Abstract
Background:
Femoroacetabular impingement (FAI) can cause hip pain and osteoarthritis in patients. Importantly, 3-dimensional (3D) bone models in dynamic hip impingement simulations can enable a patient-specific diagnosis of FAI. The manual segmentation of bone models is time-consuming; therefore, automatic segmentation should be investigated.
Purpose:
To investigate the difference between manual and automatic segmentation of magnetic resonance imaging (MRI)–based 3D bone models of the hip, (2) to correlate impingement-free hip range of motion, and (3) to determine external validation.
Study Design:
Cohort study (Diagnosis); Level of evidence, 3.
Methods:
An institutional review board–approved retrospective study involving a total of 98 hips was performed. Of these, 30 patients with symptomatic FAI (60 hips; mean age, 27 ± 9 years) and 19 asymptomatic participants (38 hips) underwent 3-T MRI of the hip including a rapid 3D T1-weighted VIBE Dixon sequence of the pelvis (192 images acquired in 32 seconds). The automatic segmentation of MRI-based 3D bone models was performed using machine learning (convolutional neural network). The Dice similarity coefficient (DSC) was calculated for 98 hips to assess the overlap and difference in MRI-based 3D bone models with 5-fold cross-validation. Automatic segmentation was assessed with 16 patients (32 hips) from another institution for external validation. Impingement-free range of motion was compared between the manual and automatic segmentation of MRI-based 3D bone models (30 patients with FAI).
Results:
The difference between the manual and automatic segmentation of MRI-based 3D bone models was <1 mm (<0.6 mm for pelvic models and <0.5 mm for femoral models), and the DSC of the 30 patients with FAI was 94.1% (pelvis) and 97.0% (proximal femur); for the 19 asymptomatic participants, the difference was <0.5 mm, and the DSC was 95.5% and 97.5%, respectively. The correlation for impingement-free flexion (r = 0.93; P < .001) was excellent (30 patients with FAI). The mean difference in flexion and internal rotation at 90° of flexion was 2.9°± 4.0° and 3.0°± 4.0°, respectively. The DSC of the 16 patients from another institution was 92.2% (pelvis) and 94.9% (proximal femur) for external validation.
Conclusion:
The automatic segmentation of MRI-based 3D bone models was as accurate as the manual segmentation of MRI-based 3D bone models for patients with FAI. It allows radiation-free and patient-specific preoperative surgical planning of hip preservation surgery and hip arthroscopic surgery for patients with FAI of a childbearing age. The automatic segmentation of MRI-based 3D bone models using deep learning was feasible with routine MRI (3D T1-weighted VIBE Dixon sequence) with a short image acquisition time (<1 minute).
Keywords
Femoroacetabular impingement (FAI) is a major cause of hip pain and osteoarthritis in patients of a childbearing age and can limit range of motion (ROM) due to early abutment between osseous prominences of the femur and the acetabulum. 14 Treatment with hip arthroscopic surgery can successfully reduce hip pain caused by intra-articular FAI,5,17 and hip arthroscopic surgery has evolved and increased in popularity in recent years. Patients with FAI typically experience pain during squats, ice hockey, or dynamic activities of daily living. 14 Although FAI is a dynamic conflict, standard 2-dimensional (2D) imaging is static and limited because it cannot visualize the exact location of the deformity 11 and differs between supine and standing.13,36 A recent study described a high prevalence of femoral and acetabular version abnormalities combined with cam- or pincer-type deformities in patients with hip pain. 23 Femoral version abnormalities have been associated with intra- and extra-articular FAI. 20
To overcome these problems, a dynamic 3-dimensional (3D) impingement simulation to calculate impingement-free ROM was introduced. 33 It allows the replication of human hip ROM and can reproduce patient-specific and painful movements of patients with symptomatic FAI. 20 Therefore, computed tomography (CT)–based 3D bone models were used for exact surgical planning of hip arthroscopic surgery 1 and open hip preservation surgery. 38 Because of the considerable radiation exposure of CT, 35 this 3D impingement simulation has not been commonly applied in clinical practice. Recently, radiation-free magnetic resonance imaging (MRI)–based 3D bone models were introduced9,38 to overcome the issue of radiation exposure. 35 MRI-based 3D bone models 16 have been used for 3D dynamic hip impingement simulations that enable the patient-specific diagnosis of intra- and/or extra-articular FAI.20,22 Yet, these MRI-based 3D bone models were obtained by time-consuming manual segmentation. Therefore, automatic segmentation using deep learning was investigated for the hip joint because good results have been reported previously with segmentation of knee cartilage and bone. 26 Previous studies10,38 have investigated the automatic bone segmentation of hip MRI and have used deep learning for the detection of fractures of the hip. 8 Yet, they described segmentation of the proximal femur only, while no segmentation of the acetabulum was performed. 10
The aim of this study was to examine the feasibility of automatic bone segmentation for MRI-based impingement simulations of patients with FAI of a childbearing age to reduce radiation exposure for future patients. We aimed (1) to investigate the difference between the manual and automatic segmentation of MRI-based 3D bone models of the hip, (2) to correlate impingement-free hip ROM, and (3) to determine external validation (with a dataset from another institution).
Methods
An institutional review board–approved retrospective study involving a total of 98 hips was performed. Of these, 30 patients with symptomatic FAI (60 hips) underwent 3-T magnetic resonance arthrography (MRA; 3D T1-weighted VIBE [volumetric interpolated breath-hold examination] Dixon sequence) of the pelvis. An additional 19 asymptomatic participants (38 hips) underwent 3-T MRI (not MRA) of the pelvis. The automatic segmentation (Figure 1) of MRI-based 3D bone models (Figure 2) using machine learning was compared with manual segmentation using water-only Dixon images. For automatic segmentation, a convolutional neural network (CNN) was used, and 5-fold cross-validation was performed. Furthermore, the generated automatic MRI-based 3D bone models were used for the aforementioned impingement simulation.

(A) Water-only 3-dimensional T1-weighted VIBE Dixon magnetic resonance imaging (MRI) and (B) automatic segmentation of a patient with symptomatic femoroacetabular impingement.

Magnetic resonance imaging (MRI)–based 3-dimensional bone model using automatic segmentation (A) without and (B) with axial images.
Participants
We evaluated symptomatic patients consecutively who had been referred to our university center for hip preservation surgery during a 2-year period (January 2017 to February 2018). Based on a history of hip pain as well as clinical and radiological findings consistent with FAI or hip dysplasia, symptomatic patients were referred for imaging. We performed the automatic segmentation of MRI-based 3D bone models and compared it to the manual segmentation of MRI-based 3D bone models. We evaluated the manual and automatic segmentation of the same patients.
The inclusion criteria for the patients were as follows: availability of standard anteroposterior (AP) radiographs, availability of standardized direct MRA of the hip including images of the entire pelvis, radiographic signs of skeletal maturity, and presence of hip pain at the time of image acquisition. The institutional imaging database was reviewed for all patients in whom direct MRA of the hip was performed between January 2017 and February 2018; this resulted in 47 patients. We had to exclude 17 patients for the following reasons: 12 patients with previous hip surgery, 3 skeletally immature patients, and 2 patients with posttraumatic conditions.
All symptomatic patients were examined clinically at our university center for hip preservation surgery. The standard clinical evaluation consisted of patient history, measurements of hip ROM, and the anterior and posterior impingement tests. 34 Routinely, we obtained AP pelvic radiographs in a standardized manner 34 and MRA of the hip as part of the diagnostic preoperative evaluation for hip preservation surgery. The mean age was 27 ± 9 years, and 60% were female (Table 1). Of the included patients, 12 had an isolated cam-type deformity, while a mixed-type (both cam and pincer types) deformity was present in 4 patients, and an isolated pincer-type deformity was present in 2 patients; some patients had a cam-type deformity combined with increased or decreased femoral version (Appendix Table A1). The definitions of cam- and pincer-type deformities were in accordance with previously published criteria on conventional AP pelvic radiographs 32 and are described in Appendix Table A2.
Asymptomatic participants (38 hips) underwent noncontrast 3-T MRI of the hip including the same 3D T1-weighted VIBE Dixon sequence of the pelvis. They were available from a previous study. 30 The mean age was 27 ± 3 years (Table 1). Patients with symptomatic FAI (16 patients; 32 hips) from another institution (Department of Radiology, District Hospital St. Johann in Tirol, Austria) underwent 1.5-T MRI of the hip including the same 3D T1-weighted VIBE Dixon sequence of the pelvis.
Demographic and Radiological Data of Study Cohort a
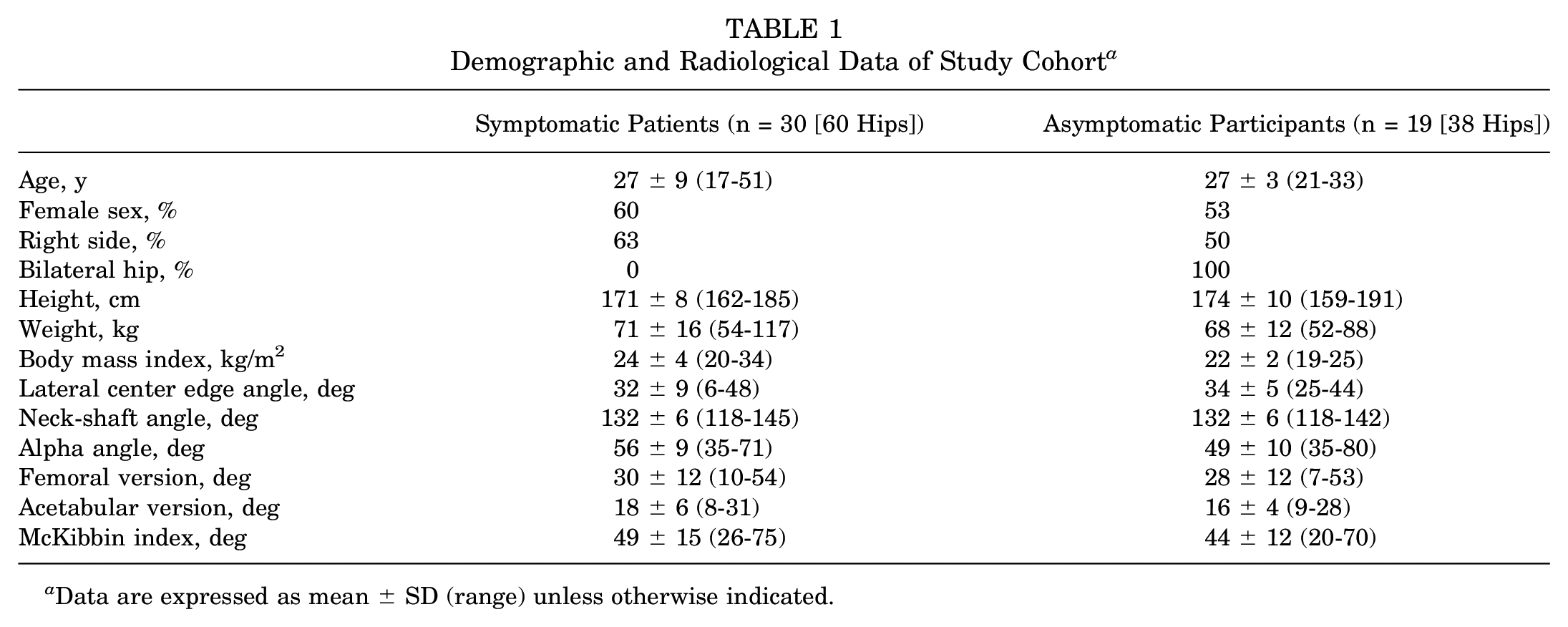
Data are expressed as mean ± SD (range) unless otherwise indicated.
Imaging Technique
We used a standardized protocol for all patients for MRA on a 3-T scanner (Siemens) with large flexible surface coils and proton density–weighted images in the coronal, sagittal, axial, and radial planes. MRA was performed with fluoroscopic guidance, and 10 to 15 mL of a gadolinium-based contrast agent was injected on the symptomatic side. In addition, we obtained bilateral 3D T1-weighted VIBE Dixon sequences of the pelvis (Figure 1A) and of the knee. The field of view of the bilateral sequence covered the entire pelvis from the anterior superior iliac spine to the level below the lesser trochanter (Figure 1A). The field of view of the bilateral proximal femur covered the entire knee joint. The total image acquisition time (multiplanar standard protocol including localizers) was 30 minutes. The 3D T1-weighted VIBE Dixon sequence (Table 2) included the entire pelvis (repetition time/first echo time/second echo time, 3.94/1.27/2.50 milliseconds; flip angle, 9°; slice thickness, 1 mm; field of view, 312 × 400 mm; matrix size, 175 × 320 mm; anisotropic voxel size, 1.2 × 1.2 × 1.0 mm; 192 images acquired in 32 seconds). A second 3D T1-weighted VIBE Dixon sequence (same parameters) for bilateral knees was acquired. This sequence is readily available on many MRI scanners worldwide.
Parameters on 3-T MRI for Bone Segmentation a

3D, 3-dimensional; DICOM, Digital Imaging and Communications in Medicine; MRI, magnetic resonance imaging; NIfTI, Neuroimaging Informatics Technology Initiative; VIBE, volumetric interpolated breath-hold examination.
For the asymptomatic participants and the symptomatic patients from another institution, MRI without contrast (not MRA) was performed, and the same 3D T1-weighted VIBE Dixon sequence (same parameters) was used for the entire pelvis and for the bilateral knees on a 3-T scanner (Siemens) with large flexible surface coils. No data preprocessing of the images was performed. There were no missing data.
Manual Segmentation of 3D Models
The manual segmentation of MRI-based 3D bone models was performed in a standardized manner. Each MRI-based 3D bone model included a 3D surface model of the entire pelvis and the proximal femur. This resulted in 98 MRI-based models of the pelvis and 196 MRI-based models of the proximal femur. Manual segmentation was performed semiautomatically for each hip joint by 2 observers (an orthopaedic resident and a radiology resident) and supervised by one radiologist (T.D.L.) with >5 years of experience in musculoskeletal radiology using a threshold-based method with Amira software (Thermo Fisher Scientific). The manual segmentation of MRI-based 3D bone models was performed using true axial water-only images with a slice thickness of 1 mm from the 3D T1-weighted VIBE Dixon sequence and took up to 3 to 4 hours. A musculoskeletal radiologist conducted a visual review of the 3D models. All of the 3D models were found to be satisfactory during the visual review.
Automatic Segmentation of 3D Models
The automatic segmentation of MRI-based 3D bone models was performed using deep learning. We developed a fully automatic segmentation method based on deep learning for 3D bone models of the hip joint from MRI (Figure 2). Deep learning is a part of machine learning and is based on neural networks, especially CNNs. The neural network architecture for the fully automatic segmentation of the hip joint on MRI is shown in Figure 3, which used the water-only Dixon images as input and a probability map as output that was utilized to derive the segmentation map. For preprocessing, images were converted to NIfTI (Neuroimaging Informatics Technology Initiative) files in an anonymized fashion. There was no missing anonymization. Automatic segmentation had a duration of 60 to 95 seconds per patient.

Architecture of the convolutional neural network for automatic segmentation of the pelvis.
The hip joint segmentation network was developed based on LP-U-Net, which was introduced in a previous study, 39 in which holistic decomposition convolution and dense upsampling convolution were applied at the beginning and end of 3D U-Net, respectively. Holistic decomposition convolution consists of a periodic down-shuffling operation, followed by a standard 3D convolution. LP-U-Net has the advantage of significantly reducing GPU (graphics processing unit) memory for subsequential processing while incorporating larger context information for better performance.
We combined the 60 hips of the symptomatic patients with the 38 hips of the healthy participants and performed standard 5-fold cross-validation. The 98 hips of 49 people were randomly divided into 5 groups: 4 groups contained 10 data points each, and 1 group contained 9 data points. Each time, 1 group was used as the test dataset (9 or 10 data points), and the remaining 4 groups were used as the training dataset. We repeated this process 5 times so that each data point was used once as part of the test dataset. All hip joint images had a voxel size of 1.2 × 1.2 × 1.0 mm3 spacing and a data point size of 320 × 320 × 250. The dataset of the 32 hips (16 patiets) from another institution was used as an unseen test set. We used the following setup: a subvolume of 128 × 224 × 288 voxels was cropped to train and test 3D LP-U-Net. The shuffling factor used in LP-U-Net was (2, 2, 2). Each subvolume was normalized as zero mean and unit variance before feeding into the network. All parameters were trained from scratch and initialized using the Gaussian distribution (µ = 0; σ = 0.01). All parameters were updated by the stochastic gradient descent with momentum algorithm (β = 0.9; weight decay = 0.005). The early stopping criterion was used, and training stopped when the accuracy on the validation dataset did not increase any more after every 1000 iterations. Each neural network was trained with a maximum of 10,000 iterations. The initial learning rate was 0.002 and halved every 1500 iterations. We used the Dice similarity coefficient (DSC), precision, recall, average surface distance (ASD), and Hausdorff distance as the main evaluation metrics. Our method was implemented with Python using TensorFlow on a workstation with a 3.6-GHz CPU (central processing unit; Intel i7) and an 11-GB graphics card (GTX 1080 Ti; Nvidia).
Dynamic Impingement Simulation
Specific software to assess impingement-free ROM and the location of hip impingement was used for the 30 patients with FAI. The software was based on a validated collision detection algorithm.28,33 The acetabular reference system was the anterior pelvic plane, while the femoral reference system was based on the proximal, the distal and the posterior femoral condyles. The following ROM parameters were calculated in 1° increments: flexion, extension, abduction, adduction, internal rotation, and external rotation at 90° of flexion and extension. The interobserver correlation coefficient was high for ROM values in a previous publication. 33 We evaluated 2 motion patterns with isolated movements: impingement-free flexion and extension. We examined whether impingement was located intra- or extra-articularly. In addition, we evaluated 2 motion patterns with combined movements: the first corresponded to the anterior impingement test, performed at 90° of flexion and 30° of internal rotation, similar to previous publications.2,21,22 Furthermore, with the second motion pattern, we aimed to reproduce the posterior impingement test. Impingement was assessed at 30° of external rotation combined with 30° of extension. The software has been described in previous publications 33 and was developed in collaboration with biomechanical engineers from the University of Bern using a validated medical research framework. 28
To fulfill the first aim of our study (to investigate the difference between manual and automatic MRI-based 3D bone models) and to calculate accuracy, the manual MRI-based 3D bone model served as the gold standard for the asymptomatic participants and the patients with FAI. We used the DSC and ASD as the outcome parameters. The DSC was calculated for 98 hips using manual and automatic MRI-based 3D bone models. To fulfill the second aim (to correlate impingement-free hip ROM), impingement-free ROM and the location of impingement were compared between the manual and automatic MRI-based 3D bone models (Appendix Table A3) of the 30 patients with FAI by 2 different observers (T.D.L. and A.B.).
Statistical Analysis
We used WinStat software (R. Fitch Software) to perform statistical analysis. The normal distribution was assessed using the Kolmogorov-Smirnov test for continuous variables. The Pearson correlation coefficient was used because the variables were normally distributed. Absolute mean differences were calculated for continuous variables. The dependent t test was used to compare continuous variables.
Results
The difference between manual and automatic MRI-based 3D bone models was <1 mm (<0.6 mm for pelvic models and <0.5 mm for femoral models). The ASD was 0.58 mm for pelvic models comparing manual and automatic MRI-based 3D bone models of the 30 patients with FAI (Table 3). The ASD was 0.44 mm for femoral models comparing manual and automatic MRI-based 3D bone models of the 30 patients with FAI (Table 3). The DSC of the 30 patients with FAI was 94.1% (pelvis) and 97.0% (proximal femur) (Table 3). The DOC of the 19 asymptomatic participants was 95.5% (pelvis) and 97.5% (proximal femur) (Table 3).
Accuracy of Automatic MRI-Based 3D Bone Models Versus Manual MRI-Based 3D Bone Models a

Data are expressed as mean ± SD (range). 3D, 3-dimensional; ASD, average surface distance; DOC, Dice overlap coefficient; MRI, magnetic resonance imaging.
The correlation for all ROM values between the manual and automatic MRI-based 3D bone models was excellent (r = 0.986; P < .001). In addition, the correlation for impingement-free flexion (r = 0.93; P < .001) and extension (r = 0.99; P < .001) was excellent for the patients with FAI between manual and automatic MRI-based 3D bone models. The mean difference in flexion and internal rotation at 90° of flexion was 2.9°± 4.0° and 3.0°± 4.0°, respectively. Intra-articular impingement occurred with similar frequency using manual and automatic MRI-based 3D bone models during the anterior impingement test (Figure 4 and Appendix Table A3). During the posterior impingement test, 73% of impingement was located intra-articularly using manual MRI-based 3D bone models compared with 67% using automatic MRI-based 3D bone models (Figure 4).

Prevalence of intra- and extra-articular hip impingement for the automatic and manual segmentation of magnetic resonance imaging (MRI)–based 3-dimensional bone models. In 30 symptomatic patients, the location of intra- and extra-articular impingement was evaluated.
The DSC of the 16 external patients with FAI was 92.2% (pelvis) and 94.9% (proximal femur) (Table 3). The ASD was 0.93 mm for pelvic models comparing manual and automatic MRI-based 3D bone models of the 16 external patients (Table 3). Manual segmentation took 3 to 4 hours per patient, while automatic segmentation had a duration of 60 to 95 seconds per patient (also for asymptomatic participants).
Discussion
A retrospective study evaluating the manual and automatic segmentation of a total of 98 hips was performed with 3D T1-weighted VIBE Dixon images. Automatic segmentation using deep learning was very time efficient and accurate compared with manual segmentation. This study is novel in that we used a full automatic radiation-free and patient-specific deep learning method in a preoperative 3D impingement simulation. The DSC was >90%, and the difference was <1 mm, for all patients with FAI; for the asymptomatic participants, slightly better parameters for accuracy were found. In addition, automatic MRI-based 3D bone models were able to be used for a hip impingement simulation. For the future use of this technique, standardized MRI protocols could be important. The imaging sequence used in the current study is readily available on many MRI scanners worldwide. Previously, MRI-based 3D models of the hip joint and pelvis have used other MRI protocols, performed manual segmentation, and applied different methods for segmentation of the 3D models, which are not standard in clinical practice. 21 Long image acquisition times, inhomogeneous bone intensity, or unclear boundaries between bone and soft tissue could possibly explain the differences.
Comparing the accuracy of our results with those from previous studies, we found comparable performance. A previous study used deep learning for automatic segmentation of the hip joint and reported comparable results. 38 For the proximal femur, others have described a DSC of 95%, precision of 95%, and recall of 95% using a deep CNN for segmentation. 10 These values are slightly lower compared with the results of the current study (DSC of 97.0% for the proximal femur). A recent study reported a DSC of 95% for automatic segmentation of the proximal femur in pediatric patients using U-Net. 15 Another study reported a lower Hausdorff distance for segmentation of the proximal femur using a random forest classifier. 9 Another study reported a slightly higher surface distance ranging from 0.55 to 0.75 mm while investigating automatic segmentation of the femoral head and the acetabulum. 7 Additionally, comparing the time needed for automatic segmentation, a previous study reported that the automatic segmentation of bone and rotator cuff tears on shoulder MRI could reduce time and effort using nnU-Net, 19 achieving a DSC of up to 97.8% for bone segmentation.
Comparing the images used for bone segmentation, previous studies have utilized 2D images with a radial orientation 9 or 2D images with a 2-mm slice thickness. 16 Others have used so-called black bone images or zero echo time images 4 for the better visualization of bone on MRI. A previous study generated 3D models of the femur using 1.5-T MRI of cadaveric specimens. 27 Another study used a FLASH Dixon MRI sequence to evaluate the osseous anatomy of patients with hip impingement. 29 We found another study that used water-only Dixon images of the shoulder joint, which are comparable to the images used in our study. 6 That study described a similar DSC for the humerus of 95%. Others have utilized T1-weighted images for shoulder segmentation 18 and reported a DSC of 91% using nnU-Net. An advantage of water-only Dixon images is their good contrast between soft tissue and bone. 12 In addition, MRA in the current study provided good contrast for the hip joint space; this was different compared with previous studies that used MRI without contrast (not MRA). MRA was performed to improve the detection of cartilage and labral lesions. However, the good results with automatic segmentation for the asymptomatic participants without contrast (ie, MRA) show that bone segmentation can be performed without MRA. Another advantage of the used sequence is the short image acquisition time (<1 minute for 192 images). A reported disadvantage is the low signal acquired in cortical bone. 12
Compared with previous studies, the images used in the current study had a shorter image acquisition time. 12 A recent review summarized current approaches for bone segmentation on MRI and found no other imaging method with a shorter image acquisition time. 12 Previous studies have used hip images with an image acquisition time of up to 9 minutes (for 128 images)37,38 or up to 10 minutes for long bones. 31 Further, 2 previous studies used gradient echo Dixon images of the hip joint and reported an image acquisition time of 3.5 minutes.27,29 The aforementioned review stated that CT segmentation can overestimate the actual bone shape, whereas MRI segmentation can underestimate it. 12
Comparing the results found for the impingement location in a dynamic impingement simulation, we found similar findings compared with previous studies using the same software.20,22 Anterior extra-articular subspine hip impingement has been described previously in patients with decreased femoral version,2,20 while posterior extra-articular hip impingement has been described in extension and was mainly located between the lesser trochanter and the ischial tuberosity.3,24 This is similar to the results found in our study (Appendix Table A3).
Automated analysis of MRI is challenging, with various factors contributing to the difficulty of bone segmentation, including image artifacts, varying image contrast, partial volume effects, and the inherent heterogeneity of image intensity. 25 Deep learning techniques, especially CNNs, improve with more data in a continuous learning process. Future studies should focus on the continuous addition of more patients to provide a larger and more variable dataset, which could help to reduce the amount of errors during segmentation and increase the likelihood of producing accurate 3D MRI models.
This study has limitations. While our dataset included a variety of common hip abnormalities, including cam- and pincer-type deformities and femoral version abnormalities (Appendix Table A1), patients with previous surgery were excluded from our dataset. Internal validation of our deep learning method may not be sufficient for its performance in future patients. Other limitations included a small sample size (30 patients with FAI) to evaluate the accuracy of a hip impingement simulation.
To the best of our knowledge, this is one of the few studies that used CNN-based automatic segmentation of MRI-based 3D bone models for patient-specific, radiation-free, and noninvasive surgical planning. Deep learning enables the fast segmentation of standard MRI of the entire pelvis. We performed external validation of our CNN with 16 patients from another institution with MRI (not MRA), which is essential before implementing such models in clinical practice. MRI (3D T1-weighted VIBE Dixon) is used routinely in our clinical examinations and could be applied in other institutions in the future because it has a short acquisition time of <1 minute.
Conclusion
The automatic segmentation of MRI-based 3D bone models using a 3D T1-weighted VIBE Dixon sequence was as accurate as manual segmentation for patients with FAI. The fast automatic segmentation of MRI-based 3D bone models combined with a short image acquisition time allows time-efficient, radiation-free, and patient-specific preoperative diagnoses and surgical planning. Because of the short image acquisition time of <1 minute, this has the potential to be used in clinical practice. This also has implications for the planning and simulation of hip preservation surgery and hip arthroscopic surgery for patients with FAI of a childbearing age.
Footnotes
Appendix
Prevalence of Intra- and Extra-articular FAI According to Manual Versus Automatic MRI-Based 3D Bone Models for Symptomatic Patients a

| Manual | Automatic | |||
|---|---|---|---|---|
| Intra-articular | Extra-articular | Intra-articular | Extra-articular | |
| Anterior impingement test | 97% | 0% | 97% | 0% |
| Maximal flexion | 90% | 30% | 93% | 27% |
| Posterior impingement test | 73% | 70% | 67% | 67% |
| Maximal extension | 63% | 33% | 57% | 33% |
Anterior impingement test signifies 90° of flexion and 30° of internal rotation. Posterior impingement test signifies 30° of extension and 30° of external rotation. 3D, 3-dimensional; FAI, femoroacetabular impingement; MRI, magnetic resonance imaging.
Final revision submitted October 22, 2024; accepted November 18, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.D.L. has received funding from the Swiss National Science Foundation (grant No. P2BEP3_195241). We thank the Swiss National Science Foundation for payment of the APC (article processing charges). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Bewilligung der KEK Bern (2018-00078).
