Abstract
Background:
Treatment of Achilles tendon injury varied from surgical to nonsurgical management, such as immobilization, step-by-step physical therapy, or injection with regenerative agents. Regenerative treatment has been studied using polydeoxyribonucleotide (PDRN), atelocollagen, prolotherapy, platelet-rich plasma, and mesenchymal stem cells. PDRN and atelocollagen monotherapy have been known to promote mitigation of fibroblasts, synthesis of collagen fibers, and secretion of growth factors for tissue remodeling.
Purpose:
To compare the therapeutic effects of PDRN, atelocollagen, and PDRN + atelocollagen treatments in rats with a partial Achilles tendon injury.
Study Design:
Controlled laboratory study.
Methods:
A total of 32 Sprague-Dawley rats, aged 8 weeks, were divided into 4 groups—normal saline, PDRN, atelocollagen, and PDRN + atelocollagen. The partial Achilles tendon injury was induced using punch biopsy tools. The treatment was administered 1 day after the injury. The biomechanical evaluation using load to failure and energy absorbed, histology related to inflammation and collagen fiber arrangement, and immunohistochemistry evaluation with collagen-I, transforming growth factor beta1, vascular endothelial growth factor (VEGF), and fibroblast growth factor were measured at 1 week and 4 weeks.
Results:
The tensile strength test conducted at week 4 after injury measured a significantly higher energy absorbed in the PDRN + atelocollagen group than that in the control group. The hematoxylin and eosin (H&E) stain showed that neutrophil invasion was significantly higher in the control group compared with the other groups. Collagen fiber arrangement relatively increased in the atelocollagen and PDRN + atelocollagen groups. After 4 weeks, H&E in all groups showed decreased neutrophil invasion and lower inflammation index. Immunohistochemistry exhibited increased levels of collagen I in the PDRN group compared with the control group and an increased level of VEGF in the PDRN + atelocollagen group compared with the other groups at week 1. After 4 weeks, a high expression of collagen I, transforming growth factor beta1, and fibroblast growth factor was observed in the PDRN and PDRN + atelocollagen groups; the expression of VEGF showed an upward trend in the atelocollagen group compared with the PDRN + atelocollagen group.
Conclusion:
The combined use of PDRN and atelocollagen resulted in increased energy absorbed by the repaired tendons and increased expression of healing growth factors in a partial tendon injury rat model. These benefits appear to be greater than the use of either agent alone. The combined use of PDRN and atelocollagen should be investigated in human patients with tendon injuries.
Clinical Relevance:
PDRN and atelocollagen are commonly used in patients with tendon injuries; however, the comparison of therapeutic effects or synergistic effects has not yet been studied.
The estimated incidence of Achilles tendon injuries ranges from 10 to 37 per 100,000 population, accounting for 20% of all tendon injuries.10,11 Achilles tendon injuries include Achilles tendinosis, Achilles tendon tendonitis, and Achilles tendon rupture, which can be partial or full thickness. The treatment for Achilles tendon injuries, especially Achilles tendon rupture, can be nonsurgical or surgical. The surgical procedures can be divided into open, mini-open, and percutaneous repair. 27 Nonsurgical treatments vary from immobilization to step-by-step physical therapy. Several tissue regeneration strategies—such as prolotherapy, polydeoxyribonucleotide (PDRN), platelet-rich plasma, mesenchymal stem cells, atelocollagen, and growth factors—are being studied. However, a comparison of the tissue regeneration or synergistic effects of combination atelocollagen with PDRN, platelet-rich plasma, mesenchymal stem cells, and growth factors has not been reported widely.17,32
PDRN is a deoxyribonucleotide polymer with chain lengths ranging between 50 and 2000 base pairs and nucleosides derived from salmon sperm.1,31 The therapeutic effects of PDRN in musculoskeletal disorders have been presented in previous in vivo and clinical studies. Kang et al 14 reported that an intraperitoneal PDRN injection and surgical repair in rats with a complete Achilles tendon rupture improved their tensile strength and collagen type I 2 and 4 weeks after treatment; the presence of other growth factors related to tendon repairs—such as transforming growth factor beta1 (TGF-β1), vascular endothelial growth factor (VEGF), and fibroblast growth factor (FGF)—also improved after 4 weeks.
Collagen plays various roles in force transmission, energy dissipation, and the prevention of mechanical failure in connective tissue. 19 A collagen molecule consists of an amino acid sequence (called a telopeptide) at both the N- and C-terminals, which confers most of the collagen’s antigenicity. Atelocollagen obtained using protease or pepsin treatment is low in immunogenicity because it is free from telopeptides.17,32 Suh et al 32 showed that rabbit models with a full-thickness tear and surgical repair treated with an implanted atelocollagen patch showed significant improvements in their tensile strength, fiber structure, and histological examination arrangements at 12 weeks after treatment. In other animal studies, rabbit models with a full-thickness patellar tendon injury or articular cartilage injury showed regenerative effects after an atelocollagen intra-tendon or intra-articular injection, respectively.22,23 In a clinical study, Kim et al 17 and Chae et al 3 indicated that atelocollagen injections in patients with a rotator cuff tear, with or without arthroscopic repair, decreased the patients’ subjective symptom scores from 2 weeks to 2 months after treatment. Jo et al 16 presented a case report showing the healing of a partial tear of a supraspinatus tendon after atelocollagen injection, which was confirmed using magnetic resonance imaging.17,24,29 However, different doses of atelocollagen did not show significant differences in patients with rotator cuff injury. 30 Furthermore, treatment with atelocollagen combined with hyaluronic acid in patients after an arthroscopic rotator cuff repair due to a full-thickness rotator cuff tear has been studied to evaluate any synergistic regeneration effects. 28 The patients in the hyaluronic acid combined with atelocollagen group required a significantly reduced number of steroid injections and experienced a decreased incidence of retearing, as confirmed through the magnetic resonance images, compared with the hyaluronic acid monotherapy and no injection groups.
In previous studies, a combination of PDRN and other therapeutic options—such as umbilical cord-mesenchymal stem cells, microcurrent, or extracorporeal shock wave therapy—has been introduced in animal studies with partial- or full-thickness tendon tears to overcome the limitations of umbilical cord-mesenchymal stem cells or to evaluate any synergistic regeneration effects that will co-promote VEGF and use a different mechanism.3,13,16,33 In clinical trials involving patients with partial- or full-thickness tendon tears, PDRN combined with prolotherapy and strength exercises of the corresponding muscles showed an improvement in the pain and tendon thickness during the ultrasound; this strategy also decreased the post-prolotherapy flare-up pain.12,18 Several factors to consider when using PDRN in combination with other therapies are that it has a short half-life (3 hours) and is easily degraded by unspecific plasma deoxyribonucleic acid nucleases or by cell membrane nucleases. 8 Therefore, repetitive treatment with PDRN is required to reach the desired tissue regeneration outcomes, which takes at least 3 to 4 weeks to reach the final phase. Atelocollagen, known for its high viscosity, may aid in prolonging the effectiveness of PDRN and reduce the necessity for repetitive injections. Moreover, the regenerative effect of atelocollagen is also reportedly related to various growth factors, which might contribute to the synergistic regenerative effects with PDRN.
According to previous studies, a comparison of the tendon regenerative effects of different biological agents, some of which are even commercialized agents, is lacking. In addition, alternative or combinational treatments with these different biological agents require further research—including their synergistic or side effects. Our study aimed to compare the regenerative effects of PDRN, atelocollagen, and a combination of PDRN and atelocollagen using biomechanical, histological, and immunohistochemical tests.
Methods
Animal Model
Approvals for all the study protocols were obtained from the Animal Review Committee (IACUC) of Jeonbuk National University (JBNU-IACUC-2021-34), and all the experiments were conducted at the Animal Research Laboratory of Biomedical Research Institute of Jeonbuk National University Hospital. The experimental animals were 8-week-old male Sprague-Dawley rats (Orient Bio) weighing 250 to 300 g. The rats were anesthetized, using an intraperitoneal injection, with a mixture of ketamine and xylazine (8:1) (ketamine, 100 mg/kg; xylazine, 10 mg/kg). After anesthetization, isoflurane (Ifran Solution; Hana Pharm) was delivered to rats with 100% oxygen using a vaporizer to maintain anesthetization. When the rats were fully anesthetized, a 1-cm incision was made on the area of the hind limb to expose the Achilles tendon. A full-thickness, partial transection injury involving >50% of the tendon was created in the midsection of the Achilles tendon on both hind legs, 1 cm above its attachment to the calcaneus, using a 1.5-mm punch biopsy tool. Subsequently, the incised skin was sutured. The rats were able to move freely in the proper enrichment environment after the injury.
Experimental Procedure
The 32 rats used in the modeling were examined 24 hours after inducing the Achilles tendon injury to verify that they were in good condition and there were no complications. These rats were randomly allocated to 4 treatment groups, with 8 rats in each group). Group 1: normal saline (0.2 mL normal saline); group 2: PDRN (0.2 mL/0.375 mg PDRN; HIDR prefilled; BMI Korea); group 3: atelocollagen (0.2 mL/6 mg atelocollagen; Regenseal; Sewon Cellontech Co); and group 4: PDRN + atelocollagen (0.1 mL/0.187 mg PDRN and 0.1 mL/3 mg atelocollagen) (Figure 1). Concerning previous studies, the concentration and dose of the injected drug were selected. 14 Also, the maximum volume that could be administered to the lesion was taken into consideration.
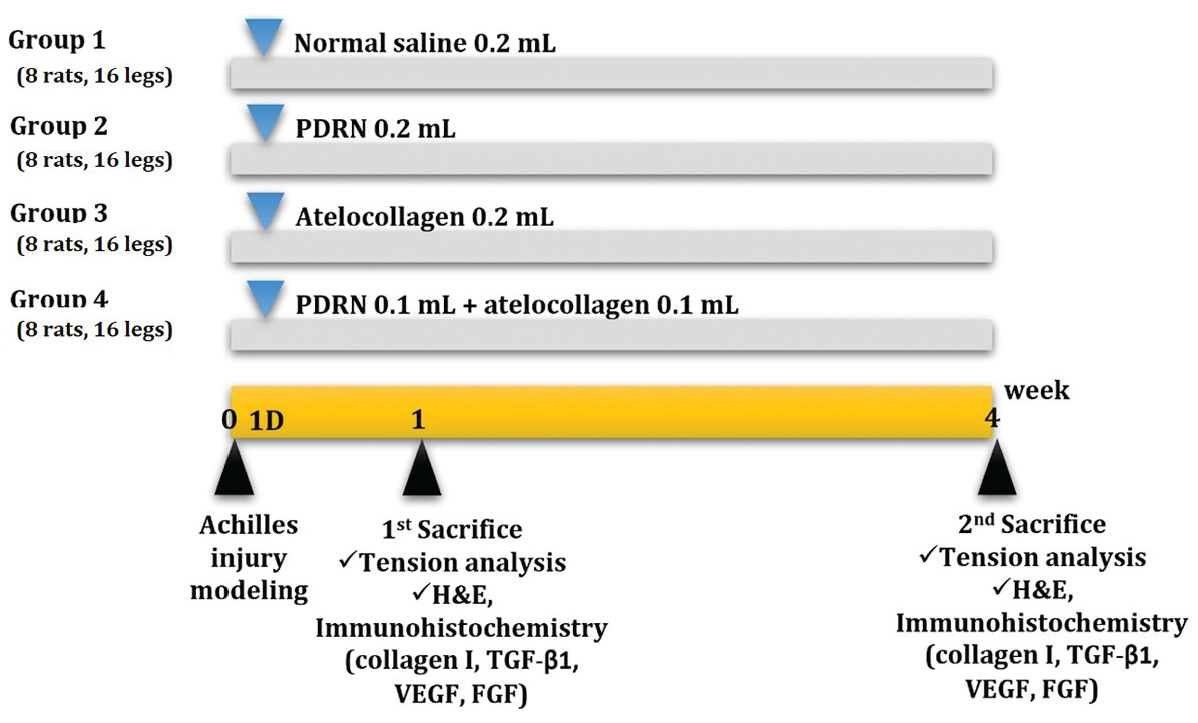
Timeline of the normal saline, PDRN, atelocollagen, and PDRN and atelocollagen combination groups. Normal saline (group 1: 0.2 mL normal saline), PDRN (group 2: 0.2 mL/0.375 mg PDRN), atelocollagen (group 3: 0.2 mL/6 mg atelocollagen), and a mixture of PDRN and atelocollagen (group 4: 0.2 mL/0.187 mg PDRN and 3 mg atelocollagen) were injected into each Achilles tendon injury a day after the injuries were created. Tension analysis, histological examinations, and immunohistochemical staining were performed at weeks 1 and 4 after the Achilles tendon injuries were made. FGF, fibroblast growth factor, H&E, hematoxylin and eosin; PDRN, polydeoxyribonucleotide; TGF-β1, transforming growth factor-beta1; VEGF, vascular endothelial growth factor.
Normal saline and the aforementioned agents were injected into the injury site of the Achilles tendon. The form of atelocollagen utilized in our study was gel-type atelocollagen. Four rats from each group were euthanized at week 1 after the modeling, and the other 4 rats in each group at week 4. The Achilles tendons were extracted from the musculotendinous junction and the attachment site of the calcaneus. The 8 tissue samples from the 4 rats’ legs per group were used at each timeline; 4 samples from 4 different rats per group were evaluated using a biomechanical test, and the other 4 samples per group were fixed in formalin for the histological and immunohistochemical examinations.
Biomechanical Evaluation
The tissue samples of the Achilles tendons from each intervention group were installed in the Instron-5569 universal testing machine (Instron). The tension test was performed at the speed of 10 mm/min, starting at 0 N, and the start length of the Achilles tendon between the clamping jaws was recorded. The computer connected to the machine produced the load-extension curve. The maximum tensile load and the energy absorbed (area under the curve) (N⋅m) were evaluated.
Histological and Immunohistochemical Examination
The fixed tendon tissues, at weeks 1 and 4 after injection, were embedded in paraffin and sliced longitudinally into sections with a thickness of 4 µm. The sections were mounted onto glass slides and stained with hematoxylin and eosin (H&E). After deparaffinization and washing 4 times with phosphate buffer, antigen retrieval for the tissue sections was performed in a Dako Target Retrieval Solution (pH 6.0; Dako) using a microwave oven. Thereafter, the sections were incubated with primary antibodies overnight (collagen type I: PA1-26204, monoclonal; TGF-β1: PA5-116055, polyclonal; VEGF: MA5-13182, monoclonal, all Invitrogen, ThermoFisher; FGF: P09038, monoclonal, Sigma-Aldrich, Merck). These sections were counterstained with hematoxylin and then mounted.
The slides were examined under an Axiophot Photomicroscope (Carl Zeiss) equipped with an AxioCam MRc5 camera (Carl Zeiss). The scoring of the immunohistochemically stained slides was performed by a professional pathologist. From the H&E stain, the neutrophil count per high-powered field, the inflammatory index (0, <10%; 1, 10%-20%; 2, 20%-30%; 3, >30%), and the grading of collagen and fibroblast contents (0, 0%; 1+, 1%-25%; 2+, 26%-50%; 3+, >50%) were evaluated. 9 Immunohistochemistry (IHC) of collagen I, VEGF, basic FGF, and TNF-ß1 was evaluated by adding up the staining intensity scores of each tissue-microarray core. The staining intensity was scored as 0 (no staining), 1 (weak staining), 2 (intermediate staining), and 3 (strong staining). 21
Statistical Analyses
All statistical analyses were performed using SPSS for Windows Version 23.0 (SPSS Inc). During the biomechanical testing, we analyzed the group effect on both the load to failure and energy absorbed among the 4 groups using the Kruskal-Wallis test. For comparisons between the 2 intervention groups, we employed the Mann-Whitney U test. In addition, to assess the time-point effect on the load to failure and energy absorbed at weeks 1 and 4 after modeling, we utilized the Wilcoxon signed-rank test. For IHC, the chi-square test was used to compare the staining scores of the 4 intervention groups. Statistically, a significant level was determined as P < .05.
Results
Biomechanical Evaluation
The mean average and maximum load (N⋅m) of the Achilles tendon at rupture were evaluated at weeks 1 and 4. At week 1 after the injury, although the mean average and maximum load had an increasing tendency in the PDRN, atelocollagen, and PDRN + atelocollagen groups, in that order, the group effect analysis of the energy absorbed and load to failure did not show any significant differences (Figure 2).

Energy absorbed and load to failure (N⋅m) of the Achilles tendon using Instron. PDRN, polydeoxyribonucleotide. *Group effect analysis between the 4 groups, P = .031. #Group effect analysis between the normal saline and PDRN + atelocollagen groups, P = .029.
At week 4 after the injury, the energy absorbed among the 4 groups showed significant differences (P = .031), and in the group effect analysis between the 2 groups, the energy absorbed was significantly higher in the PDRN + atelocollagen group compared with the normal saline group (normal saline group 19.16 N vs PDRN + atelocollagen 32.70 N; P = .029). Although the load to failure at week 4 increased in the PDRN, atelocollagen, and PDRN + atelocollagen groups, in that order, compared with the normal saline group, the group effect analysis of the load to failure at week 4 did not show any statistical significance. The energy absorbed and load to failure after 4 weeks of injury increased compared with after 1 week, but the time point effect analysis did not show any statistical significance.
Histological and Immunohistochemical Evaluations
The H&E test showed that the inflammatory index was increased in all the groups at week 1, with neutrophil infiltration being particularly severe in the normal saline group (Table 1 and Appendix Figure A1).
Histologic Analysis of Each Group a
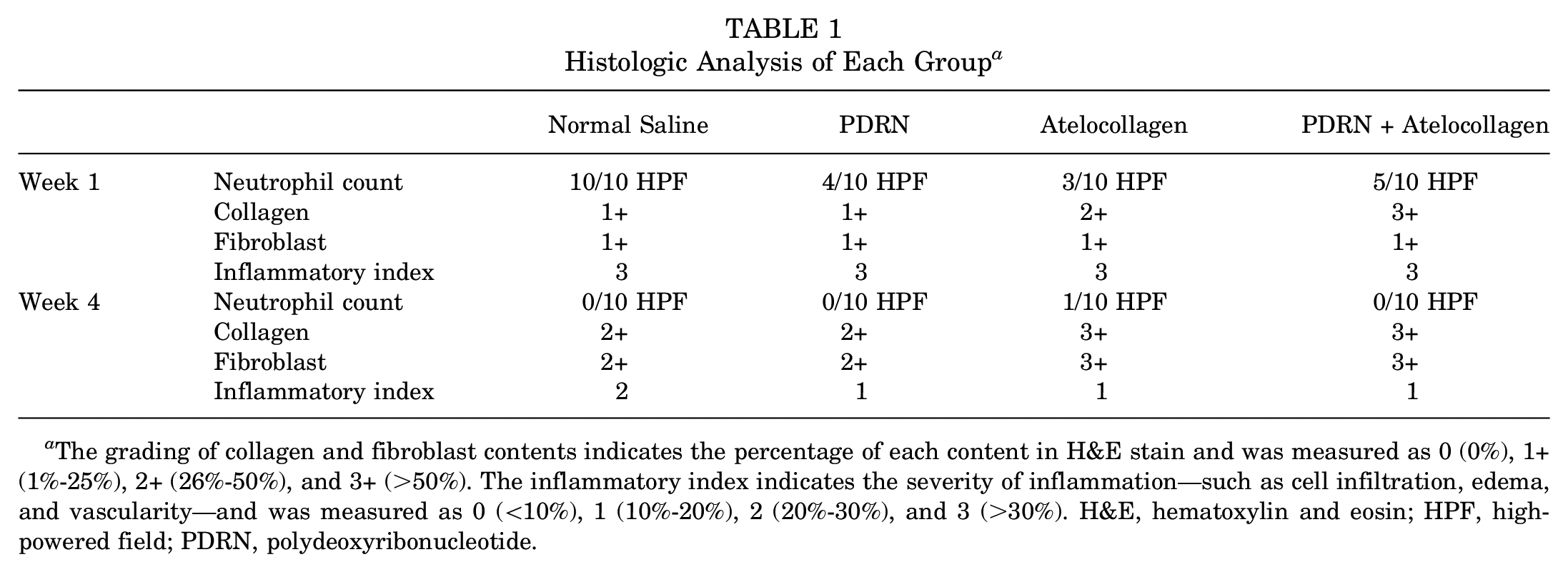
The grading of collagen and fibroblast contents indicates the percentage of each content in H&E stain and was measured as 0 (0%), 1+ (1%-25%), 2+ (26%-50%), and 3+ (>50%). The inflammatory index indicates the severity of inflammation—such as cell infiltration, edema, and vascularity—and was measured as 0 (<10%), 1 (10%-20%), 2 (20%-30%), and 3 (>30%). H&E, hematoxylin and eosin; HPF, high-powered field; PDRN, polydeoxyribonucleotide.
In addition, a denser collagen fiber arrangement was observed in the atelocollagen and PDRN + atelocollagen groups. After 4 weeks of the injury, although the neutrophil counts and inflammatory index decreased in all the groups compared with week 1 of the injury, the inflammation index in the normal saline group was slightly higher than that in the other groups. The collagen fiber and fibroblast arrangement were increased, especially in the atelocollagen and PDRN + atelocollagen groups.
During IHC, the staining intensity of collagen I increased in the PDRN groups compared with the normal saline group at week 1 of the injury. The group effect analysis between the normal saline and PDRN groups showed statistical significance (P = .028) (Figure 3 and Appendix Figures A2-A5).
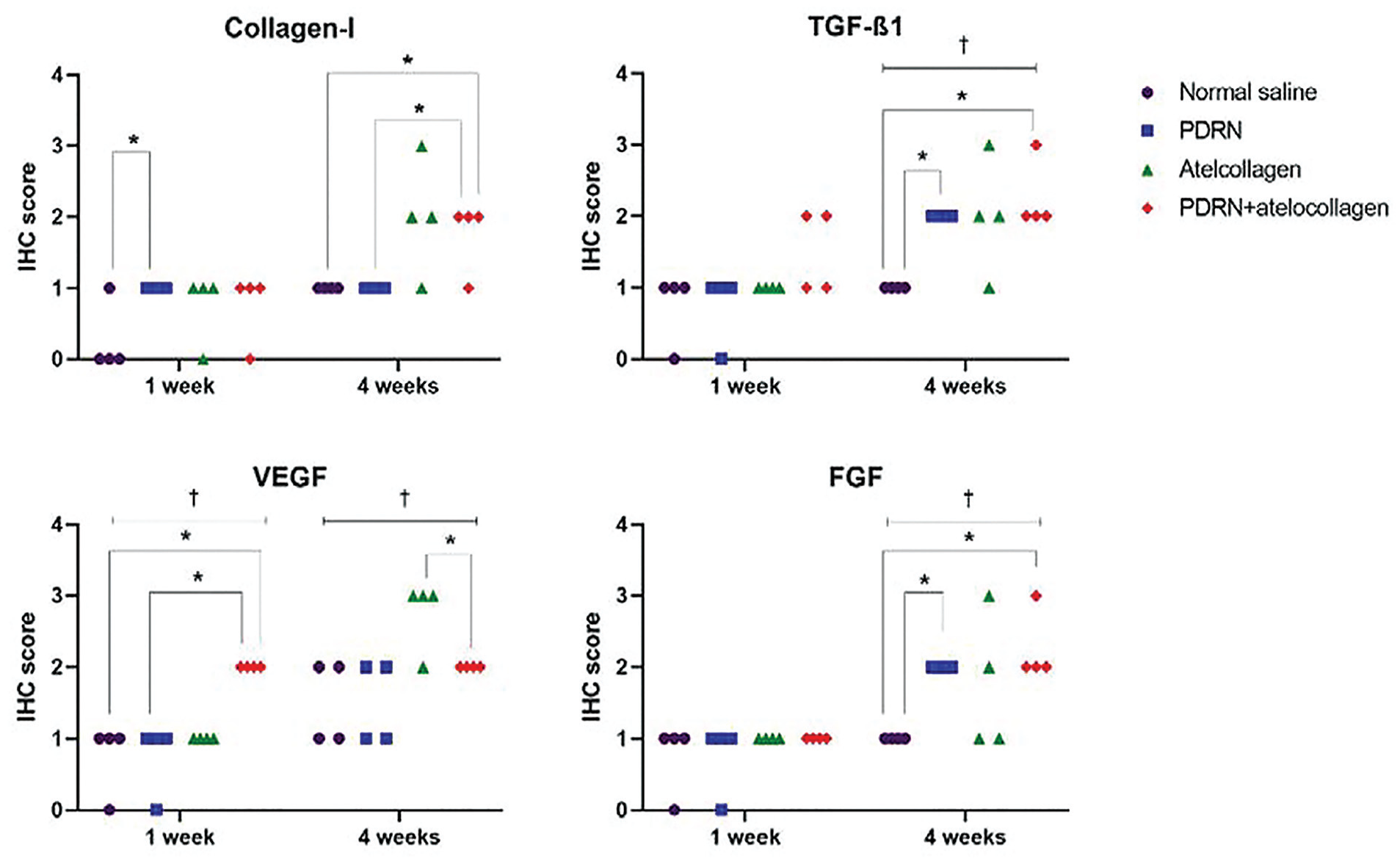
IHC analysis of each group. IHC, immunohistochemistry. The IHC score is the sum of the staining intensities and was measured as 0 (no staining), 1 (weak staining), 2 (intermediate staining), and 3 (strong staining) by a professional pathologist. FGF, fibroblast growth factor; NS, normal saline; PDRN, polydeoxyribonucleotide; TGF-β1, transforming growth factor beta1; VEGF, vascular endothelial growth factor. * and † indicate P < .05.
With regard to the TGF-β1, VEGF, and FGF levels at week 1 after the injury, the IHC score showed an inclining trend in the atelocollagen and PDRN + atelocollagen groups, in that order, compared with normal saline and PDRN groups. However, only the group effect analysis of VEGF among the 4 groups and between PDRN + atelocollagen and the other groups showed significant differences (among the 4 groups, P = .007; PDRN + atelocollagen vs normal saline, P = .018; PDRN + atelocollagen vs PDRN, P = .018; PDRN + atelocollagen vs atelocollagen, P = .005).
After 4 weeks of the injury, the intensity of collagen I increased in the atelocollagen and PDRN + atelocollagen groups, compared with the normal saline and PDRN groups, and the group effect analysis between the PDRN + atelocollagen group and the normal saline and the PDRN groups showed significant differences (PDRN + atelocollagen vs normal saline, P = .028; PDRN + atelocollagen vs PDRN, P = .028). For TGF-β1 and FGF, the staining intensity increased in the normal saline, PDRN, atelocollagen, and PDRN + atelocollagen groups; consequently, the group effect analysis among the 4 groups and between the normal saline and the PDRN and the PDRN + atelocollagen groups showed significant differences (TGF-β1 among the 4 groups, P = .025; normal saline vs PDRN, P = .005; normal saline vs PDRN + atelocollagen, P = .018; FGF among the 4 groups, P = .026; normal saline vs PDRN, P = .005; normal saline vs PDRN + atelocollagen, P = .018). For VEGF, the intensity increased in the PDRN + atelocollagen and atelocollagen groups, in that order, compared with the normal saline and PDRN groups. The group effect analysis among the 4 groups and between the PDRN + atelocollagen and atelocollagen groups showed significant differences (VEGF among the 4 groups, P = .019; PDRN + atelocollagen vs atelocollagen, P = .028).
Discussion
In this study, we compared the regenerative effects of PDRN, atelocollagen, and PDRN + atelocollagen in a rat Achilles tendon injury model using biomechanical, histological, and immunohistochemical tests at weeks 1 and 4 after the injury. Only the PDRN + atelocollagen group showed a statistically significant increase in energy absorbed by the repaired tendon. There was no significant difference in load to failure between treatment groups at either time point. During the histology test, treatment with atelocollagen decreased neutrophil infiltration 1 week after the injury and increased the collagen fiber and fibroblast levels at weeks 1 and 4. Moreover, the secretion of VEGF and collagen I at week 4 was promoted after atelocollagen management. In addition to atelocollagen, management with PDRN enhanced the collagen I levels at week 1 and TGF-β1 and FGF levels at week 4. Therefore, treatment with PDRN, atelocollagen, and PDRN combined with atelocollagen appears to increase various growth factors that are related to tendon regeneration.
Tendon restoration occurs in 3 stages. First, during the inflammatory phase, erythrocytes, platelets, neutrophils, monocytes, and macrophages migrate to the site of injury and release angiogenic factors. Second, during the proliferative phase, the fibroblasts start to synthesize collagen, proteoglycan, and a randomly arranged extracellular matrix, which is composed largely of collagen type III. Third, during the remodeling and maturation phase, collagen type III fibers are replaced by collagen type I fibers, which establishes the mechanical strength restoration of the tendon. 5 The period for the tendon restoration process varies in the literature. According to a previous study, 5 the inflammatory phase was mostly initiated 48 hours after the injury, and the proliferative phase occurred approximately 1 week after the injury. The final phase might start about 3 to 4 weeks after the proliferative phase. 15 Therefore, we designed 2 time points of outcome measurement at weeks 1 and 4 after the injury to evaluate the interval differences of change between the inflammation and proliferative phase and particularly the remodeling and maturation phase.
According to previous studies,14,25 the tensile strength is closely correlated with the synthesis of collagen type I. Administration of TGF-β1 to the rat Achilles tendon injury model increased procollagen I and III and messenger ribonucleic acid levels at week 1 after the modeling, and the tensile strength was increased at 2 and 4 weeks. 25 Moreover, daily intraperitoneal PDRN injections in the rat Achilles tendon injury models increased the tendon strength at weeks 2 and 4, in that order, compared with the normal saline group. 14 Collagen type I was significantly increased at weeks 2 and 4, and a higher expression of FGF, VEGF, and TGF-β1 was shown at week 4. Thus, tensile strength can indicate an increase in type I collagen, which is one of the most important processes in the tendon regeneration phase. In our study, the energy absorbed by the treated tendon in the PDRN + atelocollagen group at week 4 after the injury showed a significant increase compared with the normal saline group. The histology test showed higher expressions of collagen fiber and fibroblast at both weeks 1 and 4. These results may suggest that treatment with PDRN + atelocollagen can enhance the increase in fibroblast and collagen, which are related to an increase in tensile strength.
Polydeoxyribonucleotide increases VEGF expression by stimulating the adenosine receptor (A2A). The A2A receptor causes G protein activation, and thereafter protein kinase A activation via cyclic adenosine monophosphate, which stimulates VEGF and TGF-β1.2,26,35 The mechanism of the A2A receptor is also associated with collagen synthesis as well as fibroblasts and endothelial cell proliferation, which also contain synergic effects as a result of the interactions among the various growth factors.6,20,36 Moreover, the anti-inflammatory activity of PDRN is related to the A2A receptor, which reduces the histological tissue damage and inflammatory cytokines and blunts apoptotic protein expression. 8 In the present study, although the tensile strength in the PDRN groups did not show a significant increase, histology showed a decrease in neutrophil infiltration at week 1, and the IHC test showed an increase in collagen I at week 1 and TGF-β1 and FGF at week 4 after the injury. Based on previous studies, a single injection of PDRN may improve inflammation and increase growth factors but may not be sufficient to demonstrate its effects on the regeneration of type I collagen or the increase in the ability of the tendon to absorb energy.
Atelocollagen is a type of collagen molecule that has low immunogenicity due to the removal of the telopeptides. Reportedly, it can promote the synthesis of type 1 collagen and enhance tendon regeneration.17,22,23,24,30,32 In the present study, although the biomechanical parameters at week 4 did not significantly increase, the collagen fiber and fibroblast levels at week 4, according to histology, did increase. In addition, the expression of VEGF at week 4 was increased in the atelocollagen group compared with even the PDRN + atelocollagen group. In a previous animal study, which compares the tendon regeneration effect to a control group, a patch-type atelocollagen was used after surgical repair in a rat model with rotator cuff injury. 32 Gel-type atelocollagen may be more susceptible to degradation compared with the patch type, which may reduce the tendon regeneration effect. In our study, we used gel-type atelocollagen, which might have diminished the tendon regeneration and restoration of tensile strength.
Tendon healing is accomplished through both endogenous and exogenous modulation—including various growth factors from the wound itself as well as by exogenous peripheral fibroblasts. 5 Several growth factors are reportedly involved in the tendon repair process—including TGF-β1, VEGF, FGF, platelet-derived growth factor, and insulin-like growth factor 1. TGF-β is a crucial cytokine that acts in both the inflammatory and healing stages of tissue repair. TGF-β1 has a dual mechanism in tendon repair in the intrinsic tenocytes, extrinsic tendon sheath fibroblasts, and inflammatory cells. 34 Exogenous TGF-β1 in a rat Achilles tendon injury improved the tensile strength at weeks 2 and 4 after the injury. 25 Endogenous VEGF is upregulated by hypoxia-inducible factor-1, inflammatory cytokines (interleukin-1β, 6, 8), nerve growth factor, and mechanical overload, which results in angiogenesis and extracellular matrix disruption. 15 The exogenous VEGF group in a rat Achilles injury model had significantly improved tensile strength compared with the normal saline group 1 week after the modeling; this was associated with an increased expression of TGF-β. 4 Last, FGF, which is an inflammatory mediator released by blood-derived cells, plays a crucial role in the initiation of the tendon healing processes, mainly by stimulating the proliferation of the fibroblasts and thereafter promoting the synthesis of extracellular matrix proteins and the formation of granulation tissue. 7 In the present study, IHC testing revealed that the staining intensity of VEGF at week 1 significantly increased in the PDRN + atelocollagen group compared with the other groups. However, the intensity of VEGF at week 4 was higher in the atelocollagen group than in the PDRN + atelocollagen group. In addition, the intensity of TGF-β1 and FGF at week 4 was significantly higher in both the PDRN and PDRN + atelocollagen groups compared with the normal saline group. The intensity of type I collagen at week 4 also significantly increased in the PDRN + atelocollagen group. These results suggest that the increase in TGF-β1, VEGF, and FGF might contribute to an increase in collagen fibers, which are related to tendon restoration and strength. The more prominent effect observed in the PDRN + atelocollagen group may be due to a decrease in the early degradation of PDRN, attributed to the high viscosity of the atelocollagen gel. Furthermore, although the exact mechanisms of both PDRN and atelocollagen in tendon regeneration remain unclear, various growth factors and their interactions may enhance cell proliferation and angiogenesis.
The optimal doses and method of administering PDRN and atelocollagen for Achilles tendon regeneration have not yet been established. In clinical practice, local injections at the wound or injury site are commonly administered to enhance the wound-healing process. In this study, we used an intralesional injection with a PDRN dosage of 1.25 mg/kg and an atelocollagen dosage of 20 mg/kg, maintaining a total volume of 0.2 mL. This volume was chosen because the maximum capacity of a rat’s Achilles tendon is <0.2 mL. Administering a single injection with a small volume of PDRN and atelocollagen presented a challenge in our study. However, direct injection at the lesion site might mitigate the limitation. Also, we propose that the combination of PDRN and atelocollagen could potentially enhance the regeneration effect.
This study has several limitations. First, the small sample size of 8 rats per group may have introduced the risk of a type II error. Second, additional quantitative analyses—such as Enzyme-Linked Immunosorbent Assay, Western blot, and reverse transcription polymerase chain reaction—were not performed, limiting the precision of molecular measurements. To mitigate potential modeling errors, we ensured consistency through repeated practice of the modeling and sampling procedures. However, this study focused solely on unrepaired partial tendon injuries, which restricts the generalizability of the findings. Furthermore, the histological analysis relied on subjective ordinal measurements rather than continuous variables, which may have introduced bias. A control group of uninjured tendons was not included, preventing the comparison of biomechanical strength recovery to a baseline. Moreover, this study did not assess the dose-dependent effects of PDRN and atelocollagen, nor did it explore the potential benefits of patch-type atelocollagen. Future research should investigate the effect of varying dosages and the use of patch-type atelocollagen on the regeneration process to provide a more comprehensive understanding of their therapeutic potential.
Conclusion
The combined application of PDRN and atelocollagen demonstrated enhanced therapeutic efficacy in tendon healing by improving energy absorption, mitigating inflammation, and promoting the expression of collagen I, TGF-β1, VEGF, and FGF. Although PDRN and atelocollagen monotherapies reduced neutrophil infiltration and elevated growth factor levels, the combined treatment produced superior biomechanical and histological outcomes. These results highlight the synergistic potential of PDRN and atelocollagen, suggesting that their combined use may offer greater clinical benefits than either agent alone. Further investigation is warranted to validate these findings in human subjects with tendon injuries.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671251324181 – Supplemental material for Therapeutic Effects of Polydeoxyribonucleotide and Atelocollagen into Achilles Tendon Injury in a Rat Model
Supplemental material, sj-pdf-1-ojs-10.1177_23259671251324181 for Therapeutic Effects of Polydeoxyribonucleotide and Atelocollagen into Achilles Tendon Injury in a Rat Model by Da-Sol Kim, Yu Hui Won, Sung-Hee Park, Jeong-Hwan Seo, Myoung-Hwan Ko and Gi-Wook Kim in The Orthopaedic Journal of Sports Medicine
Footnotes
Final revision submitted October 30, 2024; accepted November 11, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research was supported by the Bio&Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MSIT) (No. RS-2023-00236157). Also, this work was supported by the NRF, a grant funded by the MSIT (No. 2022R1C1C1005770), and grants from the Biomedical Research Institute of Jeonbuk National University Hospital, Jeonju, Republic of Korea. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Animal Review Committee (IACUC) of Jeonbuk National University (JBNU-IACUC-2021-34).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
