Abstract
Background:
Graft choice in anterior cruciate ligament reconstruction (ACLR) is determined by surgeon preference, patient factors, and graft characteristics. An allograft is a viable option with benefits such as decreased operative times and no donor site morbidities, and it can be the preferred graft choice in certain patient populations. Despite this, there is no established standard for allograft processing.
Purpose:
To determine whether allograft processing techniques influence the risk of graft failure, patient-reported outcomes, and revision surgery in patients undergoing ACLR.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Consecutive patients from 3 fellowship-trained sports medicine surgeons undergoing ACLR with patellar tendon allografts from January 1, 2016, to January 1, 2022, were identified. Descriptive data, mechanism of injury, graft processing technique, revision history, and patient-reported outcome scores were collected. The primary endpoint was aseptic failure as defined by graft rupture on magnetic resonance imaging. Statistical analysis was performed with the χ2 test or the Fisher exact test as indicated for categorical variables and with the t test and analysis of variance as indicated for continuous variables. The significance level for statistical tests was set at 5%.
Results:
A total of 189 patients who underwent ACLR and met the inclusion/exclusion criteria were identified, with 103 Musculoskeletal Transplant Foundation (MTF), 55 Allowash XG, and 31 BioCleanse allografts. The mean age of patients was 43.4 ± 10.5 years, and 60.9% were women. No significant difference was observed between the groups with regard to sex and body mass index (P = .25 and P = .64, respectively). However, age at the time of surgery was significantly different between groups (P = .03). The mean patient follow-up time was 4.65 ± 1.40 years. Four patients experienced graft failure (MTF 1% [1/103]; Allowash XG 5.5% [3/55]; BioCleanse 0% [0/31]), with no significant difference in graft failure between groups (P = .20). There were no reported infections. Postoperative International Knee Documentation Committee (IKDC) scores (MTF, 82.6; Allowash XG, 81.7; BioCleanse, 80.2) were not different between groups (P = .60).
Conclusion:
Bone-patellar tendon-bone allografts processed using BioCleanse, Allowash XG, and the MTF protocol had similar rates of graft failure and postoperative IKDC scores. Although underpowered, the data suggest that each of these techniques can be used safely with low failure rates and good functional outcomes.
Keywords
Graft selection for anterior cruciate ligament reconstruction (ACLR) is a heavily researched and debated topic in sports medicine. While ACLR with autografts is generally favored over ACLR with allografts for young patients because of its lower failure rate,2,11,27 allografts remain a viable option with benefits such as decreased operative times and no donor site morbidities. 18 Moreover, allografts are particularly useful in older patients, where they have been shown to have a low failure rate, excellent functional outcomes, and limited disadvantages compared with autografts.5,8,11
Despite their benefits in the appropriately selected patient, allografts do pose a risk for donor-to-recipient disease transmission. 6 To decrease the risk of disease transmission, strict procedures are utilized by tissue banks, such as antibiotic washing, gamma irradiation, ethylene oxide processing, supercritical carbon dioxide exposure, aseptic processing, transportation in temperatures below zero, and various proprietary methods/protocols such as BioCleanse (RTI Biologics) and Allowash XR (Lifenet Health Inc). 6 While these sterilization techniques decrease the risk of disease transmission, they also alter graft biomechanics and have been cited as a potential reason for the increased failure rate seen when comparing autograft to allograft ACLRs. 3 Multiple meta-analyses and systematic reviews have shown similar failure rates when comparing autografts to nonsterilized allografts,3,12,26 indicating that allograft sterilization may have a role in the biomechanical and clinical difference seen between autograft and allograft ACLRs. However, it is not yet clear which sterilization process is ideal in terms of optimizing the biomechanical properties of the graft while minimizing the risk of disease transmission.
Given the lack of consensus regarding optimal allograft processing methods, allograft sterilization is not standardized across vendors. While high-dose gamma irradiation (>2 Mrad) has fallen out of favor because of dramatic effects on the biomechanical integrity of grafts,16,23 several proprietary nonirradiated or low-dose irradiation sterilization methods are common in clinical practice, including the BioCleanse, Allowash XG, and the Musculoskeletal Transplant Foundation (MTF) (MTF Biologics) protocols. Given the differing effects of various sterilization methods on the biomechanical properties of grafts, this introduces a variable that may affect outcomes and failure rates after allograft ACLR. This study aimed to determine whether common allograft processing techniques influence the risk of graft failure and revision surgery in patients undergoing ACLR. We hypothesized that there would be similar outcomes among differing processing techniques.
Methods
Following institutional review board exemption (iRISID-2022-0968), a retrospective review of all ACLRs performed by 3 fellowship-trained sports medicine surgeons (K.B.F.) from a single practice was conducted. Patients were included for analysis if they had ACL insufficiency as clinically/radiographically assessed by the attending surgeon (K.B.F.), underwent ACLR with bone-patellar tendon-bone (BPTB) allografts between January 1, 2016, to January 1, 2022, and had a minimum 2-year follow-up. BPTB allografts are among the most common grafts utilized nationally and are the most commonly used at the study institution. 20 Thus, the decision was made to specifically investigate BPTB allografts to help minimize heterogeneity within the study cohort.
Patients undergoing revision surgery and patients undergoing multiligamentous reconstructions were excluded from analysis. In addition, patients <18 years were excluded, given the high rate of allograft failure in younger athletes. For patients meeting the inclusion criteria, manual chart review was performed to extract descriptive data, mechanism of injury, operative findings, and information on concomitant pathology. Mechanism of injury was broken down into 5 categories as follows: sports; nonsport activities (hiking, recreational dance, etc); traumatic (fall, motor vehicle collision, etc); atraumatic; and unknown.
All patients underwent arthroscopic ACLR with BPTB allografts under general anesthesia. All surgeons utilized a tourniquet. An isometric single-bundle technique was used in all cases with independent drilling of tibial and femoral tunnels in the anatomic footprint. BPTB allografts were obtained from 3 different tissue banks utilizing 3 separate sterilization protocols: BioCleanse, Allowash XG, and the MTF protocol. All grafts were harvested and processed in accordance with the American Association of Tissue Banks (AATB) standards. The BioCleanse tissue sterilization process utilizes multiple cycles of pressure and vacuum wash with detergent and sterilant, hydrogen peroxide, isopropyl alcohol, and mechanical oscillations; at no point during the process is the graft irradiated. 19 The Allowash XG tissue sterilization process utilizes a patented 6-step process involving treatment with hypotonic solutions and antimicrobial agents as well as ultrasonication and centrifugation; the final step involves terminal sterilization with low-dose (<2 Mrad) irradiation. 19 MTF tissue sterilization process involves an antibiotic soak, agitation, and a water rinse; roughly 40% of grafts are preprocessed with a 1.2 to 1.8 Mrad dose of radiation before antibiotic treatment. 19 Graft processing was selected based on graft availability and vendor selection at the respective institution of the procedure. All grafts were reconstituted in saline with vancomycin at room temperature for a minimum of 10 minutes. Grafts were reshaped to fit the individual patient’s anatomy and were fixed using interference screws on the femoral and tibial side. After fixation, grafts were tensioned in extension, and the knee was cycled through a full range of motion to ensure there was no impingement. All patients underwent a standard postoperative ACL rehabilitation protocol, supervised by a physical therapist, with emphasis on range of motion and quadriceps strength. Patients were seen postoperatively by their surgeon at approximately 3, 6, 12, and 24 months in accordance with our institution’s standard ACLR follow-up protocol.
Failure was described as clinical graft failure confirmed on magnetic resonance imaging (MRI). Adverse events and complications were recorded and analyzed; charts were specifically reviewed for evidence of graft failure, knee pain, arthrofibrosis/stiffness, hematoma, neurovascular deficits, and infection. All other complications, as noted in postoperative notes, were also recorded. Major complications were defined as infection, arthrofibrosis/stiffness, and deep vein thrombosis. Patient-reported outcomes were assessed with the International Knee Documentation Committee (IKDC) Subjective Knee Form. The IKDC score was assessed preoperatively at the office visit closest to surgical intervention and postoperatively at least 2 years after the date of surgery.
The primary endpoint of this study was aseptic failure of a primary allograft ACLR. Our secondary outcomes of interest were 2-year minimum postoperative (IKDC) scores, revision rates, and complication rates. Categorical variables were characterized with percentages, and continuous variables were characterized with means and standard deviations. Categorical variables were assessed with the χ2 test or the Fisher exact test as indicated; the Fisher exact test was used when the frequency of a categorical outcome was <10. Continuous variables were assessed with the t test and analysis of variance as indicated. Statistical significance was set at .05.
Results
A total of 225 patients underwent ACLR with BPTB allografts between January 1, 2016, and January 1, 2022. A total of 189 (84%) patients had a minimum follow-up of 2 years and were thus included for the final analysis (Figure 1). Also, 170 patients completed postoperative IKDC surveys (75.6%). The mean follow-up was 4.7 ± 1.4 years (range 2.07 – 7.08). Descriptive and injury data are presented in Table 1, and further group comparisons are presented in Table 2. The mean age of patients was 43.4 ± 10.5 years (range 21 – 69), and 60.9% were women. A total of 103 patients had an MTF allograft, 55 patients had an Allowash XG allograft, and 31 patients had a BioCleanse allograft.

A CONSORT flow diagram. CONSORT, Consolidated Standards of Reporting Trials.
Overall Descriptive Data a

The bold P value indicates significance. BMI, body mass index; MTF, Musculoskeletal Transplant Foundation.
Comparing Descriptive Data Between the Technique Groupings a
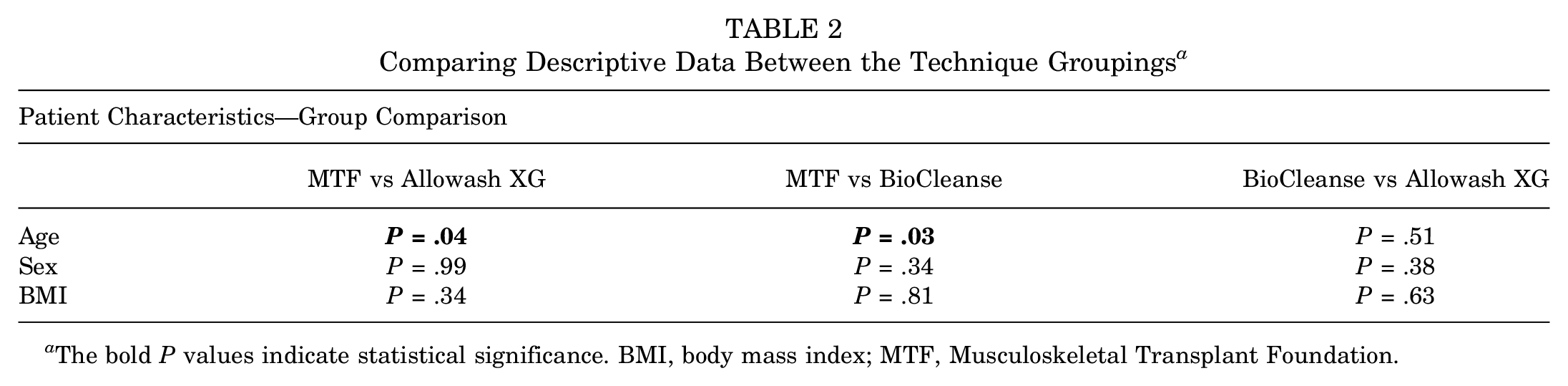
The bold P values indicate statistical significance. BMI, body mass index; MTF, Musculoskeletal Transplant Foundation.
The statistically significant difference in ages across groups is more accurate when the demographic composition of patients in each group (MTF, Allowash XG, and BioCleanse) is compared with the other respective groups. The differences in age at the time of surgery were statistically significant between MTF and Allowash XG and MTF and Biocleanse, respectively (P = .04 and P = .03). No similar statistical difference was noted in Biocleanse versus Allowash XG or with the other variables assessed (Table 2).
Injury and Surgery Characteristics
The mean time to surgery was 3.8 ± 9.2 months for all groups, and there was no significant difference in time to surgery between groups (P = .85) (Table 3). The incidence of concomitant pathology was 64% for all groups and 68.9%, 52.7%, and 67.7% for the MTF, Allowash XG, and BioCleanse groups, respectively (P = .13). There was no significant difference in the rate of concomitant procedures between groups (P = .25). The incidence of concomitant surgery was 61.9% for all groups and 66%, 55.7%, and 64.5% for the MTF, Allowash XG, and BioCleanse groups, respectively. The most common concomitant procedure was a partial meniscectomy, which was performed in 30.1% of cases (57/189), followed by meniscal repair, which was performed in 28% of cases (53/189).
Injury and Surgery Characteristics a

Data are presented as n (%) unless otherwise indicated. MTF, Musculoskeletal Transplant Foundation.
No statistically significant differences were found when comparing the injury and surgical characteristics between each group (MTF; Allowash XG; BioCleanse) with the other respective groups. The 3 variables specifically analyzed were months to surgery, concomitant pathology, and concomitant procedure (Table 4).
Comparing Injury and Surgery Characteristics Between the Technique Groupings a
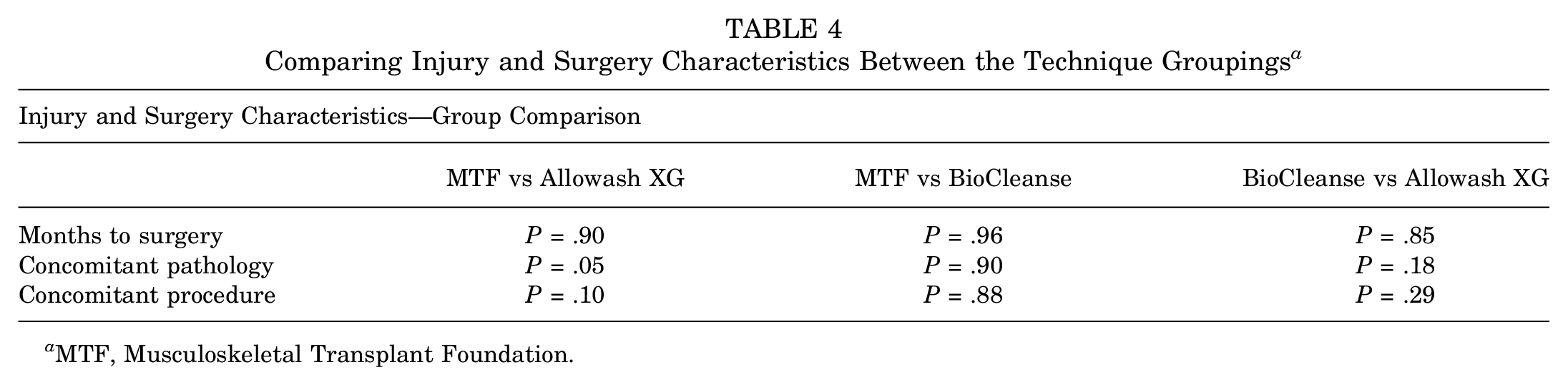
MTF, Musculoskeletal Transplant Foundation.
Outcomes: Graft Failure and Revisions
Overall, 2.1% of patients (4/189) went on to experience ACL graft failure, with 1.1% of patients (2/189) undergoing revision ACLR. Outcomes data are presented in Table 5. The overall graft failure rate was 1% (1/103) for the MTF group, 5.5% (3/55) for the Allowash XG group, and 0% (0/31) for the BioCleanse group at the final follow-up, with no significant difference in the failure rate between groups (P = .20). All 4 patients who experienced graft failure were men and the mean age of the patients was 37 years at the time of index surgery. Graft failure was diagnosed at a mean of 1.5 years after the index surgery. All 4 instances of graft failure were traumatic, with 2 resulting from soccer injuries, 1 resulting from an electric scooter accident, and 1 resulting from a slip on ice, with patients reporting subsequent knee instability. All failures were confirmed with an MRI showing complete graft rupture. All patients with confirmed graft failure we offered revision ACLR; however, 2 patients declined revision. The revision ACLR rate was 1% (2/189) overall and 0% (0/103), 3.6% (2/55), and 0% (0/31) for the MTF, Allowash XG, and BioCleanse groups, respectively (P = .11).
Graft Failure, Revision Surgery, Major Complications, and Postop IKDC Scores a

Data are presented as n (%). IKDC, International Knee Documentation Committee; MTF, Musculoskeletal Transplant Foundation; Postop, postoperative.
Outcomes: Major Complications and Postoperative IKDC
Major complications were similar between groups. Overall, 6.3% of patients (12/189) went on to experience a major complication, and 6.8% (7/103), 5.5% (3/55), and 6.5% (2/31) of patients experienced a major complication in the MTF, Allowash XG, and BioCleanse groups, respectively (P = .95). Postoperative IKDC scores were also similar between groups. The overall mean postoperative IKDC score was 81.9 ± 11.9 for all groups and 82.6 ± 11.5, 81.7 ± 9.1, and 80.2 ± 17.5 for the MTF, Allowash XG, and BioCleanse groups, respectively (P = .60) (Table 5).
When comparing the graft failure, revision surgeries, major complications, and postoperative IKDC scores between each group (MTF; Allowash XG; BioCleanse) with the other respective groups, there were no noted statistically significant differences (Table 6). The 3 variables specifically analyzed were graft failure, revision surgery, major complications, and postoperative IKDC.
Comparing Graft Failure, Revision Surgery, Major Complications, and Postop IKDC Scores Between the Technique Groupings a

IKDC, International Knee Documentation Committee; MTF, Musculoskeletal Transplant Foundation.
All Complications
The rate of all recorded complications was 13.2% (25/189) overall and 14.6% (15/103), 10.9% (6/55), and 12.9% (4/31) for the MTF, Allowash XG, and BioCleanse groups, respectively (P = .84). No significant differences were found between groups for any individual complication. The most common complication seen after ACLR was stiffness/arthrofibrosis. The rate of stiffness/arthrofibrosis was 4.8% (9/189) overall and 5.8% (6/103), 1.8% (1/55), and 6.5% (2/31) for the MTF, Allowash XG, and BioCleanse groups, respectively (P = .52). Three patients went on to experience symptomatic postoperative meniscal tears (1.6%). There were no instances of postoperative infection (Table 7).
Incidence of Complications a

MRI, magnetic resonance imaging; MTF, Musculoskeletal Transplant Foundation; Postop, postoperative.
Discussion
In this study, patients who underwent ACLR with BPTB allografts processed using BioCleanse, Allowash XG, or the MTF protocols for graft sterilization displayed statistically similar rates of clinical failure at 2 years postoperatively. Moreover, there were no significant differences in clinical outcomes between these 3 groups as determined by IKDC scores and postoperative complications. However, these similarities must be interpreted within the context of our relatively small sample size, which is likely underpowered to truly detect statistically significant differences. With high failure rates reported after ACLR with allografts and sources indicating that graft sterilization methods may affect the biomechanical strength of allografts, surgeons need to be aware of how their allografts are sterilized and whether any process affects the risk of graft failure and revision surgery.4,12,13,29 In this study, we showed a low rate of allograft failure with a 2-year minimum follow-up for grafts processed with the BioCleanse, Allowash XG, or the MTF protocols.
Allograft sterilization is a vital component of preimplantation processing to reduce the risk of disease transmission. Allograft safety is overseen nationally by the AATB and the United States Food and Drug Administration, which require sterility assurance levels of 1 × 10−6 and 1 × 10−3 for accreditation, respectively. 14 These requirements have led to the creation of numerous graft processing strategies. Three commonly used sterilization processes are BioCleanse, Allowash XG, and the MTF protocols.
Of these 3 processing methods, BioCleanse is perhaps the most studied. BioCleanse utilizes mechanical and chemical modalities, whereby oscillating pressure and vacuum are applied with chemical reagents to thoroughly perfuse the tissue. 8 In doing so, the risk of disease transmission decreases; nonetheless, there is no effect on the biomechanical or physiological properties of the allograft.1,10,15,21 Schimizzi et al 22 showed that BioCleanse processed allografts showed stiffness and failure loads similar to autografts, indicating that the BioCleanse process does not adversely affect the biomechanical properties of the allograft material. Jones et al 10 compared 40 BPTB allografts treated with and without BioCleanse processing. Mechanical testing showed no difference between the 2 groups about stiffness, creep, maximum force, or ultimate stress at failure. Mikhael et al 15 also found no difference in ultimate stress, strain, or fracture energy between cylindrical cortical bone specimens treated with BioCleanse compared with those treated with BioCleanse and 20-23kGy of gamma irradiation or left untreated. In 2013, Conrad et al 1 published results after testing 20 AlloTrue (AT) allografts, finding that those sterilized by BioCleanse had higher stiffness compared with irradiated grafts. Collectively, these studies suggest that at the time of graft implantation, allografts sterilized by the BioCleanse technique do not demonstrate any notable compromise in mechanical integrity. Despite this, some studies4,9,28 have implicated the BioCleanse process as a possible cause of graft failure in vivo, with failure rates between 0% and 31%. Engelman et al 4 showed a 31% graft failure rate with BioCleanse grafts compared with a 20% failure rate of AlloTrue grafts at 33 months postoperatively. However, this study did not control for graft type, and anterior tibial, posterior tibial, and peroneal grafts were used. Indelicato et al 9 compared 43 BioCleanse BPTB grafts with 24 aseptically processed grafts and showed no graft failure in either group, indicating they were underpowered to detect a difference. Tejwani et al 28 compared BioCleanse allografts with Allowash/AlloTrue allografts in a retrospective database study and found a higher rate of failure in the BioCleanse group (3.8% vs 2.7%). Our results stand in contrast with those of Tejwani et al, 28 suggesting a similar rate of failure between BioCleanse, Allowash XG, and MTF grafts. The difference in outcomes between our study and those of Tejwani is likely multifactorial. Like Engelman et al, 4 Tejwani et al 28 did not control for graft type in their analysis, which is concerning because they show that within their dataset, graft type is a significant contributor to failure rate with a hazard ratio of 1.79 (95% CI, 1.20-2.66) when comparing BPTB with soft tissue grafts. 28 In addition, their study was a database analysis of all allograft ACLRs done in the Kaiser Permanente system, which introduces significant heterogeneity in terms of patient characteristics, surgical procedures, and rehabilitation and follow-up protocols, which could bias their results. Our study adds to the literature by providing the largest non-database evaluation of BioCleanse grafts while controlling for graft type. Like Indelicato et al, 9 we observed similar rates of graft failure and outcome scores between BioCleanse and other allograft processing methods. However, in our cohort, no BioCleanse grafts failed. However, 1 patient with a BioCleanse graft experienced persistent knee pain for which an MRI was ordered and showed partial degeneration of the graft. This highlights the need for further studies comparing BioCleanse grafts with other common allografts with higher power; nevertheless, achieving adequate patient numbers is challenging, as our study was conducted over 8 years at a large academic clinical practice, yielding 193 participants. In addition, maintaining consistency on graft type, surgical technique, and postoperative protocols adds additional hurdles.
Somewhat less is known about Allowash XG and MTF grafts. Allowash utilizes a patented 6-step process involving treatment with hypotonic solutions and antimicrobial agents as well as ultrasonication and centrifugation; the Allowash XG process adds to this protocol by including terminal sterilization with low-dose (<2 Mrad) irradiation and has replaced the nonirradiated process since 19 2005. Greaves et al 7 showed no significant biomechanical differences between Allowash and Allowash XG grafts. Tejwani et al 28 assessed the rate of Allowash and AlloTrue grafts together and found a failure rate of 2.7%, but the Allowash XG failure rate is not well known. To that end, we were able to show an Allowash XG graft failure rate of 5.5% in a cohort of 55 patients followed for a minimum of 2 years. Moreover, we found a similar rate of graft failure between Allowash XG, BioCleanse, and MTF grafts at 2 years. However, there was a trend toward higher failure rates with Allowash XG grafts, even though this did not reach statistical significance, highlighting the need for further studies on this matter.
MTF graft processing involves an antibiotic soak, agitation, and a water rinse, with roughly 40% of grafts preprocessed with a 1.2 to 1.8 Mrad dose of radiation before antibiotic treatment. 19 While biomechanical data are lacking, several studies have reported failure rates for MTF grafts.24,25,26 Smith et al 25 reported a failure rate of 0% at 12 months, Shah et al 24 reported a failure rate of 5.65% at 40 months, and Stringham et al 26 reported a failure rate of 19% at 34 months. In a systematic review, Roberson et al 19 found a failure rate of 6.7% out of a total population of 210 knees. In our cohort, just <1% of MTF grafts failed with a minimum follow-up of 2 years. While this is lower than the rates reported in previous studies, this likely is the product of our older patient cohort when compared with other studies; our patients with MTF grafts were, on average, 41.5 years old whereas the mean age of patients was 37 years in the Smith et al 25 study, 30 years in the Shah et al 24 study, and 34 years in the Stringham et al 26 study. Given that younger age is a known risk factor for allograft failure, our results may represent a better estimate of the failure rate for older populations who more frequently undergo allograft ACLR. 17 Despite this, one must be cautious in interpreting data for relatively rare outcomes (eg, graft failure) in somewhat small data sets. It is likely that the true rate of failure falls somewhere in between the values reported by the studies listed above, which would be similar to the rate of BPTB allograft failure reported generally without segmentation by sterilization method. 2 To that end, this highlights the need for more robust studies or perhaps meta-analyses to gain a more accurate understanding of the failure rate for BPTB allografts treated via the MTF processing protocol.
Limitations
This study should be interpreted within the context of its limitations. First, we relied on clinical assessment of ACL graft integrity to determine graft failure. Only patients with specific complaints or examinations concerning graft failure underwent MRI for assessment of graft integrity. Given this, we may underreport the rate of true graft failure. Second, this was a retrospective study and as such, we are relying on data that were collected and recorded at office visits by 3 surgeons and were not able to establish a uniform assessment of each patient at all time points. However, all patients were examined with a Lachman and anterior drawer at all time points by fellowship-trained sports medicine surgeons. In addition, because of the retrospective nature of the study, we were unable to obtain preoperative IKDC scores and thus were unable to compare pre- and postoperative functional outcomes. We utilized a 2-year minimum follow-up time to help capture a large portion of complications/graft failures. However, it is possible that some patients experienced graft failure outside of our 2-year follow-up time and did not return to our practice, but given our mean follow-up of >4 years, the effect of this would likely be small. Finally, while our paper represents the largest study comparing outcome and failure rates for different types of allograft ACLR, we remain underpowered to detect significant differences between graft failure rates, given the relative rarity of this complication. This is highlighted by the fact that we had a 0% failure rate in the BioCleanse group. A sample size calculation was performed using the previously reported graft failure rates of Tejwani et al 28 and Roberson et al. 19 To compare failure rates between BioCleanse and Allowash XG, BioCleanse and MTF, and Allowash XG and MTF, our calculation suggests we would need 490 (245 patients in each group), 1854 (927 patients in each group), and 876 patients (238 patients in each group). This indicates that we are at risk of a Type 2 error. Given the low rate of ACL graft failure and the rate of allograft usage, it is not possible to capture enough patients at our institution to have the statistical power required to detect these differences. Future studies may benefit from multicenter collaboration to provide a more robust sample size.
Conclusion
BPTB allografts processed using BioCleanse, Allowash XG, and the MTF protocol have similar rates of graft failure and postoperative IKDC scores. Our data suggest that each of these techniques can be used safely with low failure rates and good functional outcomes.
Footnotes
Final revision submitted February 25, 2025; accepted March 7, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.T.K. has received support for education from Smith & Nephew. F.P.T. has received consulting fees from DJO, DePuy Synthes Products, and Medical Device Business Services; and hospitality and travel payments from MicroVention and Smith & Nephew. K.B.F. has received consulting fees from Ferring Pharmaceuticals, Vericel, and Innocoll Inc; a grant from Vericel Corporation; research support from Mitek and Johnson and Johnson; and support for education from Liberty Surgical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by the Jefferson Office of Human Research Institutional Review Board (iRISID-2022-0968).
