Abstract
Background:
Bilateral gait biomechanics change over time after anterior cruciate ligament reconstruction (ACLR) and have been suggested to contribute to the development of osteoarthritis. However, few studies have investigated the longitudinal changes in bilateral gait biomechanics in the same cohort within 24 months after ACLR.
Purpose:
To evaluate the longitudinal changes in bilateral gait biomechanics compared with the healthy control cohort within 24 months after ACLR.
Study Design:
Controlled laboratory study.
Methods:
A total of 24 patients who underwent primary unilateral ACLR surgery with autologous hamstring tendon grafts and 24 matched healthy participants were included. Also, 3-dimensional knee gait and ground-reaction forces (GRF) information were collected at 3, 6, 12, and 24 months after ACLR. Linear mixed-effects models were used to assess the influence of time and limb in ACLR participants and their interaction effect on each variable of interest. Two-way analysis of variance was used to compare intergroup and interleg dependent variables.
Results:
Three months after ACLR, most biomechanical parameters of the operated limb (peak knee extension angle [pKEA]; peak knee extension moment [pKEM]; peak knee flexion moment [pKFM]; and vertical ground-reaction force [vGRF]) were lower than both the contralateral limb and healthy controls, and gradually increased over time. By 24 months postoperation, the pKEA and the pKEM returned to normal levels. The operated limb showed significantly reduced pKEA and pKEM compared with contralateral and control groups at 3 and 6 months (all P≤ .001), with no clinically meaningful differences at 12 and 24 months. pKFM analysis revealed significant between-group effects (ACLR group vs control group) without limb or interaction effects from 3 to 24 months, indicating bilateral compensatory reduction. Regarding vGRF, GRF_peak1 demonstrated bilateral compensatory reduction at 3 months (resolved by 6 months), while GRF_peak2 showed operated limb reduction at 3 months progressing to bilateral compensation at 6 months (resolved at 12 months). Notably, both vGRF parameters exhibited bilateral compensatory reductions again at 24 months postoperation.
Conclusion:
Three months after ACLR, sagittal plane biomechanical parameters of the operated knee (pKEA, pKFM, pKEM) and GRF_peak1/2 were significantly lower than those of controls, except pKFA, which showed no intergroup difference. Longitudinal analysis revealed partial recovery patterns: GRF_peak1 was normalized by 6 months, while GRF_peak2, pKEA, and pKEM reached control levels by 12 months. However, a secondary decline emerged at 24 months, with GRF_peak1/2 values again significantly lower than those of the controls, and the pKFM remained persistently lower than that of the controls throughout the 24-month follow-up. Early postoperative gait retraining and neuromuscular control training are recommended, with sustained interventions maintained for at least 24 months.
Clinical Relevance:
Bilateral gait retraining and neuromuscular control training are recommended as early as possible and should be maintained for at least 24 months after surgery.
Anterior cruciate ligament (ACL) rupture is a widespread sports injury 36 that may contribute to knee instability and functional deficits. 17 ACL reconstruction (ACLR) surgery is a standard procedure to restore knee joint stability and function. 22 However, alterations in gait biomechanics may persist for >5 years.5,33
There are changes in the knee kinematics and kinetics in both limbs after ACLR that vary according to the time since surgery. Within 6 months postoperatively, ACLR knees were found to have greater knee flexion angles and external flexion moments during level walking compared with healthy control knees. Additionally, 6 to 12 months after surgery, ACLR knees exhibited lower flexion angles and internal knee extension moments compared with controls and their contralateral limbs during early stance. 4 Also, 6 months after ACLR, contralateral knees were found to have greater flexion angles and internal flexion moments compared with controls, 4 and 12 months after ACLR, contralateral knees showed fewer flexion angles and internal flexion moments than controls. 4 In addition, 12 to 24 months after ACLR, the operated limb demonstrated reduced knee flexion moments compared with contralateral and control limbs 9 ; 3 to 4 years after ACLR, there was no difference in operated gait biomechanics compared with contralateral and control limbs, while reduced knee external flexion moments were observed 5 to 11 years after ACLR. 9 However, previous research on early and middle-term changes in gait biomechanics after ACLR at multiple time points is mainly in meta-analyses,9,11,12,16,33 and no studies have investigated the longitudinal changes in bilateral gait biomechanics over time after ACLR in the same cohort.
After ACLR, vertical ground-reaction forces (vGRF) are typically reduced compared with the contralateral lower limb or healthy controls, 4 resulting in underload of the operated limb joints. The change in GRF appears to be related to the postoperative time after ACLR and the presence of symptoms.25,26 In their study, Pietrosimone et al 26 compared vGRF from walking between patients with and without clinically relevant knee symptoms in 3 cohorts (<12 months, 12-24 months, and >24 months after ACLR) and found that patients with clinically relevant symptoms such as joint swelling and stiffness within 12 months after ACLR had lower vGRF peaks at 0% to 26% of the stance period but higher vGRF at 34% to 65% of the stance period, compared with 12 to 24 months after ACLR. Some studies have found that lower quadriceps muscle strength reduces the vGRF after ACLR.1,38 Therefore, exploring the characteristics of gait biomechanical changes in the early stage of ACLR can further the selection and implementation of rehabilitation interventions, such as quadriceps strength training and personalized gait retraining.
The present study aimed to quantify longitudinal changes in bilateral vGRF and knee gait biomechanics over a 24-month postoperative period and to compare bilateral gait biomechanical alterations between the ACLR and control groups at 3, 6, 12, and 24 months after ACLR.
Methods
Participants
A total of 24 patients (ACLR group) who underwent unilateral ACLR at our institute between January 2014 and December 2018, and 24 healthy controls (control group) matched for sex, age, body mass index (BMI) (Table 1), and Tegner (preinjury) score were selected for gait analysis. Both patients and healthy controls were recreational sports participants.
Participant Characteristics a

Data are presented as mean ± SD. ACLR, anterior cruciate ligament reconstruction; BMI, body mass index; IKDC 2000, International Knee Documentation Committee Subjective Knee Form; NA, not applicable; Speed1, the nondominant limb in the control group or the operated limb in the ACLR group; Speed2, the dominant limb in the control group or the contralateral limb in the ACLR group.
The inclusion criteria for the ACLR group were as follows: (1) age 18 to 40 years; (2) BMI of ≥18.5 kg/m2 and <24 kg/m2; (3) unilateral isolated ACL injury without other ligament injuries; (4) those who underwent surgery using a single bundle hamstring tendon autograft; (5) those who did not undergo meniscal repair surgery but could have partial meniscectomy and total meniscectomy; (6) those with no other current orthopaedic injuries or disorders that might affect lower extremity movements and no history of orthopaedic surgery; and (7) those willing to undergo follow-up at 3, 6, 12, and 24 months after surgery. The inclusion criteria (1), (2), and (6) for the ACLR group were also applied to the healthy control group. The dominant leg in the healthy control group was defined as the leg regularly used to kick a ball, and the other leg was considered the nondominant leg.
Surgical Technique and Postoperative Rehabilitation
Five experienced orthopaedic surgeons working in our sports medicine department completed all the reconstructive surgeries. Surgery was performed as follows: an autologous hamstring tendon graft was selected. Both the semitendinosus and the gracilis tendons were prepared in 3 or 4 strands, with a total diameter of 7 to 10 mm. The tibial tunnel was located at the center of the ACL stump. The exact tibial tunnel placement was at the intersection of the free margin extension of the lateral meniscus anterior horn and the lateral slope of the medial intercondylar spine. The femoral tunnel was drilled using a transtibial technique. The femoral attachment was located in the center of the femoral stump of the ACL and proximal, high, and deep points of the posterior lateral condylar cartilage of the femur. The femoral end was fixed by Endobutton (Smith & Nephew), and the tibial end was fixed with interface screws (Smith & Nephew).
All participants with ACLR underwent similar postoperative home-based rehabilitation programs until 12 months after ACLR.10,32 The postoperative home-based rehabilitation programs were issued in written form by the Department of Sports Medicine of the authors’ hospital. On the first day after surgery, quadriceps sets, straight-leg raises, and prone hangs were initiated. All patients were allowed to walk with crutches and braces. The range of motion (ROM) increased from 0° to 90° at 3 to 7 days after ACLR, and 22 (91.67%) patients reached the target. In the fourth week, the ROM increased to 115°, and 24 (100%) patients reached the target. Closed kinetic chain exercises and full weightbearing were started in the sixth week. Patients progressed to running without bracing between 4 and 6 months; 24 (100%) patients reached the target. The training progress and functional status were regularly followed up on by a fixed therapist via telephone, video, or text message. All patients had an essentially full ROM of the knee and were not limited, and no pain while resting or walking when participating in the test. All patients were negative on the Lachman test and the pivot shift test in the operated knees, and no ACLRs were considered failures during the 2-year follow-up period (at 3, 6, 12, and 24 months after surgery).
Sample Size
Based on a previous study, 5 it was calculated that group sample size of 24 participants (12 in ACLR group 12 in control group) would provide 80% power to reject the null hypothesis of equal means when the first knee peak extension moments mean difference of the gait biomechanics is −0.63 (–3.09 minus −2.46), with standard deviations of 0.56 for the ACLR group and 0.51 for the control group at a 2-sided alpha of .05. Given an anticipated dropout rate of 20%, the total required sample size was 30 (15 in the ACLR group and 15 in the control group) (Version 11.0; PASS). A total of 24 patients and 24 healthy individuals were initially recruited; however, since 1 patient at 3 months after ACLR, 2 patients at 12 months after ACLR, and 1 patient at 24 months after ACLR could not undergo the 3-dimensional gait test due to personal reasons, data from 23, 22 and 23 patients at 3, 12, and 24 months, respectively, were used for the gait biomechanics analysis.
Ethical approval was obtained from the Ethics Committee of Peking University Third Hospital, and all participants provided written informed consent before data collection.
A 3-Dimensional Gait Test Protocol
All participants underwent a 3-dimensional (3D) gait test to collect and analyze kinetic and kinematic data. A set of passive reflective markers optimized based on the validated28,31 plug-in-gait 15 model was bilaterally attached to participants’ lower limbs to track segmental motion during walking: the anterior and posterior superior iliac spine, medial and lateral femoral epicondyles, the frontal and lateral aspects of the thigh, medial and lateral malleolus, tuberositas tibiae, the upper third and lower third of frontal aspects of the shank, posterior part of the calcaneus, heads of the first, second, and fifth metatarsal bones. During gait tests, 3D coordinate data of passive reflective markers were collected using an 8-camera motion capture system at a sample rate of 100 Hz (Vicon MX, Oxford Metrics). The GRF was collected using 2 embedded force plates at a sampling rate of 1000 Hz (AMTI, Advanced Mechanical Technology Inc).
After the standing trial, all participants were asked to walk from a specified point so that one of their feet would unintentionally step on the first force plate and the other would walk on the second force plate. The whole length of the walkway was 8.4 m. The distance before hitting the force plate was 3.6 m to establish the natural gait velocity of each participant. The distance measured after leaving the force plate was 3.6 m, used to control for deceleration. The length of each force plate was 0.6 m. A trial was considered successful when each foot stepped on the force plates at a self-selected speed, and investigators collected all kinematic and kinetic data. Each participant had 5 successful gait trials in the test. The mean value of 5 trials was used for analysis. None of the participants complained about pain during walking.
Data Reduction and Analysis
The coordinate data of the passive reflective markers were filtered at 12 Hz using a low-pass Butterworth filter, and the vGRF data were filtered at 100 Hz using a low-pass Butterworth filter. Kinematic and kinetic variables were calculated using Visual 3D software (C-motion). Knee joint angles were calculated as Cardan angles between the proximal and distal segments in flexion-extension. Knee joint moments were calculated using an inverse dynamic approach, normalized to the product of mass (kg) and height (m), and as external moments. vGRFs were normalized to the body weight (BW). Spatiotemporal variables, such as walking speed, were calculated using the following formula: Walking Speed = Lstep length/Tstep time for each side of the leg.
The peak knee flexion angle (pKFA), the peak knee extension angle (pKEA) during the gait stance phase, the peak knee flexion moment (pKFM), the peak knee extension moment (pKEM), the first peak of the vGRF (GRF_peak1), and the second peak of the vGRF (GRF_peak2) during the gait stance phase of the bilateral gait biomechanics in the ACLR cohort (ie, operated and contralateral limbs), and the uninjured control cohort (ie, dominant and nondominant limbs) were calculated.
Among the 24 participants in the uninjured control cohort, only 1 had a dominant left leg. The dominant limbs of the uninjured control cohort were matched with the contralateral limbs of the ACLR cohort for the analysis. The nondominant side of the uninjured control cohort was also matched with the operated limbs of the ACLR cohort.
Statistical Analysis
Demographic variables between the ACLR cohort (3, 6, 12, and 24 months after ACLR) and uninjured controls were compared using one-way analysis of variance (ANOVA). The Bonferroni procedure was used to adjust the type I error rate to 0.005 as the statistical significance threshold, ensuring that the overall type I error rate was no greater than 0.05.
Linear mixed-effects models were used to assess the influence of time and limb in ACLR participants and their interaction effect on each variable of interest. Speed was included as a covariate in the calculation to avoid walking speed affecting gait biomechanics.6,7 The subject was assigned as a random effect. If a significant interaction effect of time point and limb was detected, the Bonferroni-adjusted test (P < .0167) was used for pairwise comparisons between operated limbs and contralateral limbs at different time points, comparing the 4 time points in pairs on each limb.
Two-way ANOVA was used to compare the intergroup (ACLR and control groups) and interleg (operated and contralateral legs in the ACLR group; dominant and nondominant leg in the uninjured control group) dependent variables. Speed was included as a covariate in the calculation to avoid walking speed affecting gait biomechanics.6,7 If a significant interaction effect of group and leg was detected, Bonferroni-adjusted test was used for t tests, including paired t tests within each group (operated leg vs contralateral leg in the ACLR group; dominant leg vs nondominant leg in the uninjured control group) and independent t tests between groups (operated leg in the ACLR group vs nondominant leg in the uninjured control group; contralateral leg in the ACLR group vs dominant leg in the uninjured control group).
SPSS Version 26.0 (IBM) was used for all statistical analyses. The minimal clinically important difference (MCID) was defined as 3° in knee flexion angle and 0.004 BW × body height (0.0392 N·m/kg/m) in knee moment. 30 However, an MCID of the GRF is yet to be determined.
Results
As shown in Table 1, at 3 months postoperatively, the participants in the ACLR group consisted of 21 men and 2 women, with a mean age of 28.9 ± 6.5 years and a mean BMI of 25.2 ± 3.6 kg/m2. There was no significant difference (P = .378) in the Tegner score between the control group (5.69 ± 1.44) and the ACLR group before injury (6.12 ± 1.17). The IKDC 2000 scores at 3 and 6 months after ACLR were smaller than those in the control group, and the IKDC 2000 scores at 12 and 24 months after ACLR were significantly greater than those at 3 months after ACLR (all, P < .05).
The Tegner score at the 3 months after ACLR was significantly smaller than in the control group, and the Tegner scores at 12 and 24 months after ACLR were greater than that at 3 months after ACLR (all, P < .05). The Lysholm values at 3 and 6 months after ACLR were significantly smaller than in the control group (all, P < .05).
Linear mixed effects models revealed significant time and limb interaction on pKEA (P < .001), pKFM (P = .005), pKEM (P < .001), GRF_peak1 (P = .001), and GRF_peak2 (P < .001), while no significant time and limb interaction was found on pKFM (P = .929).
Significant differences were identified between time points in the operated limb (all P < .05), while there were no significant changes between time points in the contralateral limbs on pKEA, pKFM, pKEM, GRF_peak1, and GRF_peak2 (Figure 1).
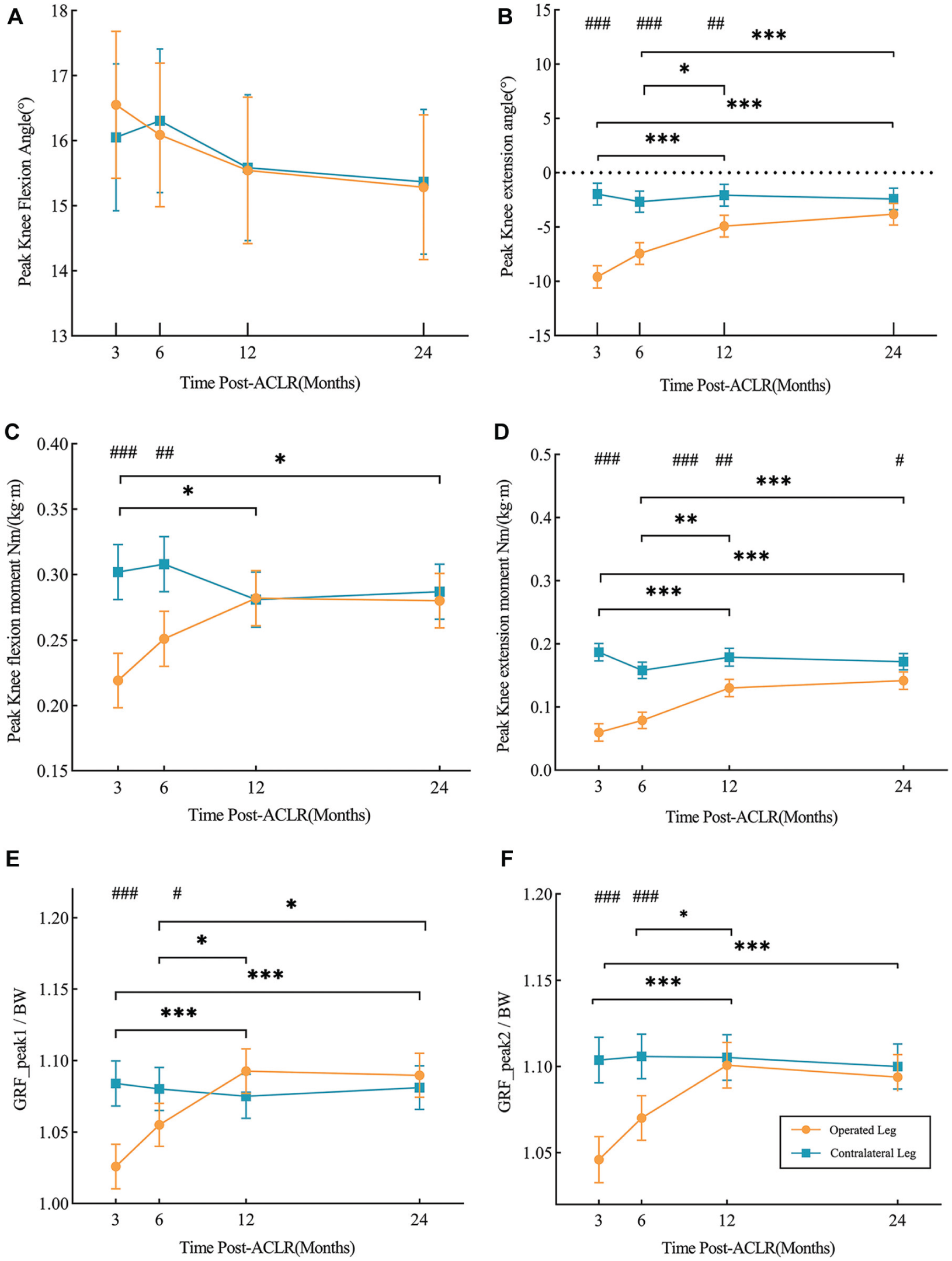
(A) The peak knee flexion angle (pKFA), (B) the peak knee extension angle (pKEA), (C) the external peak knee flexion moment (pKFM), (D) the external peak knee extension moment (pKEM), (E) the first peak of the vGRF (GRF_peak1), and (F) the second peak of the vGRF (GRF_peak1) during stance phase in the level walking at 3, 6, 12 and 24 months after ACLR. Orange indicates the operated leg in the ACLR group, and blue indicates the contralateral leg in the ACLR group. ACLR, anterior cruciate ligament reconstruction. * Indicates significant differences between time points in the operated leg of the ACLR group. # Indicates significant differences between the operated leg and the contralateral leg of the ACLR group.
In the operated limb, the pKEA (P < .001), pKFM (P = .016), pKEM (P < .001), GRF_peak1 (P < .001), and GRF_peak2 (P < .001) increased significantly 3 to 12 months after ACLR; the pKEA (P < .001), pKFM (P = .016), pKEM (P < .001), GRF_peak1 (P < .001), and GRF_peak2 (P < .001) increased significantly 3 to 24 months after ACLR; the pKEA (P = .012), pKEM (P = 0.003), GRF_peak1 (P = .028), and GRF_peak2 (P = .017) increased significantly 6 to 12 months after ACLR; the pKEA (P < .001), pKEM (P < .001), and GRF_peak1 (P = .047) increased significantly 6 to 24 months after ACLR.
Significant differences were found between limbs. The pKEA of the operated knee was significantly smaller than the contralateral knee at 3 (P < .001), 6 (P < .001), and 12 months (P = .001). However, no significant difference was found between limbs in the 24 months (P = .076). For the pKFM, the operated knee was significantly greater than the contralateral knee at 3 (P < .001) and 6 months (P = .003), while there was no significant difference between limbs at 12 (P = .961) and 24 (P = .722) months. The operated knee had significantly smaller pKEM than the contralateral knee in all time points (3 months: P < .001; 6 months: P < .001; 12 months: P = .001; 24 months: P = .035). The GRF_peak1 and the GRF_peak2 of the operated knee were significantly smaller than the contralateral knee at 3 (both P < .001) and 6 months (GRF_peak1: P = .049; GRF_peak2: P < .001), while there was no significant difference between limbs at 12 (GRF_peak1: P = .190; GRF_peak2: P = .651) and 24 months (GRF_peak1: P = .507; GRF_peak2: P = .534).
Two-way ANOVA revealed a significant leg and group interaction effect on pKEA (P < .001), pKEM (P < .001), and GRF_peak2 (P = .031) 3 months after ACLR. pKEA (P < .001), pKEM (P < .001), and GRF_peak2 (P = .002) of the operated leg were significantly smaller than those of the contralateral leg in the ACLR group; however, there were no significant differences between legs in the control groups (P = .765, P = .601, and P = .939, respectively) (Table 2). The operated leg also had significantly smaller pKEA (P < .001), pKEM (P < .001), and GRF_peak2 (P < .001) than a nondominant leg, but no significant difference was detected between the contralateral leg in the ACLR group and the dominant leg in the control group (P = .903, P = .057, P = .180, respectively) (Table 2 and Figure 2 C, D, and F). There was also no significant interaction effect of leg and group on pKFA (P = 0.875), pKFM (P = .093), and GRF_peak1(P = .090) (Table 2, Figure 2, A, B, and E). Moreover, no significant difference was observed in pKFA between legs (P = .826) and between groups (P = .953). pKFM (P = .006) and GRF_peak1 (P = .015) in the ACLR group were significantly smaller than those in the control group, but no significant difference was detected between legs (P = .079, P = .063, respectively). Nonetheless, the difference (0.029 N·m/kg/m < 0.0392 N·m/kg/m) of the pKFM between the contralateral leg in the ACLR group and the dominant leg in the control group was not clinically meaningful.
Biomechanics 3 Months After ACLR a

Data are presented as mean ± SD. ACLR, anterior cruciate ligament reconstruction; BW, body weight; pKEA, the peak knee extension angle; pKFA, the peak knee flexion angle; pKEM, the external peak knee extension moment; pKFM, the external peak knee flexion moment; GRF_peak1, the first peak of the vGRF; GRF_peak2, the second peak of the vGRF.
Indicates significant interaction and main effects.
Indicates the comparison between operated and contralateral legs in the ACLR group.
Indicates the comparison between the operated leg in the ACLR group and the nondominant leg in the control group.
Indicates the comparison between nondominant and dominant legs in the control group.
Indicates the comparison between the contralateral leg in the ACLR group and the dominant leg in the control group.

(A) The peak knee flexion angle (pKFA), (B) the peak knee extension angle (pKEA), (C) the external peak knee flexion moment (pKFM), (D) the external peak knee extension moment (pKEM), (E) the first peak of the vGRF (GRF_peak1), and (F) the second peak of the vGRF (GRF_peak2) during the stance phase in the level walking at 3, 6, 12 and 24 months after ACLR and in the control group. The data in each group were superimposed over one another; the bar with the smaller value in each group is in the front. Orange indicates the operated leg in the ACLR group, and blue indicates the contralateral leg in the ACLR group; light orange indicates the nondominant leg in the control group; light blue indicates the dominant leg in the control group. ACLR, anterior cruciate ligament reconstruction. * Indicates significant differences between the operated leg in the ACLR group and the nondominant leg in the control group. # Indicates significant differences between the ACLR group and the control group.
Significant leg and group interaction effects were detected on pKEA (P = .011) and pKEM (P = .001) 6 months after ACLR (Table 3). The operated leg also had smaller pKEA (both P = .001) and pKEM (both P < .001) than the contralateral leg in the ACLR group and the nondominant leg in the control group. However, no significant differences were found between legs in the control group (P = .750 and P = .614, respectively) and the contralateral leg in the ACLR group and the dominant leg in the control group (P = .856 and P = .363, respectively) (Table 3, Figure 2, C and D). Moreover, there was no significant interaction effect of leg and group on pKFA (P = .908), pKFM (P = .252), GRF_peak1 (P = .426), and GRF_peak2 (P = .253), and no significant difference between legs. Additionally, no significant difference was detected in pKFA (P = .356) and GRF_peak1 (P = .324) between the ACLR and control groups. pKFM (P = .016) and GRF_peak2 (P = .025) in the ACLR group were significantly smaller compared with the control group (Table 3, Figure 2, B and F). However, the difference (0.032 N·m/kg/m < 0.0392 N·m/kg/m) of the pKFM between the contralateral leg in the ACLR group and the dominant leg in the control group was not clinically meaningful.
Biomechanics 6 Months After ACLR a

Data are presented as mean ± SD. ACLR, anterior cruciate ligament reconstruction; BW, body weight; pKEA, the peak knee extension angle; pKFA, the peak knee flexion angle; pKEM, the external peak knee extension moment; pKFM, the external peak knee flexion moment; GRF_peak1, the first peak of the vGRF; GRF_peak2, the second peak of the vGRF.
Indicates significant interaction and main effects.
Indicates the comparison between operated and contralateral legs in the ACLR group.
Indicates the comparison between the operated leg in the ACLR group and the nondominant leg in the control group.
Indicates the comparison between nondominant and dominant legs in the control group.
Indicates the comparison between the contralateral leg in the ACLR group and the dominant leg in the control group.
There was no significant interaction effect of leg and group on all parameters at 12 and 24 months after ACLR, and no significant leg effect in all parameters (Tables 4 and 5). Only pKFM (P = .017) differed significantly between groups, being significantly smaller in the ACLR group than in the control group 12 months after surgery (Table 4, Figure 2B). The difference (0.068 N·m/kg/m >0.0392 N·m/kg/m) of the pKFM between the contralateral leg in the ACLR group and the dominant leg in the control group was clinically meaningful.
Biomechanics 12 Months After ACLR a
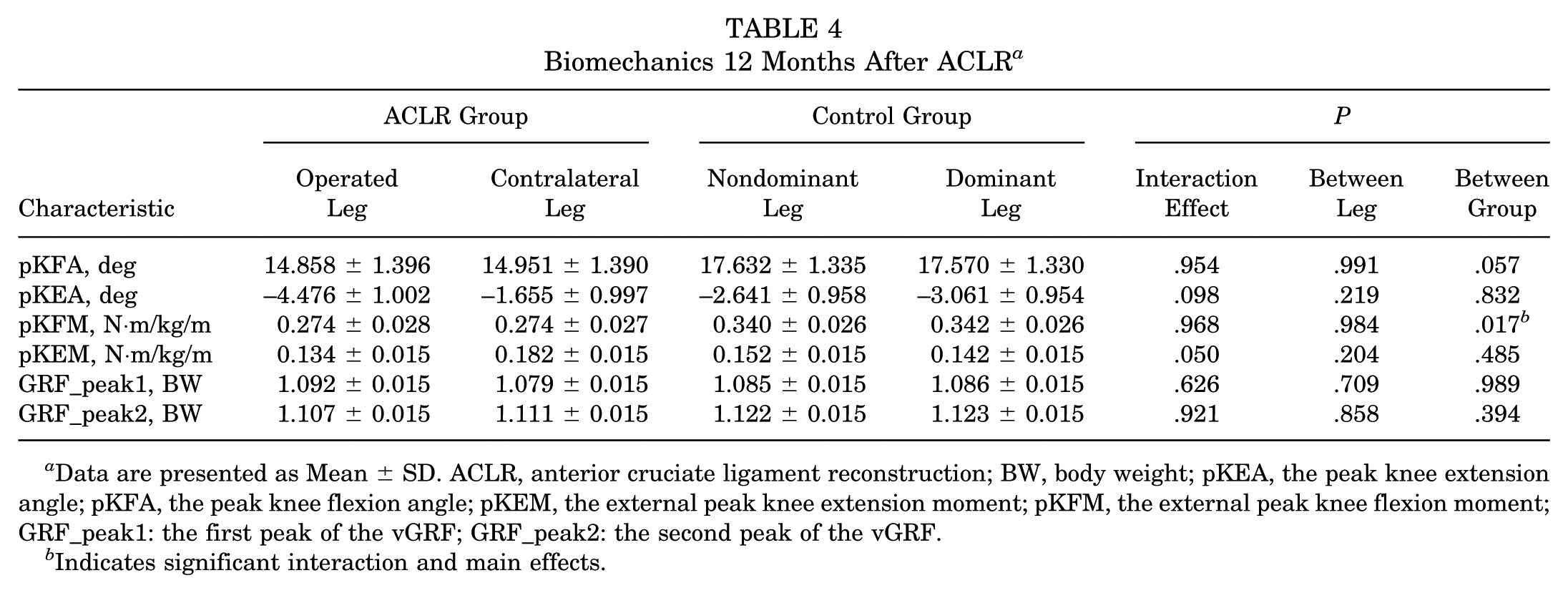
Data are presented as Mean ± SD. ACLR, anterior cruciate ligament reconstruction; BW, body weight; pKEA, the peak knee extension angle; pKFA, the peak knee flexion angle; pKEM, the external peak knee extension moment; pKFM, the external peak knee flexion moment; GRF_peak1: the first peak of the vGRF; GRF_peak2: the second peak of the vGRF.
Indicates significant interaction and main effects.
Biomechanics 24 Months After ACLR a

Data are presented as Mean ± SD. ACLR, anterior cruciate ligament reconstruction; BW, body weight; pKEA, the peak knee extension angle; pKFA, the peak knee flexion angle; pKEM, the external peak knee extension moment; KFM, the external peak knee flexion moment; GRF_peak1, the first peak of the vGRF; GRF_peak2, the second peak of the vGRF.
Indicates significant interaction and main effects.
By 24 months after ACLR, significant differences were found between groups in pKFM (P = .030), pKEM (P = .018), GRF_peak1 (P = .005), and GRF_peak2 (P = .032), and all of them in the ACLR group were significantly smaller than that in the control group (Table 5, Figure 2, B, D, E, and F). However, no significant difference was detected between groups in pKFA and pKEA. The difference (0.055 N·m/kg/m > 0.0392 N·m/kg/m) in the pKFM between contralateral leg in ACLR group and the dominant leg in control group was clinically meaningful; however, the difference (0.028 N·m/kg/m < 0.0392 N·m/kg/m) of the pKEM between contralateral leg in ACLR group and the dominant leg in control group was not clinically meaningful.
Discussion
The present study assessed bilateral biomechanical changes in level walking within 24 months after ACLR and compared the biomechanics of the operated leg with the contralateral leg and healthy controls. To the best of our knowledge, this is the first study that analyzed the longitudinal biomechanics of bilateral lower limb gait after ACLR in the same cohort at multiple consecutive time points (3, 6, 12, and 24 months).
Our findings showed that the pKFM of the reconstruction leg was significantly lower than that of the contralateral leg after ACLR. The value of the pKFM of the reconstruction leg gradually increased over time. It showed no significant differences compared with the contralateral leg at 12 and 24 months after ACLR. However, the pKFM of the ACLR group remained lower than that of the healthy controls 24 months after ACLR. Our results suggested that the knee flexion moment (KFM) in the ACLR group improved over time but did not return to normal after 24 months. During the loading response phase, eccentric contraction of the quadriceps is used to limit the speed and magnitude of knee flexion. The knee flexion 18 angle and the KFM 3 gradually increase and reach their peak to relieve the impact force from the ground. This adaptive change after ACLR in KFM may be related to decreased quadriceps muscle strength. 27 A reduced KFM of the operated leg indicated a reduction in knee joint loading. Williams et al 38 demonstrated that a reduced pKFM was associated with the degeneration of the patellofemoral joint cartilage. However, traditional rehabilitation strategies cannot completely restore the magnitude of KFM. Ito et al 13 found that diminished quadriceps neuromuscular function was characterized by abnormal sagittal plane knee moment and extensor electromyography of the involved knee early after ACLR. Therefore, the decrease in KFM may be due to the abnormal neuromuscular control ability of the quadriceps after surgery. 8 After ACL rupture, the weakened quadriceps muscle strength might be due to the loss of afferent reaction from the ACL to gamma motor neurons caused by the injury of ACL mechanoreceptors. 19 However, the impaired ACL mechanoreceptors might not be restored after the ACLR. 20 This may cause the long-term weakening of quadriceps muscle strength and affect the recovery of KFM. Suner Keklik et al 34 found that the proprioceptive sense remained poor in the ACLR group compared with the control group, even 24 months after surgery. A poorer proprioceptive sense in the ACLR limb was associated with lower internal knee extension moment (KEM), which is equal to the external KFM in our study, and lower loading rates. 2 Therefore, proprioceptive training and neurological rehabilitation interventions are needed in addition to traditional rehabilitation strategies early after ACLR. In addition, we also found that pKFM of the contralateral lower limb decreased from 6 to 12 months after ACLR, and symmetry was achieved at 12 to 24 months after surgery. Still, the difference between the pKFM of the contralateral lower limb and the control group was clinically significant at 12 to 24 months after surgery. Therefore, achieving symmetry in gait biomechanics does not necessarily represent a normal gait pattern. It has been found that there is insufficient quadriceps activation in the contralateral leg compared with the healthy control group. 23 Long-term abnormal movement patterns in operated limbs may disrupt central nervous system control of contralateral motion via neural pathways. Previous studies have demonstrated bilateral knee proprioceptive deficits after ACLR,14,29,34 findings that are supported by our electroencephalographic results, which show abnormal power spectra during contralateral movement. 24 This necessitates postoperative bilateral gait retraining and central neuromuscular control training, with contralateral limb rehabilitation synchronized to ACLR limb progress. Our data (Table 1) indicate a gradual resolution of symptoms over 24 months after surgery, suggesting functional recovery. However, persistent KFM abnormalities at 24 months reveal that functional scales have limited sensitivity in detecting residual gait biomechanical impairments despite apparent clinical recovery.
Our findings found that the pKEA and pKEM of the reconstruction leg were significantly lower than those of the contralateral leg and control groups within 24 months after ACLR. The value of the pKEA and pKEM of the reconstruction leg gradually increased over time and showed no significant differences compared with the control group at 12 months after ACLR. Although both parameters remained small compared with the contralateral leg within 24 months after ACLR and there was a significant difference in pKEM again at 24 months compared with the control group, these differences (Nondominant Leg vs Operated Leg: 0.014 N·m/kg/m; Nondominant Leg vs Contralateral Leg: 0.028 N·m/kg/m) did not reach the MCID (0.0392 N·m/kg/m). In short, gait biomechanics of the operated leg in the terminal stance period gradually recovered to the level of the contralateral leg and the control group within 24 months after ACLR.
The first and second peaks of the vGRF (GRF_peak1 and GRF_peak2) in the operated leg were found at 3 and 6 months after ACLR, compared with the contralateral leg, and the bilateral leg GRF had no significant differences 12 months after surgery. The GRF_peak1 in the ACLR group was lower than that of the control group at 3 months after ACLR and recovered to the healthy level at 6 and 12 months after surgery. The GRF_peak2 in the ACLR group was lower than that in the control group at 3 and 6 months after ACLR and recovered to a healthy level at 12 months after surgery. Although significant differences between the ACLR group and the control group appeared again 24 months after surgery, they may not have reached an MCID. Consistent with our study, previous research has found that the vGRF in the early and terminal stance of the reconstructed leg remained less than that of the contralateral leg within 12 months after ACLR. 4 Previous studies 26 have also revealed that individuals with a lesser vGRF in the stance phase might be associated with post-traumatic osteoarthritis, which suggests that lower loading in the ACLR limb can lead to degeneration of knee cartilage. Some studies have indicated that lower quadriceps muscle strength reduces vGRF1,38 after ACLR and found that the ability of the quadriceps femoris to activate neuromuscularly is impaired. 23 Quadriceps strength training should be combined with neuromuscular control training to improve gait biomechanics after ACLR.
The present study still has some limitations. First, our study only examined level walking; biomechanical changes in high-function tasks, such as running, were not analyzed. Further research should investigate the biomechanical changes that occur during running to better understand the characteristics of bilateral gait biomechanical changes after ACLR. Additionally, biomechanics were only investigated during in-laboratory gait analyses. Further research using more ecologically valid walking protocols during walking should be conducted. Third, all patients underwent a single-bundle hamstring tendon autograft ACLR. Therefore, the results of our study cannot represent the characteristics of postoperative biomechanical changes in patients with all types of grafts. Besides, almost all patients eligible for inclusion criteria in this study were men. Although there are differences in gait biomechanical characteristics in different sexes after ACLR, previous studies have confirmed that the differences mainly exist in the frontal 37 and transverse planes 37 of gait biomechanics. Further studies are still needed to characterize the longitudinal changes in gait biomechanics between sexes long after ACLR. Finally, in the current study, we did not measure the return-to-sport criteria, kinesiophobia, or functional testing.21,35 Although we mainly consider whether the return-to-sport criteria are met when involved in high-intensity sports, such as running, jumping, cutting, or pivoting sports, further studies should be conducted on whether abnormalities in some indicators of return-to-sport criteria will lead to abnormal gait biomechanics.
Conclusion
Three months after ACLR, sagittal plane biomechanical parameters of the operated knee (pKEA, pKFM, pKEM) and GRF_peak1/2 were significantly lower than those of the controls, except for pKFA, which showed no intergroup difference. Longitudinal analysis revealed partial recovery patterns: GRF_peak1 normalized by 6 months, while GRF_peak2, pKEA, and pKEM reached control levels by 12 months. However, a secondary decline emerged at 24 months, with GRF_peak1/2 values again significantly lower than those of the controls, and pKFM remained persistently lower than those of the controls throughout the 24-month follow-up. Early postoperative gait retraining and neuromuscular control training are recommended, with sustained interventions maintained for ≥24 months.
Footnotes
Final revision submitted March 24, 2025; accepted May 28, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: Financial support for this study was provided by the Beijing Municipal Natural Science Foundation (grant 7232354), the National Natural Science Foundation of China (82202821, 31900943), the Clinical Key Projects of Peking University Third Hospital (BYSY2022058), the Innovation and Transformation Fund Project of Peking University Third Hospital (HDCXZHKC2022219), the 2023 Medical Science Research Project of Hebei Province, precise injection of shoulder joint under ultrasonic guidance of platelet-rich plasma for rotator cuff injury (20230257), and the University-Industry Collaborative Education Program (230807186101349). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the institutional review board (IRB) of Peking University Third Hospital (IRB, 00006761-2012010).
