Abstract
Background:
Injections of compound betamethasone (BT) combined with lidocaine (LD) have been applied for the treatment of rotator cuff tendinopathy; however, their effects on human rotator cuff tendon cells remain unknown.
Purpose:
To explore the cytocompatibility of BT and LD on degenerative human rotator cuff tendon cells.
Study Design:
Controlled laboratory study.
Methods:
BT and LD were diluted in cell culture medium (Dulbecco’s modified Eagle medium) to match their common clinical concentrations, and degenerative rotator cuff tendon cells were cultured with the drugs for 24 hours. Cell morphology and density were monitored for 21 days after the treatment. To identify the safe concentrations, the viability, apoptosis, proliferation, and cytoskeleton of tendon cells were investigated. An enzyme-linked immunosorbent assay was performed to analyze the influence of the selected concentrations on extracellular matrix expressions.
Results:
When treated with the common clinical concentrations of 0.7 mg/mL BT and 0.5% LD, or their combination, tendon cells exhibited significant changes in cell morphology, along with reduced cell viability. Higher concentration, particularly LD concentration, exacerbated these side effects. In contrast, treatment with lower concentrations of BT, LD, or their combination did not induce notable cytotoxicity or alterations in cell morphology and extracellular matrix composition, indicating a potential safety threshold for clinical application. Compared with the effects of BT or LD alone, the combination of BT and LD enhanced cytotoxicity, which was primarily driven by LD. Based on in vitro cytocompatibility assays, the relatively safe concentrations were determined to be 0.175 mg/mL for BT, 0.05% for LD, and a combination of 0.175 mg/mL BT + 0.025% LD.
Conclusion:
BT and LD demonstrate concentration-dependent cytotoxicity in degenerative rotator cuff tendon cells, with the combined treatment of BT and LD enhancing this effect, wherein LD plays a predominant role. The relatively safe concentrations for BT, LD, and their combination resulted in only transient alterations, with complete recovery of cell morphology and viability observed.
Clinical Relevance:
This finding can serve as a potential reference for the safer clinical application of BT and LD, especially in the treatment of degenerative rotator cuff tendinopathy.
Tendinopathy is a degenerative condition caused by overuse, characterized by pain, swelling, and functional impairment.3,7,33 As one of the most common tendon diseases, 55 rotator cuff tendinopathy affects approximately one-third of individuals experiencing shoulder pain in their lifetime. 28 Glucocorticoids are commonly used in rotator cuff tendinopathy because of their short-term reduction in pain and improvement in function.11,29,33 However, studies have confirmed that these beneficial effects are transient and corticosteroids neither reverse degenerative tendon changes nor reduce long-term surgical intervention rates.29,34 When tendinopathy progresses to full-thickness rotator cuff tears, surgical repair is sometimes required to restore biomechanical integrity and improve function. 8 In this context, corticosteroid applications have expanded from nonoperative management to perioperative protocols. To alleviate postoperative pain and accelerate rehabilitation, glucocorticoids injection such as triamcinolone have sometimes been used after rotator cuff repair.19,27 Previous studies have shown that intra-articular glucocorticoid injections administered 4 to 8 weeks after rotator cuff repair surgery can reduce pain and improve the range of motion.19,26,27
Despite their effectiveness in pain relief,22,52 the clinical application of glucocorticoids is restricted by the suppression of inflammatory responses—a double-edged sword that may delay tissue repair.16,36,47,53 As a result, concerns about the potential negative effects of glucocorticoids on tendon healing have been raised.1,17,42 Animal experiments have demonstrated that repeated subacromial glucocorticoid injections can cause detrimental changes to rotator cuff structure 4 and highlighted the risk of tendon rupture,1,39 particularly after rotator cuff repair surgery.44,51,52
For rapid pain relief, glucocorticoids are usually applied in combination with local anesthetics, such as lidocaine (LD),12,15,46 but there is no consensus on ideal doses and corticosteroid type.12,25 Previous studies have indicated that LD may exacerbate the potentially harmful effects of glucocorticoids on tendon cells. 49 In China, compound betamethasone (BT) injection is a commonly used glucocorticoid to treat tendinopathy and joint adhesions or stiffness.5,21,54 Although several studies have examined the effects of corticosteroids and LD,2,9,51 the effects of BT injection on tendon cells, applied either alone or in combination with LD, are still unknown. Accordingly, the purpose of this study was to investigate the dose-dependent effects of BT and LD on the viability, apoptosis, and functional recovery of human degenerative rotator cuff tenocytes, with a focus on identifying thresholds for reversible cytotoxicity. This study hypothesized that BT and LD induce dose-dependent cytotoxicity in degenerative tenocytes, primarily through apoptosis and cytoskeletal disruption, but there exists a safe concentration to allow full functional recovery.
Methods
Study Design
An overview of the study design is illustrated in Figure 1. To evaluate the cytocompatibility of BT and LD with human degenerated rotator cuff tendon cells, the drugs were diluted with normal saline to the common clinical concentration (0.7 mg/mL BT, 0.5% LD, or their combination) and a lower concentration (0.35 mg/mL BT, 0.2% LD, or their combination) and applied to tendon cells for 24 hours.35,47 After this, cell morphology and density were observed during in vitro culture. Based on the poor cytocompatibility of the common clinical concentrations, tendon cells were treated with a concentration gradient of BT and LD. Comprehensive evaluations, including cell viability, apoptosis, proliferation, and cytoskeleton observation, were conducted to identify concentrations without significant side effects, which were defined as the safe concentrations. Finally, the production of extracellular matrix was analyzed to verify the influence of these safe concentrations.

An overview of the study design. BT, betamethasone; DMEM, Dulbecco’s modified Eagle medium; ELISA, enzyme-linked immunosorbent assay; LD, lidocaine.
Tendon Cell Culture
Degenerative rotator cuff tissues were harvested from 3 patients after obtaining informed donor consent, in compliance with national institutional ethical requirements. The ages of donors were 58, 60, and 61 years. Samples were taken from 2 men and 1 woman with degenerative rotator cuff tears requiring surgical treatment, excluding those with a smoking history, diabetes, or other systemic comorbidities. A degenerative tendon tissue sample measuring approximately 5 × 5 × 5 mm was focally harvested from the edge of the torn rotator cuff in each patient. To isolate the tendon cells, tissues were minced into small pieces, added to a digestive enzyme solution containing 3 mg/mL type 1 collagenase (Worthington) and 4 mg/mL neutral dispase (Sigma), incubated at 37°C for 2 to 3 hours, and then centrifuged at 1000 rpm for 5 minutes. After the removal of supernatant, Dulbecco’s modified Eagle medium with low glucose (DMEM-LG; Gibco) containing 10% fetal bovine serum (FBS; Gibco) was added to resuspend the sediment, which was then transferred to a culture flask and cultured in a CO2 incubator (37°C, 5% CO2). Cells from each patient were cultured and exposed to all test conditions separately. When the cells reached about 90% confluence, they were passaged at a ratio of 1:3, and cells at the fourth passage were used for the following experiments.
Cell Morphology Observation
Tendon cells of experimental groups were treated with dilutions of BT (Compound Betamethasone Injection, Schering-Plough Labo NV), 2% LD (Lidocaine Hydrochloride Injection, Hebei Tiancheng), or their combination. 50 The blank control group was maintained in normal culture medium without any drug exposure. All groups were prepared with DMEM-LG containing 10% FBS. After exposure to the drugs for 24 hours, 10 the medium was discarded, followed by rinsing 2 to 3 times with phosphate-buffered saline (PBS) and further culturing with DMEM-LG containing 10% FBS for 21 days. Continuous observation of cell morphology was conducted and recorded at key time points (2, 7, and 21 days) 20 using a phase-contrast microscope (Nikon).
Live/Dead Staining
Tendon cells were seeded on a 6-well plate at a density of 2 × 105 cells per well (3 in each group). Upon reaching confluence, the cells were exposed to different concentrations of drugs for 24 hours. Then, live/dead staining was performed using a Calcein-AM/PI Cell Viability Assay Kit (Keygen) according to the manufacturer’s protocol. The stained cells were observed using a fluorescence microscope (Eclipse Ti2; Nikon).
Cell Apoptosis
After treatment with drugs for 24 hours, tendon cells were detached, washed with PBS, and suspended in Annexin V Binding Solution (1×; Dojindo) at a concentration of 1 × 106 cells per microliter. Then, 5 µL of Annexin V FITC Binding Solution was added to 100 µL of the cell suspension, followed by the addition of 5 µL of propidium iodide solution (Dojindo). Then, the cell suspension was incubated at room temperature for 15 minutes in the dark. Finally, 400 µL of Annexin V Binding Solution (1×) was added, and cell apoptosis was detected within 1 hour using flow cytometry (CytoFLEX; Beckman). The data were analyzed with CytExpert software.
Cytoskeleton Staining
After incubation with drugs for 24 hours, cells were washed with PBS, fixed in 4% paraformaldehyde for 20 minutes, and permeabilized with 1% Triton X-100 for 5 minutes. Then, a phalloidin working solution (1:200; Sigma) was added to each sample and incubated for 30 minutes, followed by staining of the nuclei with DAPI (Beyotime). Then, the samples were observed using a laser scanning confocal microscopy (Nikon).
Cell Proliferation
Tendon cells were seeded in 96-well plates at a density of 3000 cells per well. After adhesion, cells were incubated with drugs for 24 hours, followed by culture with DMEM-LG containing 10% FBS. On days 1, 3, and 7 after the incubation with drugs, cell proliferation was measured by using a Cell Counting Kit-8 (CCK-8; Dojindo). Briefly, the cells were rinsed with serum-free DMEM-LG medium, and 300 µL of CCK-8 working solution (1:10) prepared according to the manufacturer’s instructions was added to each well. Optical density (OD) values at 450 nm were measured after the incubation at 37°C for 2 hours.
Enzyme-Linked Immunosorbent Assay
After drug treatment, the supernatant of cell cultures was collected on days 2, 7, and 21 and centrifuged at 3000 rpm for 20 minutes at room temperature. The concentrations of collagen type 1 (COL-1), collagen type 3 (COL-3), and tissue inhibitor of metalloproteinases 1 (TIMP1) were measured using an enzyme-linked immunosorbent assay (ELISA) kit (mlbio). All samples were measured at 450 nm, and the concentration of each sample was calculated according to the standard curves.
Statistical Analysis
Statistical analysis was performed using SPSS Version 25 (IBM). One-way analysis of variance (ANOVA) was used for data with a normal distribution, and Tukey multiple comparisons were used for the post hoc test. For data that did not satisfy the homogeneity of variance, Welch ANOVA and Brown-Forsythe ANOVA were used, followed by the Dunnett T3 or Tamhane T2 tests for post hoc multiple comparisons. P values <.05 were considered significant.
Results
Effect of Common Clinical Concentration of BT and LD on Tendon Cells
After treatment with BT at the common clinical concentration (0.7 mg/mL) for 24 hours, tendon cells exhibited irregular morphology with an elongated cell morphology shape on day 2, along with a significant decrease in cell density. On days 7 and 21, a noticeable recovery in cell density was observed, although cell morphology remained different from that of untreated degenerative rotator cuff cells. When treated with a lower concentration of BT (0.35 mg/mL), changes in cell morphology and density were less pronounced. Additionally, an increase in cell density was noted on days 7 and 21 for both concentrations (Figure 2).

Continuous observation of the morphology of tendon cells pretreated with the common clinical concentrations of (A) betamethasone (BT), (B) lidocaine (LD), or (C) their combination for 24 hours. Scale bar, 500 μm.
In contrast, LD at the common clinical concentration (0.5%) or a lower concentration (0.2%) induced the detachment of most cells from the substrate on day 2, and the remaining cells exhibited abnormal morphology. Particularly, the lower concentration resulted in a greater number of adherent cells. However, on day 21, neither group showed recovery of cell density (Figure 2), indicating that both concentrations were toxic to tendon cells.
When pretreated with a combination of BT and LD at the common clinical concentration (0.7 mg/mL BT + 0.5% LD) or a lower concentration (0.35 mg/mL BT + 0.2% LD), a substantial number of cells underwent shedding, resulting in a significant reduction in cell volume and density. On day 2, the lower-concentration group exhibited a higher cell density. However, neither group showed any increase in cell density at later time points. On day 21, only residual extracellular matrix and cell debris were observed in both groups, with no normal tendon cells present (Figure 2). These results revealed that both concentrations were cytotoxic to tendon cells, highlighting the need to screen for safe concentrations.
Safe Concentrations of BT and LD for Tendon Cells
BT exhibited a concentration-dependent effect on tendon cell viability, with higher concentrations resulting in an increased number of dead cells. Notably, the clinically relevant concentration (0.7 mg/mL) resulted in considerable cell death and detachment (Figure 3A). Similarly, the influence of LD on tendon cell viability was also concentration dependent. In the 0.5% LD and 0.2% LD groups, the majority of tendon cells were either dead or detached from the cell culture flask. When the concentration was <0.2%, there was a notable improvement in cell viability (Figure 3B). The combination of BT and LD also exerted a concentration-dependent effect on cell viability, showing a more pronounced cytotoxic effect than BT alone (eg, 0.7 mg/mL BT + 0.5% LD vs 0.7 mg/mL BT) or LD alone (eg, 0.7 mg/mL BT + 0.1% LD vs 0.1% LD) (Figure 3C).
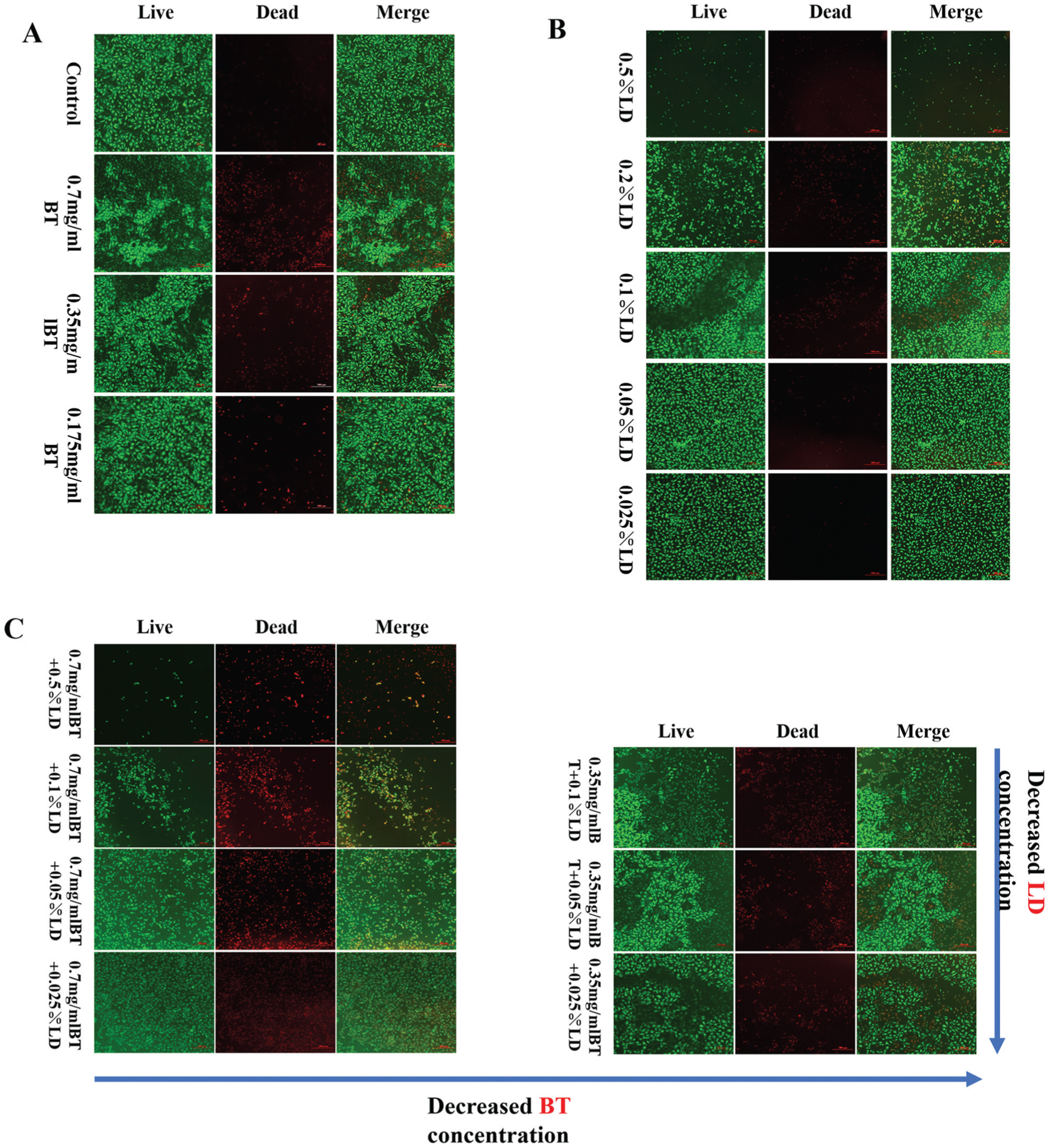
Live/dead staining of tendon cells treated with different concentrations of (A) betamethasone (BT), (B) lidocaine (LD), or (C) their combination for 24 hours. Scale bar, 500 μm.
After treatment with BT, the apoptosis of tendon cells varied with drug concentration (Table 1). All BT groups showed a significantly higher apoptosis rate than that of the control group (P < .05). Particularly, the 0.7 mg/mL BT group exhibited a higher apoptotic rate than that of the 0.125 mg/mL BT group (P < .01), but there was no statistical difference between the adjacent concentrations (ie, 0.35 mg/mL BT vs 0.125 mg/mL BT; P > .05) (Figure 4A). In the LD groups, the cell apoptosis rates were significantly higher than those of the control group (P < .05). The 0.2% LD group was higher than the other concentrations (P < .05), while no obvious difference was observed between the 0.35 mg/mL BT group and 0.125 mg/mL BT group (P > .05). At the same concentration, LD showed a higher apoptotic rate than BT. Particularly, the difference between the 0.7 mg/mL BT group and the 0.2% LD group was significant (11.24 ± 0.89% vs 23.86 ± 1.65%; P < .0001) (Figure 4B), indicating that the cytotoxic effect of LD was greater than that of BT.
Results of Cell Apoptosis a

Values are expressed as mean ± SD. BT, betamethasone; LD, lidocaine.
Comparison versus control group (P < .05).
Comparison versus 0.2% LD group (P < .05).
Comparison versus 0.7 mg/mL BT + 0.1% LD group (P < .05)
Comparison versus all other groups (P < .05).

Apoptosis of tendon cells treated with different concentrations of (A) betamethasone (BT), (B) lidocaine (LD), or (C) their combination. G1: 0.7 mg/mL BT + 0.2% LD; G2: 0.35 mg/mL BT + 0.2% LD; G3: 0.7 mg/mL BT + 0.1% LD; G4: 0.35 mg/mL BT + 0.1% LD; G5: 0.7 mg/mL BT + 0.05% LD; G6: 0.35 mg/mL BT + 0.05% LD. ns, not significant (P > .05). *P < .05.
When BT and LD were used in combination, the apoptosis rates of the 0.7 mg/mL BT + 0.2% LD group and the 0.35 mg/mL BT + 0.2% LD group were much higher than those of the other groups (P < .05). However, there was no significant difference between these 2 groups (P > .05). Additionally, no statistical difference was observed among the other groups (P > .05). Comparing the results of the 0.7 mg/mL BT + 0.2% LD, 0.7 mg/mL BT + 0.1% LD, and 0.7 mg/mL BT + 0.05% LD groups, it is evident that variation in LD concentration had a pronounced effect on cell apoptosis. When the concentration of LD was ≤ 0.1%, the apoptosis rate decreased significantly. However, changing the concentration of BT did not have a similar effect on the cell apoptosis rate. For instance, the apoptosis rates of the 0.7 mg/mL BT + 0.2% LD group and the 0.35 mg/mL BT + 0.2% LD group were comparable (P > .05) (Figure 4C). These results revealed that a combination of BT and LD promoted the apoptosis of tendon cells, which was driven primarily by LD.
After treatment with BT, the cytoskeleton of tendon cells underwent contraction, resulting in a decreased cell volume and morphological changes, characterized by irregular and filamentous alterations in the cytoskeleton. As the concentration of BT increased, the cytoskeletal changes became more pronounced. LD induced an overall contraction of the cytoskeleton, with some cells even experiencing disintegration, although the majority of cells maintained the integrity of their cytoskeleton. Changes in LD concentration exerted a significant effect on the cytoskeleton. When BT and LD were administrated in combination, the changes in the cytoskeleton were more pronounced, characterized by varying degrees of cell contraction and disintegration. Although changes in either BT concentration (eg, 0.35 mg/mL BT + 0.05% LD group vs 0.175 mg/mL BT + 0.05% LD) or LD concentration (eg, 0.175 mg/mL BT + 0.05% LD group vs 0.175 mg/mL BT + 0.025% LD group) resulted in cytoskeleton alterations, the influence of LD concentration was more prominent (Figure 5).

Cytoskeleton of tendon cells treated with different concentrations of betamethasone (BT), lidocaine (LD), or their combination for 24 hours. Scale bar, 100 μm.
According to the cell proliferation results, significant differences were observed between the control group and the 0.7 mg/mL BT group on days 1 and 7, further confirming the obvious cytotoxicity of the common clinical concentration of BT. Notably, the OD value of the 0.175 mg/mL BT group was significantly higher than those of the 0.7 mg/mL BT and 0.35 mg/mL BT groups on day 7, indicating that the 0.175 mg/mL BT group possessed better cytocompatibility (Figure 6A).

The proliferation of tendon cells after pretreatment with (A) betamethasone (BT), (B) lidocaine (LD), or (C) their combination for 24 hours. G1: 0.7 mg/mL BT + 0.05% LD; G2: 0.35 mg/mL BT + 0.05% LD; G3: 0.175 mg/mL BT + 0.05% LD; G4: 0.175 mg/mL BT + 0.025% LD. *P < .05; **P < .01; ***P < .001; ****P < .0001. OD, optical density.
Similarly, when compared with the control group, the 0.2% LD and 0.1% LD groups showed reduced proliferation at all time points. Among the LD groups, the concentration of 0.05% exhibited better proliferation than the other 2 groups (Figure 6B). As shown in Figure 6C, the combination of BT and LD at high concentrations, such as the 0.7 mg/mL BT + 0.05% LD group and the 0.35 mg/mL BT + 0.05% LD group, showed significantly reduced cell proliferation on day 7. Compared with the 0.7 mg/mL BT + 0.05% LD group, the 0.175 mg/mL BT + 0.025% LD group showed much better proliferation, especially at day 1 (P < .05), indicating the good cytocompatibility of this group.
Based on the results of tendon cell viability, apoptosis, proliferation, and cytoskeleton observation, the following concentrations were selected for further analysis: 0.175 mg/mL BT, 0.05% LD, and 0.175 mg/mL BT + 0.025% LD.
Effect of BT and LD at Safe Concentrations on Tendon Cells
To explore whether the selected concentrations were safe for rotator cuff tendon cells, the cells were treated with drugs for 24 hours, followed by continuous observation of cell morphology during subsequent culture in normal cell culture medium for 21 days. On days 2, 7, and 21, the morphology of cells was recorded. As shown in Figure 7A, the morphology of tendon cells did not change significantly after treatment with the drugs. Furthermore, the cell density increased gradually in each group, revealing good cytocompatibility of these concentrations.

(A) Morphology of tendon cells after pretreatment with the selected concentrations of drugs for 24 hours. Scale bar, 500 μm. (B) Level of extracellular matrix (collagen type 1 [COL-1], collagen type 3 [COL-3], tissue inhibitor of metalloproteinases 1 [TIMP1]) produced by tendon cells after pretreatment with the selected concentrations of drugs. BT, betamethasone; LD, lidocaine.
After observing cell morphology, the production of extracellular matrix was determined. As shown in Figure 7B, 0.175 mg/mL BT treatment exhibited a gradual increase in the total amount of COL-1, with no significant difference between the BT group and the control group (P > .05). Similarly, 0.05% LD treatment showed a comparable COL-1 amount to that of the control group (P > .05). Additionally, the 0.175 mg/mL BT + 0.025% LD group did not show any significant alterations in total COL-1 levels, remaining similar to the control group at each time point (P > .05).
In addition to COL-1, the total amounts of COL-3 and TIMP1 remained relatively stable in each group. Notably, no statistical differences were observed among the control, 0.175 mg/mL BT, 0.05% LD, and 0.175 mg/mL BT + 0.025% LD groups at all time points (P > .05) (Figure 7B). These results clearly demonstrate that the selected concentrations were safe for rotator cuff tendon cells.
Discussion
This study observed that exposing degenerative rotator cuff tendon cells to a common clinical concentration of BT reduced cell viability and induced morphological changes that remained irreversible even after prolonged culture. Previous studies have shown that glucocorticoids can elicit acute deleterious effects on healthy tendons 6 and inhibit the proliferation of tendon cells in a dose-dependent manner. 57 Triamcinolone acetonide, another glucocorticoid in the same category as BT, exerted side effects on tendon cells and changed their morphology. When treated with high concentrations of triamcinolone acetonide, changes in the tendon cell morphology were irreversible; however, when exposed to low concentrations of triamcinolone acetonide, tendon cells could recover the normal morphology. 20 This is similar to the observation in this study that rotator cuff tendon cells treated with the safe concentration of BT exhibited normal morphology.
The toxicity of LD on tendon cells is influenced by drug dosage, concentration, and duration of exposure.23,49 Similarly, the results demonstrate that the common clinical concentration of LD resulted in substantial shrinkage and detachment of rotator cuff tendon cells, showing a sharp decline in cell viability. When combined with BT, LD further exacerbated BT’s deleterious effects on tendon cells, consistent with previous findings. 58 This synergistic cytotoxicity likely arises from the convergence of 2 distinct injury pathways. LD exposure impairs mitochondrial function, causing mitochondrial DNA damage, ATP depletion, 18 and dysregulation of cell cycle proteins and collagen expression, 10 while glucocorticoids suppress the transcriptional programs required for cell survival and extracellular matrix synthesis,56,57 ultimately diminishing tenocyte proliferation and matrix production. As a result, energy-depleted cells are unable to activate growth and repair pathways that are inhibited by steroids, rendering them particularly vulnerable to injury.
After analyzing the critical time points at which notable changes in tendon cell morphology and density occurred, it was observed that the potential harmful effects of the drugs on cells can be determined 1 to 2 days after administration, the reversibility of the drug’s effects can be assessed 7 to 12 days posttreatment, and any long-term changes in cell status compared with pretreatment can be determined at approximately 21 days after administration. These findings are quite similar to those of a previous study in which the time-dependent effects of triamcinolone acetonide on human rotator cuff–derived cells were investigated. 20
Animal models have demonstrated that corticosteroid treatment compromises the biomechanical properties of tendons,24,32,38 which may be related to alterations in the extracellular matrix.14,43 Additionally, previous studies have reported that corticosteroids modify the extracellular matrix composition of tenocytes,41,45 with more pronounced effects observed at higher doses and shorter intervals of administration. 41 Similarly, glucocorticoids have been reported to significantly inhibit the expression of COL-1 in tendon cells while increasing the expression of TIMP1, suggesting a reduced capacity for tendon repair and inferior mechanical performance. 51 In this study, to verify the safety of the selected concentrations of drugs, the total amounts of COL-1, COL-3, and TIMP1 were measured. The results demonstrate that, after treatment with the selected concentrations of drugs, the expression levels of these proteins were stable, further confirming the safety of these concentrations at the molecular level.
Interestingly, in clinical practice, the common concentration of BT has not been shown to cause significant structural or functional damage to the rotator cuff tendons.13,30 This phenomenon may be attributable to the dilution by synovial fluid31,37,48 or absorption of the medications into the bursa, bloodstream, and so forth. However, the exact cause of this phenomenon remains unclear. In cases of tendon pathology secondary to adhesive capsulitis, inadequate dilution of the injectant can result in BT and LD doses exceeding safe concentrations, which may adversely affect tendon healing.11,40 Therefore, diluting BT or LD to a safe concentration may be warranted, but further clinical studies are required to verify the concentration.
Limitations
This study has certain limitations, including the possibility that the use of multiple patients for harvesting tissue impacted the results. The patients in this study ranged in age from 58 to 61 years, which may not be representative of other age groups. Larger sample sizes and a range of age groups should be included in future studies. Second, quantitative assessments of cell density or percentage cell viability at common concentrations were not performed to more accurately evaluate drug-induced cytotoxicity. Furthermore, because results from in vitro studies may not directly translate to in vivo or clinical contexts, future in vivo research is warranted. Specifically, appropriate animal models should be incorporated to validate the safe concentrations of BT, LD, and their combinations for therapeutic effect in tendinopathy.
Conclusion
BT and LD, at clinically relevant concentrations, induced notable morphological changes in rotator cuff tendon cells and significantly reduced their viability in vitro. Adverse effects increased with higher concentrations, with LD demonstrating significant cytotoxicity even at low concentrations. When BT was applied in combination with LD, the cytotoxic effects were additive, with the concentration of LD playing a more critical role in the combined treatment. Safe concentrations were determined to be 0.175 mg/mL for BT, 0.05% for LD, and 0.175 mg/mL BT combined with 0.025% LD for their combination. These concentrations preserved the normal morphology of tendon cells and supported the synthesis of extracellular matrix.
Footnotes
Final revision submitted June 2, 2025; accepted June 24, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by the National Natural Science Foundation of China (82072514, 82272569), the Tibet Autonomous Region Science and Technology Plan Joint Funding Project (XZ202201ZY0041G), and the Key Program of the Science and Technology Department of Sichuan Province, China (2024YFFK0299). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
