Abstract
Background:
Uniform, data-driven guidelines for oral opioid prescriptions after anterior cruciate ligament reconstruction (ACLR) are lacking.
Purpose:
To provide a concise, systematic review of literature quantifying postoperative opioid pill consumption in patients who underwent ACLR.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
Using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, a literature search was performed using Ovid MEDLINE, Ovid Embase, Scopus, and Cochrane CENTRAL databases from inception to August 12, 2022. Search terms included “anterior cruciate ligament reconstruction,” “opiate alkaloids,” “hydrocodone,” “oxycodone,” and “narcotics.” In total, 752 unique studies were identified. Studies were included if they quantified postoperative oral opioid consumption for ≥4 postoperative days. Exclusion criteria included pediatric studies, abstracts or conference papers, reviews, unpublished randomized controlled trials, case reports, and papers not in the English language. For each included study, the following data were extracted: number of pills in initial prescription, type of pill prescribed, and size of any additional prescriptions given after initial prescription. We also recorded the intervention or context for which prescription size was being investigated. The methodological index for non-randomized studies criteria were used to evaluate the quality of all included studies.
Results:
A total of 20 articles were included in the study. For all studies, regardless of postoperative period length, oral opioid consumption ranged from 2.3 ± 2.9 tablets of 5-mg oxycodone (17.3 MMEs) to 32.2 ± 15.9 tablets of 5-mg oxycodone (242 MMEs). Specifically, for studies reporting on oral opioid consumption within the first 7 postoperative days, consumption ranged from 3.0 tablets of 5-mg oxycodone to 32.2 tablets of 5-mg oxycodone. No study reported consumption >35 tablets. Effective ways to decrease postoperative opioid consumption included preoperative adductor canal blocks, intraoperative bupivacaine local anesthetic injection, and postoperative multimodal analgesic regimens.
Conclusion:
Our review demonstrated that several pre- and postoperative interventions, including preoperative regional nerve blocks, use of infiltrative local anesthetics like bupivacaine or a similar agent, as well as multimodal analgesic regimens, can decrease perioperative pain and therefore decrease postoperative oral opioid consumption. Due to the heterogeneity of the available literature, no conclusions can be made as to what single nonopioid analgesic, or combination thereof, is optimal. However, this systematic review sets the foundation for future studies investigating opioid consumption in settings of nonopioid analgesic medications such as ketorolac and multimodal analgesic regimens.
As of 2019, an estimated 6 to 8 million Americans struggled with opioid use disorder. 20 In 2020, the Centers for Disease Control and Prevention (CDC) reported that 75% of overdose-related deaths involved an opioid and that prescription opioid–related deaths increased by 17%. 27 Excessive opioid prescribing has been determined to be a significant contributor to the ongoing opioid epidemic.22,30,43 Orthopaedic surgeons are a major contributor to overprescribing, as they prescribe the third most number of postoperative opioids compared with other medical specialties.22,30,43 To curtail excessive prescription writing, orthopaedics as a field must understand patient postoperative opioid requirements and define data-informed postoperative opioid prescribing practices.
Orthopaedic research, especially for anterior cruciate ligament (ACL) reconstruction (ACLR), has always had a keen interest in finding perioperative and postoperative regimens to minimize patient pain.32,34,39 Most recently, research has emphasized the use of nonopioid regimens and the quantification of postoperative opioid consumption.5,17,34 Despite the growing body of opioid-focused ACLR research, there is no consensus on appropriate prescribing practices for opioids after ACLR. To our knowledge, no study has systematically organized recent literature on post-ACLR patient opioid consumption. The primary aims of this systematic review are to (1) identify the ranges of consumption previously recorded in the literature and (2) provide general recommendations for prescribing based on identifiable consumptions patterns. We hypothesize that substantial variability will exist across all studies reporting opioid consumption.
Methods
We performed this systematic review according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Studies were included if they quantified the number of opioid pills orally consumed by patients after ACLR. Studies that included minor secondary procedures such as meniscal repair or chondroplasty with ACLR were included. Studies were excluded if ACLR was performed with osteotomies, multiple other reconstructive surgeries, or revision surgery; opioid pill consumption was reported for <4 days postoperatively; data were reported for pediatric patients only; no extractable data were found for pill count; paper was a review article, incomplete randomized control trial, abstract, or conference poster; and article was not written in the English language.
Search Strategy
A search strategy was developed in conjunction with a medical librarian (T.K). The search was conducted in Ovid MEDLINE, Ovid Embase, Scopus, and Cochrane CENTRAL databases from inception to August 12, 2022. A combination of controlled vocabulary and keywords, including but not limited to “anterior cruciate ligament reconstruction,” “opiate alkaloids,” “hydrocodone,” “oxycodone,” and “narcotics,” was used along with truncation and adjacency operators to search for relevant literature. No date, language, or publication type restrictions were used in the search strategy.
Study Selection
A total of 1668 studies were identified from all databases. All studies were transferred to the Covidence application (Veritas Health Innovation Ltd), and 3 authors (M.E.H., M.S.R., M.J.S.) were granted access. After 916 duplicates were removed, 2 medical student authors (M.E.H., M.S.R.) independently screened the abstracts and titles of the remaining 752 articles. Discrepancies between authors were resolved by the senior author (M.J.S.). A total of 149 articles were selected for full-text review. The same 2 reviewers then screened these articles with discrepancies arbitrated by the senior author. Of these, 18 articles were eligible for inclusion in this review. Two articles were added post hoc, as they were published outside of the collection period but deemed to be relevant to the aims.17,18
Data Items and Quality Assessment
The extracted data included title of article, author name(s), publication year, surgical intervention, opioid pill count for a given intervention, opioid pill type, pain management techniques, graft type, visual analog scale, surgical complications, and outcome measurements. For easier cross-study comparison, opioid tablets were converted to morphine milligram equivalents (MMEs) using CDC guidelines when possible. 9 If only MME was provided, conversion was made to tablets of 5-mg oxycodone. Otherwise, initial type and concentration of opioid tablet were retained and reported from the original study. When opioid tablets were recorded numerically on graphs, total consumption was calculated by summing values and converting to MMEs. Each study was reviewed according to the methodological index for non-randomized studies (MINORS) criteria, which reliably assesses the methodological quality of nonrandomized surgical studies. 40
Results
This systematic review identified 1668 studies from all databases. After removal of duplicates, 752 studies were identified with potential for inclusion. Of these, 603 were excluded based on abstract and title review, leaving 149 studies for full-text review (Figure 1). A total of 131 studies was excluded for the following reasons: 36 did not record oral opioid consumption for ≥4 days, 28 did not report outcome of interest, 19 were incomplete randomized controlled trials either still enrolling patients or without published data, 12 had unextractable data, 12 were abstracts, 8 were irretrievable, 4 did not have ACL-specific outcomes, 4 did not use oral administration of postoperative opioids, 3 were retracted since publication, 3 utilized the wrong study design, 1 studied the wrong patient population, and 1 reported the same data as an already-included study. After review, 18 studies remained. ‡ Two studies were added post hoc because they were outside the original search period.17,18 This allowed for a total of 20 studies for analysis.
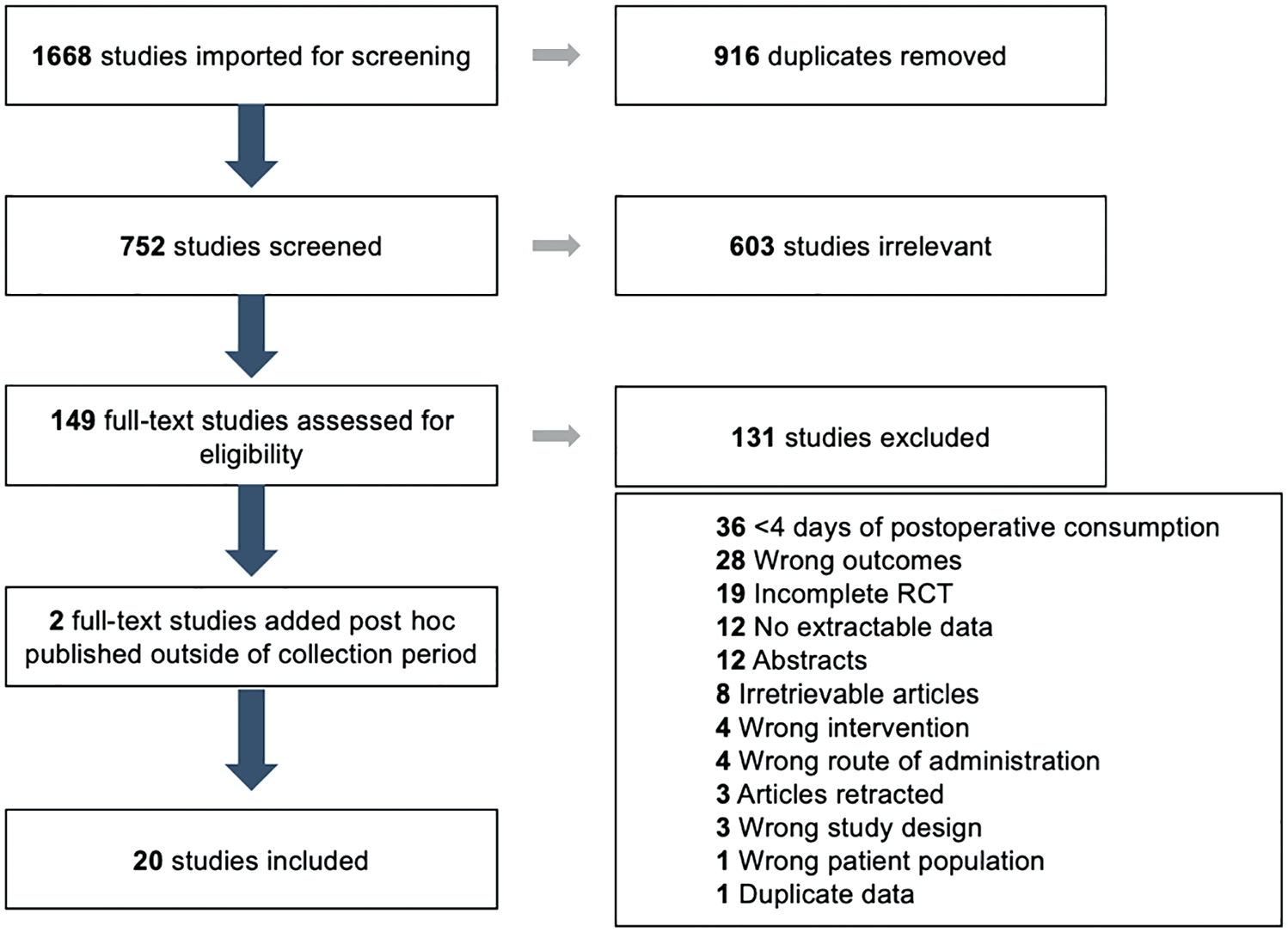
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram for the study selection process.
Local Anesthetic Infiltration
As shown in Table 1, there were 5 studies that related local anesthetic infiltration with opioid consumption.2,6,7,33,42 Four of these studies reported opioid consumption within the first postoperative week,2,6,33,42 finding that opioid consumption ranged from 3.0 tablets of 5-mg oxycodone (22.5 MMEs) 42 to 22.7 tablets of 5-mg oxycodone (113.6 MMEs). 33 The local anesthetic regimen that minimized opioid consumption was intra-articular infiltration with 9.5-mL 0.5% bupivacaine, 1:200,000 epinephrine, and 5 mg morphine. 42 Three of the 5 studies reported on patient adverse effects and found no significant complications for graft failure, laxity, or functional outcome associated with their regimens.2,7,42
Summary of Included Studies Quantifying Opioid Consumption in Patients Receiving Local Anesthesia a
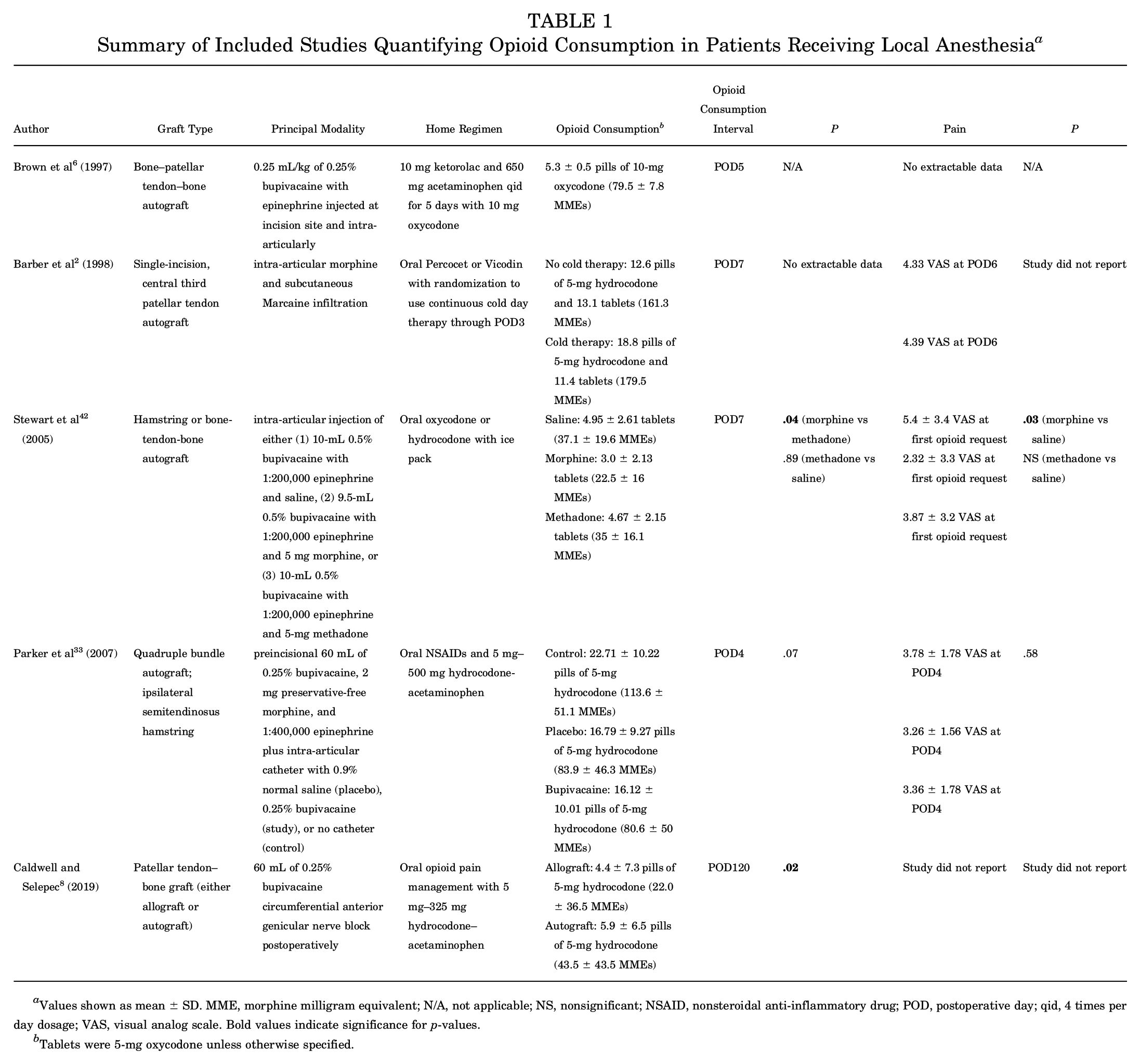
Values shown as mean ± SD. MME, morphine milligram equivalent; N/A, not applicable; NS, nonsignificant; NSAID, nonsteroidal anti-inflammatory drug; POD, postoperative day; qid, 4 times per day dosage; VAS, visual analog scale. Bold values indicate significance for p-values.
Tablets were 5-mg oxycodone unless otherwise specified.
Regional Nerve Block
As shown in Table 2, there were 11 studies that related perioperative usage of regional nerve block with opioid consumption. § Six of these studies17-19,29,38,45 reported opioid consumption throughout the first 1 to 2 postoperative weeks with opioid consumption ranging from 5.5 tablets of 5-mg oxycodone (41.3 MMEs) 29 to 23.5 tablets of 5-mg hydrocodone (117.3 MMEs). 19 Four studies reported opioid consumption throughout the entire postoperative period with opioid consumption ranging from 2.3 tablets of 5-mg oxycodone (7.7 MMEs) to 17.4 tablets of 5-mg oxycodone (130.9 MMEs).11,13,23,31 Adductor canal blocks (ACBs) were most common and were effective perioperative pain management techniques across studies. ‖ The regional nerve block and postoperative regimen that minimized opioid consumption was an ACB with 10 mg of ketorolac qid, 5 mg diazepam qid, 400 mg gabapentin tid, 1000 mg acetaminophen tid with 10 rescue tablets of 5-mg oxycodone for postoperative days 1 to 5 then 5 mg diazepam tid, 1000 mg acetaminophen tid, meloxicam bid with gabapentin weaning for postoperative days 6 to 14. 31 Notably, this regimen contained diazepam, a controlled substance, and gabapentin, which required careful weaning as noted per protocol. DeMaio et al 11 employed a multimodal pain regimen that did not contain any controlled substances nor drugs requiring weaning, and the regimen remained constant across the postoperative period. Furthermore, DeMaio et al achieved a comparably low postoperative opioid consumption throughout the entire postoperative period: 3.8 (28.6 MMEs) to 6.0 (45.0 MMEs) tablets of 5-mg oxycodone depending on nerve block used.
Summary of Included Studies Quantifying Opioid Consumption in Patients Receiving Regional Anesthesia or Nerve Block a

Values shown as mean ± SD, unless otherwise specified. Bold values indicate significance for p-values. ACB, adductor canal block; bid, 2 times per day dosage; FNB, femoral nerve block; LFCN, lateral femoral cutaneous nerve block; MME, morphine milligram equivalent; N/A, not applicable; NS, nonsignificant; NSAID, nonsteroidal anti-inflammatory drug; PCI, posterior capsular injection; POD, postoperative day; qid, 4 times per day dosage; tid, 3 times per day dosage; TXA, tranexamic acid; VAS, visual analog scale.
Tablets were 5-mg oxycodone unless otherwise specified.
Mean [95% CI].
The 1 study that commented on adverse outcomes found no significant complications for graft failure, laxity, or functional outcomes. 29
Surgical Technique
As shown in Table 3, there were 4 studies that related surgical technique with opioid consumption.3,24 -26 All 4 studies reported on soft tissue techniques with bony techniques notably absent. Across the 4 studies that specifically assessed opioid consumption in the context of surgical technique, opioid consumption was measured throughout the first 1 to 2 postoperative weeks, and opioid consumption was found to range from 2.8 opioid pills (no MME conversion available) 26 to 32.2 tablets of 5-mg oxycodone (242 MMEs). 25 The postoperative regimen that minimized opioid consumption was thirty 5 mg–325 mg oxycodone/acetaminophen or 30 mg–15 mg–300 mg codeine phosphate-caffeine-acetaminophen with 30 naproxen tablets. 26
Summary of Included Studies Quantifying Opioid Consumption Based on Surgical Technique a

Values shown as mean ± SD. Bold values indicate significance for p-values. BEAR, Bridge-Enhanced Anterior Cruciate Ligament Repair; KSS, Knee Society Score; MME, morphine milligram equivalent; POD, postoperative day; VAS, visual analog scale.
Tablets were 5-mg oxycodone unless otherwise specified.
Quality Assessment
The median MINORS score for the 6 noncomparative studies (indicated in Table 4 by dashes in the comparative studies cells) was 13.5 (range, 11-15) out of a possible total 16 points (Table 4). The median MINORS score for the 14 comparative studies was 22.5 (range, 17-24) out of a possible total 24 points. A score of 2 for a given criterion denotes that the item of interest was reported and adequate. A score of 1 denotes that the item of interest was reported but inadequate while a score of 0 denotes that the item was not reported in the study. The most common reasons for point deductions (receiving a score of 1 or 0) were failure to include adequate prospective study size calculations (9 studies, 45% of all studies), loss to follow-up exceeding 5% (7 studies, 35%), and unbiased end of study endpoint (6 studies, 30%). Specifically, in comparative studies, point reductions were most often earned for failing to have baseline equivalence between compared groups (2 studies, 14% of comparative studies), having an inadequate control group (2 studies, 14%), and lacking a contemporary control group (1 study, 7%).
Summary of MINORS Criteria a

Criteria: 1 = clearly stated aim; 2 = inclusion of consecutive patients; 3 = prospective collection of data; 4 = endpoint appropriate for study aim; 5 = unbiased assessment of study endpoint; 6 = follow-up period for study appropriate; 7 = loss to follow-up <5%; 8 = prospective calculation of study size included; 9 = adequate control group; 10 = contemporary control group; 11 = baseline equivalence between compared groups; 12 = adequate statistical analysis. Dashes indicate nonapplicability. The items are scored o (not reported in study), 1 (reported but inadequate), or 2 (reported and adequate). MINORS, methodological index for non-randomized studies.
Discussion
The major finding of this review was that, across all 20 included studies, postoperative opioid consumption ranged considerably from 2.3 ± 2.9 tablets of 5-mg oxycodone 31 (17.3 MMEs) to 32.2 ± 15.9 tablets of 5-mg oxycodone (242 MMEs). 25 Eight of the 20 included studies reported postoperative opioid consumption of ≤10 tablets of opioid medication, suggesting that consumption can be limited without affecting patient postoperative pain. ¶ Various perioperative pain management techniques, including preoperative regional nerve blocks, use of infiltrative local anesthetic, and multimodal analgesic regimens, can decrease pain and therefore help mitigate oral opioid usage postoperatively.
Four out of the 5 studies relating local anesthetic infiltration with opioid consumption employed perioperative bupivacaine injections.6,7,33,42 Three out of the 4 studies using bupivacaine reported opioid consumption <6 tablets of 5-mg oxycodone (9 MMEs) within the first postoperative week, suggesting that intraoperative usage mitigates postoperative pain and decreases postoperative opioid requirements. Systematic reviews have further supported the use of liposomal bupivacaine for lowering both postoperative pain within the first 12 hours and total postoperative opioid consumption.10,44 While effective, bupivacaine may exhibit chondrotoxic effects, proving problematic in the ACLR patient population due to these patients’ propensities for developing early osteoarthritis.16,21 When used at concentrations >0.5%, bupivacaine is the most chondrotoxic locally infiltrative agent. 37 Instead, perioperative use of ropivacaine as a locally infiltrative anesthetic is recommended by senior authors because it demonstrates similar efficacy to bupivacaine while minimizing chondrotoxicity. 16
One study of note in the cohort reporting on locally infiltrative anesthetics was published in 1997 and reported 5.3 tablets of 10-mg oxycodone (79.5 MMEs) consumed within the first 5 postoperative days. 6 The pain management regimen used in this study was minimal, including 10 mg of ketorolac with 650 mg of acetaminophen qid and 10 mg of rescue opioids. This is 8.3 tablets of 5-mg oxycodone (62.2 MMEs) more than the study that reported the least postoperative opioid consumption. In their 1997 study, Brown et al 6 were able to achieve low rates of opioid consumption without the addition of any scheduled substances or complicated pain management regimens. This demonstrates how little forward progress has been made in pain management techniques for ACLR.
The reason for minimal progress may result from the historical boom and then bust approach taken to opioid prescriptions. A perfect storm of events led to massive increases in opioid prescribing at the end of the 1990s: OxyContin was approved by the Food and Drug Administration in 1995 and increased in availability throughout the late 1990s and early 2000s 43 ; pain was recognized as a “fifth vital sign” by the American Pain Society in 1996, 8 and efforts were made by the Joint Commission to standardize assessment and treatment of pain in 2000. 35 Studies included here and published in the 2000s correlate with these historic events and report the highest opioid consumptions (Figure 2). Only when the threat of the opioid epidemic emerged, and the US Department of Public Health declared the misuse of prescription opioids an epidemic in 2013 did the studies included here attempt to quantify and minimize opioid consumption. Thus, studies published after 2015 reported a relative decrease in opioid consumption, although few studies reported opioid consumption as low as Brown et al 6 in 1997. Looking forward, we should look to the model from Brown et al to build a successful, streamlined postoperative pain management regimen.

Nine of the 11 studies that related the perioperative usage of regional nerve blocks with opioid consumption utilized ACBs. # While femoral nerve blocks (FNBs) are popularly used in ACLRs, recent literature has found that ACBs exhibit similar efficacy.12,41 However, compared with FNBs, ACBs have not been found to directly decrease postoperative opioid consumption.12,34,41 The primary source of benefit for using ACBs is preservation of quadriceps function in the early postoperative period. 12 Early recovery of quadriceps function after ACLR is crucial and has been correlated with improved functional performance, gait performance, and return to sport.4,14 While evidence correlating ACBs and FNBs with opioid consumption remains lacking, the clear clinical benefit of ACBs makes it the strongest recommendation for ACLR use.
Multimodal postoperative analgesic regimens were a common theme across all studies. Moutzouros et al 31 recorded the lowest opioid consumption throughout the full operative period with their 2-phase regimen. However, their regimen included diazepam, which is a controlled substance, and gabapentin, which requires weaning at certain doses. DeMaio et al 11 used a monophasic regimen without scheduled substances and observed similarly low levels of consumed opioids. A systematic review of pain management after outpatient ACLR found that, while there was significant evidence on multimodal regimens, the body of research was unable to agree upon 1 optimal regimen; gabapentin, zolpidem, ketorolac, and ibuprofen were all found to decrease opioid consumption. 39
In this review, ketorolac was included in postoperative regimens of 3 studies (Table 5).6,29,31 Ketorolac presents an alternative to opioid-based pain management that may help reduce postoperative narcotic consumption in patients undergoing ACLR. Ketorolac is a stronger alternative to naproxen and can be used for short-term control of pain that would usually require opioids. 36 Its potency may have the added benefit of minimizing other pain medications taken concurrently, thus simplifying regimens to the benefit of the patient. Mengers et al 29 demonstrated that ketorolac reduces opioid consumption within the first 5 postoperative days and significantly lowers patient-perceived pain compared with opioids. Barber and Gladu 1 showed similar efficacy for ketorolac for short-term postoperative pain relief, with patients reporting significantly better pain management when given ketorolac than hydrocodone. Additionally, ketorolac was shown to have mostly mild adverse side effects limited to nausea, dizziness, and headache. 1 When used postoperatively, ketorolac is safe with respect to bone healing and has not been shown to affect perioperative blood loss. 28 Together, these data lend support to prescribing ketorolac to reduce opioid use in postoperative ACLR pain management. However, if we aim to implement considerable change in prescription practices in this patient population, a high-quality, double-blind, randomized controlled trial must be performed to demonstrate a similar efficacy and safety profile of ketorolac to opioids.
Summary of Included Studies Utilizing Ketorolac as part of Postoperative Multimodal Pain Regimen a

Values shown as mean ± SD. ACB, adductor canal block; bid, 2 times per day dosage; FNB, femoral nerve block; MME, morphine milligram equivalent; N/A, not applicable; POD, postoperative day; qid, 4 times per day dosage; tid, 3 times per day dosage; VAS, visual analog scale.
Tablets were 5-mg oxycodone unless otherwise specified.
Limitations
The most considerable limitation to this systematic review is that surgical techniques and approaches to perioperative pain management were heterogeneous among the included studies. This is most evidenced by the wide range of postoperative opioid consumption across the 4 studies that investigated ACLR surgical techniques (2.8-32.2 tablets), highlighting how these differences likely led to various degrees of postoperative pain and influenced the quantity of opioids patients consumed. This introduced inherent bias to our study, prohibiting us from performing a meta-analysis and direct comparison of the studies. A second limitation is that only 55% of studies included an adequate power analysis in their methodologies, making it unclear if the remaining 45% of studies had adequate sample sizes to identify significant differences. However, our findings do highlight the ability of multimodal pain regimens to decrease opiate consumption in postoperative ACLR patients, setting the stage for future large, randomized controlled trials comparing nonopioid analgesics with opioids in this patient population.
Conclusion
In conclusion, our review demonstrated that several pre- and postoperative interventions, including preoperative regional nerve blocks, use of infiltrative local anesthetics like bupivacaine or a similar agent, and multimodal analgesic regimens, can decrease perioperative pain and therefore decrease postoperative oral opioid consumption. On the basis of this, the authors recommend a multimodal perioperative pain management regimen consisting of a preoperative adductor canal nerve block and intraoperative ropivacaine local anesthetic injection to areas not covered by the regional block. Postoperative pain medication should consist of 10 tablets of 5-mg oxycodone, scheduled acetaminophen, and a scheduled nonsteroidal anti-inflammatory drug with our preference for ketorolac. We propose that a well-designed double-blind randomized controlled trial evaluating effective nonopioid prescription medication such as ketorolac can demonstrate further reduction in opioid consumption and standardize prescribing protocols.
Footnotes
Final revision submitted March 21, 2025; accepted March 31, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: K.P.S. has received support for education from Summit Surgical, consulting fees from Novo Pedics and Flexion Therapeutics, and honoraria from Novo Pedics and PRO-T. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
