Abstract
Background:
Improved patient outcomes and decreased patellar instability have been reported after medial patellofemoral ligament (MPFL) reconstruction for recurrent lateral patellar dislocation; however, there is a lack of comparative evidence on functional outcomes associated with different femoral attachment sites for the MPFL graft.
Purpose:
To identify differences in MPFL reconstruction graft isometry with femoral tunnel malpositioning, specifically evaluating isometric differences as the femoral position is moved anterior, posterior, proximal, and distal relative to the Schöttle point, the femoral radiographic landmark of the MPFL.
Study Design:
Descriptive laboratory study.
Methods:
A biomechanical study evaluating 11 fresh-frozen cadaveric knees was conducted. Nonelastic suture, used as an analog to the MPFL graft, was anchored with the knee at 30° flexion at the Schöttle point and at 5 and 10 mm anterior, posterior, superior, and distal to the Schöttle point. A draw wire displacement sensor was used to evaluate length changes of the MPFL graft analog through 0° to 120° knee flexion. Knee flexion position was continuously measured using a motion tracking system. Pairwise t tests with Bonferroni correction were used to compare isometry between the Schöttle point and the nonanatomic femoral insertion points.
Results:
Grafts placed at the Schöttle point proved mildly anisometric, with tightening in extension and loosening in flexion. Similarly, grafts placed distally and posteriorly also demonstrated tightening in extension and loosening in flexion. Grafts placed anteriorly and proximally demonstrated tightening in flexion. Pairwise comparisons relative to the Schöttle point found that grafts placed proximally or distally demonstrated significant differences in total MPFL excursion magnitude (10 mm proximal: 0.36 [P = .03], 5 mm distal: 0.14 [P = .01], 10 mm distal: 0.22 [P < .001]).
Conclusion:
When deviating from the Schöttle point, posterior and distal femoral tunnel positionings minimized the risk of MPFL graft tightening during knee flexion. Errant anterior and proximal positioning were concerning for MPFL overconstraint, and proximal tunnel placement was most at-risk.
Clinical Relevance:
An understanding of the effects that femoral tunnel malpositioning has on graft isometry is crucial to minimizing instability or overconstraint, which leads to anterior knee pain, increased patellofemoral contact pressures, or graft failure.
The medial patellofemoral ligament (MPFL) originates on the distal femur between the adductor tubercle and medial epicondyle and inserts onto the superomedial patella.10,28,31 The femoral attachment site of the MPFL, approximated by the Schöttle point, is located on lateral radiographs a mean of 1 mm anterior to the extension of the posterior femoral cortex, 2.5 mm distal to the posterior origin of the medial femoral condyle, and proximal to the posterior Blumensaat line. 28 The anatomic MPFL is tightest in knee extension, and previous studies have shown it remains largely isometric throughout early knee flexion angles.12,13,14,26 The MPFL serves to guide the patella into the trochlear groove, functioning as the primary medial constraint in knee flexion angles of 0° to 30°.5,11,21,23 As the knee flexes further and the patella successfully tracks into the trochlear groove, the MPFL becomes a secondary source of medial patellar stabilization with the trochlear groove providing primary stabilization. 1
An MPFL injury during an acute patellar dislocation predisposes patients to developing recurrent lateral patellar dislocations.3,6,9,20,30 Numerous studies have shown improved patient functional outcomes and decreased patellar instability following MPFL reconstruction indicated for treatment of recurrent lateral patellar dislocation.18,19,25,27,32,37 In MPFL reconstruction, the placement of the femoral tunnel is tremendously important, as nonanatomic positioning of the graft on the femoral side may cause over or undertensioning of the graft at various flexion angles. It is important to recognize that the surgical graft should not be placed in a position where the graft becomes too tight as the knee flexes. Graft tightening with knee flexion increases tension on the graft, potentially leading to graft stretching/failure. Inappropriate graft tensioning may also result in increased medial patellofemoral joint contact pressures, knee pain, and reduced knee range of motion, and an inappropriately loose graft may contribute to recurrent patellar instability.2,4,7,8,35
A previous cadaveric study demonstrated MPFL length changes when attachment sites were changed relative to the anatomic location on both the femur and the patella. 36 This confirmed prior knowledge that the anatomic MPFL was tightest in extension, and the length decreased throughout early knee flexion until approximately 30°, at which point it was relatively isometric throughout remaining degrees of flexion. They also found that throughout 0° to 110° knee flexion, the most isometric femoral attachment site was the anatomic femoral MPFL attachment site. Kernkamp et al 14 further studied isometry of the MPFL as attachment sites were altered and found the most isometric femoral attachment site to be posterior and proximal to the anatomic femoral MPFL attachment site. These studies provide important insight as to the biomechanics of the MPFL but are limited in clinical utility, as identifying the precise femoral anatomic attachment site for the MPFL intraoperatively is challenging.
The purpose of the current study was to identify differences in MPFL reconstruction graft isometry with femoral malpositioning, specifically evaluating isometric differences as the femoral position is moved anterior, posterior, proximal, and distal relative to the Schöttle point. The Schöttle point is a useful radiographic marker that is often used intraoperatively to determine the MPFL reconstruction site. We hypothesized that femoral tunnel malpositioning either proximal or anterior to the Schöttle point would result in significant graft tightening with knee flexion. Further, we hypothesized femoral tunnel malpositioning either distal or posterior to the Schöttle point would exhibit similar anisometric patterns to anatomic MPFL placement with tightening in knee extension and loosening in knee flexion.
Methods
Eleven fresh-frozen cadaveric knee specimens were obtained with an age range of 53 to 75 years (6 female, 4 male; age and sex were unavailable for 1 specimen due to lost documentation). The specimens showed no evidence of prior surgical intervention. Specimens were kept at −20°C until thawed just before dissection and testing. All specimens were then carefully dissected to remove skin and subcutaneous tissues, and the MPFL was identified. The insertion of the MPFL was carefully isolated and its position was identified on the patella. 21 A drill hole was placed in the center of the patellar attachment site. A nonelastic Ethibond (Ethicon) suture was placed into the drill hole and secured using a suture anchor. A No. 0 Ethibond Krackow stitch (Ethicon) was placed in the quadriceps tendon with free ends of the suture attached to a 10-lb. weight hung over a pulley to simulate baseline muscle tension. The Ethibond suture previously secured to the patella was then passed between the second and third soft tissue layers of the medial knee replicating the path of the MPFL reconstruction allograft.
The Schöttle point was identified using fluoroscopy (Figure 1) by drawing reference lines according to previously described methodology. 28 The fluoroscopic machine (OEC 9900 Elite Mobile C-Arm; GE Healthcare) was positioned at a standardized distance from the testing apparatus, and the specimen was rotated such that the posterior and distal aspects of the femoral condyles were aligned when imaged from the lateral side. A 1.6 mm–diameter Kirschner wire (K-wire) was placed through the medial femoral cortex at the Schottle point. A 3-dimensional (3D) printed template guide was used to identify points in 4 directions (anterior, posterior, proximal, distal) measuring 5 or 10 mm away from the Schöttle point. 28 This template guide was then placed over the K-wire at the Schöttle point and rotated to position the guide holes in the anterior, posterior, proximal, and distal directions with respect to the femur.
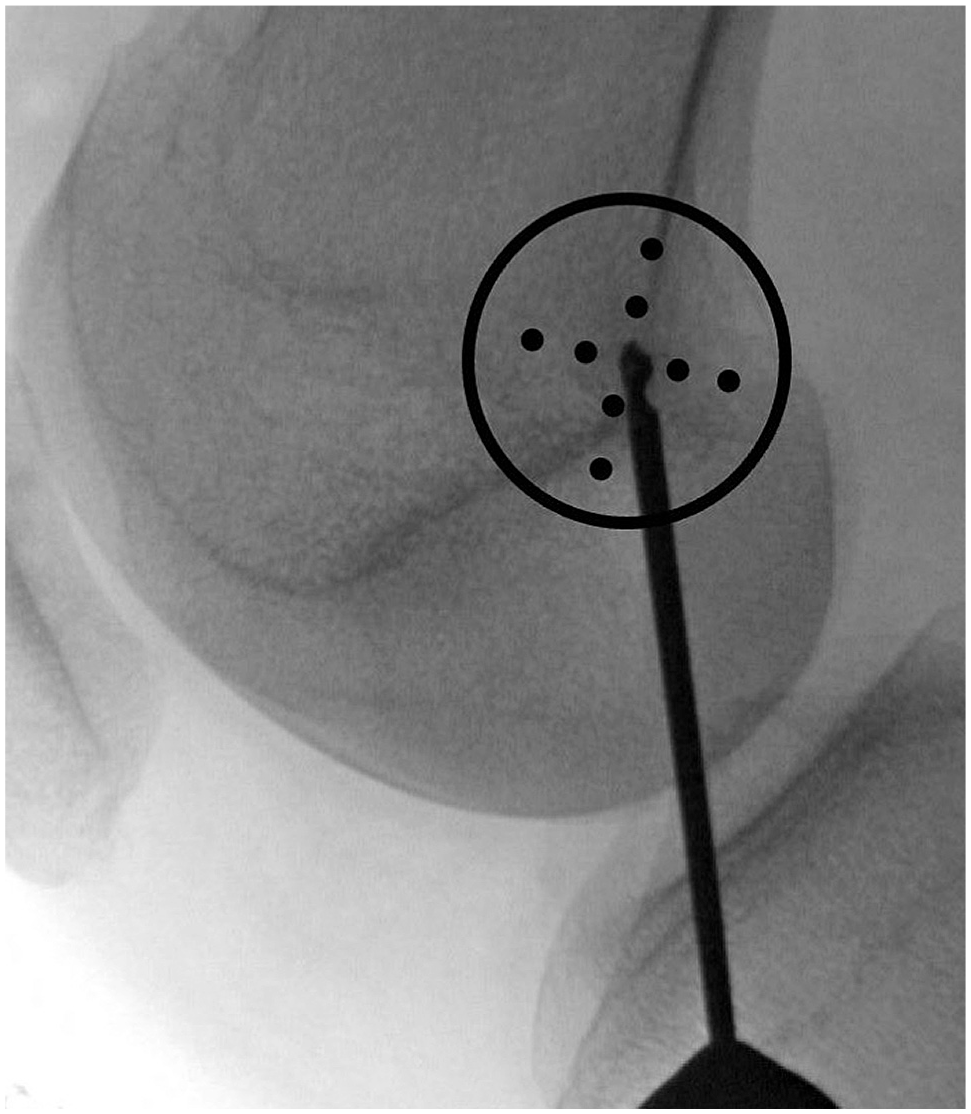
Lateral radiograph of right knee depicting the Schöttle point at the tip of the drill and the experimental points of 5 mm and 10 mm anterior, posterior, proximal, and distal to the Schöttle point.
Optitrak motion capture sensors (NaturalPoint, Inc) were mounted to the femur, patella, and tibia using rigid standoff mounts to track precise bony position data during experimental range of motion. A second 1.6 mm–diameter K-wire was then placed through one of the templated holes on the 3D-printed guide at 5 mm and 10 mm anterior, posterior, proximal or distal to the Schöttle point (Figure 2). The Ethibond suture simulating the MPFL reconstruction graft was then draped over the K-wire and secured to a mounted draw wire sensor (Epsilon 50 mm wire sensor; Micro-Epsilon). The knee was then manually taken through 2 complete flexion and extension cycles from full extension to 120° flexion. Bony position and knee flexion angle data were collected using the Optitrak 3D motion tracking system. Change in anatomic distance between the patellar insertion of the MPFL and the experimental femoral origin position was measured using the draw wire sensor.

Experimental apparatus setup of a right cadaveric knee specimen. The 3-dimensionally printed template is centered at the Schöttle point, and Optitrak Image Capture System sensors are mounted to the femur, patella, and tibia.
Data Collection
For each specimen and each femoral insertion position, the mean draw wire length at each flexion angle was calculated from the mean of 2 full flexion-extension cycles of 0° to 120°. In previous research, 30° knee flexion with the patella centered in the trochlear groove has been commonly used as the surgical position in which the graft is tensioned. 22 Therefore, in our analysis, the draw wire length at 30° knee flexion was used as the baseline length for comparison. The change in draw wire length (ΔL) was calculated as the difference in millimeters between the length of the graft at various flexion angles and the length of the graft at 30° of flexion.
To directly compare the magnitude of draw wire length change among the various femoral insertion positions, a ratio of the draw wire length at each flexion angle compared with the baseline draw wire length at 30° flexion (normalized length ratio [NLR]) was calculated. Mean baseline draw wire length at 30° knee flexion for each femoral insertion point was represented by NLR = 1. Values above NLR = 1 represented a lengthening of the anatomic distance between the patellar origin and experimental femoral insertion point along the anatomic path of the reconstructed MPFL and thus a tensioning of an inelastic, rigidly fixed graft. Conversely, values below NLR = 1 represent a shortening of the anatomic distance between the patellar origin and experimental femoral insertion point and thus a relaxation of an inelastic, rigidly fixed graft. The NLR for each experimental condition is a unitless number that models the tension on the graft while accounting for anatomic variations and baseline graft lengths, and the values given can be compared across specimens and between graft insertion experimental positions.
Finally, the total normalized excursion (NEX) of the draw wire was calculated as the difference between the minimum and maximum NLR for each cycle and was averaged over all flexion-extension cycles for a given specimen and insertion position. This value represents the magnitude of overall length change that a graft undergoes during the entirety of the flexion-extension cycle.
Statistical Analysis
Both NLR and ΔL were reported as the means of all specimens and flexion-extension cycles. Standard deviations of all measurements were propagated through development of the normalized mean. To determine whether or not the NEX of the graft placed at various positions differed from those placed at the center location, a pairwise t test adjusting for multiple comparisons using the Bonferroni method was used. The difference between NEX at various positions and the NEX at the center location was calculated and visually presented in a bar chart with 95% CI error bars calculated.
Results
The mean change in graft length, ΔL, at 0°, 60°, 90°, and 120° at various femoral insertion points is demonstrated in Figure 3. Grafts placed distally and posteriorly demonstrated similar isometry to grafts placed at the Schöttle point, with tightening in extension and loosening in flexion. Grafts placed anteriorly and proximally demonstrated tightening in flexion, with grafts placed proximally further demonstrating loosening during extension.

Graft excursion (change in draw wire length, mm) as measured from baseline wire length at 30° knee flexion. Bars above 0 mm represent graft tightening, whereas bars below 0 mm represent loosening. MPFL, medial patellofemoral ligament.
NLR as a function of flexion angle at each experimental femoral insertion position is demonstrated by the plots in Figure 4. Figure 4A shows the length response curve of a graft fixed at the Schöttle point, demonstrating mild graft tightening in extension and mild graft relaxation in flexion past 30°. Femoral insertion points anterior and proximal to the Schöttle point demonstrated graft tightening in flexion (Figure 4, B-E). The magnitude of tightening in flexion with anterior or proximal placement of the femoral insertion point was directly related to the magnitude of displacement from the Schöttle point. In contrast, femoral insertion points distal and posterior (both 5 mm and 10 mm) to the Schöttle point showed graft tightening in extension and loosening in flexion, similar to the length change patterns observed with grafts fixed at the Schöttle point (Figure 4, F-I).
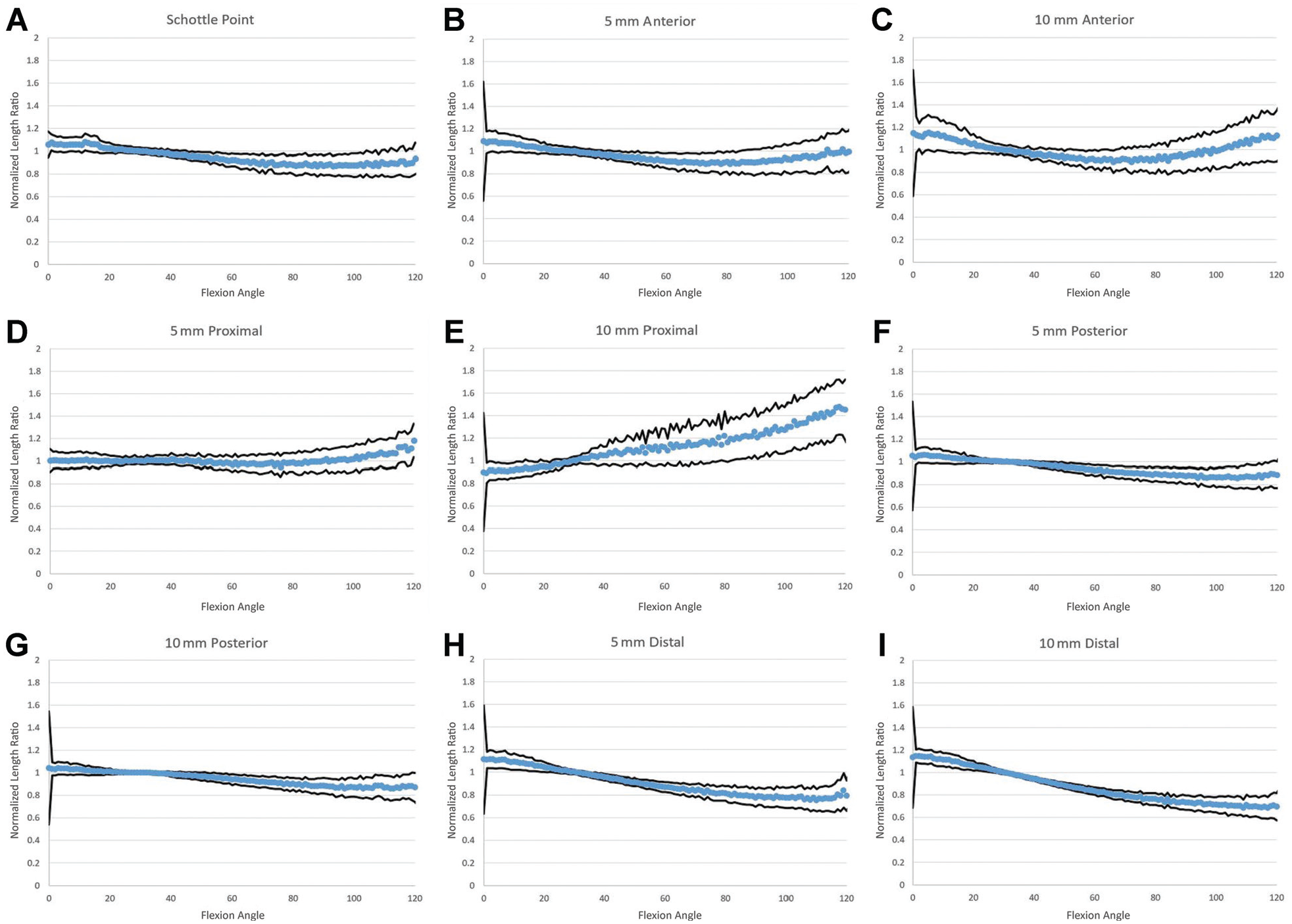
Normalized length ratio vs flexion angle at (A) the Schöttle point and all experimental points: (B) 5 mm anterior, (C) 10 mm anterior, (D) 5 mm proximal, (E) 10 mm proximal, (F) 5 mm posterior, (G) 10 mm posterior, (H) 5 mm distal, and (I) 10 mm distal. Shown are the mean values of all specimens, with black lines indicating ±1 SD.
Statistical analysis of the magnitude of NEX showed that grafts placed 5 mm proximal, 5 mm and 10 mm anterior, and 5 mm and 10 mm posterior to the Schöttle point demonstrated similar magnitudes of excursion compared with grafts placed at the Schöttle point (P > .05) (Table 1 and Figure 5). The mean NEX of grafts placed 5 mm distal, 10 mm distal, and 10 mm proximal were significantly higher than that of the Schöttle point (P = .01, P < .001, and P = .03 respectively).
Pairwise Comparisons of MPFL Excursion (NEX) for the 8 Off-Center Locations Relative to the Schöttle Point a

Boldface P values indicate statistical significance (P < .05). NEX, normalized excursion.
Difference = NEXlocation− NEXSchöttle Point.

Pairwise comparison of total normalized excursion (NEX) at each experimental point subtracted by NEX at the Schöttle point, with error bars representing 95% CIs. Values near zero represent similar magnitudes of graft excursion compared with grafts placed at the Schöttle point. *Statistically significant (P < .05).
Discussion
In this study, we aimed to identify the consequences of malpositioning of the femoral insertion point intraoperatively by experimentally moving this point anterior, posterior, proximal, or distal to the Schöttle point. Grafts placed at the Schöttle point demonstrated mild anisometry with lengthening in knee extension and loosening in deep knee flexion. Alteration of the femoral attachment site posterior or distal to the Schöttle point resulted in a similar anisometric length change pattern of lengthening in knee extension and loosening in deep knee flexion. These tension-flexion relationships are considered desirable, as the MPFL is the primary constraint of the patella in extension and therefore tightening of the graft during this portion of movement ensures proper constraint of the patella. We found that proximal positioning of the femoral insertion point causes loosening in extension and tightening in flexion, and anterior positioning of the femoral insertion point causes tightening in both knee extension and deep flexion, both commensurate with the magnitude of displacement from the Schöttle point. These tension-flexion relationships are less desirable, as they may lead to overtightening in flexion when the patella is primarily constrained by the trochlear groove rather than the MPFL. The present study further showed that distal and proximal attachment sites demonstrated significantly greater total magnitude of excursion compared with a graft placed at the Schöttle point.
Multiple prior studies7,10,13,14,26,31,36 have been performed to identify the appropriate femoral location of a single-bundle MPFL graft. The most commonly cited insertion point, described by Schöttle in 2007, 28 or the modification described by Redfern in 2010, 24 is based on cadaveric dissections and identification of the anatomic insertion point of the native MPFL and gives a radiographic technique for identifying the graft insertion point that most closely represents native anatomy and reportedly remains isometric through knee range of motion. Similar to several other studies,14,36 our data show that the native MPFL, as modeled by graft attachment at the Schöttle point, is an anisometric structure with relative lengthening in extension and relaxation in flexion. This finding suggests that achieving perfect isometry of the MPFL reconstruction graft may not be anatomically consistent or even desirable.
Because of anatomic differences between individuals, and a level of imprecision inherent in the radiographic localization technique, it is expected that small variations in femoral graft placement location will occur among patients, even with careful surgical attention. Several cadaveric and in vivo studies have been performed to evaluate the effect that variations in placement have on the isometry of the MPFL.12,26,33,34,36,38,39 One such cadaveric study by Stephen et al 35 in 2014 showed that proximal or distal femoral tunnel placement, or overtensioning of the graft can increase patellofemoral contact pressures and patellar tilting. However, Stephen et al 35 did not address anterior or posterior displacement of the femoral tunnel; furthermore, they modulated graft tension as an independent variable and did not evaluate variation in graft tension using a fixed femoral insertion point over a flexion-extension cycle. A computational modeling study by Kernkamp et al 14 used knee computed tomographic data and biplanar fluoroscopy to simulate knee range of motion and evaluate the length between patellar and femoral insertion points of an MPFL reconstruction graft using a 3D wrapping algorithm. Their results showed that the most isometric femoral MPFL attachment point was located posterior and proximal to the anatomic MPFL insertion point and that length changes were more responsive to changes in the proximal-distal directions compared with the anterior to posterior direction. Our findings build on Kernkamp et al and a litany of existing literature by providing cadaveric biomechanical data to support that proximal/distal changes in the femoral insertion point of the MPFL have a significant impact on the total magnitude of graft excursion.36,39 The present study reveals the overall tension-flexion profile of various MPFL reconstruction placements, rather than simply the magnitudes of excursion reported in previous studies. Further, it is differentiated from previous literature because it provides baseline length data from the more clinically relevant radiographic MPFL attachment site.
This study has particular importance in the pediatric patient population. When performing an MPFL reconstruction in an immature patient, avoiding the physis is prioritized, as a significant amount of literature exists on the consequences of surgical violation of the physis.15,16 Additionally, studies have suggested that the position of the physis, and its relationship with the Schöttle point, changes during growth 29 and that the distance from the physis varies as one moves posteriorly. 17 This study has shown that the positioning of the graft affects the isometry of the construct; therefore, careful intraoperative consideration of the physis and the effect that subsequent growth may have on the isometry of the construct should be utilized to determine optimal placement of the MPFL graft. In the pediatric skeletally immature knee, femoral MPFL placement can be adjusted either distal or posterior to avoid the physis with epiphyseal drilling.
Limitations
Our results provide surgeons with a biomechanical basis for how to choose the position of the femoral insertion point of an MPFL graft. However, our study does have several limitations. First, as a biomechanical study on cadaveric tissue, correlation with clinical outcomes such as increased patellofemoral contact pressures resulting in flexion pain or risk for dislocation in extension is difficult. The in vivo mechanics of the patellofemoral joint are subject to the soft tissues about the knee as well as overall lower extremity alignment, and while this model isolates the MPFL as the structure of interest, the necessary dissection of surrounding soft tissues may alter patellar tracking during range of motion testing. Second, the draw wire and suture construct used in this study to approximate the MPFL and estimate tension behave biomechanically differently than the partially elastic biologic tendon graft material. While the length between the patellar and femoral anchor points in vivo are a function of an individual patient's anatomy, the partially elastic ductile properties of tendon material allow it to absorb energy in deformation compared with the nonductile, nonelastic suture material used as a graft analog in this study. Therefore, the relationship between draw wire length in this study and the influence of graft tension on patellofemoral forces in vivo must serve only as an estimate in an imperfect biomechanical model. Further studies to evaluate this biomechanical model in specimens with a more anatomic soft tissue envelope or using anatomic graft tissue and direct tension measurements may provide more clinically translatable results. A study using these methods but directly evaluating graft tensioning forces in the patellofemoral joint with different femoral insertion points may result in a more clinically relatable evaluation of how this technique parameter affects anterior knee pain after MPFL reconstruction. Third, this study did not take into consideration variation in the patellar graft positioning and only evaluated femoral location. Last, the cadavers used in this study were older than a typical patellofemoral instability population; it is a possibility that different biomechanical results would be seen in younger cadaveric specimens. Further, patellofemoral pathology such as patella alta, variations in trochlear groove geometry, or lateral tibial tuberosity positioning may also influence the length that the graft travels from its origin to its insertion point on the patella.
Conclusion
If deviating from the Schöttle point, posterior and distal femoral tunnel positionings minimize the risk of MPFL graft tightening during knee flexion. Errant anterior and proximal positioning are concerning directions for MPFL overconstraint, and proximal tunnel placement should be avoided, as it is most at risk.
Footnotes
Acknowledgements
The authors acknowledge Jennifer Herrick for her expertise in the statistical analysis for this study.
Final revision submitted June 17, 2024; accepted June 24, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.S.G. has received education payments from Arthrex and hospitality payments from Stryker. D.I. has received grant support from Arthrex, education payments from Arthrex, and hospitality payments from Skeletal Dynamics and Acumed. R.T.B. has received education payments from Arthrex, consulting fees from DePuy/Medical Device Business Services, nonconsulting fees from Arthrex, royalties from Arthrex, and acquisitions payments from DePuy Synthes. S.K.A. has received consulting fees from Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
