Abstract
Background:
Recent studies have used the anterior cruciate ligament reconstruction (ACLR) mouse model to study the tendon-bone healing process. Although mechanical load has an important effect on ACL graft healing, only a limited number of studies have investigated the effect of treadmill exercise.
Purpose/Hypothesis:
This study aimed to investigate the effects of different mechanical loading stimuli on the tendon-bone interface (TBI) healing process after ACLR in mice. It was hypothesized that moderate loading would positively affect TBI healing after ACLR.
Study Design:
Controlled laboratory study.
Methods:
A total of 58 C57BL/6 male mice (12 weeks old) underwent ACLR of the right knee. The first 30 mice were assigned to 5 exercise groups (n = 6 per group) to evaluate the different exercise regimens (study arm 1). After the optimal exercise regimen was identified, an additional 28 mice were included, and 14 mice in the exercise regimen were compared with 14 free cage controls (study arm 2). Starting 7 days after surgery, treadmill exercise was performed 5 days per week for 3 weeks. All animals were euthanized 4 weeks after surgery. Microfocus computed tomography (μCT) imaging, biomechanical testing, and histological tendon–bone tunnel healing (TBTH) scoring were performed. Statistical analyses were performed using the Wilcoxon signed rank test, the Mann-Whitney U test, and 1-way analysis of variance with a post hoc Tukey honestly significant difference test. Statistical significance was defined as P < .05.
Results:
In study arm 1, no significant intergroup differences were observed in the μCT analysis (P = .18-.72) and histological assessment (P = .21). For study arm 2, the 10-m/min, 50-minute treadmill exercise was selected. Further μCT and biomechanical testing in study arm 2 revealed no significant intergroup differences. However, histological evaluation revealed significantly higher TBTH scores in the treadmill group as compared with the free cage activity group. This difference was most pronounced in the extra-articular region of the femoral tunnel (mean ± SD, 4.28 ± 2.15 vs 8.18 ± 2.98; P = .027) and the intra-articular region of the tibial tunnel (7.17 ± 7.07 vs 16.62 ± 5.42; P = .027).
Conclusion:
Our study demonstrated that moderate treadmill exercise after ACLR positively affects TBI healing in the femoral and tibial tunnels in the murine model. Future studies are necessary to objectively quantify the optimal type, magnitude, and duration of loading.
Clinical Relevance:
There may be a role for a moderate-load rehabilitation protocol to improve healing of the tendon graft–bone interface in patients after ACLR.
Keywords
Graft failure after anterior cruciate ligament reconstruction (ACLR) remains a significant clinical issue. According to a systematic review containing 8 studies that included >10,000 patients each, the failure rates range from 2.1% to 3.6%. 29 The function of an ACLR graft is critically dependent on the secure healing of the tendon graft in a bone tunnel, and biological healing at the tendon-bone interface (TBI) plays a critical role in determining long-term graft survivorship. Therefore, optimizing the early healing environment may reduce the risk of graft failure. Recent studies have used a mouse model of ACLR to study the tendon-bone healing process and the development of posttraumatic osteoarthritis.5,13,18 Mice have several advantages for studying the molecular mechanisms underlying physiologic and pathologic conditions, including anatomic similarity to humans, 6 rapid progress in mouse genomics,2,14,22,26 and the availability of transgenic and knockout mice. 8 In addition, mouse models provide time- and cost-effectiveness, ease of care and physical handling, and disease assessment in translational osteoarthritis studies.10,21
Several biological and biomechanical variables have important effects on ACL graft-to-bone healing. Although mechanical load is known to have an important effect on anterior cruciate ligament (ACL) graft healing, 4 only a limited number of studies have investigated the effect of running activity on ACLR graft healing.19,20,25,27,28 Previous studies have provided preliminary information on the effects of treadmill running on mouse knees. Oka et al19,20 examined a treadmill speed of 18 m/min for 30 min/d, 3 d/wk, expecting it to be within a comfortable range. This expectation was based on the fact that studies in mice typically do not involve treadmill use but instead use a rotating cylinder, where mice typically perform active exercise at a rate of 15 to 25 m/min. This protocol was reported to be beneficial for cartilage in normal knees but harmful to cartilage in ACL-transected knees.19,20 Using a treadmill at 13 m/min for 50 min/d, 5 d/wk, has been reported to have beneficial effects on growth factors and tendon progenitor cells, whereas using a treadmill for 4 h/d under the same conditions was found to be harmful to tendon progenitor cell differentiation.27,28 These studies suggest that moderate treadmill use may maintain tendon homeostasis by promoting the differentiation of tendon progenitor cells into tenocytes, whereas intense treadmill use may induce nontenocyte differentiation of tenocytes.
Treadmill exercise can promote histological and biochemical changes that benefit the joints of rats with osteoarthritis. Saito et al 23 examined the effects of running exercise on articular cartilage in Wistar rats; however, few studies have reported its beneficial effects.7,9 For example, improved preservation of proteoglycan content in the superficial and intermediate regions of joint cartilage was observed with a protocol of 13 m/min for 50 min/d, 3 d/wk, for 60 days. 7 Calibrated slight or moderate exercise exerted a beneficial influence on the severity of chondral lesions in rats that had undergone ACL transection (osteoarthritis model, 18 m/min for 15 or 30 min/d, 5 days/wk, for 28 days). Conversely, higher levels of loading (the same protocol at 60 min/d) abolished this chondroprotective effect. 9 Treadmill exercise in rats has been shown to effectively maintain the integrity of the cartilage–subchondral bone unit. Exercise-induced modifications of posttraumatic osteoarthritis-related gut microbial shifts may be involved in the exercise-induced amelioration of posttraumatic osteoarthritis. 11
Although previous studies have investigated the effects of treadmill exercise on osteoarthritis models in rats and mice, no studies have examined the effects of different exercise regimens on ACLR graft healing in mice. In this study, we explored the effects of different mechanical loading stimuli on the TBI healing process after ACLR in mice. We aimed to identify loading protocols that are beneficial for promoting TBI healing in bone tunnels. We hypothesized that moderate loading positively affects TBI healing after ACLR.
Methods
Study Overview
The study protocol was approved by the Institutional Animal Care and Use Committee (2020-0024). A total of 58 C57BL/6 male mice (12 weeks old) underwent ACLR of the right knee as previously described.5,15 The first 30 mice were assigned to 5 exercise groups (6 per group) to evaluate the different exercise regimens (study arm 1; Figure 1A). Based on the results of this study and previous reports,20,28 the optimal exercise regimen was identified. Subsequently, an additional 28 mice were included in the next experimental group (study arm 2; Figure 1B).
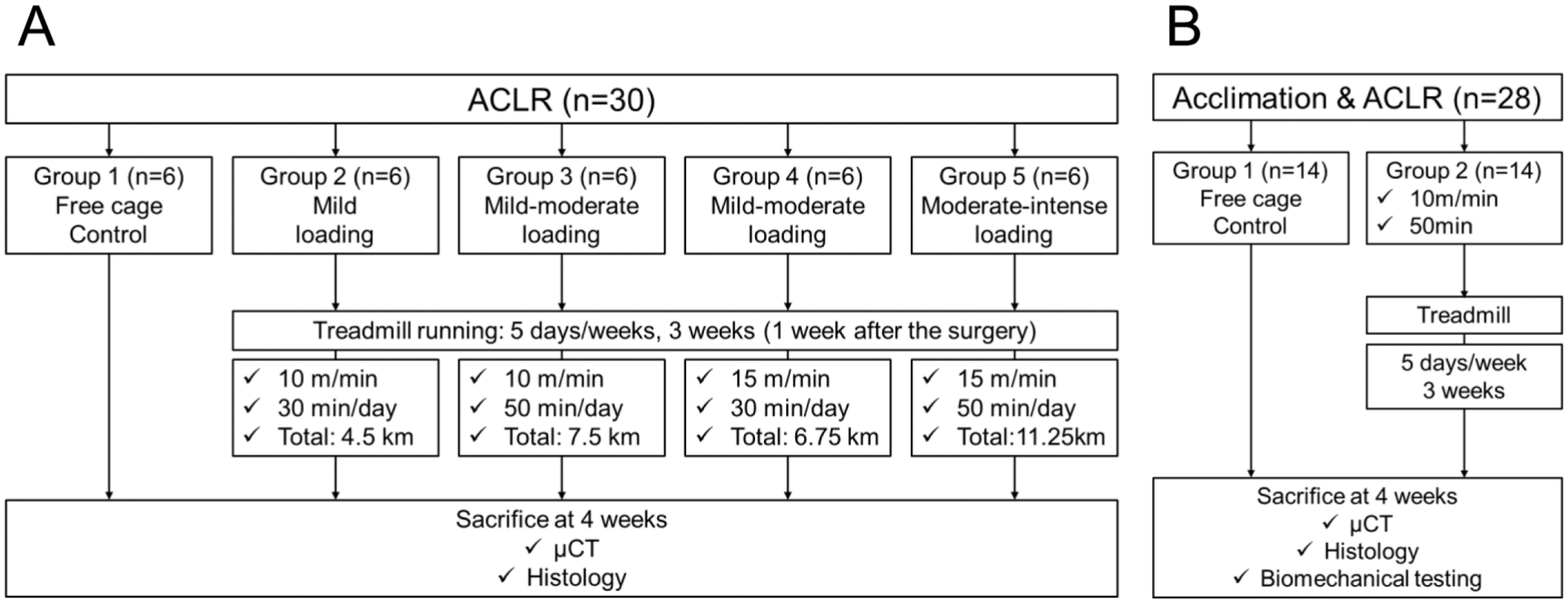
In vivo study design, including the number of animals per group and their outcome measurements: (A) flowchart of arm 1 and (B) flowchart of arm 2. ACLR, anterior cruciate ligament reconstruction; μCT, microfocus computed tomography.
Before surgery, all mice were acclimated to treadmill running based on a 5-day acclimation protocol (Table 1).
Acclimation Protocol of the Treadmill Exercise

The mice were allowed free activity in the cage after each procedure. Seven days after ACLR surgery, treadmill exercises were performed 5 d/wk for 3 weeks. All animals were euthanized 4 weeks after surgery. Animals in arm 1 were evaluated by microfocus computed tomography (μCT) and histology (sagittal slices of the entire tibia). Arm 2 mice were evaluated by μCT, histology (axial slices of the femoral and tibial intra- and extra-articular parts), and biomechanical testing (Figure 1).
Surgical Procedure
ACLR was performed as previously described. 5 Under general anesthesia consisting of 2%-3% isoflurane inhalation with oxygen at a flow rate of 2 to 3 L/min, the mice were placed in the supine position on a custom-made table. The ipsilateral flexor digitorum longus tendon was procured as an autograft with 6-0 Prolene (Ethicon) applied to the proximal end and with a microvascular clip applied to the distal end. A standard medial parapatellar arthrotomy was performed to access the knee joint. The femoral and tibial tunnels were first drilled with a 23-gauge needle (0.64 mm), after which the graft was passed through both tunnels. The clip provided extracortical fixation on the femoral side. Tibial fixation relied on a transosseous 4-0 Ethibond suture (Ethicon) being passed through the anterior aspect of the tibia and tied over the graft while the graft was tensioned to 5 N with the knee extended, using a 5-N weight and a stand with a pulley. Postoperatively, the animals received buprenorphine for pain management, and comfort and recovery were assessed multiple times per day for at least 72 hours.
Radiographic Imaging
μCT scanning and evaluation were performed using a μCT 45 system (Scanco Medical AG) per the following parameters: 12.2-μm voxel size, 55 kVp, 0.5-mm aluminum filter, 1200-mgHA/cm3 beam hardening correction, 25.2-mm field of view/diameter, and 400-ms integration time. The Scanco μCT software (Integrity Server, 64 bit; HP) was used for image viewing, analysis, and thresholding. The bone tunnel was identified; a region of interest was defined as the bone tunnel diameter (0.646 mm; specified by the voxel size); and bone volume fraction (bone volume/total volume [BV/TV]), bone mineral density, and trabecular number/thickness were evaluated in the tibial tunnel. The entire tunnel was individually measured on the μCT images. The tunnel was then divided into 3 equal-length subregions: extra-articular, middle, and intra-articular.
Histology
Histological assessment was performed on tissues fixed in 10% formalin (Sigma-Aldrich) for 48 hours, decalcified by Immunocal (StatLab), dehydrated, and embedded in paraffin. Subsequently, 6-μm-thick sagittal or axial sections were stained with hematoxylin and eosin. All sections were stained simultaneously under standard conditions to control the consistency of staining intensity. Images were obtained using an Axioscan 7 slide scanner (ZEISS).
TBI healing was evaluated per a well-established histological scoring system. 16 The tendon–bone tunnel healing (TBTH) scoring system comprises 5 items on graft status, fiber type, and interface connectivity, evaluated on a 5- or 6-point scale. These items included (1) graft degeneration, (2) graft remodeling, (3) percentage of fibrous tissue between the tendon and bone, (4) percentage of collateral connections between the tendon and bone, and (5) percentage of head-to-head connections between the tendon and bone. In addition to brightfield light microscopy, polarized microscopy was used to evaluate the interface connectivity (Nos. 4 and 5). The tunnel was divided into extra- and intra-articular subregions by the ACLR surgeon (Y.O.), as illustrated in Figure 2. Meticulous attention is required when the procedure is performed because of the extremely small size of both the femur and tibia and the short length of their bone tunnels. After decalcification and hydration, the following steps were performed: (1) the femur and tibia were separated; (2) the specimen was cut along line 1, which is perpendicular to the estimated (created) bone tunnel shown in Figure 2; (3) a cut was made along line 2, followed by line 3. Subsequently, paraffin embedding was performed so that histological sections could be prepared from the surface of lines 1 and 2 but not line 3. Sectioning continued until a slice was obtained in which the entire circumference of the graft was surrounded by bone (allowing for TBTH scoring), after which staining was performed. Three blinded investigators (Y.O., Y.S., L.A.S.) quantified TBI healing in the tunnel using the TBTH score, and the 3 scores were then averaged. The interrater reliability for study arm 1 was moderate (0.71), while that for study arm 2 ranged from moderate to excellent (femoral tunnel, 0.85 for extra-articular region and 0.85 for intra-articular region; tibial tunnel, 0.95 for extra-articular region and 0.99 for intra-articular region).

Preparation of histological sections corresponding to the extra- and intra-articular subregions of the (A) femur and (B) tibia. The specimen was first cut along line 1, which is perpendicular to the estimated bone tunnel, followed by cuts along lines 2 and 3. Paraffin embedding was then performed so that histological sections could be prepared from the surfaces of lines 1 and 2 but not line 3. Arrows indicate the cutting directions for histological sectioning. Black dotted line, estimated bone tunnel; green solid outline, femoral articular surface; blue dashed line, tibial plateau.
Biomechanical Testing
Biomechanical testing was performed with a materials testing machine (ELF 3200; EnduraTEC). The knee joints were carefully dissected to isolate the ACL grafts. Customized fixtures were designed to ensure that the ACL was aligned along the loading direction. The specimens were subjected to load until failure at a rate of 5.0 mm/min. Data were recorded for load to failure (newtons) and displacement (millimeters), and the stiffness was calculated from the slope of the load-displacement curve. Before the experiment, 18 murine cadaveric knees were used to determine the optimal speed for biomechanical testing.
Statistical Analyses
Data are reported as mean and standard deviation. Statistical analyses were performed in EZR software (Saitama Medical Center, Jichi Medical University) 12 and Prism 6 software (GraphPad Software Inc). Intra- and intergroup differences were evaluated by the Wilcoxon signed rank test and the Mann-Whitney U test, respectively. Differences among >2 groups were evaluated by 1-way analysis of variance with a post hoc Tukey honestly significant difference test. Statistical significance was defined as P < .05. We performed an a priori power analysis, which suggested that 11 subjects per group would be required to achieve 80% power for histological analysis (free cage control group vs 10-m/min, 50-minute treadmill exercise group). However, due to ethical and practical constraints, we used 6 per group, which yields a power of 0.561 and 0.556 for the extra-articular part of the femur and the intra-articular part of the tibia.
Results
Arm 1: Evaluation of Different Exercise Regimens
No mice in arm 1 were euthanized before the estimated date. No significant differences were observed in the μCT analysis (entire tibia and each of the 3 parts: extra-articular, middle, and intra-articular; P = .18-.72; Figure 3) or the histological assessment (sagittal slices of the entire tibia, P = .21; Figure 4) among the groups (BV/TV, bone mineral density, and trabecular number/thickness in μCT analysis). Although the sample size was limited, the most relevant trend was observed in the 10-m/min, 50-minute treadmill group (7.5-km total distance) and 15-m/min, 30-minute treadmill group (6.7-km total distance) as compared with the free cage activity group.

Microfocus computed tomography analysis of bone tunnel. (A) Bone volume fraction (bone volume/total volume [BV/TV]) of the entire tibia. No significant differences were observed among the groups. (B) BV/TV in each of the 3 parts of the tibia. No significant differences were observed among the groups. Data are presented as mean (SD).

Tendon–bone tunnel healing score of the sagittal slices in the entire tibia. No significant differences were observed among the groups. Data are presented as mean (SD).
We proceeded with the following experiment based on the 10-m/min, 50-minute treadmill group, as this distance closely aligns with previous studies (13 km, 20 10 km 28 ) and because BV/TV in the intra-articular part was relatively higher as compared with the 15-m/min, 30-minute treadmill group.
Arm 2: Free Cage Control Group vs 10-m/min, 50-Minute Treadmill Exercise Group
No mice in arm 2 were euthanized before the estimated date. There were no significant intergroup differences in the μCT measurements of BV/TV, bone mineral density, and trabecular number/thickness (Figure 5) and no significant differences in failure load and stiffness on biomechanical testing (Figure 6). In the free cage group, there were 7 cases of femoral-side pullout failure and 1 case of midsubstance failure. In the treadmill group, there were 4 cases of femoral-side pullout failure, 2 cases of tibial-side pullout failure, and 2 cases of midsubstance failure.
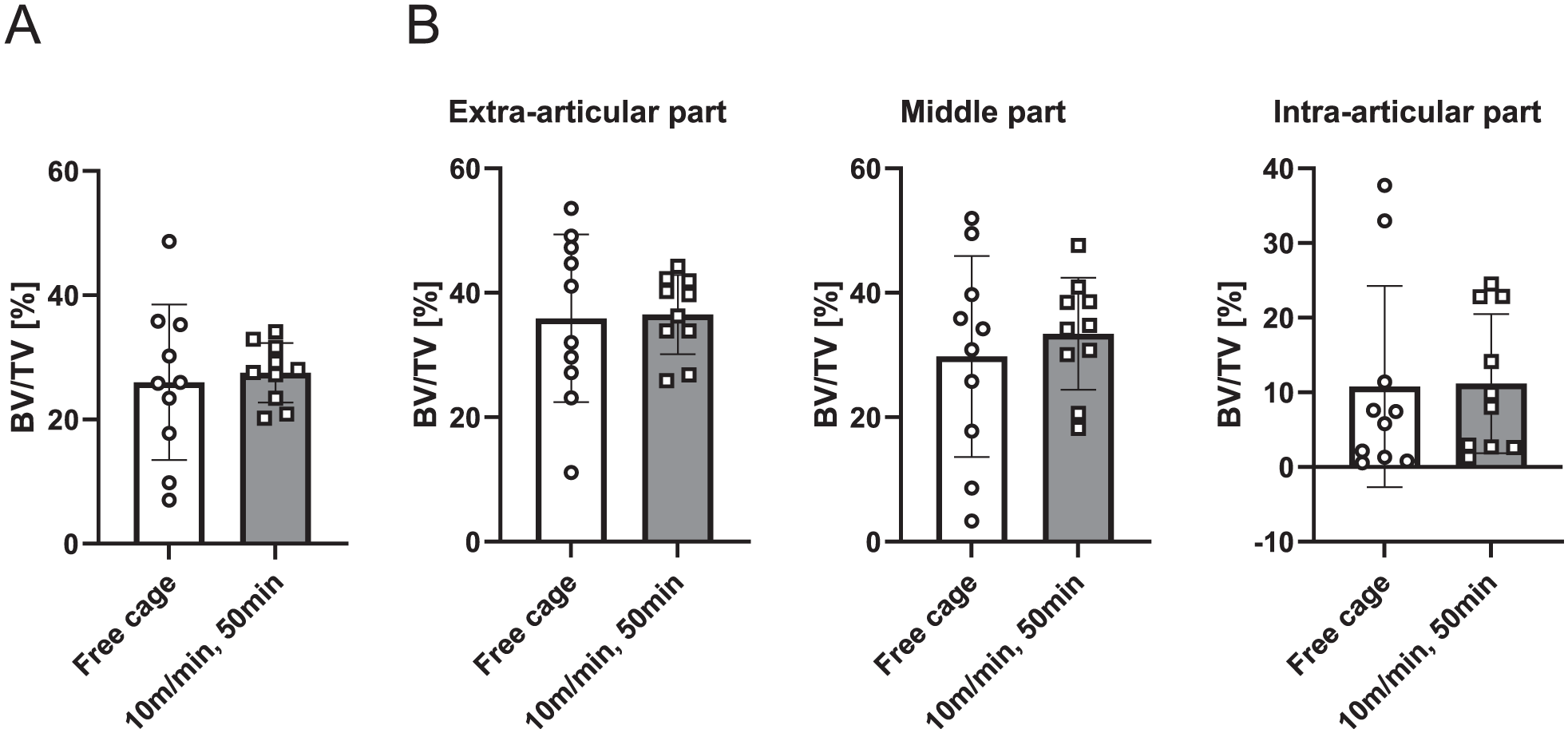
Microfocus computed tomography analysis of bone tunnel. (A) Bone volume fraction (bone volume/total volume [BV/TV]) of the entire tibia. No significant differences were observed among the groups. (B) BV/TV in each of the 3 parts of the tibia. No significant differences were observed among the groups. Data are presented as mean (SD).
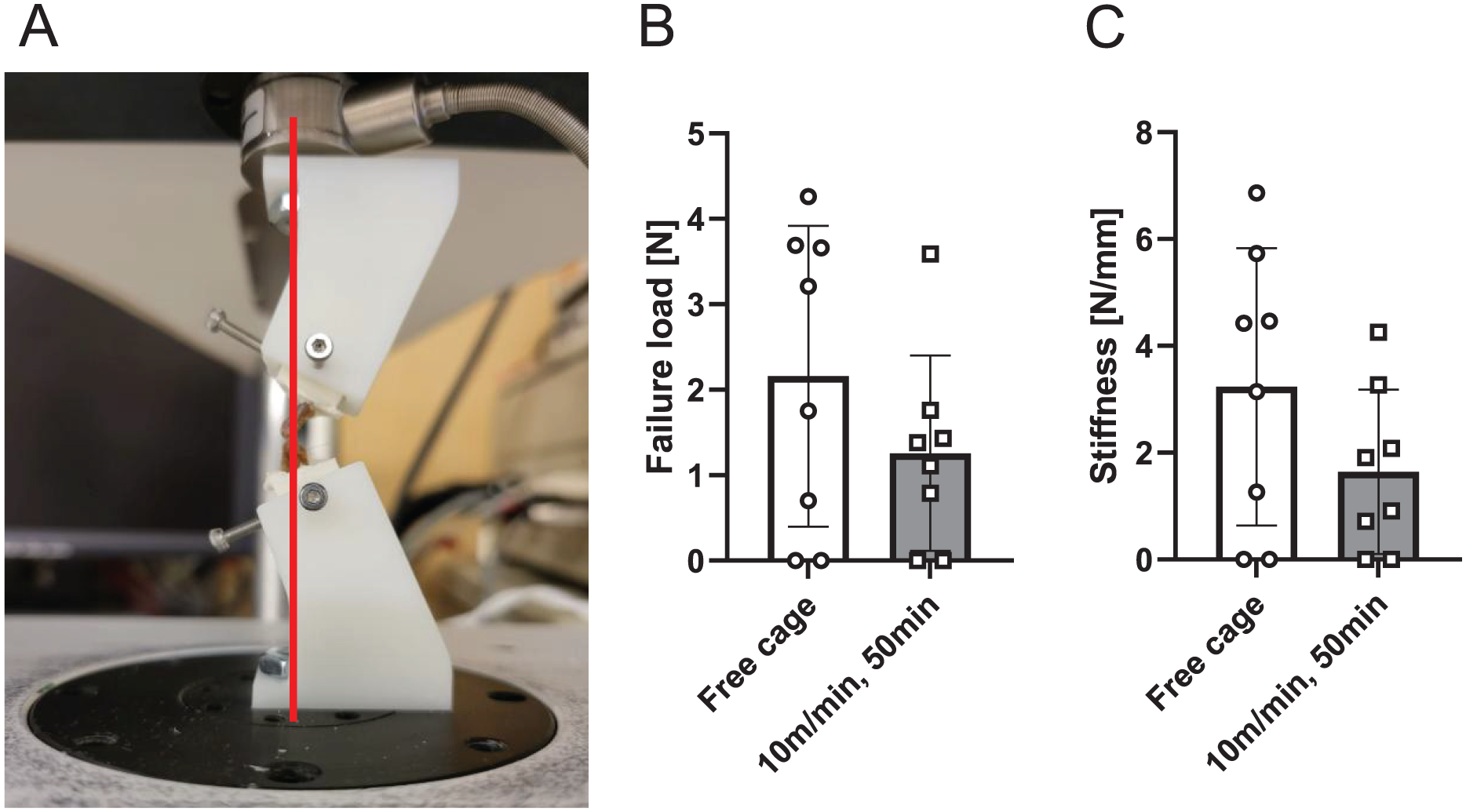
Biomechanical testing. (A) Biomechanical test setting: the loading axis is indicated by the red line. (B) Failure load of the tendon graft. No significant differences were observed among the groups. (C) Stiffness of the tendon graft. No significant differences were observed among the groups. Data are presented as mean (SD).
However, histological evaluation of axial sections revealed significantly higher TBTH scores in the treadmill group as compared with the free cage activity group (Figure 7A). This difference was most pronounced in the extra-articular region of the femoral tunnel (4.28 ± 2.15 vs 8.18 ± 2.98; P = .027) and the intra-articular part of the tibial tunnel (7.17 ± 7.07 vs 16.62 ± 5.42; P = .027). In the free cage group (Figure 7B), significantly higher TBTH scores were observed in the extra-articular part of the tibia than in the extra-articular part of the femur (4.28 ± 2.15 vs 17.17 ± 6.74; P = .0012) and the intra-articular part of the tibia (17.17 ± 6.74 vs 7.17 ± 7.07; P = .012). Additionally, the treadmill group (Figure 7C) had a higher TBTH score in the tibia than in the femur in the extra-articular part (8.18 ± 2.98 vs 19.50 ± 1.52; P < .0001) and intra-articular part (6.45 ± 3.18 vs 16.62 ± 5.42; P < .001). Representative histological findings are shown in Figure 7D.
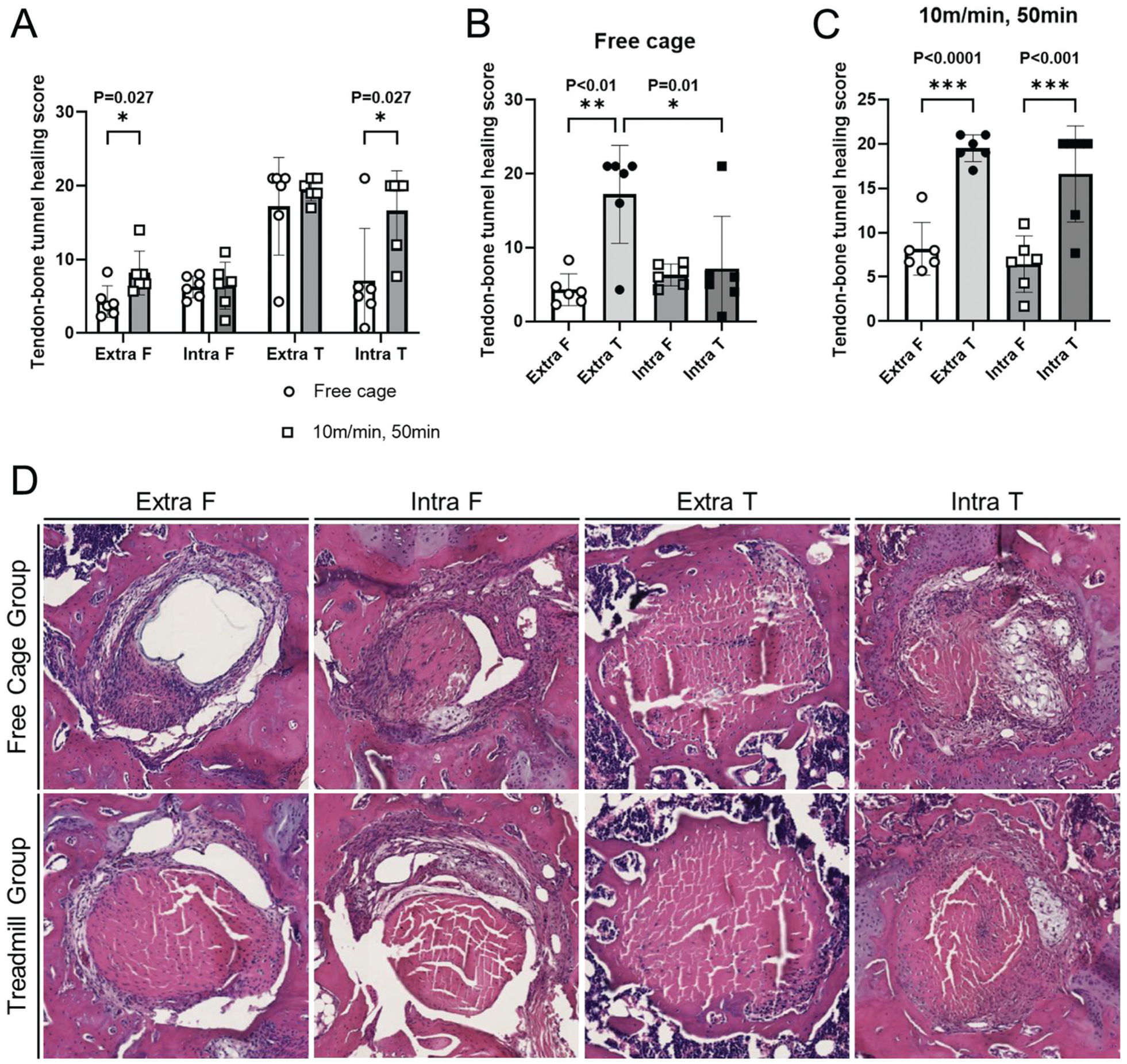
Tendon–bone tunnel healing (TBTH) score of the femur and tibia. (A) Comparison between free cage control and 10-m/min, 50-minute treadmill exercise groups. Significant differences were observed in the extra-articular region of the femoral tunnel (4.28 ± 2.15 vs 8.18 ± 2.98; P = .027) and the intra-articular part of the tibial tunnel (7.17 ± 7.07 vs 16.62 ± 5.42; P = .027). (B) Comparison within the free cage control group. The TBTH scores were significantly higher in the extra-articular part of the tibia than in the extra-articular part of the femur (4.28 ± 2.15 vs 17.17 ± 6.74; P = .0012) and in the intra-articular part of the tibia (17.17 ± 6.74 vs 7.17 ± 7.07; P = .012). (C) Comparison within the 10-m/min, 50-minute treadmill exercise group. The TBTH scores were significantly higher in the tibia than in the femur in the extra-articular part (8.18 ± 2.98 vs 19.50 ± 1.52; P < .0001) and intra-articular part (6.45 ± 3.18 vs 16.62 ± 5.42; P < .001). (D) Representative histological findings of the femur and tibia. In the femoral tunnel, a tendon graft–bone connection was not consistently observed. Some specimens in the free cage group showed histological signs of graft degeneration. In the tibial tunnel, part of the tendon graft was completely replaced by new bone ingrowth in the extra-articular part of both groups and the intra-articular part of the 10-m/min, 50-minute treadmill exercise group. Data are presented as mean (SD). extra, extra-articular part; F, femur; intra, intra-articular part; T, tibia.
Discussion
The major findings of our study demonstrated that animals treated with a moderate treadmill exercise protocol after ACLR resulted in better histological findings of graft-to-bone healing in the extra-articular part of the femoral tunnel and the intra-articular part of the tibial tunnel as compared with the free cage control group. The treadmill group had significantly higher TBTH scores than the free cage activity group. This difference was most pronounced in the extra-articular region of the femoral tunnel (P = .027) and the intra-articular part of the tibial tunnel (P = .027). These findings suggest that moderate treadmill exercise after ACLR positively affects TBI healing in the femoral and tibial tunnels, thus confirming our hypothesis. To our knowledge, this is the first study to identify a beneficial treadmill protocol for TBI healing after ACLR in mice.
Within the treadmill group (10 m/min for 50 minutes), the intra- and extra-articular parts of the tibia exhibited a significantly higher TBTH score than the femur. Based on histological analysis of the treadmill group, the healing process began earlier in the tibia than in the femur. This may be partly attributed to the tibial tunnel being relatively longer than the femoral tunnel in this murine ACLR model or possibly due to the greater relative graft-tunnel motion in the femoral tunnel, which can lead to bone resorption around the tunnel (widening). 3 The angle between the graft and the tunnel is much higher on the femoral side, and bending and contact forces are highest at the femoral intra-articular aperture, as suggested by 3-dimensional and finite element research. 24 This increased “bending” moment may lead to increased micromotion of the tendon in the tunnel, which could delay or impair graft-to-bone healing. Additionally, our data suggest that healing begins in the extra-articular part earlier than in the intra-articular part, based on the histological analysis of the free cage group. Significantly higher TBTH scores were observed in the extra-articular tibia than in the intra-articular tibia. The fixation position was outside the extra-articular tunnel exit (“suspensory fixation”), and this rigid and stable fixation would decrease graft-tunnel micromotion in the extra-articular part of the tunnel.
The lack of a significant difference in the biomechanical test results (failure load and stiffness) between the groups could be attributed to the incomplete healing of the graft in the femoral tunnel across all groups, given the relatively short 4-week period. Failures during biomechanical testing were most often observed on the femoral side. In addition to the histological results, this finding suggests that healing begins faster on the tibial side than on the femoral side. There were no significant differences in the biomechanical results in this study, and the histological results showed a significantly lower TBTH score in the femoral tunnel than in the tibial tunnel. It appears that the promotion of healing solely through treadmill exercise (moderate mechanical stimulation) was insufficient to achieve adequate healing on the femoral side at 4 weeks in the mouse ACLR model, although treadmill exercise was more beneficial for TBI healing than free cage activity. Another possible reason is insufficient remodeling of the intra-articular part of the graft (“ligamentization”). At 4 weeks, the graft was likely still in the proliferative phase of healing before remodeling.
The absence of a significant difference in the μCT assessment between the groups could be attributed to the early time point, as new bone formation and remodeling will occur over time. Analysis at earlier and later time points (1 or 2 weeks postoperatively and 8-12 weeks) could reveal significant differences in the microstructural values (eg, BV/TV) among the groups.
In this study, we began the treadmill exercise 7 days after surgery, allowing normal cage activity for the first 7 days. Previous studies from our laboratory have evaluated the effect of the timing of onset of mechanical loading of the joint after ACLR.4,5,18 We have consistently found that delayed mobilization after an initial period of immobilization results in superior healing. For example, in a rat ACLR model, the animals were initially immobilized with an external fixator; then, the joints were subjected to controlled cyclic axial distraction using a computerized controlled loading device. 1 Delayed initiation of cyclic axial loading on postoperative day 10 resulted in a load to failure of the femur-ACL-tibia complex that was significantly greater than that resulting from immediate loading or prolonged immobilization. In a mouse ACLR model, an external fixator was placed across the knee at the time of surgery and removed when mechanical loading was scheduled to commence. 5 After removal of the external fixator, animals were permitted free, unrestricted cage activity. A 5-day period of immobilization without weightbearing improved biomechanical strength of the healing TBI, while prolonged periods without mechanical load and motion decreased the ultimate load to failure in this murine model. We also used this model to examine the presence of inflammatory mediators at the healing TBI using near-infrared in vivo imaging with a fluorescent probe that detects matrix metalloproteinase 2, 3, 9, and 13. 18 We demonstrated that short-term postoperative immobilization (5 and 14 days) led to less matrix metalloproteinase activity around the bone tunnel as compared with immediate cage activity or prolonged immobilization. 18 The current study is the first to use treadmill activity to impart mechanical load on the healing graft, more closely simulating the clinical scenario. Additional study could evaluate the effect of treadmill activity beginning after an initial period of knee immobilization, in contrast to the normal cage activity allowed for the first 7 days in this study.
Further studies are warranted to achieve a more robust sample size and investigate alternative loading regimens, such as periods of immobilization followed by remobilization, 5 and higher-intensity treadmill running (20 m/min) to evaluate TBI healing after ACLR. Previous work from our laboratory demonstrated that a 5-day immobilization period using external fixation had a positive effect on graft healing in a murine ACLR model. 18 As complete immobilization with an external fixator is not as clinically relevant, ongoing studies in our laboratory now use a custom-designed, 3-dimensionally printed knee brace that provides more clinically relevant immobilization 17 and does not provoke excessive inflammation, as previously described. 18
Limitations
The main limitation of this study is the lack of histological analysis in arm 1 (assessment of the femoral tunnel and the axial slice of the tibia) and the lack of femoral tunnel assessment by μCT in both study arms. Furthermore, the joint was not immobilized after ACLR, and the precise magnitude and type of mechanical load (tension, shear, compression) at the healing TBI is unknown. The evaluation focused solely on the effects of treadmill exercise at speeds of 10 and 15 m/min. Potential adverse effects, such as changes in range of motion or gait patterns, were not assessed, although they may have been affected by the intervention. In addition, higher speeds (eg, 20 or 25 m/min) should have been evaluated as well, because their effects on TBI healing—whether beneficial or detrimental—remain unclear. We still do not know the optimal type, magnitude, and duration of loading that optimizes or prohibits healing, nor have we elucidated the underlying biological responses, such as gene expression changes. Further studies are necessary to objectively quantify. Other limitations include the relatively small sample size without sufficient power, short follow-up, and evaluation at a single time point, as we focused on the acute phase of healing (4 weeks).
Conclusion
Our study demonstrated that after ACLR moderate treadmill exercise (10 m/min for 50 min/d, 5 d/wk for 3 weeks) positively affected TBI healing in the femoral and tibial tunnels in the murine model. The healing process was initiated earlier in the tibia than in the femur in the mechanical loading protocol used herein. There may be a role for the moderate-load rehabilitation protocol to improve healing of the tendon graft–bone interface in patients after ACLR.
Footnotes
Acknowledgements
The authors also recognize the valuable support of Liang Ying (Orthopaedic Soft Tissue Research Program, Hospital for Special Surgery at that time) for technical support with histological analysis.
Final revision submitted July 24, 2025; accepted September 12, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding for this study was provided by the Virginia Toulmin Foundation and AOSSM Osteoarthritis Grant. Y.O. has received financial support from the Uehara Memorial Foundation, Kodama Memorial Prize, Japan Orthopaedics Traumatology Foundation, International Travel Award from the Japanese Society for Matrix Biology and Medicine, and the Japan Society for the Promotion of Science through the Grants-in-Aid for Scientific Research (KAKENHI 24K23295 and 25K19973). S.A.R. has received consulting fees from Teladoc, Inc, DonJoy/Enovis, and Novartis Pharmaceuticals. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Institutional Animal Care and Use Committee of Weill Cornell Medical College (2020-0024).
