlated with the grade of CET abnormality and CET tear size (all P < .05). In addition, the area of the RHSP and the grade of LCL complex abnormality were significantly correlated (all P < .05).
Correlation of MRI Features of RHSP With MRI Features of CET and LCL a
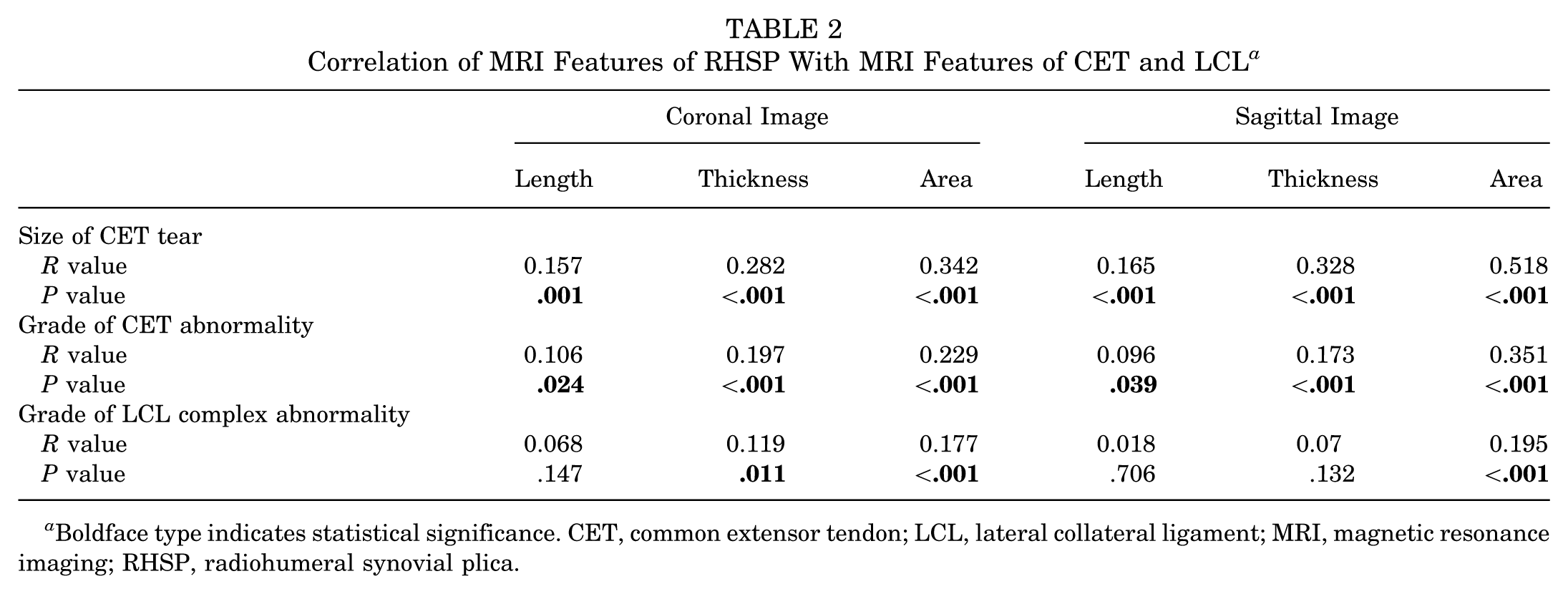
Boldface type indicates statistical significance. CET, common extensor tendon; LCL, lateral collateral ligament; MRI, magnetic resonance imaging; RHSP, radiohumeral synovial plica.
Predictors of Prognosis for Nonoperative Treatment
A total of 6 variables with P < .05 were entered into the multivariate logistic analysis. The final analysis showed that sex (OR, 0.475; 95% CI, 0.278-0.814; P = .007), the presence of RHSP syndrome (OR, 4.653; 95% CI, 2.704-8.07; P < .001), the size of CET tear (OR, 1.358; 95% CI, 1.197-1.541; P < .001), the grade of LCL complex abnormality (grade 1: OR, 1.979; 95% CI, 1.186-3.302; P = .009; grade 2: OR, 10.276; 95% CI, 1.117-94.530; P = .04), and the presence of muscle edema (OR, 4.445; 95% CI, 1.880-10.510; P = .001) were independently predictive of failure of nonoperative treatment (Table 3). The AUC of model 1 (0.839; 95% CI, 0.796-0.881) was significantly higher than that of model 2 (0.805; 95% CI, 0.760-0.851) (P = .024) (Figure 4). We have developed a nomogram of model 1 (Figure 5).
Logistic Regression Analysis to Predict Failure of Nonoperative Treatment of Lateral Epicondylitis a
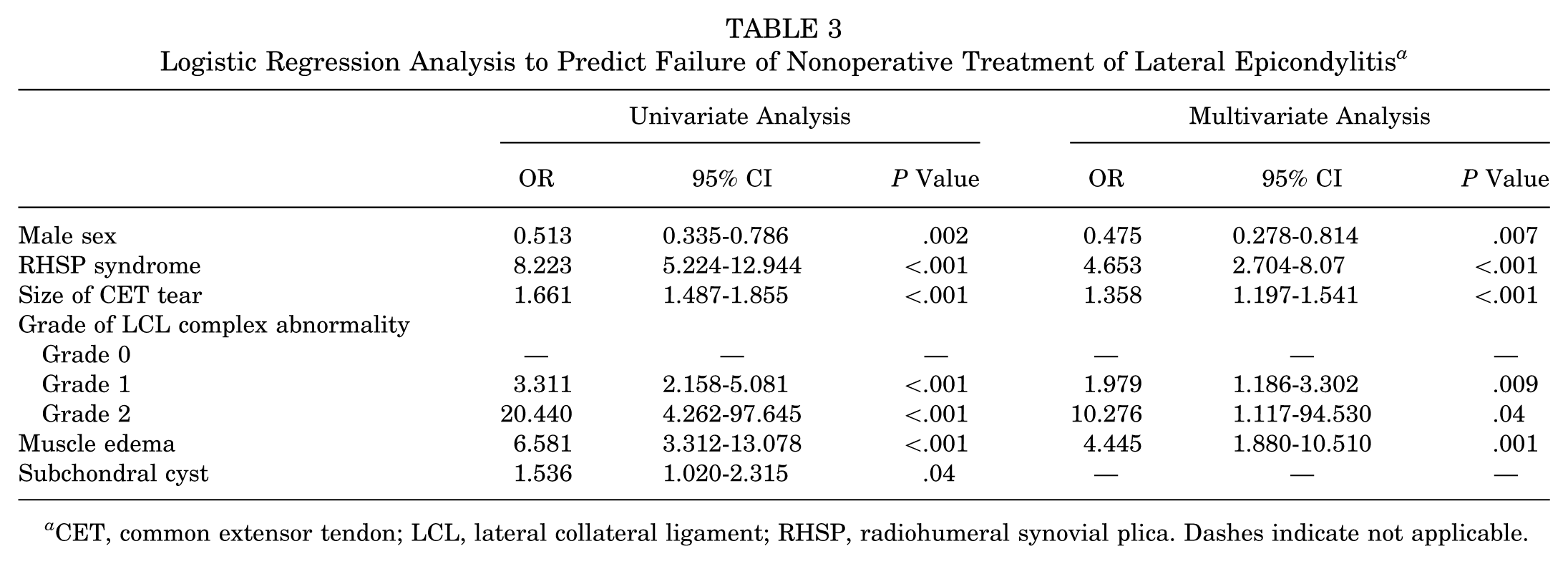
CET, common extensor tendon; LCL, lateral collateral ligament; RHSP, radiohumeral synovial plica. Dashes indicate not applicable.

Receiver operating characteristic curves of model 1 (red line) and model 2 (blue line) for predicting the failure of nonoperative treatment in lateral epicondylitis. The area under the curve (AUC) of model 1 (with the parameter “RHSP syndrome”) was significantly higher than that of model 2 (without the parameter “RHSP syndrome”) (P = .024). This suggests that the inclusion of the RHSP syndrome variable significantly improves the predictive ability of the model.

The nomogram for model 1 was used to predict the failure of nonoperative treatment in lateral epicondylitis. The value of each variable was given a score on the point scale axis. A total score could be easily calculated by adding each single score, and by projecting the total score to the total point scale, we were able to estimate the probability of failure of nonoperative treatment in patients with lateral epicondylitis. CET, common extensor tendon; LCL, lateral collateral ligament; RHSP, radiohumeral synovial plica.
Discussion
The main findings of the present study were as follows. (1) From a structural perspective, the RHSP is a contiguous structure with the radiohumeral articular capsule and subtly integrates with the LCL complex and CET, forming an enthesis at the lateral epicondyle. (2) From a clinical perspective, the presence of RHSP syndrome may increase the confidence to predict the failure of nonoperative treatment in patients with LE.
Currently, there is a lack of standardization in treatment decision-making for patients with LE, which relies heavily on the personal experience of clinicians. 23 Without timely and appropriate treatment, patients are at higher risk of further exacerbation of CET damage. Moreover, the longer the duration of symptoms, the lower the likelihood of successful recovery with nonoperative or surgical treatment.27,37 Current research has recognized the importance of developing a rational treatment plan and accurate early prediction of the treatment outcome. Studies have found that persistent pain, CET abnormalities, and muscle edema are associated with failure of nonoperative treatment.17,21,27,31 However, little is known about the role of RHSP syndrome in guiding treatment planning for patients with LE. Arthroscopy is the gold standard for diagnosis of RHSP, but it is invasive and expensive. Therefore, among noninvasive methods, MRI is the most reliable tool for the assessment of RHSP as it is highly consistent with the observations made by arthroscopy and cadaveric specimens.6,13,25 Our study is the largest investigation of RHSP by MRI in the existing literature and the first to assess the relationship between MRI parameters of RHSP and treatment options for patients with LE.
RHSP is reported to be a common anatomic finding, occurring in 86% to 100% of specimens.14,19,28 Our study showed a 100% prevalence of posterior RHSP in the examined specimens. Previous studies of RHSP have focused on asymptomatic patients and RHSP syndrome. As our methodology for measuring RHSP dimensions was consistent with that of previous studies,9,16,29,35 a direct comparison of the results is feasible. In a study of RHSP in 60 normal elbows using MRI, the median anteroposterior length was 4.3 mm, the mediolateral length was 3.9 mm, and the craniocaudal thickness was 1.9 mm. 16 In another study measuring RHSP, the mediolateral and sagittal dimensions of the RHSP in the asymptomatic group were 3.8 mm and 4.7 mm, respectively. 9 Our results demonstrate that RHSP dimensions in patients with LE are substantially larger than both the normal values reported in the literature and those measured in our own cadaveric specimens. According to previous studies, symptomatic RHSP tends to be thick and fibrotic with signs of chronic synovitis.
RHSP syndrome is considered if the RHSP area in the sagittal plane is >20 mm2 using MRI based on established literature.2,30,35 Specifically, Ruiz de Luzuriaga et al 35 compared RHSP areas between patients with RHSP syndrome and asymptomatic volunteers, finding that more than half of the patients with RHSP syndrome had a cross-sectional area >20 mm2. Similarly, Aydoğmuş et al 4 recognized 20 mm2 as the threshold indicating an abnormal RHSP. The prevalence of RHSP syndrome in LE was 39.4% in our study, which is higher than the prevalence previously found in asymptomatic populations. 35 Previous studies have analyzed the incidence of RHSP syndrome in patients with chronic LE and proposed a relationship between ineffective nonoperative treatment in patients and RHSP syndrome.6,13 Consequently, surgical intervention is often recommended for chronic LE with concomitant RHSP syndrome,3,22,26,34 with satisfactory outcomes reported.20,39 However, to the best of our knowledge, no study has directly analyzed the correlation between RHSP syndrome and nonoperative treatment prognosis. Our research is the first to specifically address this evidence gap.
Furthermore, we developed a simple, intuitive, and highly accurate nomogram model. This nomogram helps orthopaedic surgeons to personalize the prediction of each patient's outcome of nonoperative treatment based on 5 factors—sex, the presence of RHSP syndrome, the size of the CET tear, abnormalities of the LCL complex, and the presence of muscle edema—to timely determine whether a patient needs early surgical treatment and to further refine the patient's treatment plan, avoiding unnecessary pain and saving time and money.
Moreover, comparison of the 2 models revealed that the inclusion of RHSP syndrome significantly enhanced predictive power (AUC, 0.839 vs 0.805; P = .024). While the absolute increase of 0.034 may appear modest, it holds clinical importance. This enhancement reflects the model's improved capacity to identify a distinct patient subgroup—those with LE complicated by RHSP syndrome—who are predisposed to nonoperative treatment failure due to the additional pathology. Early identification of these patients using the nomogram can facilitate a timely shift in management strategy toward interventions that address both the CET and the RHSP, thereby personalizing treatment and potentially improving outcomes for this challenging population. The reasons for the close association of RHSP with LE are analyzed in the following points. In the cadaveric study, we found that the RHSP, unlike the annular ligament, the lateral portion of which is subtly mingled with the LCL complex, eventually forms an enthesis with the CET on the lateral epicondyle. Therefore, primary lesions of LE (eg, angiofibroblastic hyperplasia of CET origin) may affect the hypertrophy of the RHSP due to the anatomic location connection. 38 Mechanically, the RHSP is located within the radiohumeral joint at the maximal extension, covering the posterior half of the radial head that in such a position is not articulated with the capitulum. 12 Elbow extension increases tension on the humeral capsule, and forearm pronation places tension on the CET, forcing the RHSP to displace into the radiopatellar joint. 20 Repetitive piston radial head motion causes considerable compressive stress, which in turn leads to hypertrophy of the RHSP. 19 The tension on the CET may push the pathological RHSP into the radiocapitellar joint and lead to refractory lateral elbow pain.14,32,34 Furthermore, we found that in LE, the size of the RHSP was positively correlated with lesions of the CET and LCL complexes. Thus, LE may involve lesions not only of CET origin but also of RHSP. Therefore, if long-term nonoperative treatment fails in patients with LE or if the prognosis for arthroscopic surgery is poor, clinicians need to consider the possibility of concurrent RHSP syndrome and the need for concomitant treatment of RHSP to ameliorate the symptoms of chronic lateral elbow pain.22,26,34
In addition, the muscular endurance ratio of the wrist extensors was lower in women than in men, suggesting that women need to exert greater effort to maintain the wrist extension position against resistance; therefore, sex differences in endurance may be associated with the refractoriness of LE. 18 The CET is the main site of repetitive stress injury in LE, and therefore CET abnormalities are strongly associated with the failure of nonoperative treatment.10,33 Furthermore, the LCL complex and the CET are anatomically closely related, and thus LCL complex injury often occurs with progressive injury of CET.8,24 Finally, muscle edema may be associated with compensation due to abnormal motion or compensation associated with CET pathology. Consequently, patients in the nonoperative treatment failure group exhibited a higher incidence of muscle edema, likely reflecting greater mechanical stress or aberrant movement patterns. 11
Limitations
There are some limitations to this study. First, our database is retrospective and may have confounding variables that were not collected or included. In addition, there is a lack of long-term follow-up on the prognosis of surgical treatment of patients with LE. Furthermore, because no RHSP-negative specimens were identified in this study, it was not possible to assess the negative predictive value of MRI. However, this finding concurs with previous literature reporting a similarly high prevalence of RHSP in specimens. Therefore, future studies should expand sample sizes and include confirmed negative cases to comprehensively evaluate the diagnostic performance of MRI.
Conclusion
The treatment process for LE can be long and frustrating for patients, especially because there is no standard treatment. Clinicians should consider earlier surgical intervention for female patients with larger CET tears, abnormalities in the LCL complex, and muscle edema, and especially those with concomitant RHSP syndrome, as these factors indicate poor outcomes with prolonged nonoperative treatment.
Footnotes
Supplemental Tables
Reproducibility analysis of some MRI parameters
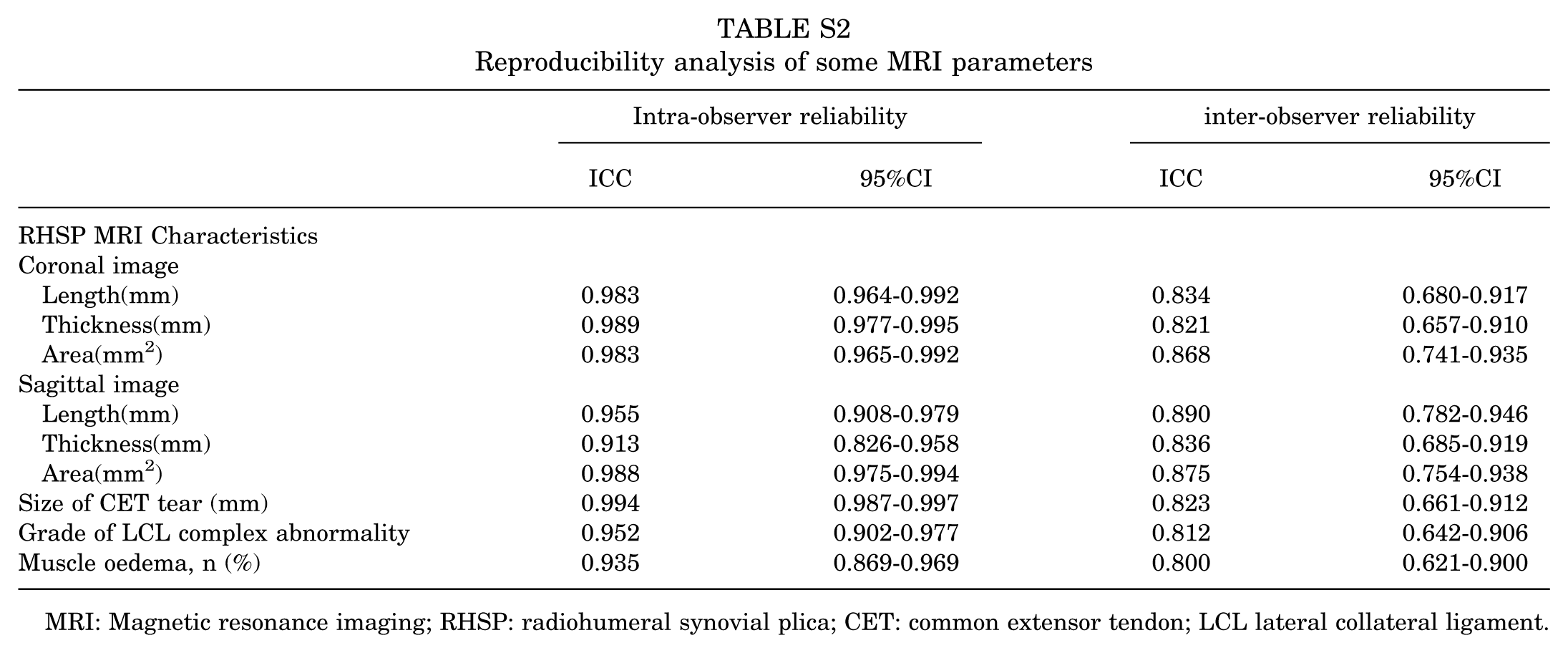
| Intra-observer reliability | inter-observer reliability | |||
|---|---|---|---|---|
| ICC | 95%CI | ICC | 95%CI | |
| RHSP MRI Characteristics | ||||
| Coronal image | ||||
| Length(mm) | 0.983 | 0.964-0.992 | 0.834 | 0.680-0.917 |
| Thickness(mm) | 0.989 | 0.977-0.995 | 0.821 | 0.657-0.910 |
| Area(mm2) | 0.983 | 0.965-0.992 | 0.868 | 0.741-0.935 |
| Sagittal image | ||||
| Length(mm) | 0.955 | 0.908-0.979 | 0.890 | 0.782-0.946 |
| Thickness(mm) | 0.913 | 0.826-0.958 | 0.836 | 0.685-0.919 |
| Area(mm2) | 0.988 | 0.975-0.994 | 0.875 | 0.754-0.938 |
| Size of CET tear (mm) | 0.994 | 0.987-0.997 | 0.823 | 0.661-0.912 |
| Grade of LCL complex abnormality | 0.952 | 0.902-0.977 | 0.812 | 0.642-0.906 |
| Muscle oedema, n (%) | 0.935 | 0.869-0.969 | 0.800 | 0.621-0.900 |
MRI: Magnetic resonance imaging; RHSP: radiohumeral synovial plica; CET: common extensor tendon; LCL lateral collateral ligament.
Final revision submitted October 14, 2025; accepted December 5, 2025.
This study was supported by the National Natural Science Foundation of China (grant numbers: 82572168, 82171921, 81771809).
The authors declared that they have no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the ethics committee of Beijing Jishuitan Hospital, Capital Medical University (ID 20210309).
