Abstract
Background:
There is an opportunity to implement improved techniques for the repair of radial meniscus tears; previous studies have shown a benefit to the addition of rebar or “ripstop” sutures parallel to the tear, forming “hashtag constructs” to strengthen the repair of radial meniscus tears. Studies have not directly compared different types of rebar constructs.
Purpose/Hypothesis:
The study purpose is to evaluate the biomechanical properties of different suture repair patterns for lateral meniscus tears, including patterns with and without rebar sutures, as well as all-inside and capsule-based constructs. It was hypothesized that the patterns with reinforcing rebar sutures will have a significantly higher load to failure (N).
Study Design:
Controlled laboratory study.
Methods:
A total of 68 fresh-frozen lateral human menisci were randomized into 8 groups in 2 rounds of testing. The first round compared rebar and nonrebar suture constructs: 2 simple repairs (double horizontal [DH], cross-stitch [CS]), 1 “hashtag” construct with 2 reinforcing rebar-type sutures parallel to the tear and 2 sutures crossing the tear (all-inside rebar 1 [AIR1]), and an experimental hybrid construct (oblique box [OB]). The second round focused on 4 rebar suture repair constructs that included the box-type “hashtag” suture repair, comparing all-inside and capsule-based repairs: the same all-inside rebar 2 (AIR2) construct as in round 1, 1 all-capsule rebar (ACR) construct with knots tied on the lateral surface, and 2 combined all-inside and capsule-based rebar constructs (combined rebar 1 [CR1] and combined rebar 2 [CR2]). The repaired menisci underwent cyclic loading and load-to-failure testing. Ultimate failure load data were analyzed using analysis of variance testing to compare multiple groups, along with Tukey-adjusted P values for pairwise testing.
Results:
In the first round, the rebar suture construct (AIR1) sustained a higher ultimate load to failure than the other 3 constructs, at 168.6 N, compared with DH (60.3 N), CS (58.2 N), and OB (102.5 N) (analysis of variance P < .001, pairwise P < .001, P < .001, and P < .002, respectively). In the second round, there was no significant difference in mean ultimate failure load among the 4 rebar constructs, with AIR2 (149.7 N), ACR (148.9 N), CR1 (133.7 N), and CR2 (161.7 N) (P = .596). In pairwise testing, all of the rebar suture constructs (AIR1, AIR2, ACR, CR1, CR2) failed at significantly higher ultimate failure load when compared with the nonrebar suture repair constructs (DH, CS) (P≤ .001 for each).
Conclusion:
Our cadaver study demonstrated that in a model of lateral meniscus radial tear repair, constructs using rebar sutures parallel to the tear provided reinforcement for the sutures spanning the tear and resulted in a higher load to failure when compared with nonrebar suture repair constructs.
Clinical Relevance:
Surgeons should consider adding parallel rebar sutures to their suture constructs when repairing radial tears arthroscopically to improve the strength of the construct, while maintaining flexibility for various techniques.
Meniscal injuries are a common orthopaedic injury, particularly in the young and physically active population, with an estimated incidence rate in the United States as high as 8.27 per 1000 in this population.7,9,13 Asymptomatic, degenerative tears are more common in older adults, whereas symptomatic, acute traumatic tears are more common in younger, active patients.9,15,21 Meniscal tears often are treated surgically due to symptoms, including pain, effusion, stiffness, and mechanical symptoms, such as locking, catching, and clicking. 25
Menisci are composed primarily of type 1 collagen bundles with a complex network of fibers, most of which are oriented circumferentially and provide resistance to compression. 7 Meniscus tears are classified by anatomic location and morphology, including but not limited to horizontal, radial, complex, bucket-handle, and root tears. 7 Radial tears disrupt the circumferentially oriented collagen fibers, disrupting the meniscal hoop strength and resulting in a significant loss of function. 12 Radial tears are associated with higher-grade cartilage damage and osteoarthritis; complete radial tears have been described as functionally similar to complete meniscectomy.3,4,12 Radial tears are more common in younger patients and typically occur with higher-energy traumatic mechanisms; they are often associated with anterior cruciate ligament rupture. 9 With this background, there is impetus to continue to innovate and study techniques for radial meniscus repair.
The basic techniques for meniscus repair include outside-in, inside-out, and all-inside. 14 While these have been studied, there remains a lack of consensus and conflicting analyses on which technique, if any, is superior. 1 Radial meniscus tears are known to be particularly difficult to successfully repair because of the orientation of the collagen fibers, as detailed above. A systematic review examining clinical outcomes of patients with repair of radial meniscus tears found improvement in patient-reported outcomes postoperatively, with 62% complete healing among patients assessed with second-look arthroscopy. 20 Because of the historically variable rates of healing when radial meniscus tears are treated with either all-inside or inside-out horizontal mattress repairs, in 2012, Nakata et al 24 introduced the concept of a rebar or ripstop inside-out technique.8,22 It involves the placement of reinforcing sutures parallel to the tear, followed by more typical repair sutures crossing the tear and the reinforcing sutures; the reinforcing sutures parallel to the tear act as a rebar or ripstop to prevent the crossing sutures from pulling through the meniscus tissue. An example of an arthroscopic meniscus repair using rebar sutures can be seen in Figure 1.
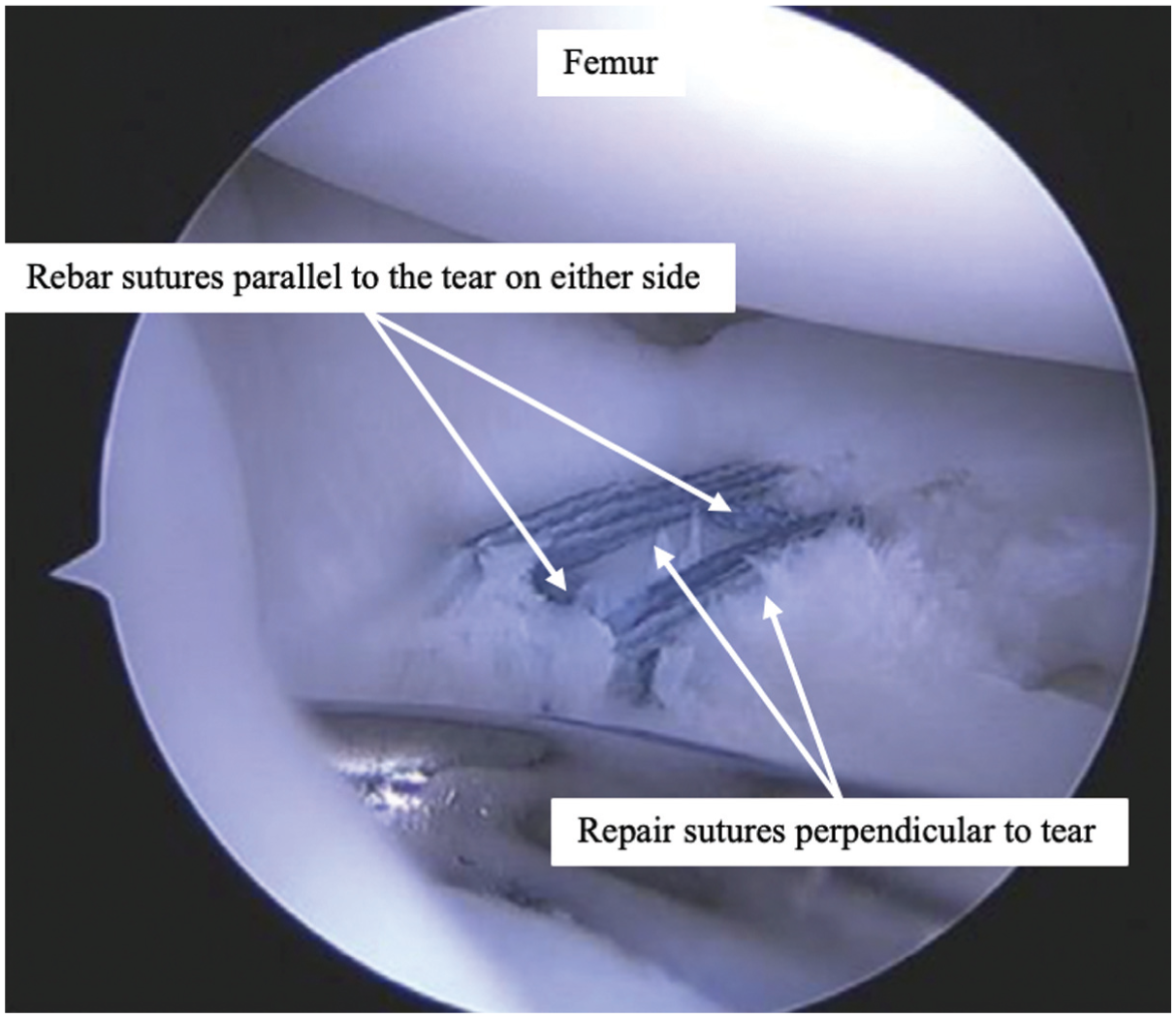
Example of an in vivo all-capsule rebar repair construct for a radial meniscus tear (lateral knee compartment visualized from the anterolateral portal).
This technique concept has gained popularity over the past decade and has been studied by multiple groups. 16 Primarily, previous studies have compared the traditional double horizontal or double cross pattern with a rebar-reinforced horizontal mattress in porcine and human cadaver menisci, finding promising results with a higher ultimate failure load for the rebar-reinforced construct.6,17,23 These studies have not compared multiple different rebar repair constructs. Additionally, these studies have not compared different all-inside and capsule-based techniques, which have evolved over time with the introduction of devices that facilitate the placement of all-inside meniscus and capsule-based repair sutures.
The purpose of this study is to evaluate the biomechanical properties of radial meniscus repair constructs, to help determine which might provide the strongest repair construct for young, active patients. In the first round of testing, 4 different all-inside repair patterns for radial meniscus testing were compared, including rebar and nonrebar constructs. In the second round of testing, we compared 4 “hashtag” constructs using rebar sutures in various repair techniques, including all-inside, all-capsule, and combined repair patterns. Our hypothesis is that there is a difference in load to failure between the rebar and nonrebar constructs.
Methods
Meniscus Preparation
An institutional review board determined that approval was not necessary for this study, as it did not involve patient identifiers, the use of genetic information, or contact with the patient's family, as per guidelines from the US Department of Health and Human Services. A tissue harvesting facility (AlloSource) sourced the cadaveric tissue, which had obtained family consent for use in research purposes prior to conducting this study.
In total, 68 fresh-frozen, adult human, lateral menisci were donated. Each sample consisted of an intact lateral meniscus attached to a tibial bone block with capsule material removed. All samples were kept frozen from time of shipment until day of testing.
Each sample was thawed, prepared, repaired, and tested in a continuous sequence. Each meniscus sample was thawed in a warm water bath to room temperature. After thawing, each sample was removed from its labeled bag and photographed before and after dissecting away the bone block.
Next, a radial tear was created with a No. 11 scalpel blade at the midbody of the meniscus. Approximately 2 mm of tissue was left intact at the medial edge of the meniscus to assist with approximation during suture repairs. After the repair was complete, the remaining tissue bridge was cut to complete the tear.
Repair Constructs
In this study, all-inside suture repair refers to sutures with the knots tied intra-articularly, on the superior or inferior surface of the meniscus. In vivo, these are placed and tied intra-articularly via arthroscopic portals. All-capsule suture repair refers to suture constructs with the knots tied extra-articularly, against the capsule. In vivo, this has typically been done via a medial or lateral incision with careful dissection down the capsule, with sutures passed “inside-out,” meaning they start intra-articularly and the needle is passed through the meniscus and out the capsule, or “outside-in.” More recently, the development of suture deploy devices, such as the FiberStitch (Arthrex), has allowed the placement of sutures with capsular suture anchors to be deployed through arthroscopic portals without the need for an additional open incision. While these devices were not used in this study, the sutures would be considered capsular because they anchored extra-articularly on the capsule.
In the first round of testing, 4 all-inside repair constructs were used, including 2 nonrebar constructs, 1 true rebar construct, and 1 experimental design that had performed favorably during preliminary testing on rubber meniscus models (Figure 2). The nonrebar constructs include the double horizontal (DH) construct, with 2 sutures crossing the tear in a standard perpendicular fashion, and the cross-stitch (CS) construct, with 2 sutures crossing the tear and crossing each other to form an “X.” The all-inside rebar 1 (AIR1) construct consists of 2 reinforcing sutures placed parallel to the tear, followed by 2 horizontal sutures crossing the tear and the reinforcing sutures. The experimental oblique box (OB) construct consisted of reinforcing sutures forming a trapezoidal box on either side of the tear, followed by the crossing double horizontal sutures.
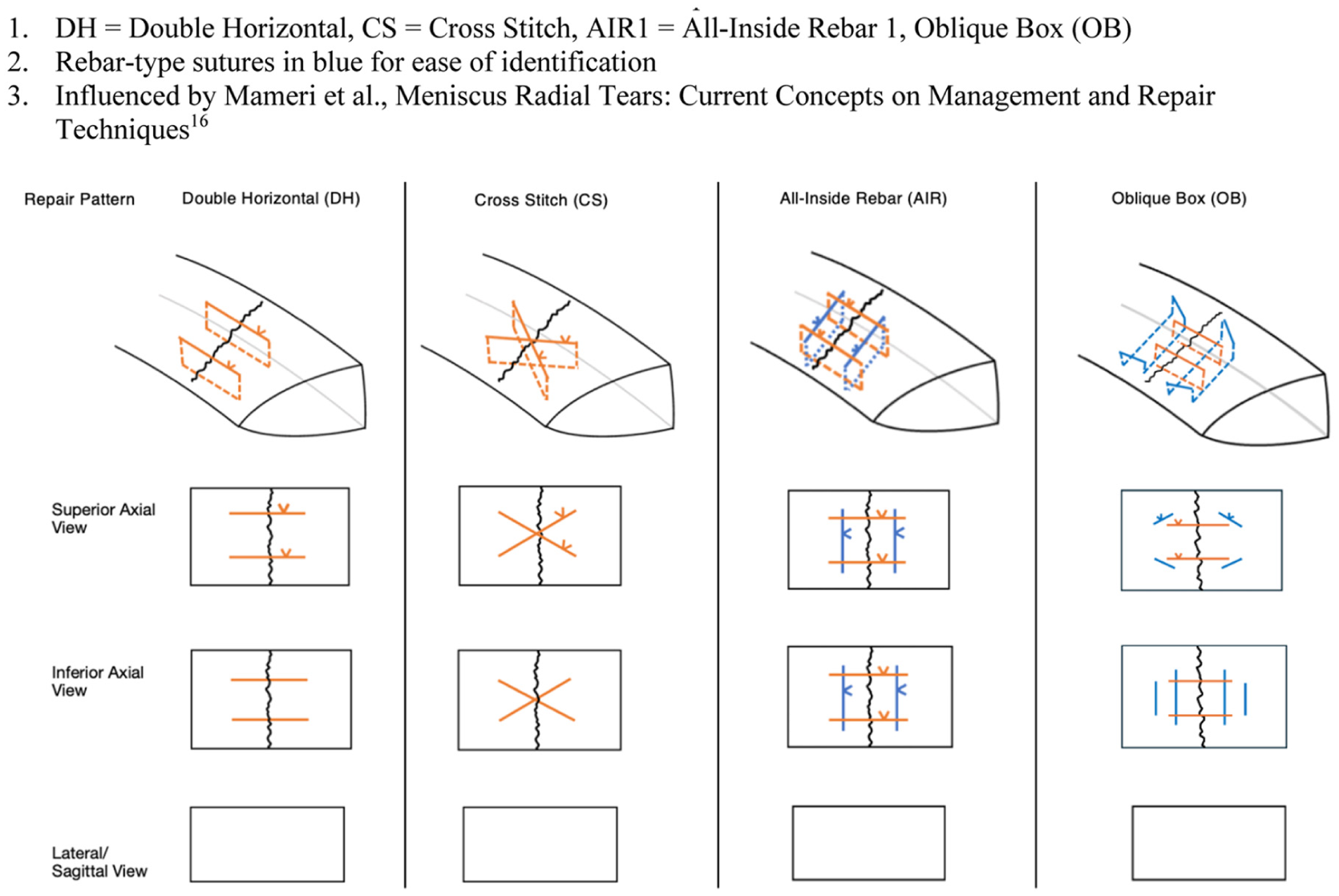
Round 1 diagrams of the radial meniscus repair constructs. Rebar-type sutures in blue for ease of identification. Influenced by Mameri et al. 16
In the second round of testing, 4 different “hashtag” repair constructs using rebar sutures were tested (Figure 3). The all-inside rebar construct from the first round was repeated to ensure our methods remained consistent (all-inside rebar 2 [AIR2]). The second repair (all-capsule rebar [ACR]) used an all-capsule-based construct, which can be completed via inside-out or outside-in arthroscopic techniques in vivo. The remaining 2 repair constructs used a combination of all-inside and capsule-based techniques. The third construct (combined rebar 1 [CR1]) includes capsule-based rebar sutures with all-inside repair sutures across the tear. The fourth construct (combined rebar 2 [CR2]) specifically includes rebar sutures that transverse the superior and inferior meniscal surface but are tied in a capsule-based fashion, avoiding the knots within the joint, with all-inside repair sutures across the tear.
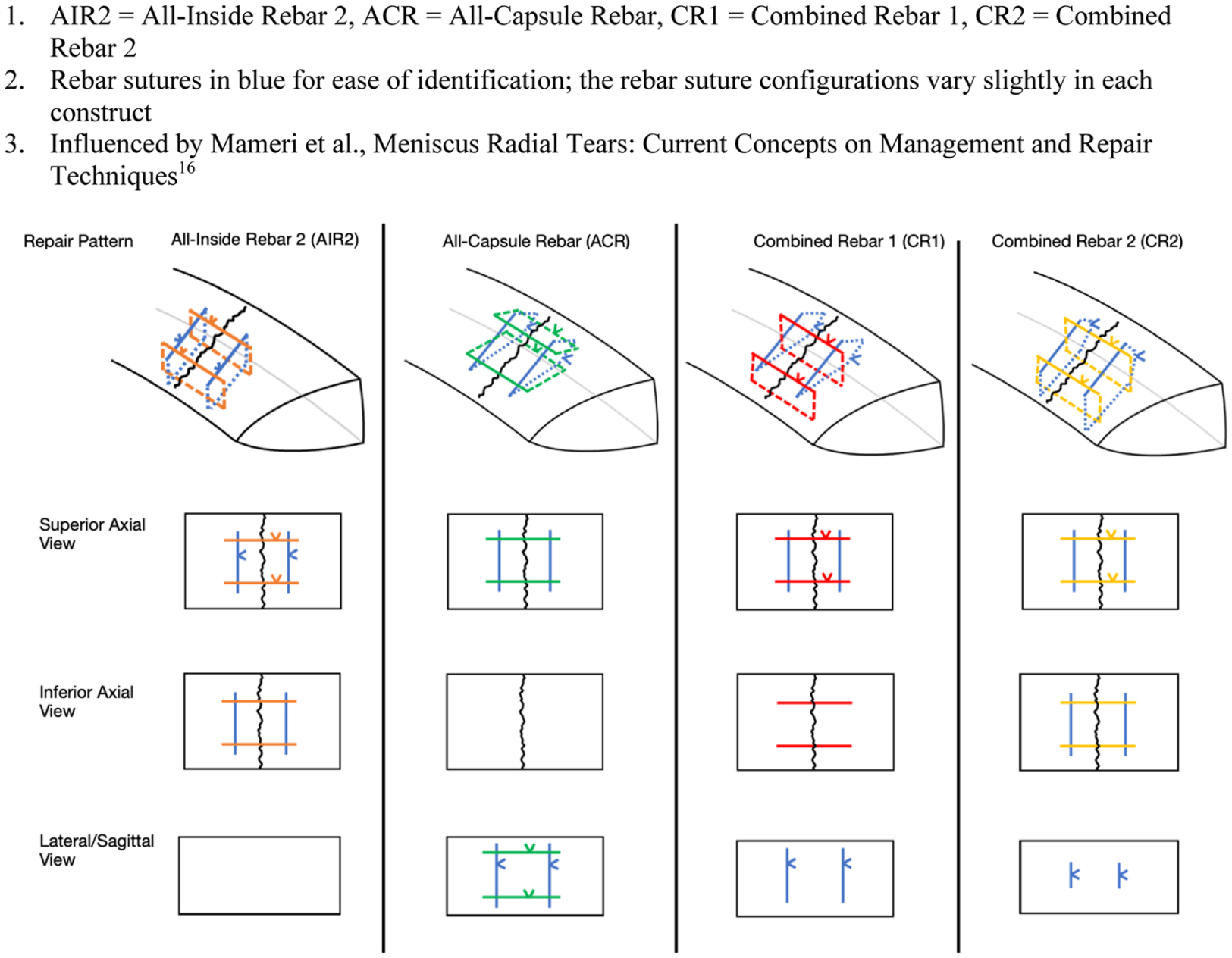
Round 2 diagrams of the radial meniscus tear repair constructs using rebar sutures. Rebar sutures in blue for ease of identification; the rebar suture configurations vary slightly in each construct. Influenced by Mameri et al. 16
Repairs were done using a 2-0 Ultrabraid (Smith & Nephew) suture and a straight needle. In each case, the rebar sutures were placed first, followed by the repair sutures crossing the tear.
All repairs were completed by 1 third-year orthopaedic surgery resident and one fourth-year medical student, with guidance from the principal investigator on this study, a board-certified orthopaedic sports medicine surgeon (K.H.M., D.R.W.B., K.G.S.). Photographs were taken of each repaired meniscus from the superior axial view; for round 2, additional photographs of the inferior axial and lateral or capsule views were taken when relevant (Figure 4).

Photos of menisci after rebar suture repair for radial meniscus tear. (A) Double horizontal superior axial view. (B) Cross-stitch superior axial view. (C) All-inside rebar 1 superior axial view. (D) Oblique box superior axial view. (E1) All-inside rebar 2 (AIR2) superior axial view. (E2) AIR2 inferior axial view. (F1) All-capsule rebar (ACR) superior axial view. (F2) ACR sagittal/lateral view. (G1) Combined rebar 1 (CR1) superior axial view. (G2) CR1 inferior axial view. (H1) Combined rebar 2 (CR2) superior axial view. (H2) CR2 inferior axial view.
Biomechanical Testing
All menisci were tested at room temperature using a servomechanical load testing system (Instron Model 5944; Instron) and custom clamps. We aimed to largely replicate the biomechanical testing protocols of various authors.2,5,17,19,26
To prevent slippage, 100-grit sandpaper was glued to each end of the meniscus to provide additional traction between the tissue and the clamp surface. The sandpaper and glue did not touch the sutures or the meniscal tissue in the area of repair. Each sample was loaded with the radial tear perpendicular to the force vector of the testing system (Figure 5).

Repaired radial meniscus tear setup for testing in the Instron machine.
Each sample underwent cyclic preconditioning, consisting of 20 cycles of loading between 2 and 5 N, immediately followed by cyclic loading of 5 to 30 N for 500 cycles at 1 Hz. After completion of cyclic loading, samples rested for 30 minutes with no load applied. After this resting period, the samples underwent load to failure with force increased at a constant displacement rate of 5 mm per minute until failure. Throughout the biomechanical testing, the menisci were sprayed with room temperature saline to ensure they remained moist and did not become brittle from drying out.
Data Recording and Analysis
Data were recorded as load displacement curves by Instron's built-in software Bluehill (Bluehill for Instron). Failure was automatically recorded by Bluehill as a decrease of at least 20% from maximum load. Ultimate failure load (N) was determined by the peak of the load displacement curve. Mode of failure was visually observed, video recorded, and photographed, with subjective notes recorded within the Bluehill platform and on an Excel spreadsheet.
With data from previous biomechanical studies of radial tear repairs, an a priori power analysis (G*Power; University of Dusseldorf) determined that 8 samples per group would achieve a power of 0.8 with a type I error rate of .05 and an effect size of 0.7.17,19,26,28
The biomechanical data were analyzed using analysis of variance (ANOVA) to compare the mean failure loads of multiple groups. Pairwise tests report Tukey-adjusted P values. Means, standard deviations, and P values are all reported. Statistical significance was set at P < .05.
Results
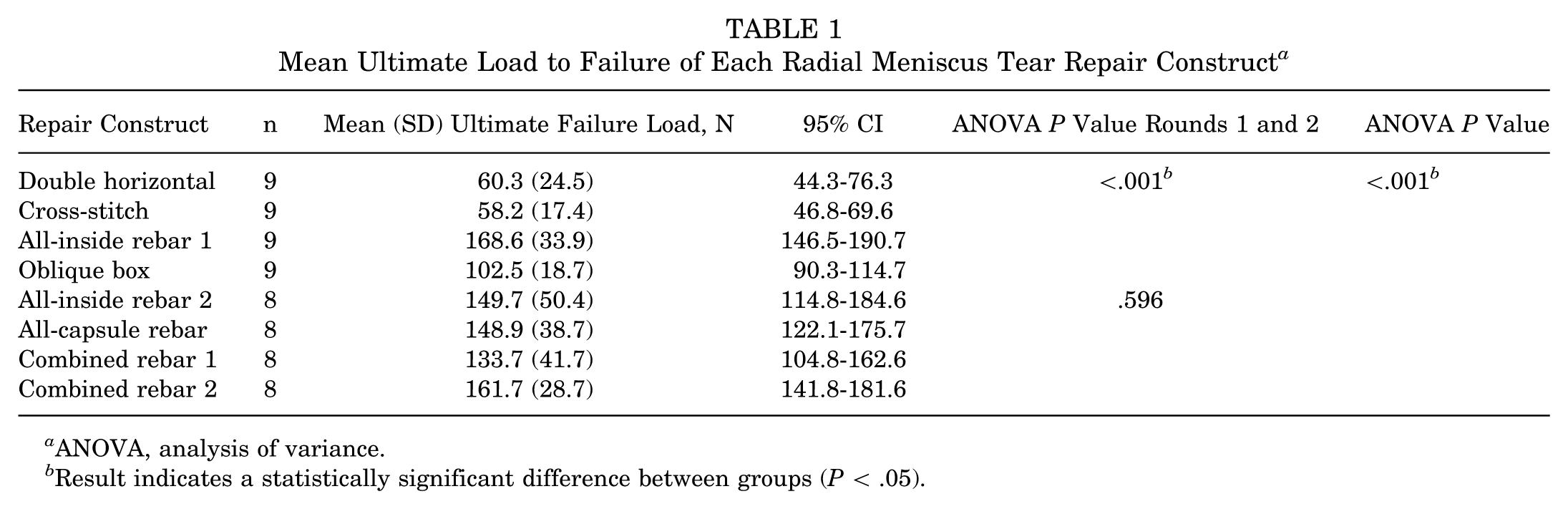
A total of 68 cadaveric lateral menisci were tested in 2 rounds. The first round consisted of 4 groups of 9, and the second round consisted of 4 groups of 8. Ultimate failure load was the primary outcome measure collected and analyzed. Mean ultimate failure load for each group is shown in Table 1.
Mean Ultimate Load to Failure of Each Radial Meniscus Tear Repair Construct a
ANOVA, analysis of variance.
Result indicates a statistically significant difference between groups (P < .05).
For the first round, we tested 4 all-inside rebar and nonrebar constructs. The mean ultimate failure load was 60.3 N (95% CI, 44.3-76.3 N) for DH, 58.2 N (95% CI, 46.8-69.6 N) for CS, 168.6 N (95% CI, 146.5-190.7 N) for AIR1, and 102.5 N (95% CI, 90.3-114.7 N) for OB.
For the second round, we tested 4 rebar constructs using different suture techniques, including all-inside, all-capsule, and combined approaches. The mean ultimate failure load was 149.7 N (95% CI, 114.8-184.6 N) for AIR2, 148.9 N (95% CI, 122.1-175.7 N) for ACR, 133.7 N (95% CI, 104.8-162.6 N) for CR1, and 161.7 N (95% CI, 141.8-181.6 N) for CR2.
For the first round, there was a statistically significant difference in mean ultimate load to failure among the 4 constructs, with P < .001. The AIR1 construct performed significantly better than each of the other constructs when compared individually.
For the second round, ANOVA testing showed no significant difference among the 4 rebar repair constructs, with P = .596.
When all 8 constructs are compared together, there is a significant difference among the groups, with an ANOVA P < .001. This is visually represented in a box-and-whiskers plot (Figure 6).
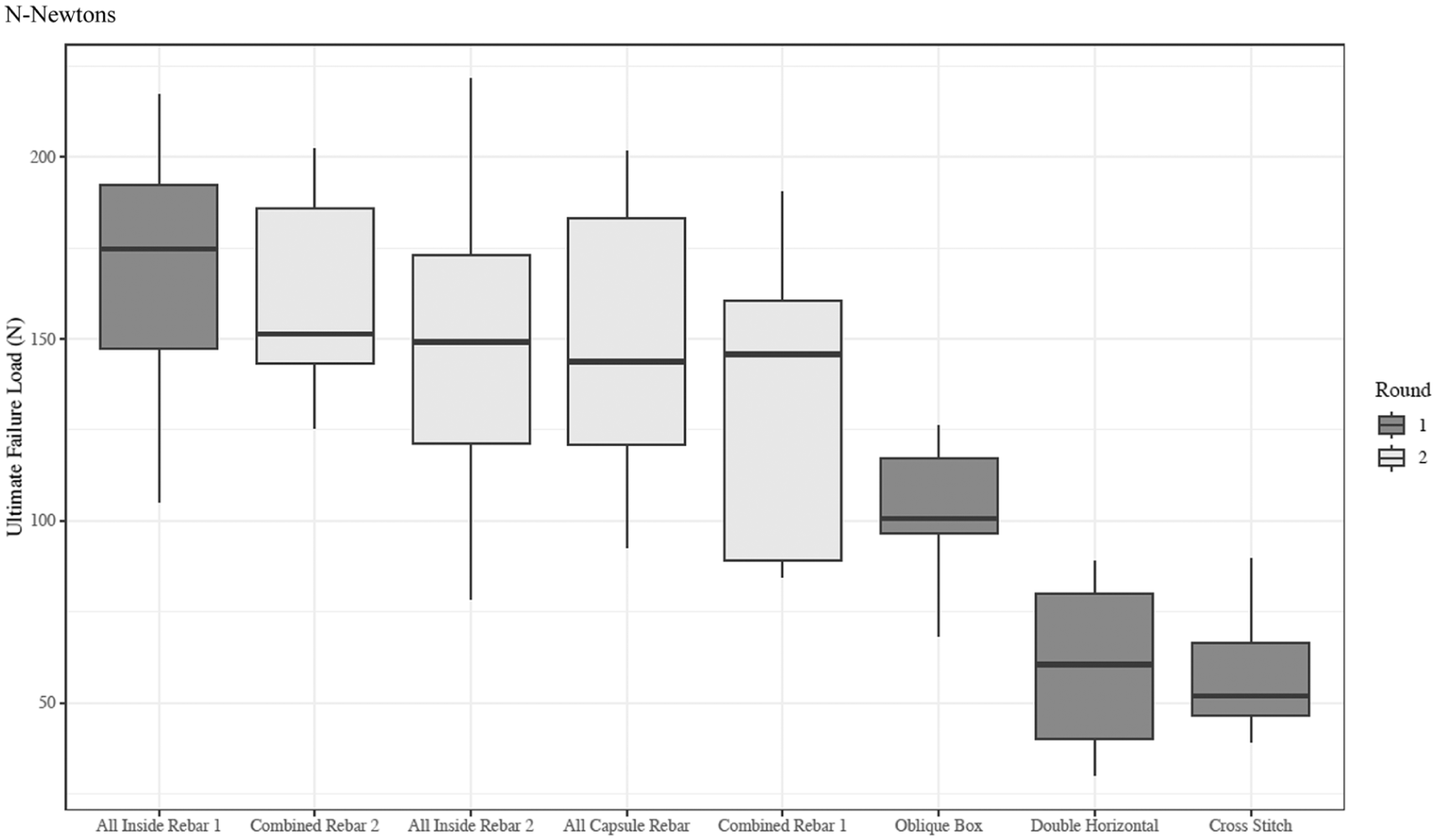
Mean ultimate failure load by suture repair construct for radial meniscus tears, box-and-whiskers plot.
Pairwise testing with Tukey adjusted P values can be seen in Table 2. Each of the rebar-reinforced constructs (AIR1, AIR2, ACR, CR1, CR2) has a statistically significantly higher load to failure when compared with the nonrebar DH or CS constructs. The experimental OB construct failed at a lower load than the AIR1 and CR2 constructs and did not have a statistically significant difference when compared with DH, CS, AIR2, ACR, and CR1.
Pairwise Testing of Each Radial Meniscus Tear Repair Construct Using Tukey Adjusted P Values
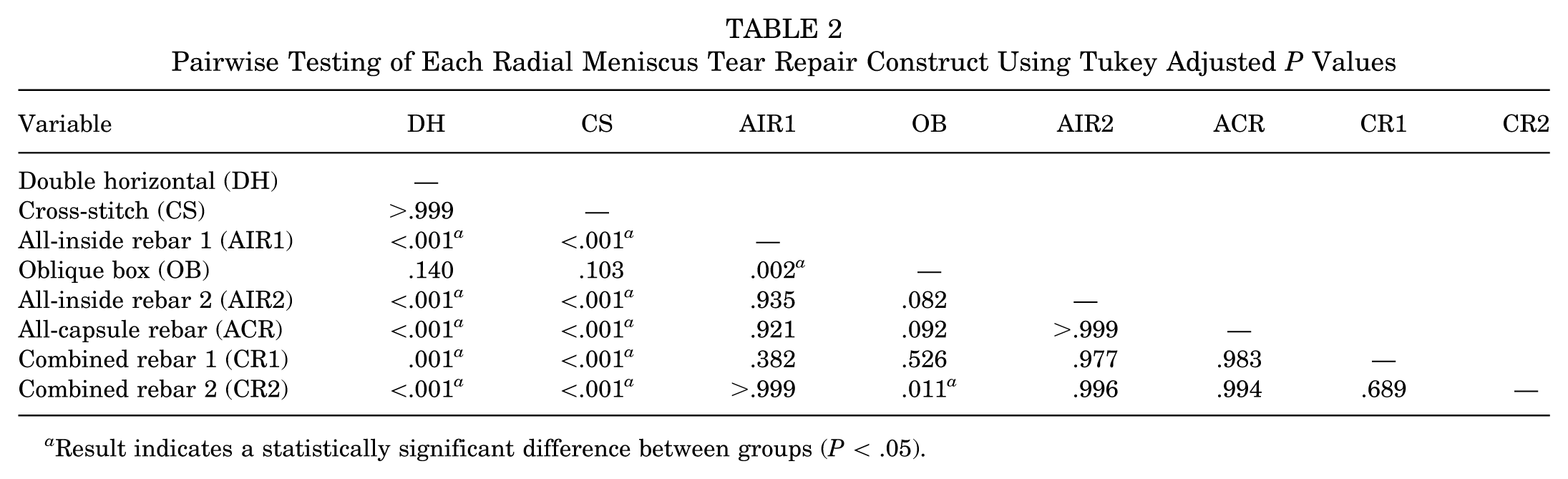
Result indicates a statistically significant difference between groups (P < .05).
The mode of failure was observed and recorded qualitatively for each sample. It was similar for all samples, with a cutout or “cheesewiring” of the horizontal sutures. For the rebar-reinforced constructs, the cutout or “cheesewiring” was initially caught and delayed by the vertical rebar sutures. The horizontal sutures eventually slipped around the rebar suture and then continued to cut or “cheesewire” through the meniscus until failure (Figure 7).

Example of radial meniscus sample after testing, with eventual failure of repair by cutout or “cheesewiring” of the suture.
Discussion
The major findings of our research demonstrated high ultimate failure loads for all rebar-reinforced suture constructs (AIR1, 168.6 ± 33.9 N; AIR2, 149.7 ± 50.4 N; ACR, 148.9 ± 38.7 N; CR1, 133.7 ± 41.7 N; CR2, 161.7 ± 28.7 N), with no significant difference among the rebar-reinforced constructs (P = .596) and a significant difference when compared with the nonrebar constructs (DH, 60.3 ± 24.5 N; CS, 58.2 ± 17.4 N; P < .001). The OB construct with trapezoid-shaped reinforcing sutures effectively has rebar sutures only on the inferior surface; it had mixed results and did not perform significantly better than the nonrebar constructs. This pattern would also be more difficult to re-create arthroscopically. Therefore, we do not believe this construct is worth further study or discussion.
Our study compares the biomechanical strength of different suture repair constructs for lateral meniscus radial tear repair. While previous studies have demonstrated the efficacy of rebar reinforcement in meniscus repairs, none have directly compared different combinations of all-inside and inside-out techniques. Using human adult cadaveric lateral menisci, we found that all 4 rebar-reinforced repair constructs demonstrated significantly higher ultimate failure loads than repairs without rebar. Importantly, we found that all variations of our all-inside and inside-out rebar-reinforced repairs had a higher load to failure than nonreinforced repairs, giving surgeons flexibility of technique intraoperatively. Our results suggest that the presence of the rebar stitch perpendicular to the circumferential collagen fibers, rather than the technique used to secure it, is the critical factor in improving strength.
Because of the historically variable rates of healing when radial meniscus tears are treated with either all-inside or inside-out horizontal mattress repairs, in 2012, Nakata et al 24 introduced the concept of a rebar or ripstop inside-out technique.8,22 This technique concept has gained popularity over the past decade and has been studied by multiple groups. Our findings align with previous studies demonstrating the superiority of rebar-reinforced repairs over nonreinforced repairs using inside-out techniques. Nakanishi et al 23 found that ultimate failure loads of 2 rebar-reinforced “tie-grip” (145.2 ± 39.1 N) and “cross tie-grip sutures” (154.9 ± 29.0 N) constructs were significantly greater than nonreinforced double horizontal and cross patterns in porcine cadaveric menisci. The authors also found that displacement after 500 loading cycles was significantly smaller in the rebar “cross tie-grip” group than in all other configurations. 23 In a study of medial and lateral human cadaveric meniscus, Massey et al 17 found that the load to failure of their rebar-reinforced repair (124.1 N ± 27) was significantly higher than without rebar (horizontal mattress 85.5 ± 22.0 N and cross-stitch 76.2 ± 28.8 N). The authors also noted that 50% of their rebar-reinforced specimens failed via suture breakage, compared with suture cut-through that occurred in 100% of the nonreinforced constructs. 17 In another human cadaveric study of radial medial meniscus tears, Buckley et al 6 compared horizontal mattress repair augmented with rebar sutures, a transtibial cortical suspension augmentation, or both; while the authors found no statistical difference, both rebar-reinforced constructs had higher average loads to failure (349 N and 365 N) than the nonreinforced group (259 N).
Stender et al 26 also studied variations on rebar-reinforced constructs using inside-out techniques in human cadaveric lateral menisci, with 2 rebar-reinforced constructs (hashtag and crosstag) to a crisscross horizontal mattress (cross-suture). In contrast to our results, the authors found that load to failure of the rebar-reinforced technique was statistically similar (hashtag, 86.08 ± 23.58 N) or lower (crosstag, 62.50 ± 12.15 N) compared with the crisscross horizontal mattress (cross-suture, 81.43 ± 14.31 N). 26 However, cyclic loading demonstrated smaller displacement of the rebar constructs compared with the nonreinforced (2.42 ± 1.13 mm and 3.13 ± 1.77 mm vs 4.78 ± 1.65 mm), and the authors noted that cyclic loading may more closely resemble in vivo forces on meniscus repair. 11
In studying all-inside techniques, Branch et al 5 evaluated a rebar-reinforced (“Mason-Allen”) construct in human cadavers compared with all-inside and inside-out horizontal mattress patterns. The authors found that their rebar-reinforced technique had a load to failure of 86 ± 19 N, greater than inside-out horizontal mattresses (64 ± 20 N) but not all-inside horizontal mattresses (75 ± 16 N).
While there has been some variability in the reported biomechanical strength of all-inside and inside-out rebar-reinforced meniscus repairs, our results speak to the importance of the rebar stitch in improving radial meniscus repair strength. Rebar reinforcement may biomechanically enhance repair strength by distributing forces more evenly across the meniscus repair and reducing stress concentration at the horizontal mattress suture holes. 17
Clinically, several authors have reported encouraging outcomes in patients using inside-out rebar constructs. Nakata et al 24 reported on a case series of 27 patients who underwent rebar-reinforced medial or lateral meniscus repair. At a mean follow-up of 18 months, 89% of patients reported being free from symptoms in the tibiofemoral joint; second-look arthroscopy at a mean 9-month follow-up found that 93% of the meniscal tears were completely or partially healed. A series of 17 patients treated with rebar repair augmented with bone marrow aspirate concentrate found that all patients, at an average 36.3-month follow-up, had significant improvements in pain, Lysholm, and International Knee Documentation Committee scores and returned to their preinjury level of activity. 18 In a series of 18 patients who underwent rebar repairs of the lateral meniscus, Tsujii et al 27 performed a second-look arthroscopy and found 61% of patients had achieved complete or partial healing 6 months postoperatively. We are unaware of clinical outcome studies on patients undergoing all-inside rebar-reinforced repairs.
Limitations
Limitations include the use of cadaveric tissue, the controlled laboratory setting, and the absence of in vivo loading conditions. As is typical with cadaveric studies, any freeze-thaw cycle may alter the structural integrity of the meniscus compared with living tissue. However, we attempted to minimize this factor by subjecting our meniscus sample to only 1 freeze-thaw cycle. For our repairs, we isolated the menisci and used straight needles to perform repairs with digital calipers. This improved the consistency between our samples but did not replicate the difficulty surgeons face when performing these repairs arthroscopically. Importantly, the location of the meniscus tear in vivo may also make arthroscopic repair more difficult, particularly when they occur anterior to the popliteal hiatus. Our biomechanical testing apparatus is designed to test a pure distraction force across the repair at time zero. In patients, meniscus repairs encounter shear and compressive forces that are influenced by any healing that has taken place postoperatively, conditions that are challenging to re-create in a laboratory setting. 17 Finally, the placement of suture knots on the superior surface of the meniscus raises the theoretical possibility of articular cartilage abrasion, although this has not been observed clinically. 10
Conclusion
Our cadaver study demonstrated that in a model of lateral meniscus radial tear repair, constructs using rebar sutures parallel to the tear provided reinforcement for the sutures spanning the tear and resulted in a higher load to failure than nonrebar suture repair constructs. Surgeons should consider adding parallel rebar sutures to their suture constructs when repairing radial tears arthroscopically to improve the strength of the construct, with flexibility to use different techniques.
Footnotes
Final revision submitted August 8, 2025; accepted September 8, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: AlloSource donated cadaveric tissue used in the study. This organization had no other involvement in the study. M.T. is a board or committee member of the American Orthopaedic Society for Sports Medicine, Pediatric Research in Sports Medicine Society, and ROCK; is a consultant for Abys Medical; and has received other financial or material support from AlloSource–ROCK Group and Vericel–ROCK Group. H.E. has received speaking fees from OrthoPediatrics; has received grants from the Pediatric Orthopaedic Society of North America, Department of Defense, Washington University in St Louis, and American Orthopaedic Society for Sports Medicine; is a board member of American Academy of Orthopaedic Surgeons and Pediatric Research in Sports Medicine (PRiSM); has received nonfinancial support from Arthrex and Pylant Medical; and is a board or committee member of TOA, PRiSM, and American Academy of Orthopaedic Surgeons. Y.-M.Y. is a paid consultant for Smith & Nephew. T.G. is a board or committee member of the American Academy of Orthopaedic Surgeons, American Academy of Pediatrics Section on Orthopaedics, International Pediatric Orthopaedic Symposium, Pediatric Orthopaedic Society of North America, PRISM Pediatric Research in Sports Medicine, and Research on Osteochondritis Dissecans of the Knee; has received research support from AlloSource and Vericel; and is an associate editor for the American Journal of Sports Medicine. S.L.S. is a board or committee member of AANA, American Academy of Orthopaedic Surgeons, ACLSG, American Orthopaedic Society for Sports Medicine, Biologic Association, ICRS, and ISAKOS; is a member of the AO Sports Medicine Taskforce; is a paid educational consultant for Arthrex, Kinamed, and LifeNet; is a paid advisory board member for Ostesys, Reparel, Sarcio, Sparta Medical, Vericel, and Vivorte; is a member of the design team and receives royalties from ConMed and DJO; holds stock options with LinkX, Osteosys, Moximed, Sarcio, Reparel, and Vivorte; and receives research support from JRF, Smith & Nephew, Aesculap Biologics LLC, University of Pittsburg, Miach Orthopaedics, and Organogenesis. K.G.S. is a board or committee member of the American Academy of Orthopaedic Surgeons, American Orthopaedic Society for Sports Medicine, Pediatric Orthopaedic Society of North America, PRISM Pediatric Research Sports Medicine, and ROCK–Research for Osteochondritis Dissecans of the Knee; holds stock or stock options with nView and Sarcio; is an unpaid consultant for nView and Sarcio; and has received research support from Ossur and Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
