Abstract
Background:
The medial collateral ligament (MCL) is one of the most commonly injured ligaments in the knee, accounting for 40% of all knee injuries. Autologous platelet concentrate, such as platelet-rich plasma (PRP) and platelet-rich fibrin (PRF), has been proposed as a novel therapy in ligament injury.
Purpose:
To evaluate the effectiveness of PRP and PRF in recovery after MCL injury.
Study Design:
Controlled laboratory study.
Methods:
This study was a randomized, single-blind, in vivo study on MCL injury in Wistar rats (Rattus norvegicus). Rats that were sick, had a decrease in body weight of >10% after adaptation in the laboratory, or died were excluded. A total of 30 male Wistar rats were divided into 3 groups: group 1 (n=10) received no intervention, group 2 (n=7) received PRP, and group 3 (n=10) received PRF. The variables that were investigated included tensile strength and histopathological appearance. All statistical analysis was performed using SPSS 22.0 using the appropriate test based on the variable.
Results:
This study found that there was a significant difference in tensile strength across all groups (P = .004), with a significantly higher value in the PRF group compared with the PRP group (mean difference, 2.76; P = .003). There was no significant correlation between tensile strength and ligament length (P = .34). Based on the histopathological data, this study found that PRF resulted in a higher amount of collagen cell orientation when compared with PRP and control (P = .049).
Conclusion:
This study found that PRF was associated with greater tensile strength when compared with PRP and control. However, more studies with a longer observation period and further trials, especially in humans, should be conducted to confirm this finding.
Clinical Relevance:
PRF injection can be a novel therapy consideration for ligament injury, with a better result in both function and histological appearance when compared with PRP.
Medial collateral ligament (MCL) is an important primary stabilizer in the medial side of the knee joint that resists valgus stress, rotational force, and anterior translation force in the tibia. Injury to the MCL is common, occurring in approximately 40% of all knee injuries. 4 In observational studies of athletes, MCL tears comprise 7.9% of all knee injuries. 14
Depending on the degree of injury, operative and nonoperative care may be considered in isolated MCL injuries. Prolonged immobilization in nonoperative treatment results in bone resorption at the ligament's insertion site and MCL collagen degradation and may be a reason to start early mobilization in treating patients with MCL injury.10,22 This, in turn, may be an indication for operative management.
Autologous platelet concentrate has been widely used as a simple, cost-effective method to allow a high concentration of growth factors in injured tissue. Examples of autologous platelet concentrate include platelet-rich plasma (PRP) and platelet-rich fibrin (PRF).5-9 PRP consists of growth factors and bioactive proteins that may influence the recovery of injured tendon, ligament, muscle, and bone.8,15,23 PRP has been used clinically in humans, as the increase of autologous growth factors and secretory protein may improve the recovery process at a cellular level.11,17 PRP contains growth factors that stimulate cellular anabolism, inflammatory mediators, and modulators that exert anti-inflammatory effects, as well as fibrinogen that acts as a biomaterial scaffold. Platelets in the blood are a source of various growth factors, coagulation factors, adhesion molecules, cytokines, chemokines, and integrins. After activation, platelets in PRP can release a variety of growth factors at concentrations significantly higher than baseline blood levels, including transforming growth factor–β, platelet-derived growth factor, insulin-like growth factor, basic fibroblast growth factors, vascular endothelial growth factor, epidermal growth factors, and many others. These anabolic cytokines are chondropromoting and chondroprotective, stimulating chondrocyte and multipotent mesenchymal stem cell proliferation, promoting chondrocyte synthesis of aggrecan and collagen type II, driving mesenchymal stem cell chondrogenic differentiation, preventing chondrocyte and mesenchymal stem cell apoptosis, and diminishing the catabolic effects of inflammatory cytokines. PRP also contains plasma proteins, which are critical components in the healing mechanism of connective tissues. Plasma contains fibrinogen and other clotting factors, which can be activated to form a provisional fibrin scaffold for cells to adhere, migrate, and proliferate.18,25,26
PRF is a biomaterial fibrin that serves as a binding site for platelet and growth factors, which are essential for tissue recovery. An increase in local growth factor in specific tissue may lead to tissue regeneration facilitated by PRF. PRF has been used in combination with bone graft materials to reduce recovery time and improve bone regeneration in maxillofacial surgery.12,20,23
There are several animal model studies that have investigated the efficacy of PRP and PRF as treatments for ligament injuries. A systematic review and meta-analysis of animal model studies compared with placebo by Kunze et al 13 concluded that in cases of ligamentous injuries, PRP treatment significantly enhances cell viability, gene expression, ligament repair, and mechanical component improvement compared with the control. Regarding PRF application in animals, studies on ligaments are still limited. An animal study by Wong et al 24 on tendons reported that the implantation of PRF facilitates tissue healing in a partial-tendon defect animal model. However, few studies have directly compared the outcomes of different autologous platelet concentrate models—namely, PRP and PRF—in ligament injury recovery, particularly in the recovery of the MCL. Additionally, studies involving these 2 treatments generally have heterogeneous results and variable preparation procedures for implementation. Therefore, this study aimed to evaluate the effectiveness of PRP and PRF in MCL injury.
Methods
Experimental Materials
This is a randomized, single-blinded trial in an animal model to evaluate the outcomes of PRP and PRF application in MCL injury. The protocol of this study has received ethical approval from Hasanuddin University Research Ethics Committee.
We included male Rattus norvegicus Wistar rats aged 8 to 10 weeks with a mean body weight of 250 g to 300 g and in good health (ie, normal hair, active movement, adequate oral intake). We excluded sick rats, rats with a decrease in body weight of >10% after adaptation in the laboratory, or any death during trials. All Wistar rats were conditioned in a cage with dimensions of 31 × 10 × 40 cm with wire bedding roof and a plastic-based drink container.
PRF Preparation
Wistar rats were anesthetized via inhaled ether. Blood (3 mL) was obtained from the plexus infraorbitalis using a non–ethylenediaminetetraacetic acid (non-EDTA) tube, followed by centrifugation at 2700 rpm for 12 minutes. The centrifuged blood resulted in 3 layers: the first layer consisted of platelet-poor plasma, the second layer consisted of PRF, and the third layer consisted of erythrocyte sedimentation. The PRF layer was extracted using a syringe and laboratory forceps.
PRP Preparation
Blood (3 mL) was obtained from the plexus infraorbitalis of anesthetized Wistar rats using an non-EDTA tube, followed by centrifugation at 3000 rpm for 3 minutes. The centrifuged blood resulted in 3 layers: the first layer consisted of platelet-poor plasma, the second layer consisted of buffy coat, and the third layer consisted of erythrocyte sedimentation. The 2 upmost layers were centrifuged at 3000 rpm for 10 minutes, resulting in a buffy coat. Platelet concentrate was activated using thrombin or chloride calcium 10% with a ratio of 9:1 to trigger platelet activation and fibrin polymerization.
MCL Injury Model
MCL injury in Wistar rats was surgically induced using the femoral approach, which involved making an incision along the medial side of the thigh near the femur to access the MCL. The rats were sedated, and anesthesia was administered using a combination of ketamine and xylazine (0.15-0.2 mL/rat). Cauterization with a horizontal incision was performed to minimize bleeding. The MCL injury was created by making a vertical incision in the middle third of the ligament. This incision was carefully directed longitudinally, parallel to the length of the ligament fibers, and involved a partial-thickness cut, intentionally designed to replicate a moderate injury without completely severing the ligament. After injury, the wound was closed using absorbable sutures, and the rats were monitored for any signs of distress or complications. For postinjury management, the rats were kept in individual cages to limit movement and prevent further injury. Pain management was provided using analgesics, and the animals were allowed to recover with standard care procedures. In the control group, a sham incision was performed. This involved making an identical incision in the skin and soft tissue overlying the MCL without actually injuring the ligament itself, allowing for a direct comparison between the injured and control groups in terms of healing and response to treatment.
Intervention
A total of 30 male Wistar rats were labeled with numbers 1 through 30 and were categorized into 3 groups. Group 1 (n=10) was control without platelet concentration, group 2 (n=7) received PRP administration, and group 3 (n=10) received PRF administration. Postoperatively, each rat was given a long-acting injectable antibiotic suspension (Itramox-150 LA) for 7 days. Analgesia was not given, as it could influence the inflammatory process.
Histopathological Examination and Biomechanical Testing
The rats were observed for 1 month and then euthanized. Wistar rats were anesthetized using ether before euthanasia. Euthanasia was done using cardiac embolization with a 3-mL syringe. After death was confirmed, surgery/necropsy was performed for further biomechanical testing and histopathological examination.
Initially, the MCL, along with femoral and tibial bone blocks, was surgically extracted from the Wistar rats to ensure that the ligament was removed with its attachment points intact (Figure 1). This approach allowed for accurate biomechanical testing. The extracted femoral and tibial bone tissues were then subjected to biomechanical testing using an industrial electronic tensile tester. This device is designed to measure the tensile strength and tensile length of materials, providing precise and reliable data on the mechanical properties of the ligament-bone complex. The measurements were recorded and analyzed using TRAPEZIUM LITE X software (Shimadzu Corp), which offered detailed documentation and data analysis capabilities.

Femoral and tibial tissue extracted from Wistar rat from each group (from left to right: control (K7), PRF, and PRP). PRF, platelet-rich fibrin; PRP, platelet-rich plasma. The experiment used a total of 54 legs, 27 legs for tensile test and 27 for histopathological test. 3 rats did not survive until the end of the experiment.
After testing, the femoral and tibial bone tissue samples were preserved in 10% formalin. The preservation process involved a dehydration sequence using progressively higher concentrations of alcohol (70%, 80%, 90%, and 95%) for 24 hours, followed by treatment with 100% alcohol 3 times for 1 hour each. Clearing was performed using xylol with an initial room temperature for 15 minutes, followed by incubation at 56°C for 60 minutes. The samples were then embedded in paraffin and sectioned using a microtome for histopathological examination, which was conducted using hematoxylin-eosin staining to assess tissue morphology and any pathological changes.
Statistical Analysis
The results of this study are presented in both the text and the tables. Categorical data (ie, macroscopic and microscopic data) were analyzed using nonparametric Kolmogorov-Smirnov test. For continuous data (ie, microscopic and biomechanical tensile data), 1-way analysis of variance was used if the data had a normal distribution, and Kruskal-Wallis test as used if the data had a skewed distribution. Spearman rho analysis was performed to find a correlation between macroscopic data, microscopic data, and biomechanical tensile data with a 95% CI. If the P value was <.05, it was considered statistically significant.
Results
Histopathological Examination
A total of 30 Wistar rat subjects were included in the trial, with 1 rat from the PRP group excluded during the trial caused by sudden death during postoperative care. The histopathological examination between all groups can be seen in Table 1.
Patient Characteristics and Overall Histopathological Examination a

PRF, platelet-rich fibrin; PRP, platelet-rich plasma.
Kolmogorov-Smirnov analysis.
After 1 month of observation, more ligament cell hypertrophy was seen in the PRF group compared with the other 2 groups. However, this difference was not statistically significant (P = .21). The cellular hypertrophy in PRF groups can be seen in Figure 2.

Histopathological examination (40× magnification) in the platelet-rich fibrin sample showed cell hypertrophy (H) and inflammation represented (R) with an increase in basophil matrix (black arrowheads).
The inflammatory process and increase in basophil matrix at 1 month of observation were seen in all groups. However, this difference was not statistically significant (P = .99). The inflammatory process and matrix quality in PRP groups can be seen in Figure 3.

Histopathological examination (40× magnification) in the platelet-rich plasma sample showed inflammatory response represented (R) by increase in basophil matrix (black arrowheads) and increase in vascularization (V) (white arrowhead).
The vascularization process at 1 month of observation across all groups showed a higher amount in the PRF group when compared with the other 2 groups. However, this difference was not statistically significant (P = .35). The vascularization in PRF groups can be seen in Figure 4.

Histopathological examination (40× magnification) in the platelet-rich fibrin sample showed inflammatory response, represented (R) by increase in basophil matrix (black arrowhead), and increase in vascularization (V) (black arrowhead).
The collagen cell orientation that was observed by regularity of the cell at 1 month of observation across all groups showed a higher amount in the PRF group when compared with PRP and controls. While the difference in cellular orientation was statistically significant (P = .049), the P value was quite borderline, so the results should be interpreted with caution. The cellular orientation can be seen in Figure 5.
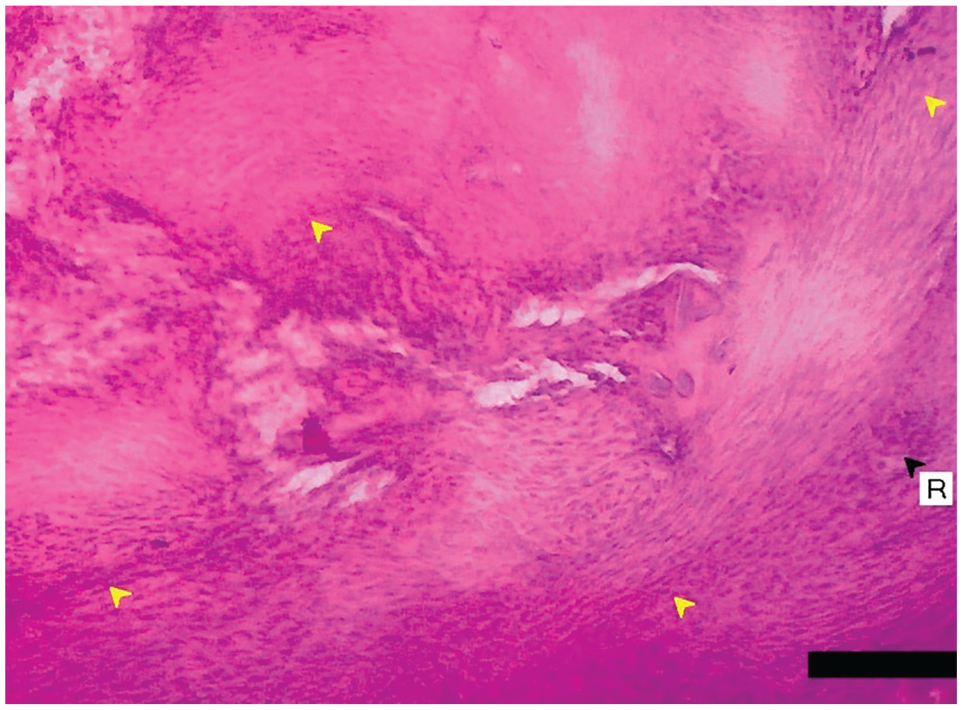
Histopathological examination (40×x magnification) showed regular collagen orientation (yellow arrowheads) and inflammation represented (R) by increase in basophil matrix (black arrowhead).
There was no significant difference in the vacuolization process at 1 month of observation across all groups (P = .14).
Biomechanical Testing
Tensile strength and ligament length were measured after 1 month of observation. The linear comparison between groups can be seen in Figure 6.

Linear comparison of tensile strength between groups. The axis of the chart was measured in Newtons (N) (× 10–4, while the ordinate of the chart was measured in cm. PRF, platelet-rich fibrin; PRP, platelet-rich plasma.
There was a significant difference in tensile strength across all groups (P = .004), with a significantly higher value in the PRF group when compared with the PRP group (mean difference [MD], 2.76; P = .003) as shown in Table 2. No significant difference was seen in ligament length in all groups (MD, 0.57; MD 0.67; P = .14) as shown in Table 3.
Comparison of Tensile Strength Between Control, PRF, and PRP Groups a

PRF, platelet-rich fibrin; PRP, platelet-rich plasma.
PRF vs. PRP: Mean difference 2.76; P = .003.
Comparison of Ligament Length Between Control, PRF, and PRP Groups a

PRF, platelet-rich fibrin; PRP, platelet-rich plasma.
PRF vs. control: Mean difference 0.67.
PRP vs control: Mean difference 0.57.
A regression analysis was conducted to determine the correlation between tensile strength and ligament length. The linear regression showed that there was no significant correlation between tensile strength and ligament length (P = .34). Every change of 0.0001 N in tensile strength was associated with an increase in ligament length of 6.7% (P = .44).
Discussion
Within the past 40 years, the use of platelet derivates in musculoskeletal injury or disorders has been widely used in medicine. The mechanism of action of PRP and PRF can be observed via histopathological examination of sample tissues. Many parameters can be used to measure the regeneration process, including ligamentous cell hypertrophy, increase in basophil matrix, vascularization, and collagen cellular orientation. Previous studies have shown no significant difference in histopathological examination between any groups at the 14- and 28-day follow-up period. A study by Xie et al 26 found qualitatively that the cellularity level did not differ between the PRP group and the control group after 14 days. However, the PRF group showed a more advanced neovascularization process with a decrease in inflammatory cell infiltration without the presence of granuloma when compared with control. In our study, we found that the PRF group had a significantly higher regularity in the cellular orientation of collagen compared with other groups (P = .049) even though the P value was quite borderline, so the results should be interpreted with caution. We also found in the histopathological examination that the PRF samples showed a more organized tissue with abundant fibrocytes and fewer fibroblasts, indicating a partial tissue recovery process. The study by Xie et al found that the difference in cell type, amount of vascular tissue, and collagen fiber structure suggested a more progressive tissue recovery and repair in the PRF group when compared with control and PRP. 26 Moreover, a study conducted by Alviti et al 1 showed an increase in vascularization, which may aid the recovery process after PRF administration in patients with Achilles tendon rupture. A study conducted by Wong et al 24 showed that the histological analysis of samples treated with PRF showed a more regular collagen fiber with minimal formation of cartilage. PRF increased tenocyte viability and phenotypic tenogenic differentiation in vitro.
Other parameters that can be used to evaluate ligament repair include the measurement of tensile strength in the graft. No study has evaluated and compared the effectiveness of PRP and PRF in ligamentous tensile strength using the Newton unit. The tensile test in the MCL ligament in our study showed a significantly higher tensile strength in the PRF group compared with PRP and control (P = .004). PRF may improve regeneration capacity in soft tissue compared with PRP, which may lead to recovery to normal function in a shorter time. Although there was no significant difference in the ligament length across all groups, the ligament length was higher in the PRF group compared with PRP and control. The linear regression showed a significant correlation between tensile strength and ligament length, in which every 0.0001 N change of tensile strength was associated with an increase in ligament length by 6.7%. This was supported by the study conducted by Anitua et al, 2 which showed that the platelet in the fibrin matrix could increase tendon cell proliferation in sheep Achilles tendon, characterized by higher collagen type I and growth factor (ie, vascular endothelial growth factor and human growth factor) synthesis. Another study conducted by Visser et al 21 demonstrated a higher concentration of transforming growth factor-beta 1 (TGF-β1) elution and increased tendon cell proliferation after administration of PRF when compared with whole blood coagulation factors in dog tendons in vitro. Beyzadeoglu et al 3 also reported a higher integration and graft maturation in the reconstruction of a proximal one-third ACL hamstring injury using autologous PRF compared with those without PRF. Regarding the tensile test in the MCL ligament in our study, we found an interesting result that the lowest minimum tensile strength of 2.50 (× 10–4 N) was observed in the PRP group. We believe this does not necessarily imply that PRP weakened the ligament. The observed minimum tensile strength could be due to variability in the biological response among individual rats, differences in the specific injury model, or variations in the application and integration of PRP treatment. Additionally, the minimum tensile strength represents a lower bound of the data and does not account for the overall distribution or mean tensile strength within the PRP group. A more comprehensive assessment of the PRP group’s performance should consider the full range of tensile strength measurements and compare them with the control and PRF groups to determine the overall effectiveness of the treatment.
The advantages of PRF are that it is a completely autologous product, there is minimal blood manipulation without biochemical handling, no bovine thrombin requirements, extended growth factor release compared with other platelet concentrates, high flexibility and elasticity, and low expense. The shortcomings of PRF are that the success of PRF preparation mainly depends on the speed of blood handling, the fact that the membrane should be used immediately because the structural integrity of PRF modulates over time, storage of PRF membrane is not possible due to potential bacterial contamination and dehydration, and the quantity of PRF is low because it is an autologous product. 19
In summary, we found that PRF was superior compared with PRP and control. The superiority of PRF may stem from the fact that (1) preparation of PRF did not involve any additional materials (eg, anticoagulant, thrombin, or calcium chloride), in which tissue repair cascade was maintained by eliminating thrombin-related risk; (2) PRF improves immunoregulation and antimicrobial response, which aids in tissue repair; (3) PRF forms thick fibrin network spontaneously, allowing for a slower degradation process and production of many growth factors in the desired area; and (4) production and standardization of PRF were more accurate and economical as compared with PRP. 16 Our study showed that PRF has a higher repair capacity and recovery rate compared with PRP in a Wistar (Rattus norvegicus) rat MCL injury model.
Limitations
Limitations in this study are the following: (1) This research is still limited to experimental animals, although hopefully, it can be a stepping stone for further research that can be applied to humans. (2) The outcomes assessed in this study are only limited to 1 month; long-term research is needed with a variety of outcomes, not limited to histopathology but also including immunohistochemical features and knee function after therapy. (3) PRP studies in humans have yielded conflicting results for decades, and there is a general agreement that research should specify whether the PRP is leukocyte-rich or -poor and report the platelet count on a CBC. In our study, we did not differentiate between leukocyte-rich and -poor PRP or report platelet counts. This decision was made because the focus of our research was on the comparative effectiveness of PRP and PRF in a specific animal model rather than the detailed characterization of PRP subtypes. Additionally, the variability in clinical outcomes attributed to PRP may be influenced by multiple factors beyond leukocyte content and platelet count. While these factors are important for comprehensive understanding, our study aimed to provide a preliminary evaluation of PRP and PRF efficacy. Future research should address these aspects to enhance the interpretation and application of PRP therapies in clinical settings. Thus, further study is needed to support the use of PRP and PRF in orthopaedic surgery.
Conclusion
This study showed the effectiveness of PRF in improving the recovery process of MCL injury in a Wistar Rattus norvegicus rat model. No significant difference was seen between PRP and control. PRF may aid in cytokine distribution to increase cell viability, proliferation, differentiation, and extracellular matrix synthesis, which leads in the acceleration of growth and repair quality. This further resulted in earlier functional rehabilitation and a faster recovery period.
Footnotes
Acknowledgements
We would like to acknowledge Hasanuddin University, Makassar, for all the support in producing this research.
Final revision submitted November 5, 2024; accepted November 12, 2024.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Hasanuddin University Research Ethics Committee (261/UN4.6.4.5.31/PP36/2024).
