Abstract
Background:
Chronic hamstring tears can result in challenging surgical repairs. Allografts may be used to augment the repair; however, there is limited clinical evidence supporting their use.
Purposes:
To (1) evaluate clinical outcomes of patients who underwent proximal hamstring repair with allograft augmentation or interposition and (2) compare them to matched controls who underwent primary hamstring repair.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A total of 117 patients who underwent proximal hamstring repair were included, consisting of 19 allograft augmentations, 20 allograft interpositions, and 78 primary repairs. Allograft cohorts were propensity-matched with those who underwent primary repair in a 1 to 2 ratio. Clinical outcomes were obtained at the final follow-up. Data were extracted using continuous variables, with normality reported as mean values ± standard deviation, and non-normality reported as median values with interquartile ranges (IQR).
Results:
The median follow-up of the augmentation cohort was 1.9 [IQR, 0.8-3.9] years, while it was 3.2 [IQR, 1.6-5.1] years (P = .182) for the primary repair cohort. The mean follow-up for the interposition cohort was 3.5 ± 2.4 years, while it was 4.2 ± 2.5 years for the primary repair cohort (P = .227). Forty-two percent of the augmentation cohort had undergone previous surgery, compared with 5% of the interposition cohort. The time from injury to surgery for the augmentation and primary repair cohorts was 6.4 and 1.3 months, respectively (P < .001). For the interposition and primary repair cohorts, the durations were 17.8 and 1 months, respectively (P < .001). Clinical outcomes, including the Tegner Activity Scale (3 vs 3), pain with activity (47.4% vs 23.7%), and muscle spasms (36.8% vs 26.3%), did not show significant differences between the augmentation and primary repair cohorts. The augmentation cohort had a significantly worse Lower Extremity Functional Scale (LEFS) score (64 vs 75; P < .001) and were more likely to experience pain with sitting (52.6% vs 21.1%; P = .034) than the primary repair cohort. There was no significant difference in clinical outcomes between the interposition and primary repair cohorts (LEFS: 69 vs 75.5, Tegner Activity Scale: 4 vs 4; pain with sitting: 26.3% vs 27.5%; pain with activity: 42.1% vs 30%; muscle spasms: 26.3% vs 30%). Revision rates were similar between cohorts (P≥ .255).
Conclusion:
Proximal hamstring repair for chronic tears with allograft augmentation or interposition is a reproducible procedure that results in satisfactory, yet poorer clinical outcomes compared with patients who underwent primary hamstring repair. These procedures should be considered in symptomatic patients with chronic tears that are not amenable to primary repair.
Keywords
The hamstring muscles are the most commonly injured in the body and typically occur during activities involving rapid acceleration or deceleration, forced hip hyperflexion, and knee extension.6,8 This can result in strains or partial tears, most of which recover entirely without long-term sequelae. 17 Complete avulsions of the hamstring tendons from the ischial tuberosity can also occur, but are less common. 10 In contrast, these injuries typically require prompt medical and surgical care, and even so, return to normal function may not be achieved. 2
For proximal hamstring avulsions, there is a lack of consensus on the indication and timing of surgery.11,14 Nonoperative treatment occasionally results in poor outcomes with significant pain, weakness, and muscular atrophy,4,15 while some recommend surgical repair if ≥2 tendons are avulsed and retracted ≥2 cm. 1,5
While hamstring tendons are commonly able to be repaired back to their origin on the ischial tuberosity, this is not always feasible. For example, patients with chronic tears or a history of previous surgeries may have muscle atrophy, substantial scar tissue, or significant tendon retraction, making it difficult to perform a primary repair. 18 In cases where primary repair can be achieved but is prone to retearing, augmentation with an allograft can be performed to strengthen the repair. Alternatively, when a tear has a large degree of retraction and cannot be repaired directly, an allograft may be interposed between the retracted hamstring tendon and the ischial tuberosity.
While these presentations are infrequently encountered, these techniques provide a treatment option for patients with chronic hamstring dysfunction and intractable pain. To date, limited clinical data support the use of allografts, as only case reports and small case series are currently reported in the literature.9,12,13 This study aimed to evaluate clinical outcomes of patients who underwent proximal hamstring repair with allograft augmentation or interposition and compare them with matched controls who underwent primary hamstring repair. We hypothesized that allograft augmentation and interposition would result in satisfactory and comparable clinical outcomes to primary repair.
Methods
All patients who underwent proximal hamstring repair across 2 academic institutions by 2 board-certified orthopaedic surgeons (K.J.E. and A.J.K.) from 2013 to 2023 were retrospectively reviewed after approval from our institutional review board (IRB No. 23-008165). The inclusion criteria consisted of patients who underwent primary proximal hamstring repair, proximal hamstring repair with allograft augmentation, and proximal hamstring repair with allograft interposition. Patients were excluded if they had incomplete outcome scores or had a comorbidity that would severely affect functional outcomes after surgery (Figure 1). Complete postoperative clinical outcomes were obtained for all patients included in the study.
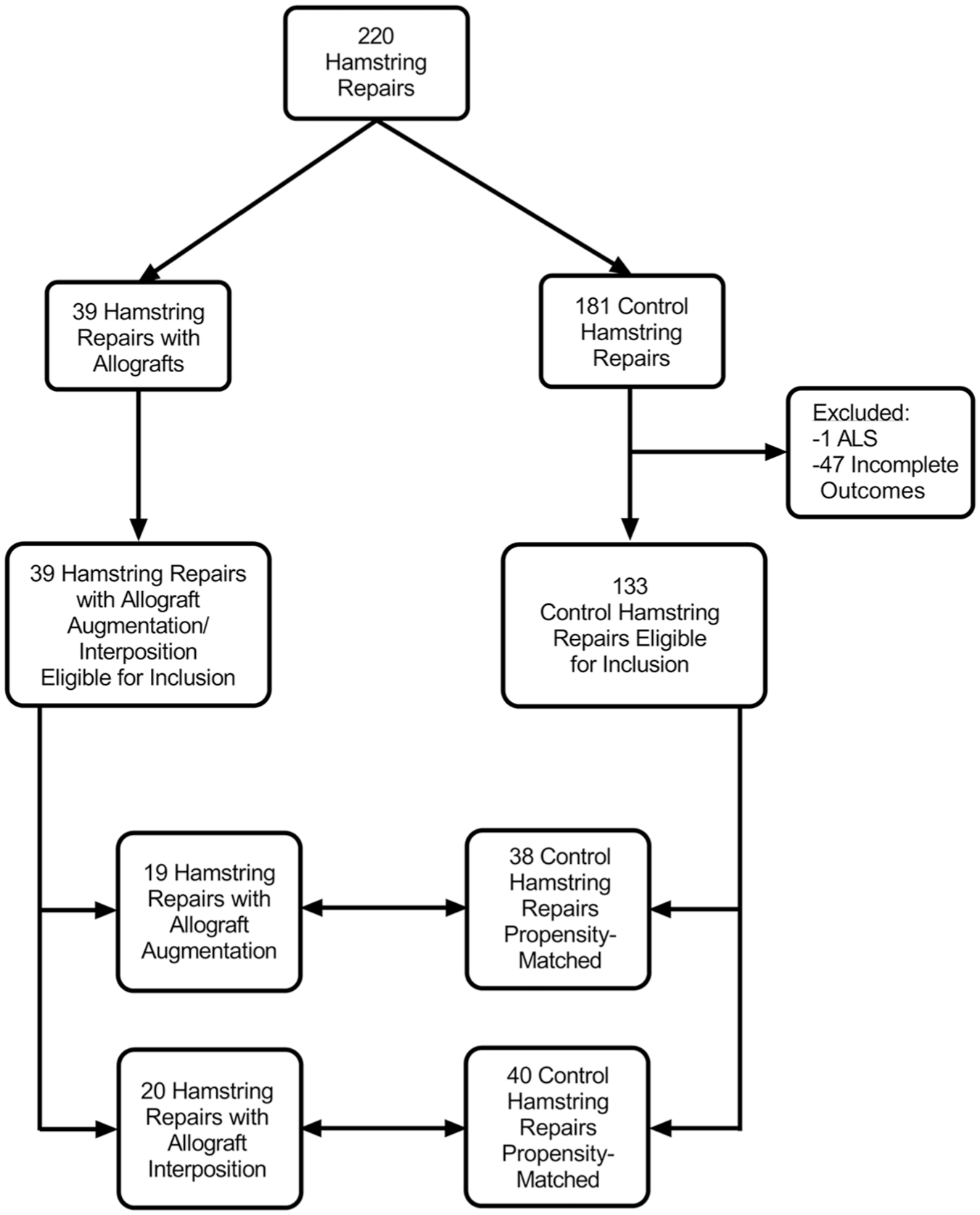
Patient screening and enrollment based on inclusion criteria. ALS, amyotrophic lateral sclerosis.
Clinical and demographic data, including age, sex, body mass index (BMI), laterality, previous surgery, time from injury to surgery, and length of follow-up, were obtained from medical records and surveys. In addition, complete tears of any of the hamstring tendons (biceps femoris, semimembranosus, and semitendinosus), the amount of retraction, the number of suture anchors, and whether the patient received intraoperative neurolysis were recorded (Figure 2). Tears were considered acute if they were repaired within 2 months of the injury and chronic if repaired ≥2 months after the time of injury. All patients who underwent allograft augmentation and interposition received a soft tissue Achilles tendon allograft.

(A) Anatomy of uninjured hamstring tendons. (B) Tears of ≥1 hamstring tendons with at least 1 tendon remaining intact at the ischial tuberosity. (C) Complete tears of all 3 hamstring tendons from the ischial tuberosity with minimal retraction. (D) Complete tears of all 3 hamstring tendons as well as significant retraction from the ischial tuberosity. BFlh, biceps femoris long head; CT, conjoint tendon; IT, ischial tuberosity; SM, semimembranosus; SN, sciatic nerve; ST, semitendinosus; STL, sacrotuberous ligament.
Patients who underwent proximal hamstring repair with either allograft augmentation or interposition were compared with patients who underwent primary proximal hamstring repair. To minimize baseline differences between comparison cohorts, propensity score matching was performed. Specifically, 19 patients who underwent proximal hamstring repair with allograft augmentation and 20 patients who underwent proximal hamstring repair with allograft interposition were propensity-matched in a 1 to 2 ratio based on age, sex, and BMI with patients who underwent primary proximal hamstring repair. The standardized mean difference between the augmentation and primary repair cohorts was 0.004, while it was 0.017 for the interposition and primary repair cohorts, indicating satisfactory matching. 20
Postoperative outcomes, including the Lower Extremity Functional Scale (LEFS), Tegner Activity Scale, pain with sitting, pain with activity, presence of muscle spasms, and subsequent reoperations, were obtained at the final follow-up.
Surgical Technique
Patients were placed in the prone position, and their lower extremities were prepared and draped in a sterile fashion. A 6 to 8 cm vertical longitudinal incision was made distal to the gluteal crease. Full-thickness skin flaps were developed, the deep fascia of the thigh was split in line with the incision, and the gluteus maximus was mobilized proximally. Depending on the degree of scar tissue surrounding the sciatic nerve, neurolysis was performed. After which, the sciatic nerve was carefully protected for the remainder of the case. In cases with preoperative sensory abnormalities or motor weakness attributed to the sciatic nerve, a peripheral neurosurgeon (R.J.S.) was available to perform sciatic nerve neurolysis.
The torn hamstring tendons were then carefully inspected for quality. Degenerative portions of the tendon were excised using scissors or a scalpel. Once a healthy tendon remained, it was assessed for quantity and quality to determine its suitability for repair. If robust, then direct repair was performed (Figure 3). Typically, 3 suture anchors were placed in a triangular configuration. Suture anchors varied throughout the study period, dependent on contemporaneous technology. Initially, metal corkscrew anchors were used, followed by Biocorkscrew anchors (Arthrex, Inc), and finally, all-suture anchors (Arthrex, Inc) in the later part of the study period. A combination of Krakow sutures was used to pulley the tendon against the bone, and mattress sutures were used to increase the surface area of compression.

Primary proximal hamstring repair of torn tendons to the ischial tuberosity.
If the tendon had poor remaining quality but was not retracted, it was augmented with an Achilles tendon allograft. For allograft augmentation, the Achilles tendon was sized and placed on the dorsal aspect of the native hamstring tendon to maintain native tendon to native bone healing. Krackow and mattress sutures were utilized to “unitize” the augmented allograft and the native tendon. Then sutures from the suture anchors were used to secure both the tendons and the allograft as one unit (Figure 4).

Proximal hamstring repair to the ischial tuberosity with the addition of an Achilles tendon allograft to augment and strengthen the repair.
If the tendon was of poor quality and retracted after all releases were performed, then an interposition construct was created. The size of the spanning defect was measured with the knee and the hip in full extension. For allograft sizing of the Achilles interposition graft, 5 cm was added to the length of the measured gap for distal overlap with the native tendon, and 2 cm was added proximally for attachment to the ischial tuberosity bone with suture anchors (overall length of allograft = length of gap in full extension + 5 cm + 2 cm). The allograft was then sutured to the native distal tendon stump with Krackow and mattress sutures (Figure 5). The sutures from the suture anchors in the ischial tuberosity were then placed solely in the allograft.

Proximal hamstring repair with an Achilles allograft to interpose the gap between the retracted proximal hamstring and the ischial tuberosity.
All of these constructs were tied to the knee in a flexed position. Afterwards, the sciatic nerve and its branches were carefully examined, thorough irrigation was performed, and the subcutaneous tissues and skin were closed in a layered fashion. A locked hinged knee brace was applied with the knee in 90° of flexion.
Postoperative rehabilitation included 6 weeks in a hinged knee brace with the knee flexed at 90°. Lovenox was used for deep vein thrombosis prophylaxis while the patient was in the knee brace. At 3 weeks, passive range of motion was permitted from full to 80° of flexion. After 6 weeks, the knee brace was discontinued, and patients were permitted to begin weightbearing as tolerated. Progressive strengthening was then allowed, and return to full activities was anticipated at 16 to 24 weeks with the guidance of a physical therapist.
Statistical Analysis
A Shapiro-Wilk test was performed to determine normality for all patient characteristic variables and outcome scores. Data were extracted using continuous variables, with normality reported as the mean ± standard deviation, and non-normality reported as the median with interquartile ranges (IQR). Categorical variables were reported as frequencies with percentages. Differences between continuous variables were evaluated either using a Student t test or a Mann-Whitney U test. Differences between categorical variables were assessed using a χ2 (chi-square) test or a Fisher exact test. P < .05 was considered statistically significant.
Results
Allograft Augmentation
The demographic characteristics of patients who underwent allograft augmentation, as well as their matched controls who underwent primary proximal hamstring repair, are listed in Table 1. There were no significant differences in age, sex, or BMI between cohorts, supporting satisfactory matching. The median follow-up of the augmentation cohort was 1.9 [IQR, 0.8-3.9] years, while it was 3.2 [IQR, 1.6-5.1] years for the primary repair cohort (P = .182). Additionally, 42% (8/19) of patients in the augmentation cohort had undergone a previous proximal hamstring repair, with 1 patient having undergone 2 previous repairs. The time from injury to surgery was significantly longer for the augmentation cohort compared with those who underwent primary repair (6.4 [IQR, 4.9-12.1] vs 1.3 [IQR, 0.9-5.0] months; P < .001). All patients (19/19) in the allograft cohort had chronic tears, compared with 44.7% (17/38) of the primary repair cohort (P < .001).
Demographic Characteristics Patients Who Underwent Proximal Hamstring Repair With Allograft Augmentation and Matched Controls Who Underwent Primary Proximal Hamstring Repair a

Data are presented as mean ± SD, median [IQR], %, or n. The bold P values indicate statistical significance. This table shows characteristics of patients who underwent proximal hamstring repair with allograft augmentation and matched controls who underwent primary proximal hamstring repair. BMI, body mass index; IQR, interquartile range.
Functional outcomes—including Tegner Activity Scale (3 [IQR, 1.5-4] vs 3 [IQR, 3-5.8]; P = .155), pain with activity (47.4% vs 23.7%; P = .131), and muscle spasms (36.8% vs 26.3%; P = .609)— were not statistically different between the augmentation and primary repair cohorts. The augmentation cohort had a significantly lower LEFS (64 [IQR, 49-69.50] vs 75 [IQR, 70.3-79]; P < .001) and were significantly more likely to have pain with sitting (52.6% vs 21.1%; P = .034) than the primary repair cohort (Table 2). One patient in the allograft cohort underwent subsequent irrigation and debridement for infection at 3 months, while 2 patients in the primary repair cohort underwent subsequent irrigation and debridement at 3 weeks and 8 years.
Clinical Outcomes of Patients Who Underwent Proximal Hamstring Repair With Allograft Augmentation and Matched Controls Who Underwent Primary Proximal Hamstring Repair a

Data are presented as median [IQR] or %. Bold P values represent statistical significance. IQR, interquartile range; LEFS, Lower Extremity Functional Scale.
Allograft Interposition
The demographic characteristics of patients who underwent allograft interposition, as well as their matched controls who underwent primary proximal hamstring repair, are listed in Table 3. There were no significant differences in age, sex, or BMI between cohorts, supporting satisfactory matching. The mean follow-up for the interposition cohort was 3.5 ± 2.4 years, while it was 4.2 ± 2.5 years for the primary repair cohort (P = .227). One patient (5%) in the interposition cohort had undergone a previous primary proximal hamstring repair. The time from injury to surgery was significantly longer for the interposition cohort (17.8 [IQR, 9- 40] vs 1 [IQR, 0.5-3] months; P < .001). All patients (20/20) in the interposition cohort had chronic tears, compared with 32.5% (13/40) of the primary repair cohort (P < .001).
Demographics Characteristics of Patients Who Underwent Proximal Hamstring Repair With Allograft Interposition and Matched Controls Who Underwent Primary Proximal Hamstring Repair a

Data are presented as mean ± SD, median [IQR], or %. Bold P values represent statistical significance. BMI, body mass index; IQR, interquartile range.
There were no significant differences in clinical outcomes between the interposition and primary repair cohorts (Table 4). One patient in the interposition cohort underwent subsequent revision proximal hamstring repair with allograft interposition at 1.1 years, and 1 patient in the primary repair cohort underwent hardware removal 6 months after ischial tuberosity fracture repair.
Clinical Outcomes of Patients Who Underwent Proximal Hamstring Repair With Allograft Interposition and Matched Controls Who Underwent Primary Proximal Hamstring Repair a

Data are presented as median [IQR] or %. IQR, interquartile range; LEFS, Lower Extremity Functional Scale.
Discussion
In this study, we investigated the clinical outcomes of patients who underwent proximal hamstring repair with allograft augmentation or interposition and compared them with matched controls who underwent primary hamstring repair. We found that these procedures demonstrated favorable outcomes. However, these procedures resulted in poorer clinical outcomes compared with patients who underwent primary repair. This may have been due to multiple factors. Both allograft cohorts had a significantly longer time from injury to surgery, whereas a large proportion of patients in the augmentation cohort had undergone previous surgical intervention. Ultimately, this led to substantially greater muscle atrophy, scar tissue deposition, retraction, and increased pain with activity. Nevertheless, these reproducible techniques demonstrated satisfactory clinical outcomes and low reoperation rates for patients with irreparable proximal hamstring tears.
While the existing literature is limited, the available published studies have similarly demonstrated favorable outcomes after allograft interposition. For example, Rust et al 16 found that allograft interposition in 14 patients resulted in comparable outcomes to those who underwent primary repair for a chronic hamstring injury of more than 6 weeks. However, they had worse outcomes than those who underwent acute repair within 6 weeks of the injury. Notably, our interposition cohort had no significantly different outcomes compared with the matched primary repair cohort, which had a median time from injury to surgery of 1 month. Rust et al also demonstrated that a retraction of >5 to 6 cm was predictive of the need for allograft interposition. In our interposition cohort, all patients had a retraction of ≥7 cm with a median of 11.5 cm.
Marx et al 12 reported on 2 patients who underwent allograft interposition and found that, at a final follow-up of 2 years, both patients had returned to a more active lifestyle, with significant improvements in participation in sports and overall function. Notably, they considered allograft interposition to be a salvage procedure, as the patients did not fully recover to normal function and still exhibited hamstring weakness. Ebert et al 7 reported on 6 patients who underwent proximal hamstring interposition using distal hamstring autografts and found that at a 2-year follow-up, 83% of patients were satisfied with the results of the surgery, as well as their ability to return to recreational and sporting activities.
To our knowledge, Folsom and Larson 9 reported the only case of hamstring allograft augmentation in the literature. They examined 5 patients with chronic hamstring ruptures, 1 of whom was treated with an Achilles allograft augmentation, 3 were treated with interposition, and 1 was treated with primary repair. They found that 80% of these patients were able to return to sports, while 60% achieved a full recovery. In our study, the augmentation cohort had worse functional outcomes compared with the matched controls, which met the minimally clinically important difference for LEFS. 3 However, it is important to note that compared with the primary repair and interposition cohorts, almost half of the augmentation cohort underwent previous surgical intervention, with a majority undergoing concomitant sciatic nerve neurolysis.
Numerous studies in the literature have demonstrated that the timing of a repair can affect outcomes. Bowman et al. determined that patients with excellent or good results after surgical treatment had an average delay of 2.4 months to surgery. In contrast, patients with moderate or poor results had a mean delay of 11.7 months. 17 A separate study found that patients who underwent surgical repair ≥3 months after injury were 29 times more likely to experience a poor outcome. 17 A systematic review by Bodendorfer et al 4 demonstrated that patients who underwent repair within 2 months had higher patient satisfaction, less pain, greater strength, and better patient-reported outcome measures than those who underwent repair for a chronic tear. In contrast, a separate systematic review found no difference in outcomes based on time from injury to surgical repair. 19
Based on the findings in the present study as well as previous literature,9,16 for chronic tears of ≥6 months, we recommend having an allograft available. It is important to strengthen the repair in cases of poor tendon quality and significant retraction, as the risk of reoperation will be reduced and better outcomes will be achieved. For instance, only 1 patient from both allograft cohorts underwent subsequent revision due to a retear. In addition, among patients who underwent allograft interposition, only 1 had a previous surgery, resulting in comparable outcomes between the interposition and primary repair cohorts. Conversely, nearly half of the patients who received allograft augmentation had undergone previous surgery, potentially contributing to inferior outcomes compared with those who underwent primary repair.
Limitations
The present study has limitations. The sample sizes of the allograft cohorts were relatively small; however, these procedures are rare. Preoperative clinical outcome scores were not available, which would have permitted direct evaluation of the effect that these procedures had on patients’ functional status. Moreover, our institutions serve as tertiary referral centers, with numerous proximal hamstring repairs being performed annually by surgeons with substantial expertise for complex hamstring injuries. Therefore, these outcomes may not be generalizable to lower-volume orthopaedic surgeons. Last, our results are susceptible to the biases inherent in retrospective reviews—including not identifying all eligible patients and incomplete record-keeping.
Conclusion
Proximal hamstring repair for chronic tears with allograft augmentation or interposition is a reproducible procedure that results in satisfactory, yet poorer, clinical outcomes compared with patients who underwent primary hamstring repair. These procedures should be considered in symptomatic patients with chronic tears that are not amenable to primary repair.
Footnotes
Final revision submitted July 5, 2025; accepted July 26, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.J.S. has received consulting fees from DePuy Synthes Products, Inc. K.J.E. has received support for education from Goode Surgical and hospitality payments from Arthrex, Inc. M.H. has received support for education from Smith & Nephew, Inc, Arthrex, Inc, Medwest Associates, Foundation Medical, LLC, and Vericel; honoraria from Encore Medical, LP; hospitality payments from Orthalign and Stryker; a grant from Medical Device Business Services; and consulting fees from Vericel Corporation. A.J.K. has received nonconsulting fees from Arthrex, Inc; consulting fees from Arthrex, Inc, JFR Ortho, and Responsive Arthroscopy LLC; royalties from Arthrex, Inc; travel and lodging from Arthrex, Inc; and honoraria from JFR Ortho. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Mayo Clinic (23-008165).
