Abstract
Background:
Spiked ligament staples (SLSs) and biocomposite suture anchors (BSAs) are commonly used onlay fixation devices for modified Lemaire lateral extra-articular tenodesis (LET). However, hardware prominence, tunnel penetration, footprint size, and irritation are documented issues. Knotless all-suture anchors (ASAs) are a promising alternative that mitigates these issues.
Purpose:
To compare the biomechanical behavior of LET using ASAs with SLSs and BSAs under simulated anterolateral forces.
Study Design:
Controlled laboratory study.
Methods:
Human iliotibial band grafts were harvested, whipstitched, and fixed onto fresh-frozen porcine femurs using ASAs, SLSs, or BSAs (n = 6/group). Each femur was placed in a custom fixture at the base of an electromechanical testing machine, oriented at 60° with iliotibial band grafts parallel to the load axis. Cyclic loading and load-to-failure testing were performed. Five millimeters of displacement was determined as the critical threshold for failure and survivorship analysis. Biomechanical outcomes included cyclic displacement, survivorship, and ultimate load.
Results:
Cyclic displacement values after all load ranges were 4.25 ± 0.28 mm, 3.94 ± 0.71 mm, and 8.14 ± 1.68 mm for ASA, SLS, and BSA, respectively. No significant differences in cyclic displacement were found between ASA and SLS, whereas significant differences were found in each load range between BSA and both ASA and SLS (P < .05). Survivorship was 100% for ASA, 83% for SLS, and 0% for BSA. The ultimate loads were 257 ± 40 N, 300 ± 47 N, and 205 ± 24 N for ASA, SLS, and BSA, respectively; only SLS and BSA were significantly different (P = .002).
Conclusion:
ASA onlay fixation for LET demonstrated comparable displacement to SLS and significantly lower displacement to BSA with greater survival during cyclic loading. ASA had comparable failure loading, with all groups exceeding the native anterolateral ligament failure load requirements. Therefore, ASA is a biomechanically viable alternative to SLS and BSA for iliotibial band fixation during LET.
Clinical Relevance:
ASAs are a biomechanically viable alternative to spiked staples and biocomposite suture anchors for LET onlay fixation, potentially mitigating complications associated with conventional techniques.
Keywords
As the incidence of anterior cruciate ligament (ACL) reconstruction (ACLR) has increased, there has been a concomitant rise in the number of revision ACLR procedures. 19 Of the numerous predictors for ACLR failure, a residual positive pivot shift has been consistently associated with failure leading to revision surgery.1,3 This is further supported by clinical studies reporting higher revision rates for patients returning to pivoting sports such as soccer and basketball. 9 Numerous adjustments to surgical techniques have been proposed to address the problem of persistent rotatory laxity after ACLR, including different graft choices, more anatomic placement of tunnels, and double-bundle reconstruction, but the improved biomechanical rotatory control that these techniques offer is not substantial enough and has not resulted in improved clinical outcomes.23,39
Lateral extra-articular tenodesis (LET) has re-emerged as a popular adjunct procedure for addressing residual anterolateral rotatory instability, with a recent study revealing increased adoption by the ACL Study Group from 2016 to 2023 in revision and primary ACLR.3,8,9,12,24,32 Biomechanically, LET has been convincingly demonstrated to reduce anterolateral rotatory laxity and offload the ACL graft, restoring normal knee stability.7,21,28,34,38 Clinically, LET has been shown to reduce revision and rerevision in both the adult and the adolescent populations, particularly with soft tissue grafts.4,9,29,32
Numerous LET techniques have been described, with onlay staple fixation being the most common and utilized in the Stability I trial.9,14,17 While both cost-effective and clinically validated, the use of a staple for fixation can lead to hardware irritation on the iliotibial (IT) band, and, more concerningly, staple penetration into the femoral ACL tunnel. 25 More recently, other techniques have been described, including inlay techniques with interference screws, onlay techniques with suture anchors, and techniques without additional anchors. Each of these presents unique challenges, including the risk of fixation loss with less stable fixation, and the risk of tunnel convergence with inlay techniques.13,36,37 A potential advantage of an onlay, all–suture anchor (ASA) technique is the possibility of secure fixation, while avoiding some of the challenges proposed by other published methods, including hardware irritation or tunnel penetration. Accordingly, the goal of the present study was to compare the biomechanical behavior of an onlay, ASA LET with that of onlay spiked ligament staple (SLS) and onlay biocomposite suture anchor (BSA) technique. We hypothesized that ASA would demonstrate comparable fixation strength with SLS and BSA with comparable survival rates under cyclic loading.
Methods
Specimen Preparation
Fifteen fresh-frozen male human cadaveric knees (mean age, 63 years; range, 51-71 years) were used to harvest IT band grafts, sectioned to 1 cm–wide by 15 cm–long strips with a scalpel. 30 The knees were set out to thaw at room temperature for approximately 24 hours, and whole IT band tendons were harvested, individually wrapped in saline-soaked gauze, and placed into plastic bags before storing them in a freezer at −20°C. Tendons were randomly selected on test days and thawed in a warm water bath for 30 to 60 minutes before sectioning off central strips. An additional strip was sectioned out of 3 of the specimens to obtain a total of 18 grafts. Afterward, each tendon strip was individually whipstitched using 0.9-mm SutureTape (Arthrex Inc). The first pass was made 40 mm distal to the graft end, and 7 additional passes were made proximally, 5 mm apart in a staggered fashion, before a final locking pass was made to complete IT band graft preparation.
Eighteen fresh-frozen porcine femurs (Animal Biotech Industries Inc) were used for this experiment, considering the accessibility and homogeneity of porcine specimens, along with their conventional use in biomechanical studies to simulate human knees.6,10,26 The femurs were thawed at room temperature and dissected of all soft tissue. Each femur was cut approximately 7 cm from the proximal trochlear edge, and 2 inch–diameter PVC was used to pot femurs 2 inches deep in resin. Femoral condyles were kept hydrated with saline while the resin set. After setting, potted femurs were wrapped in saline-soaked gauze and stored in a freezer at −20°C until test day, during which they were thawed in a warm water bath for 30 to 60 minutes.
The origin of the lateral collateral ligament (LCL) was marked and selected as the fixation point for each specimen. LET procedures traditionally fix the graft proximal and posterior to the LCL’s origin in humans. 16 However, considering the porcine nature of the specimens, the exact location should not affect the results, and a consistent location was prioritized. The LCL origin was identified by a prior porcine dissection, and it was found to be approximately 1.5 cm anterior and 0.5 cm distal to a depression found on the lateral side of the femur (Figure 1). Additionally, a reference line was marked on the lateral face, beginning at the point of fixation and ending at the edge of the femoral condyle, and angled at 60° with respect to the femur’s longitudinal axis. This line served to guide the placement of the IT band during fixation, ensuring consistent and proper alignment for testing.

The porcine lateral collateral ligament was dissected and its origin was marked. The origin was recorded with respect to a lateral depression on the femur with (A) anterior and (B) distal measurements to establish a consistent location for fixation.
After marking the femur, a bone quality measurement tool (OsteoProbe; Active Life Scientific Inc) was used to confirm homogeneity across all specimens.5,31 The device provides a score (bone material strength index value [BMSi]) based on the bone’s ability to resist microscopic challenge, with higher values indicating greater bone quality.18,20,33 After securing each femur in a vice, 5 indentations were made around the point of fixation, and on a reference block, to generate respective scores per the manufacturer’s instructions, completing porcine femoral preparation.
Onlay Techniques
Three different onlay fixation devices were tested for this study: 2.6-mm double-loaded knotless ASA (FiberTak; Arthrex Inc; n = 6), 8.0 mm–wide SLS (Arthrex Inc; n = 6), and 3.0×14.5–mm BSA (SutureTak; Arthrex Inc; n = 6). The ASA was secured into the femur per manufacturer's instructions, and the distal end of a whipstitched IT band was then passed through both ASA loops from anterior to posterior, ensuring IT band alignment with the 60° line marked on the femur (Figure 2A). Both loops were individually cinched 30 mm from the proximal end of the graft, over the third whipstitch, and a digital force gauge (Mark-10) was used to ensure a minimum of 120 N for each cinch (122.5 ± 2.1 N) to properly set the anchor and promote adequate anchor bunching against the cortical layer. Five alternating half-hitch knots were tied over the cinches using the trailing suture tails from the ASA. For SLS samples, whipstitched IT bands were placed along the marked femoral line and secured orthogonally to that line over the LCL origin, fixing staples 30 mm from the proximal end of the graft until they were flush with the bone (Figure 2B). For BSA samples, suture anchors were secured into the LCL’s origin per manufacturer instructions, resulting in 2 suture tails exiting the fixation point (Figure 2C). Whipstitched IT bands were placed along the marked femoral line, and 5 alternating half-hitch knots were tied 30 mm from the proximal end of the graft using BSA suture tails. The order in which the devices were fixed onto the femurs was randomized on test day.

Onlay fixation for the (A) all-suture anchor, (B) spiked ligament staple, and (C) biocomposite suture anchor techniques. The grafts were cortically fixed on the femur (1), then folded over the repair and sutured onto themselves for final fixation (2).
Final preparation for all constructs occurred on an electromechanical testing machine (ElectroPuls E10000; Instron), where the grafts were held at 20 N of tension for 10 minutes. During this time, the proximal ends of the grafts were flipped over respective fixation devices and sutured back onto themselves by passing each trailing SutureTape tail through the distal portions of the grafts individually before tying 5 alternating half-hitch knots to complete the constructs.
Biomechanical Testing
Each femur was placed into a custom fixture, oriented at 60° (±1°) to simulate a common flexion angle during LET graft fixation. 14 The fixutre was clamped to the base of the Instron, equipped with a 1-kN load cell (Figure 3A). IT bands were aligned parallel to the central load axis, simulating the worst-case scenario, and clamped with 50 mm of exposed tendon length from the implant using a hanging vise and custom, 3-dimensional printed clamps (Figure 3B). After pretensioning and final preparation of the constructs, specimens were cyclically loaded through the following load ranges for 100 cycles, each, at 0.5 Hz: 10 to 50 N, 10 to 75 N, 10 to 100 N, and 10 to 125 N. The rationale for testing to 100 cycles per load range was based on the principle that the majority of the elongation should occur within this range, with progressively smaller differences in elongation as the cycle count approaches and exceedes 100. 35 The load ranges were based on anterolateral ligament (ALL) forces during squat and gait (ACL-intact) and LET graft force after ACLR.15,22 After cyclic loading, samples were brought down to 10 N before loading to failure at 25 mm/min. 10 Five millimeters of cyclic displacement, based on preliminary testing and postoperative knee kinematics after ACLR with LET, was chosen as the critical threshold for failure and survivorship analysis. 2 Biomechanical outcomes included cyclic displacement (measured after each load range), survivorship, ultimate failure, and failure mode.

Final construct for all–suture anchor group. (A) All constructs were placed into a custom fixture with the iliotibial band graft flipped distally and sutured back onto itself. (B) Key features for the biomechanical setup are also denoted.
Statistical Methods
A power analysis for cyclic displacement was performed based on results from Glasbrenner et al, 10 revealing a minimum sample size of 4 to yield 80% power in detecting a 3-mm minimum difference between group means (α < .05). Mean and standard deviation values are used to describe all data. One-way analysis of variance (ANOVA) with a post hoc Tukey test was used for data that passed equal variance, whereas Welch ANOVA with a post hoc Games-Howell test was used for data that failed equal variance (P < .05). Multiple pairwise log-rank tests were performed to determine significance relating to survivorship among groups with a Bonferroni correction applied to reduce type 1 error (P < .02). Statistical analyses were performed using SigmaPlot (Version 14.0; Systat Software Inc), Minitab (Version 22; Mintab LLC), and JMP (Version 18; SAS Institute Inc).
Results
The mean BMSi values for ASA, SLS, and BSA femurs were 46.5 ± 6.0, 42.7 ± 6.9, and 41.1 ± 6.5, respectively, with no significant differences found among the groups.
Cyclic Displacement
Cyclic displacement values after all load ranges were 4.25 ± 0.28 mm, 3.94 ± 0.71 mm, and 8.14 ± 1.68 mm for ASA, SLS, and BSA, respectively. There were no statistically significant differences in cyclic displacement found between ASA and SLS after each load range (Δ = 0.31 mm; 95% CI, −0.62 to 1.25 mm, after 10-125 N load range) whereas significant differences were found between BSA and both ASA (Δ = 3.89 mm; 95% CI, 1.66 to 6.11 mm, after 10-125 N load range) and SLS (Δ = 4.20 mm; 95% CI, 1.98 to 6.42 mm, after 10-125 N load range) after each load range (P < .05 all comparisons) (Figure 4).
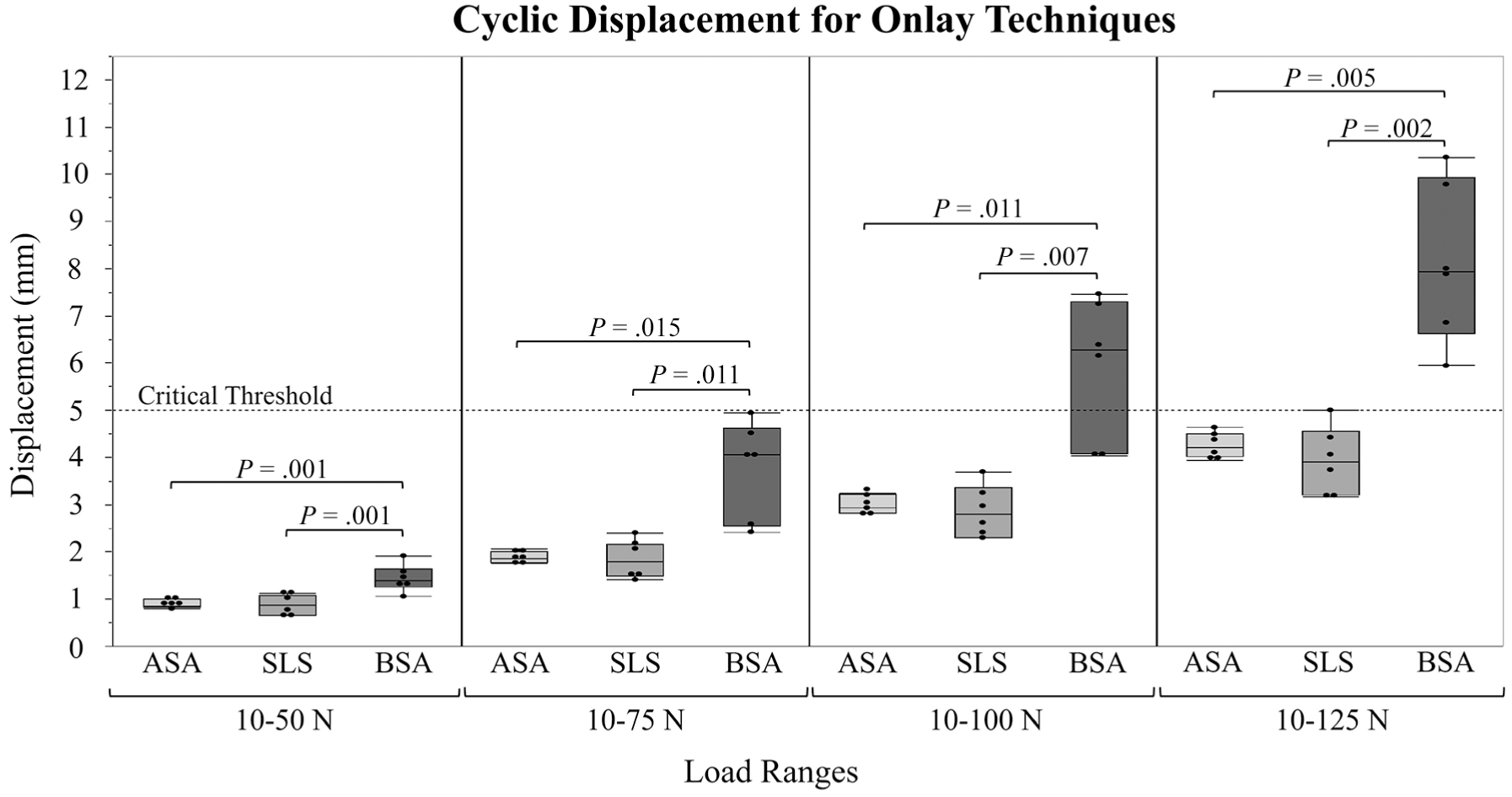
Box plots of the cyclic displacement for each device (all-suture anchor [ASA]; spiked ligament staple [SLS]; biocomposite suture anchor [BSA]) after each load range with critical threshold and respective P values denoted.
Survivorship
Survivorship, defined as cyclic displacement below a threshold of 5.00 mm for each load cycle, was 100% for all 3 groups at 10 to 50 N, 100% for all 3 groups at 10 to 75 N, 100% for ASA and SLS and 67% for BSA at 10 to 100 N and 100% for ASA, 83% for SLS, and 0% for BSA at 10 to 125 N (Figure 5). Based on the pairwise log-rank tests between techniques, with Bonferroni correction applied (P < .02), there was no significant difference between ASA and SLS; however, there was a significant difference between BSA and both ASA (P < .001) and SLS (P = .002).

Kaplan-Meier curves for each device (all-suture anchor [ASA]; spiked ligament staple [SLS]; biocomposite suture anchor [BSA]) after each load range. The final survival probability for ASA, SLS, and BSA were 100%, 83%, and 0%, respectively.
Ultimate Load and Failure Modes
The ultimate loads were 257 ± 40 N, 300 ± 47 N, and 205 ± 24 N for ASA, SLS, and BSA, respectively, with only SLS and BSA demonstrating a significant difference (Δ = 95 N; 95% CI, 38-153 N; P = .002) (Figure 6). All failure loads exceeded the ALL’s failure load of 175 N for a relative comparison of adequate strength in the anterolateral compartment. 16 Failure modes included implant failure (2/6) and graft failure (4/6) for ASA, graft failure (6/6) for SLS, and implant failure (5/6) and graft failure (1/6) for BSA (Figure 7).

Ultimate load for each device (all-suture anchor [ASA]; spiked ligament staple [SLS]; biocomposite suture anchor [BSA]) after loading to failure with the native anterolateral ligament (ALL) maximal load and P value denoted.
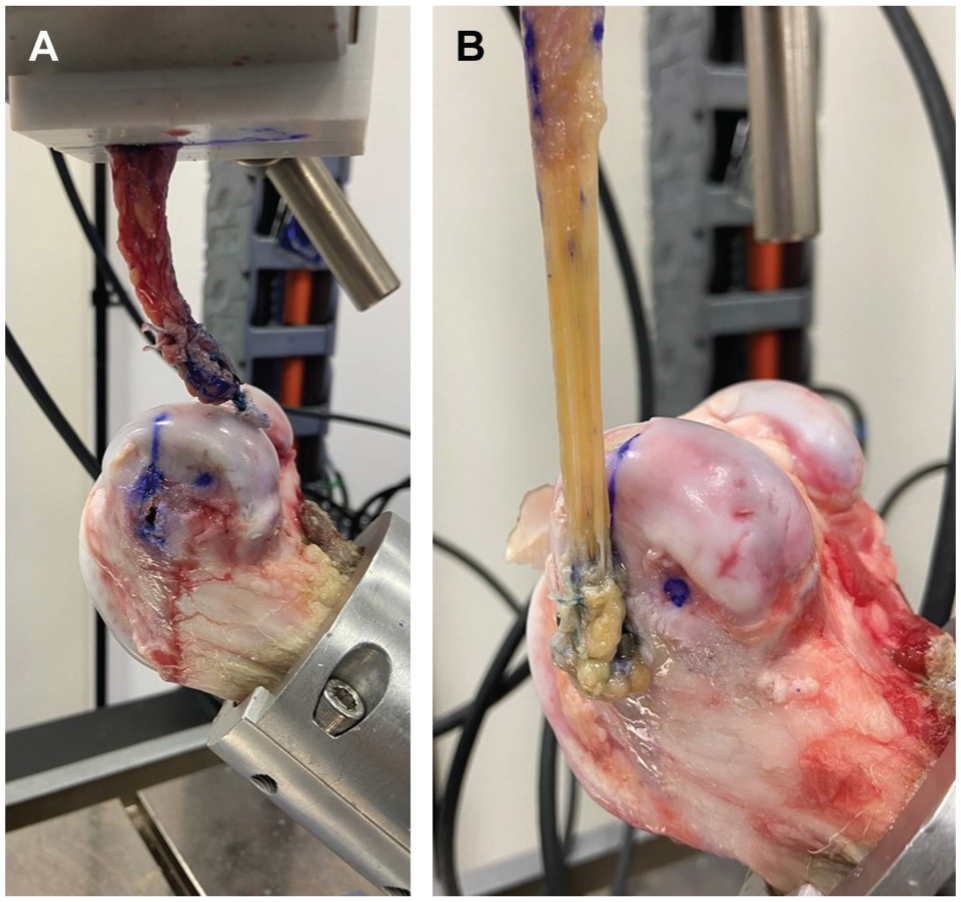
Failure modes include (A) implant failure (shown for all-suture anchor) and (B) graft failure (shown for spiked ligament staple).
Discussion
The principle finding of this study was that onlay ASA fixation demonstrated comparable biomechanical properties with the standard-of-care spiked ligament staple and superior biomechanical properties to biocomposite suture anchor fixation for a lateral extra-articular tenodesis procedure. Given these biomechanical findings and the clinical knowledge that LET with ASA requires a smaller incision and results in less hardware irritation compared with staples, ASA onlay fixation represents a biomechanically superior and clinically favorable treatment strategy for LET.
Biomechanical literature comparing various fixation techniques for extra-articular knee ligaments is limited. In the present study, mean cyclic displacement and ultimate load values for ASA, SLS, and BSA resemble those found by Glasbrenner et al, 10 who used a similar test setup to evaluate medial collateral ligament (MCL) reconstruction stability in porcine tibiae using suture anchor (Corkscrew FT II; Arthrex Inc), bone staple (8-mm wide; Smith & Nephew), and interference screw (MegaFix; Karl Storz) fixation with porcine flexor tendons. All constructs were cyclically loaded to peak forces of 50 N and 100 N for 500 cycles each, and after 1000 total cycles, the mean elongation values were 6.4 ± 0.9 mm, 3.4 ± 1.0 mm, and 3.9 ± 1.2 mm, and mean load-to-failure values were 228 ± 49 N, 376 ± 120 N, and 313 ± 100 N, respectively. The mean SLS values in the present study (3.94 mm and 300 N) were in a similar range to the staple values found by Glasbrenner et al. However, mean ASA cyclic displacement (4.25 mm) and ultimate load (257 N) values were lower and higher, respectively, in the current study than the suture anchor values found by Glasbrenner et al. Mean BSA values (8.14 mm and 205 N) more closely resembled suture anchor outcomes. This may be attributed to the difference in knotted versus knotless fixation techniques, as knotted fixation (BSA and suture anchors) can experience knot loosening due to inconsistent knot security, which has been shown to vary considerably among experienced surgeons. 11 Additionally, knotted constructs offer primary fixation at a single point. In contrast, ASA fixation utilizes preconverted knotless loops that provide independent fixation at 2 points, potentially reducing the likelihood of loosening due to the additional fixation point. Between the knotted fixation techniques, suture anchor fixation did exhibit a lower mean cyclic elongation and higher mean ultimate load value than BSA fixation, which may be attributed to the additional knots and Krackow suturing incorporated into the grafts. If these aspects were included in BSA fixation, it might improve biomechanical performance by decreasing cyclic displacement and increasing the ultimate load without needing to consider a second BSA to attain adequate fixation. Additionally, Deichsel et al 6 evaluated MCL stability in porcine tibiae using 4 different bone staple designs with the same test methods as Glasbrenner et al. Reported elongation and load-to-failure values for the 8 mm–wide spiked staple group were 3.4 ± 1.2 mm and 364 ± 85 N, respectively, which were in a similar range to values reported by Glasbrenner et al and those reported in the present study for SLS. Omar et al 27 also evaluated MCL stability using several different fixation techniques, reporting cyclic displacement values of 11.9 ± 5.2 mm and 8.3 ± 1.6 mm for single and double suture anchor repair, respectively. Load-to-failure values for respective techniques were approximately 300 N ± 70 N and 456 ± 54 N. ASA and BSA values in the present study were lower for both outcomes. However, these differences may be attributed to the difference in primary fixation and loading protocol.
Clinically, there are potential benefits to onlay ASA fixation. The initial pilot hole for anchor placement can be reestablished to prevent ACL femoral tunnel convergence. Once inserted, the deformable body of the anchor bunches against the inner aspect of the lateral femoral cortex, eliminating hardware prominence and reducing irritation deep to the IT band. Additionally, tensioning of the IT band can be customized to the patient, potentially avoiding lateral compartment overconstraint. However, from a clinical outcome perspective, publications comparing onlay ASA fixation with other techniques (staple fixation, transosseous fixation) are fairly limited, likely due to ASA-only techniques being a relatively new strategy for IT band fixation during the LET procedure. Behrendt et al 2 published a nonrandomized multisurgeon clinical study comparing outcomes of revision ACLR with concomitant LET using either onlay anchor fixation (n = 26 patients) or transosseous fixation (n = 26 patients) for the LET, with a mean follow-up of 13.7 ± 3.4 months. The overall outcomes were similar between groups with respect to patient-reported outcomes, physical examination findings, and failure rates (n = 1 in the anchor group), suggesting equivalent outcomes using an onlay suture anchor technique to a transosseous interference screw technique. Certainly, additional clinical comparative studies are needed to determine the potential advantages of one fixation strategy over another for IT band fixation during the LET procedure.
Limitations
As with any biomechanical study, specimen age and quality limit the ability to extrapolate these findings to real-life clinical scenarios, as patients undergoing LET are typically younger than our mean specimen age. Additionally, this experiment was conducted using a porcine cadaveric model. However, the random assignment of IT bands to fixation groups and homogeneous bone quality in femurs mitigated potential variability in the study’s findings due to specimen quality. In addition, as with any cadaveric, time-zero study, graft healing and dynamic stabilization of the knee cannot be accounted for. Further, we did not perform concurrent ACLR surgery on any of the specimens, as would typically be performed in a real-life scenario, or simulate in vivo forces, choosing to focus on applying a unidirectional force. Overall, while these cadaveric biomechanical findings may not be reflective of in vivo biomechanical, and thus clinical outcomes, they do provide robust comparative data to the 2 other commonly used IT band fixation strategies during the LET procedure and demonstrate that onlay ASA fixation is a viable treatment option.
Conclusion
ASA onlay fixation for LET demonstrated comparable displacement to SLS and significantly lower displacement to BSA with greater survival during cyclic loading. ASA had comparable failure loading, with all groups exceeding the native anterolateral ligament failure load requirement. Therefore, ASA is a biomechanically viable alternative to SLS and BSA for IT band fixation during LET.
Footnotes
Final revision submitted March 20, 2025; accepted May 27, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by Arthrex Inc (grant No. AIRR-0121). M.D.T. is employed by Arthrex Inc. R.M.F. is a board or committee member of the American Academy of Orthopaedic Surgeons; AOSSM; American Shoulder and Elbow Surgeons; Arthroscopy Association of North America; International Cartilage Regeneration & Joint Preservation Society; and International Society of Arthroscopy, Knee Surgery, and Orthopaedic Sports Medicine; is a paid consultant for Allosource, Arthrex Inc, and JRF; is a paid presenter or speaker for Allosource, Arthrex Inc, JRF, and Ossur; receives research support from Arthrex Inc and Smith & Nephew; has received publishing royalties and financial or material support from Elsevier; is an editorial or governing board member of Journal of Shoulder and Elbow Surgery and Orthopedics Today; has received consulting fees from Arthrex Inc; has received travel and lodging from Arthrex Inc, Joint Restoration Foundation Inc, and Synthes GmbH; has received support for education from Arthrex Inc and Gemini Mountain Medical LLC; and has served as faculty or a speaker for a medical education program for Bodycad USA and Synthes GmbH. O.L.H. is employed by Arthrex Inc. C.A.W. is employed by Arthrex Inc and is a board or committee member of Orthopaedic Research Society. B.C.W. participates in other professional activities for Arthrex Inc, LifeNet Health Inc, and Pacira Pharmaceuticals Incorporated; has received consulting fees from Arthrex Inc and Pacira Pharmaceuticals Incorporated; has received travel and lodging from Arthrex Inc; and has received support for education from Fortis Surgical LLC. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
