Abstract
Background:
The Banff Patellar Instability Instrument 2.0 (BPII 2.0) and Norwich Patellar Instability (NPI) score are reliable, valid patient-reported outcome (PRO) measures for evaluating patellofemoral instability. Both have been translated and culturally adapted to various languages to document PROs accurately. However, no validated Arabic versions of these PRO measures exist.
Purpose:
To translate the BPII 2.0 and NPI score into Arabic and assess the reliability and validity of the translated questionnaires.
Study Design:
Cohort study (diagnosis); Level of evidence, 3.
Methods:
The NPI score and BPII 2.0 were translated into Arabic based on standard guidelines recommended by the Consensus-based Standards for the selection of health Measurement Instruments. Patients who underwent patellofemoral stabilization surgery were included in this study. Respondents completed the translated versions of the questionnaires 1 week apart. The validity of the translation was assessed using Cronbach alpha, and Pearson correlation coefficient (r) was used to determine the relationship between the Arabic versions of the NPI score, BPII 2.0, and Kujala Score (KS). Test-retest reliability was assessed using the intraclass correlation coefficient.
Results:
The translated versions were assessed in 83 patients and found reliable. The response rate was 94.3%. A negative correlation was found between the KS and NPI score (r = −0.80; P < .05) and a positive correlation with BPII (r = 0.84; P < .05). A significant negative correlation was noted between the NPI and the BPII (r = −0.71). For first-time responses, Cronbach alpha for the NPI score was 0.80, with an intraclass correlation coefficient of 0.80 (0.68-0.83), while the BPII was 0.91 (0.87-0.93) and the KS was 0.80 (0.73-0.86). No floor or ceiling effects were observed.
Conclusion:
The study demonstrated that the translated Arabic versions of the BPII and NPI score are valid and reliable for measuring PROs in Arabic-speaking patients with patellofemoral instability.
Keywords
Patellofemoral pain and instability are becoming more common with the increased interest in sports and physical activity. Disease-specific functional outcomes and quality-of-life measures are being developed and validated to assess the impact of the disease and treatment interventions on patients' quality of life. Traditionally used patient-centered knee outcomes, such as the Lysholm knee score, Knee injury and Osteoarthritis Outcome Score (KOOS), and International Knee Documentation Committee (IKDC) Subjective Knee Evaluation, do not specifically assess quality of life and instability for patients with patellofemoral pain and instability.7,14,23,25
The Kujala Score (KS) was developed to evaluate subjective symptoms and functional limitations in those with patellofemoral disorders. 19 However, it is limited in assessing the daily life impact and severity of patellofemoral instability (PFI), as only 1 question specifically addresses the symptoms of PFI.1,2,19 Consequently, new patient-reported outcome (PRO) measures (PROMs), such as the Banff Patellar Instability Instrument 2.0 (BPII) and the Norwich Patellar Instability (NPI) score, have been developed. The BPII for patellar instability was developed by Hiemstra et al 16 in 2013 as a tool to comprehensively assess the quality of life of patients with patellar instability. It is based on the Anterior Cruciate Ligament Quality of Life scale. 22 In 2016, Lafave et al 20 validated a revised version after reducing the number of items in the questionnaire for easier administration and named it BPII 2.0. 16 The Banff questionnaire attempts to generate a holistic view of patient outcomes by assessing the physical and psychosocial effects of patellar instability.15,16 The NPI score was published in 2013 to improve the PROs in patients with PFI. It focuses on physical function, the frequency of dislocation during specific activities, and its effect on activities of daily living. This score is useful for assessing surgical and rehabilitative outcomes. 26
Studies have reported a 9% to 38.3% incidence of patellofemoral pain in the Arabic-speaking population.3,4,21 An Arabic-validated outcome score for assessing quality of life and interventions for PFI is lacking. This study aimed to reliably translate and validate the Arabic version of BPII 2.0 and NPI for use in Arabic-speaking populations. We hypothesized that Arabic versions of the BPII and NPI score are valid and reliable for measuring PROs in Arabic-speaking patients with PFI.
Methods
Study Population and Translation
This study was conducted from November 2023 to August 2024 after approval by the King Saud University institutional review board. The sample size was calculated to be ≥62 patients based on the variables contained in the Banff questionnaire.8,13 A convenience sampling method was used. Hiemstra et al 17 reported that both the NPI and the BPII 2.0 showed a good level of psychometric evaluation for assessing PFI; hence, these scores were chosen to be translated to Arabic.
The translation of the NPI score and BPII into Arabic was based on guidelines provided by Guillemin et al 11 and Beaton et al. 6 The translation aimed to generate a linguistically valid translation that was understandable to the Arabic-speaking population and equivalent to the original questionnaires. The translation process was initiated by obtaining approval from the original developers of the NPI score 26 and the BPII 2.0 20 and explaining the purpose of the questionnaire.
The questions were initially translated from English into Arabic by 2 bilingual orthopaedic surgeons (A.Z.A. and F.A.) and 2 physical therapists (A.S.A. and D.A.) who were fluent in English and Arabic, with Arabic as their first language. Non-professional translators were chosen for the first translation because of the clinical nature of their work, which involved frequent patient interaction; hence, they were considered apt to create a jargon-free translation as opposed to a professional translator with no medical background. They synthesized 2 Arabic translation versions of each questionnaire. The 2 versions of the 2 questionnaires were compared by the 2 surgeons (A.Z.A. and F.A.) along with 2 professional translators, both native Arabic speakers fluent in English. One professional translator had expertise in medical terminology, and another specialized in physical therapy–related translation. The committee addressed discrepancies and resolved disparities between translations. Any unsuitable language was improved, and the final versions for both questionnaires were prepared.
Two additional translators, fluent in Arabic and native English speakers, who were blinded to the original translation and had no knowledge of the subject matter, independently back-translated the Arabic-translated versions of both questionnaires into English.
The translation team (expert committee), consisting of 8 members, 2 orthopaedic surgeons (A.Z.A. and F.A.), 2 physical therapists (A.S.A. and D.A.) and 4 professional translators of which 2 were native Arabic and 2 were native English speakers), reevaluated the translations against each other and the original questionnaire. Discrepancies were analyzed for linguistic accuracy and cultural relevance, and modifications were made as necessary. This process synthesized the Arabic prefinal BPII and NPI questionnaires, prepared along with a detailed written report on the challenges encountered. Final versions were reviewed by a professional Arabic writing and language editor to correct any grammar or typographical errors.
The questionnaire was pretested in 20 patients identified from the records at the study hospital; all patients included in the pretest had been diagnosed with and operated for PFI. They were invited by phone to provide consent to participate in the study and asked to respond to the prefinal versions to assess the face and content validity of the Arabic versions, which were modified from previously used questionnaires. The responses were analyzed for clarity, understanding, and acceptability. Feedback from the participants on comprehensibility and relevance was solicited and analyzed. An expert committee assessed and refined the translations based on the feedback.
The Arabic versions of the Banff questionnaire and NPI score were submitted to the original developers for final approval. All steps of the translation process, information regarding discrepancies, and committee consensus were submitted. The authors approved the versions of the questionnaire without any additional changes. The Banff Arabic translation was named “Banff Arabic Final Version (BPII), and the NPI questionnaire was named “Norwich Arabic Final Version” (NPI). The process of translation from the draft of the first versions to the final translated manuscript took 12 weeks. A summary of this process is shown in Figure 1. The translated versions of the NPI score are provided in Supplemental Material 1 and the BPII 2.0 in Supplemental Material 2, available separately.
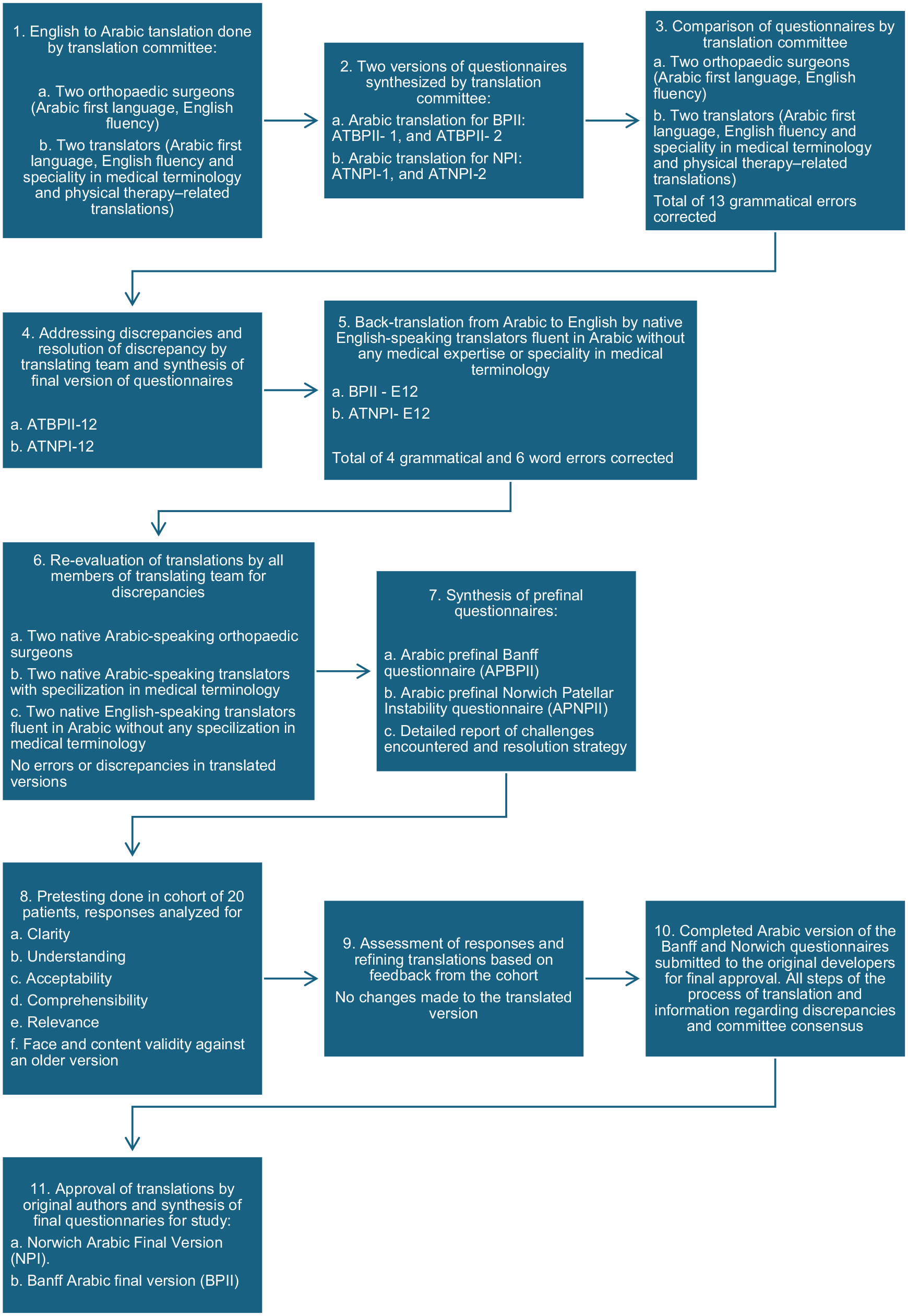
Flowchart of the questionnaire translations from English to Arabic. ATBPII, Arabic translation of Banff Patellar Instability Instrument; ATNPI, Arabic translation of Norwich Patellar Instability score; BPII, Banff Patellar Instability Instrument; E, English; NPI, Norwich Patellar Instability score.
Once the Arabic-translated questionnaire was finalized, patients >18 years of age who had undergone patellofemoral stabilization surgery and consented to participate in the study were contacted. Patients who were unable to answer the questionnaire themselves were excluded, as they may not have been participating in many activities even in the preoperative stage because of their illness, which would have led to a higher rate of unanswered questions. Patients who had participated in the pretest validation process were not included in this study. Upon presenting to the outpatient clinic, a detailed clinical assessment was performed, after which patients were asked to respond to the questionnaire. A research team member assisted the patients or provided any clarifications during questionnaire administration. To minimise the chance of recall bias, the participants were contacted via telephone within 1 week to request that they respond to the questionnaire again after determining no change in their clinical condition regarding their operated knees within 1 week. In case a patient stated a change in condition from the previous week the patient would be excluded from the study, however we did not encounter any such patients.
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics for Windows Version 26. All data were entered into Microsoft Excel 2019, and each variable was assigned a code for analysis. Descriptive statistics were calculated as percentages, means, standard deviations, and ranges. The Shapiro-Wilk test was used to assess data normality. Cronbach alpha was used to evaluate the reliability of the translated version, and values ranging from 0.7 to 0.95 were accepted. Test-retest reliability was assessed using the intraclass correlation coefficient (ICC), and a P value of <.05 was considered significant. Interclass correlation coefficient values of <0.50 were considered poor agreement, 0.50-0.75 was moderate agreement, 0.76-0.90 represented good agreement, and values >0.90 were considered excellent agreement. Pearson correlation coefficient (r) was computed to determine the validity of the translated questionnaire. The KS has already been translated and validated in Arabic, forming the basis for this comparison. 12
Results
A total of 88 patients were contacted to participate in the survey. Of these, 5 patients missed one of the survey questionnaires and were excluded, resulting in a final assessment of 83 patients (response rate, 94.3%). The mean age of participants was 19.99 ± 1.96 years (range, 18-27 years). All patients had undergone surgery for PFI, with a mean duration from surgery to evaluation of 18.69 ± 12.73 months (range, 7-73 months). Table 1 shows the demographic characteristics of the study groups.
Demographic Data of the Study Group (N = 83) a

MPFL, medial patellofemoral ligament.
The translated versions demonstrated good reliability, with acceptable Cronbach alpha values for each questionnaire. On examining the internal consistency of the questionnaires for first-time responses, Cronbach alpha for the NPI score was 0.80 with an ICC of 0.80 (95% CI, 0.68-0.83; P < .05) for the initial assessment. The BPII was 0.91 with an ICC of 0.91 (95% CI, 0.87-0.93; P < .05), and the KS was 0.80 with an ICC of 0.80 (95% CI, 0.73-0.86; P < .05). The scores showed good test-retest reliability. The ICC for the NPI score was 0.99 (95% CI, 0.99-1.0), BPII was 0.98 (95% CI, 0.97-0.98), and KS was 0.98 (95% CI, 0.98-0.99); all values were statistically significant (P < .05) (Table 2). The patients scored a mean of 19.39 ± 8.8 for the NPI score on first completion and 19.3 ± 8.9 after 1 week. For the BPII, patients scored 84.7 ± 9.4 on the initial test and 83.3 ± 7.0 after 1 week. The mean KS was 84.23 ± 10.65 and 83.99 ± 10.42 at the initial assessment and 1 week, respectively (Table 2). The construct validity was calculated using Pearson r. A significant negative correlation was found between the KS and NPI score (r = −0.80; P < .05) and a strong positive correlation with BPII (r = 0.84; P < .05). Additionally, a significant negative correlation was noted between the NPI score and the BPII (r = −0.71) (Table 3). Floor and ceiling effects were not observed in this study, as no patient scored the minimum NPI score in either assessment. For the BPII, no patient had the maximum score at the initial assessment (range, 61.3-93.9) or the 1-week assessment. No patients scored maximum or minimum KS (Table 2). Because the total number of maximum and minimum scores was <15% of the study population, it implies that floor and ceiling effects were not observed.
Descriptive Analysis and Validity Testing of the Translated Scores Showing Good Validity and Intra- and Interclass Correlation a
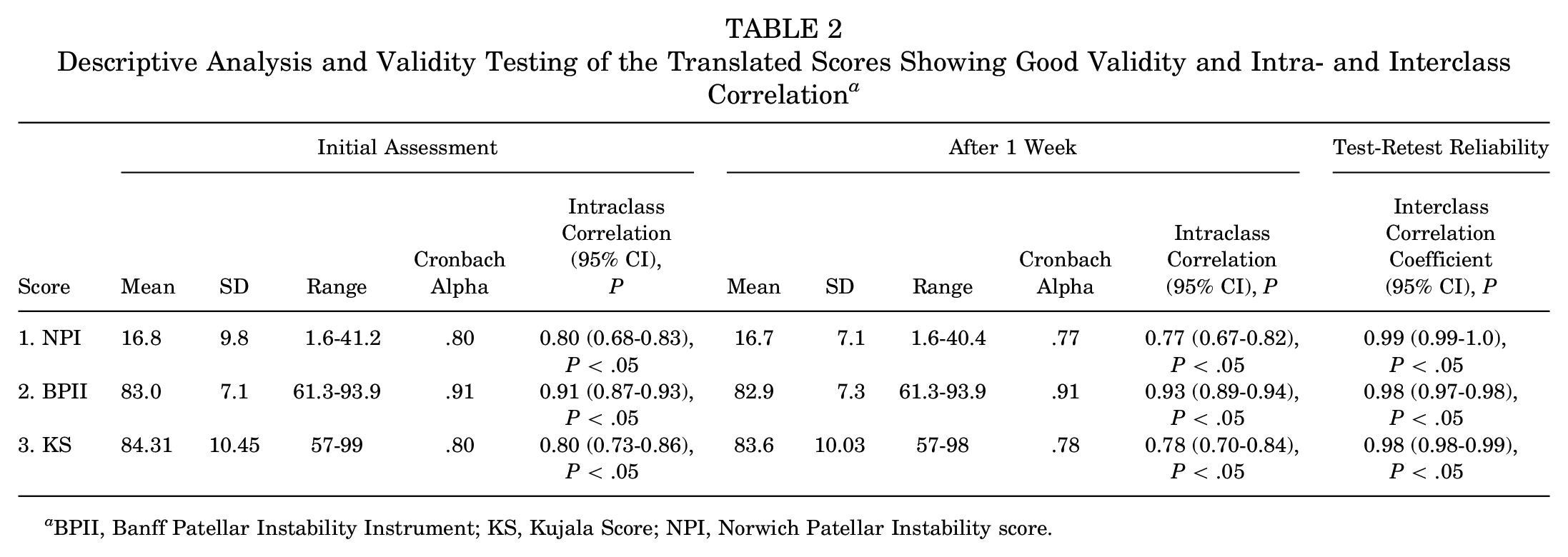
BPII, Banff Patellar Instability Instrument; KS, Kujala Score; NPI, Norwich Patellar Instability score.
Correlation coefficients for NPI score, BPII and KS a

BAFV, Banff Arabic Final Version; BPII, Banff Patellar Instability Instrument; KS, Kujala Score; NAFV, Norwich Arabic Final Version; NPI, Norwich Patellar Instability score.
Discussion
The findings of our study indicate that the translated Arabic versions of the NPI score and the BPII 2.0 are valid, reliable, and could be culturally adapted for Arabic-speaking populations. The NPI and BPII demonstrated good validity with a Cronbach alpha of .81 and .91, respectively. Additionally, a statistically significant ICC confirms the reliability of the Arabic translations of both questionnaires. The NPI score showed a significant negative correlation with both BPII (r = −0.71; P < .05) and KS (r = −0.80; P < .05). The NPI score measures the degree of patellar instability experienced by the patient; a higher score signifies greater instability, explaining the negative correlations with the KS and BPII. 15 The BPII exhibited a significant positive correlation with the KS and a significant negative correlation with the NPI score, indicating a valid translation. A high Cronbach alpha and significant ICC, along with a statistically significant correlation coefficient in the absence of a floor and ceiling effect, indicate that the Arabic translations were successful and can be reliably used for the clinical assessment of patients with PFI. The participants answered all the questions, confirming they understood the translated version well. With a trend toward an active lifestyle and increased participation in sports among the Arabic population, the identification of patellofemoral pain and instability has increased, with reported incidences ranging from 9% to 38.3%.3,4,21 The translated versions of the questionnaires should enable both surgeons and patients to better understand and assess the outcome of surgical interventions as well as their implications on the quality of life of Arabic-speaking populations.
The NPI score is disease specific in measuring PFI; it assesses physical function and frequency of dislocation. The questionnaire is a 19-item scoring system based on a 5-point Likert scale for assessing daily activities. The scoring system is relatively complex, and the final score ranges from 0 to 250. The final score is converted into a percentage. A higher score signifies greater instability in the patient. 26 Arrebola et al 5 reported a moderate correlation with the Brazilian-Portuguese version of both the Lysholm score (r = −0.56; P < .01) and KS (r = −0.57; P < .01). Yilmaz et al 29 translated the NPI score into Turkish with a reported Cronbach alpha of .84 for their version and excellent test-retest reliability (ICC, 0.91; P < .05). Van Sambeeck et al 27 concluded that their Dutch translation was appropriate for patients with patellar instability. They reported excellent internal consistency (Cronbach alpha = .97) and a high correlation with other scores such as the 36-Item Short Form Health Survey and KS. This study also reported good internal consistency (Cronbach alpha = .80) and excellent test-retest reliability, indicating that the translation was accurate and reliable (Table 2).
The BPII was developed by Hiemstra et al 16 in 2013 to assess the quality of life of patients with PFI, evaluating the functional, social, and economic aspects in addition to the physical symptoms of these patients. A higher score signifies a better quality of life. The Dutch translation by Van Seembeck et al 27 reported good internal consistency and a high Cronbach alpha (.97). Additionally, Hysing-Dahl et al 18 reported that the Norwegian version of the BPII has good face and content validity, as well as excellent internal consistency (Cronbach alpha = .95). After factor analysis and a reduction in the number of questions, the BPII 2.0 was developed by Lafave et al. 20 The BPII 2.0 has been translated and validated in Turkish, French, Spanish, and Finnish.17,18,27-29 Rhatomy et al 24 reported good internal consistency and high test-retest reliability for their Indonesian version of the questionnaire. This study found the translated version to have excellent internal consistency (0.91) with an ICC of 0.91 (95% CI, 0.87-0.93; P < .05), comparable with the values by the other authors, indicating successful translation of the questionnaire.
Hiemstra et al 15 also successfully validated the BPII 2.0, NPI, and KS. Yilmaz et al 29 demonstrated an excellent correlation of BPII 2.0 with the Turkish versions of the KS and Lysholm scores. Van Seembeck et al 27 successfully translated the BPII 2.0 and NPI score into Dutch in a single study. The results of this study demonstrated that both the NPI score and BPII showed excellent correlation with the already validated Arabic version of the KS using similar methods (Table 3). This translated questionnaire can effectively report PROs using the NPI score and BPII for Arabic-speaking patients with PFI. A comparison with other commonly used scores, such as the KOOS patellofemoral subscale and the IKDC Subjective Knee Evaluation, was not done in this study as they did not correlate with KS despite being validated to measure PFI.9,17,23
Limitations
This cross-sectional study is subject to limitations inherent to this design. The relatively small sample size of 83 patients, narrow age range, chronicity of the disease, and type of procedure could have affected the outcomes. The variable postoperative follow-up of the participants is also a notable limitation. A variable postoperative period may increase the variation of responses (eg, a patient who has been operated on recently is expected to score lower in quality-of-life scores as compared with a patient in whom a significant time since surgery has passed). Sample size calculations yielded a sample size of 62 based on the variables in the Banff score for a power of 0.80. This study has reported results in 83 patients, but a larger sample size would yield a wider age range for the study, and a more consistent follow-up period may have yielded higher power for the study. This study was conducted in patients who had already been operated on for PFI, but a comparison of the pre- and postoperative scores could have further validated the accuracy of translation. 10 It could provide insight into the change in the patient's condition in all the scores. This study used only the KS because it is more specific to the patellofemoral joint, excluding other PROMs, such as the Lysholm knee score. The Lysholm knee score has demonstrated good convergent validity and strong positive correlation with the KS, and a comparison could have further improved the validation of this translation.17,23
Conclusion
Our study demonstrated that the translated Arabic versions of the NPI score and BPII are reliable and valid for measuring PROs in patients with PFI, enabling accurate assessment in the Arabic-speaking population. Future studies should further validate the versions of BPII and NPI in this study for nonoperatively managed patients as well.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671251340989 – Supplemental material for Reliability and Validity of the Arabic Translated Version of the Norwich Patellar Instability Score and the Banff Patellar Instability Instrument 2.0
Supplemental material, sj-pdf-1-ojs-10.1177_23259671251340989 for Reliability and Validity of the Arabic Translated Version of the Norwich Patellar Instability Score and the Banff Patellar Instability Instrument 2.0 by Abdulaziz Z. Alomar, Faisal A. Alghamdi, Ahmed S. Alhowimel, Faris Alodaibi and Dalia Alimam in Orthopaedic Journal of Sports Medicine
Supplemental Material
sj-pdf-2-ojs-10.1177_23259671251340989 – Supplemental material for Reliability and Validity of the Arabic Translated Version of the Norwich Patellar Instability Score and the Banff Patellar Instability Instrument 2.0
Supplemental material, sj-pdf-2-ojs-10.1177_23259671251340989 for Reliability and Validity of the Arabic Translated Version of the Norwich Patellar Instability Score and the Banff Patellar Instability Instrument 2.0 by Abdulaziz Z. Alomar, Faisal A. Alghamdi, Ahmed S. Alhowimel, Faris Alodaibi and Dalia Alimam in Orthopaedic Journal of Sports Medicine
Footnotes
Acknowledgements
The authors are grateful to the College of Medicine Research Center, Deanship of Scientific Research, King Saud University, for supporting our project.
Final revision submitted January 10, 2025; accepted February 12, 2025.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from King Saud University institutional review board (No. 24/1528).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
