Abstract
Background:
Additional anterolateral ligament (ALL) reconstruction has been associated with improved clinical outcomes and reduced failure rates in anterior cruciate ligament (ACL) reconstruction. Despite the excellent clinical results reported, there is still a heated debate about its indications. Currently, the indications are mainly based on the patient’s clinical criteria and not the imaging diagnosis of the injury of this ligament.
Purpose:
To compare the clinical outcomes of patients undergoing isolated ACL reconstruction and combined ACL and ALL reconstruction conditioned to intraoperative ultrasound-guided diagnosis of the ALL lesion.
Study design:
Cohort study; Level of evidence, 3.
Methods:
A retrospective analysis of prospectively collected data was performed. Patients undergoing primary ACL reconstruction between January 2017 and January 2022 were included. Patients were excluded if they had a previous history of ipsilateral knee surgery or if they underwent other concomitant procedures, including multiligament reconstruction surgery or osteotomy. The decision to perform an ALL reconstruction in addition to isolated ACL reconstruction was based on ultrasound diagnosis of this lesion. At the end of the study period, further knee injury and any other reoperations or complications were recorded and compared between patients who had isolated ACL reconstruction (no-ALLR group) and combined ACL and ALL. Propensity score matching was performed between groups. A multivariable analysis using the penalized Cox model was performed to explore the relationship between the graft rupture, surgical procedure type, and potential explanatory variables.
Results:
A total of 339 patients met the predefined eligibility criteria; 146 ACL reconstructions were performed in an isolated manner (no-ALLR group) and 193 were combined with ALL (ALLR group). After matching, 130 patients were allocated to each group. The mean follow-up was 36.7 months. The overall graft failure rate was 4.6% (6.9% in the no-ALLR group and 2.3% in the ALLR group; P = .076). The adjusted Cox regression showed that graft failure rate was nonsignificantly different between the groups (adjusted hazard ratio [HR], 0.36 [95% CI, 0.096-1.364]; P = .133). Younger age (≤20 years) was associated with graft failure (adjusted HR, 0.29 [95% CI, 0.121-0.719]; P = .007).
Conclusion:
Combined ACL and ALL reconstruction conditioned to intraoperative ultrasound-guided diagnosis of the ALL lesion has an equivalent ACL graft failure rate to isolated ACL. Intraoperative ultrasound diagnosis of an ALL injury may be an indication for the addition of an ALL reconstruction.
Keywords
The remarkable long-term clinical outcomes of combined anterior cruciate ligament (ACL) and anterolateral ligament (ALL) reconstruction have been the subject of a growing body of literature in the last decade, with better ACL graft survivorship, lower overall rates of reoperation, and lower rates of revision ACL reconstruction.17,23,27,29,43-45,49,50 Despite the excellent clinical results reported, there is still a heated debate about the indications for this combined use. A consensus established the guidelines to be followed in ALL reconstruction (ALLR), 48 but at that time, the imaging studies of ALL were not yet well developed, so to date the indications are mainly based on the patient’s clinical criteria1,19,34,54 and concomitant procedures such as repair of the medial meniscus. 52
In the years that followed those meetings, imaging studies of ALL have increased in number and quality.5,7-9,15,18,25,28,40,56 Magnetic resonance imaging (MRI) and ultrasound (US) are the main imaging methods for the evaluation of ALL. Although MRI is the gold standard for diagnosing most ligament injuries in orthopaedics, an inconsistency regarding the visualization of the ALL in its entirety on MRI is reported in the literature.8,13,25,56 US is a dynamic, low-cost, nonirradiating, and widely available option, and added to that, some authors have reported that the entire ALL is visible on US with 100% sensitivity.12,15 In the course of scientific evolution, it is paramount that this diagnostic test be included in the decision tree of ALLR indication. Furthermore, there is still a lack of studies to support new consensus meetings. 47
The aim of this study was to compare the clinical outcomes of patients undergoing isolated ACL reconstruction and combined ACL and ALL reconstruction conditioned to intraoperative US-guided diagnosis of the ALL lesion. Our hypothesis was that the failure rate would be equivalent between groups.
Methods
Institutional review board approval (Toulouse University Hospital: RnIPH 2021-64) was granted for this study, and all patients gave informed consent to participate. A retrospective analysis of prospectively collected data was performed. Patients undergoing primary ACL reconstruction by a single surgeon (E.C.) between January 2017 and January 2022 were considered for study inclusion. Patients were excluded if they had a previous history of ipsilateral knee surgery or if they underwent other concomitant procedures, including multiligament reconstruction surgery or osteotomy.
The decision to perform an ALLR in addition to isolated ACL reconstruction was based on US diagnosis (Aplio 500 Toshiba; 14-MHz probe; Toshiba) of the ALL lesion performed the day of the arthroscopy procedure with the patient under anesthesia, according to a previously validated protocol.8,9
ALL Lesion US-Guided Intraoperative Diagnostic Protocol
Exploration of the ALL is performed with a US high-frequency probe (Aplio 500 Toshiba; 14-MHz probe), with the knee at 90° of flexion and internal rotation of the foot. This position applies tension on the ALL favoring its visualization. First, the surgeon locates the iliotibial band (ITB) at its insertion on the Gerdy tubercle, and then moving the probe posteriorly, it is possible to visualize the tibial insertion of the ALL. Then, a 20° counterclockwise rotation (for a right knee) of the probe reveals the major axis of the ALL, allowing us to track the ALL proximally to its insertion on the femur. At this point, bone damage at the tibial insertion of the ALL (Segond fracture) can be found. 9 Once the ALL has been identified, dynamic testing is performed. When applying internal rotation of the knee, increased tension on this structure will be clearly visible if the ALL is intact. This does not occur if the ALL is injured. Furthermore, US analysis allows for short percutaneous incisions to be made exactly at the desired location for further tunnel placements (Figure 1). 7
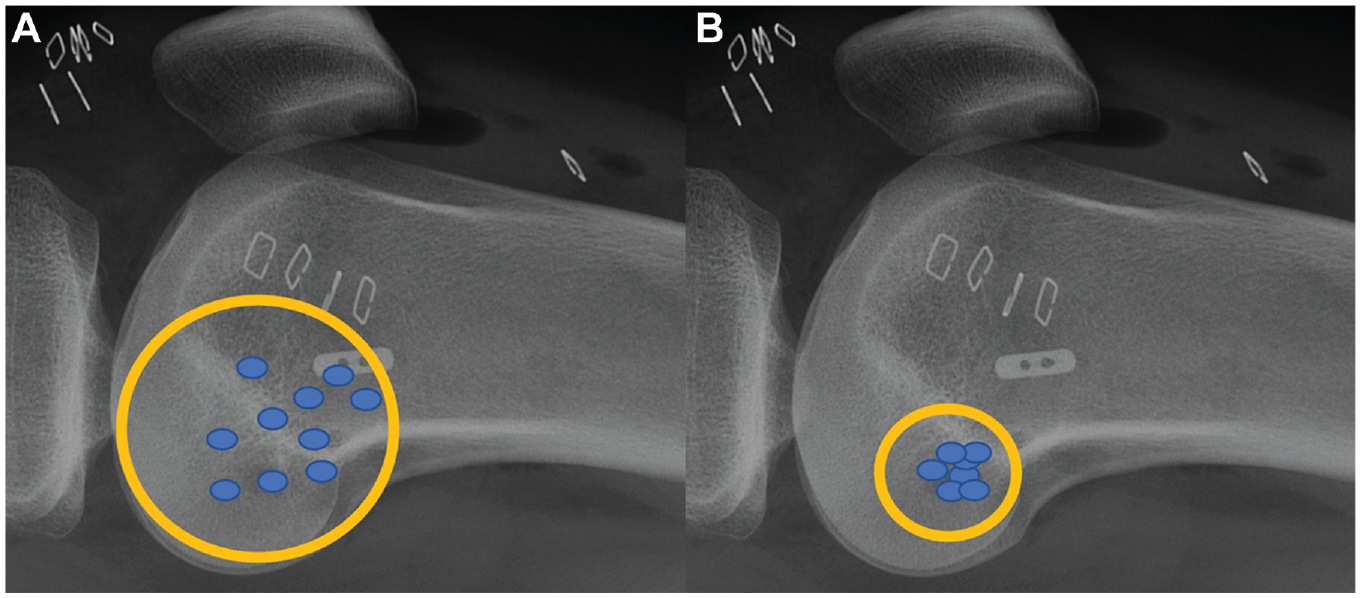
Femoral positioning of the anterolateral ligament graft posterior and proximal to the lateral epicondyle (A) with palpation alone and (B) with ultrasound guidance. 5
Combined ACL and ALL Reconstruction
Once the ALL lesion is confirmed using US, a vertical 2-cm incision is made slightly medial to the anterior tibial tuberosity. The semitendinosus and gracilis tendons are harvested with an open tendon stripper and are then cut close to their tibial insertion. For the ACL graft, the semitendinosus tendon is prepared in 4 strands on a TightRope device (Arthrex) with a FiberWire suture (No. 2; Arthrex) at the distal end. Both grafts are soaked in vancomycin solution.
The tibial tunnel is drilled from the hamstring incision with an outside-in guide. Then a 10 mm–long femoral tunnel is drilled with an inside-out guide. 10 The ACL graft is passed from distal to proximal, the TightRope fixation system (Arthrex) is fixed on the femoral cortex, and the graft is tightened and fixed at the tibia tunnel in 30° of flexion, with a BioComposite interference screw (Arthrex).
Two incisions are made after fixation of the ACL graft: the first one just posterior and proximal to the lateral epicondyle and the second one midway between the Gerdy tubercle and the fibular head (Figure 2). The ITB is incised starting at the proximal incision.

Lateral aspect of the left knee. Two incisions were made after fixation of the anterior cruciate ligament graft: the first one just posterior and proximal to the lateral epicondyle (LE) and the second one midway between the Gerdy tubercle (GT) and the fibular head.
A 5.5-mm suture anchor with 2 No. 2 Hi-Fi sutures (CrossFT; ConMed) is fixed to the femoral cortex. A Kelly clamp is introduced through the proximal incision, deep to the ITB and superficial to the lateral collateral ligament, toward the distal incision. The gracilis tendon is prepared and folded in 2 strands pulled with the clamp from distal to proximal, with the 2 free ends hanging distally. The proximal end of the graft is sutured on the femoral anchor by passing a strand of each suture in the fold. With the knee in full extension, the distal part of the graft is tightened and secured with a 6 × 20-mm Spiked Ligament Staple (Arthrex), which is impacted posterior to the Gerdy tubercle. The free end of the graft is cut flush with the ligament staple.
Isolated ACL Reconstruction
Once the integrity of the ALL is confirmed using US, the ACL reconstruction is performed using the same technique described in the previous section.
Rehabilitation
All patients underwent the same postoperative rehabilitation protocol. Full weightbearing was allowed immediately after the surgery. A cryotherapy knee brace (Excell’Ice Cryotherapy Articulated Knee Splint) was used in every case. Physical therapy started the day after surgery. Patients who received a meniscal repair were advised to limit their range of motion to between 0° and 90° for 6 weeks after the surgery. Running was allowed in the third month postoperatively. Return to pivoting and contact sports was allowed once the results of the isokinetic tests were satisfactory (>85% muscle strength in hamstrings and quadriceps compared with the contralateral side), generally around the seventh month.
Clinical Assessment and Follow-up
All patients were reviewed by the surgical team or a sports medicine physician at 3 and 6 weeks and 3, 6, 12, and 24 months postoperatively. Physical examination was performed to assess the stability of the operated knee.
The following patient data were collected: age, sex, time to surgery, date of last follow-up visit, preoperative pivot-shift grade, and meniscal status. For the preoperative pivot shift, the number of patients with grade 0 (none), 1 (slight), 2 (definite), or 3 (locking) was determined for each technique. For the meniscal status, the presence of a meniscal tear was recorded, along with the meniscus affected (medial, lateral, or both). In patients who had a meniscal tear, the operative records were consulted to determine if the damaged meniscus had been repaired or if a meniscectomy had been performed.
The outcomes of interest were the graft rupture rate, contralateral ACL tear rate, complication rate, Lysholm score, 43 International Knee Documentation Committee (IKDC) Subjective Knee Form score, 30 Self Knee Value (SKV), 38 and Marx Activity Rating Scale score. 39 These outcomes were collected by the independent surgeon (T.R.) who called or reviewed the eligible patients. The types of complications were tabulated.
Statistical Analysis
Propensity score matching was undertaken to reduce possible treatment selection bias and allow the creation of 2 groups (ALL and no ALL) in which confounding factors were balanced. A propensity score was determined for each patient based on the following criteria: age at the time of surgery (categorized as ≤20, 20 to 30, and >30 years), type of sport (categorized as no pivot, pivot: no contact, and pivot: contact), meniscal status (lesion and no lesion), body mass index (BMI) (World Health Organization categories), 20 and Tegner Activity Scale score. 4 Evaluation of covariate balance was determined by calculation of absolute standardized differences, with a prespecified threshold of <0.25 indicating that the groups were similar enough and adequately matched for reliable comparison.
Descriptive data analysis was conducted depending on the nature of the criteria considered. Descriptive statistics included the number of nonmissing observations, mean with standard deviation, for continuous variables and number of nonmissing observations with frequency (percentage) for categorical variables.
Categorical endpoints were compared between groups using the Pearson chi-square test or likelihood ratio chi-square test when necessary. The t test for 2-paired samples/2-tailed test was used to compare the distribution of continuous variables.
The Cox proportional hazards model, including multivariable analysis, was performed to explore the relationship between graft survivorship and potential explanatory variables. Survivorship analyses typically study the time between inclusion and graft failure. The Kaplan-Meier method was performed to illustrate the survival probability.
All reported P values were 2-sided and the significance threshold was <.05. Statistical analyses were performed using Stata software 17.0 (StataCorp). An a priori sample size calculation was not performed because all patients eligible for study participation were included.
Results
Patient and Clinical Characteristics
A total of 739 ACL reconstructions were identified during the study period. After applying the eligibility criteria, 339 patients were included in the analysis (Figure 3). Overall, 146 ACL reconstructions were performed in an isolated manner (no-ALLR group) and 193 were combined with ALL (ALLR group). After matching, 130 patients were allocated to each group. The mean follow-up was 36.7 months (34.8 months for the ALLR group and 38.7 months for the isolated no-ALLR group; P < .05). Table 1 presents the propensity score matching variables and their respective standardized coefficient. The predetermined threshold of 0.25 was met for all variables. Table 2 outlines the patient data and detailed analysis of each group.

Flowchart of patient inclusion. ACL, anterior cruciate ligament; ALL, anterolateral ligament.
Propensity Score With Standardized Coefficients a

BMI, body mass index; TAS, Tegner Activity Scale.
Patient Data a

Data are presented as n (%) unless otherwise indicated. ALLR, anterolateral ligament reconstruction; BMI, body mass index.
Pearson chi-square, t test for 2-paired samples/2-tailed test.
Likelihood ratio chi-square test.
Kruskal-Wallis equality-of-populations rank test.
Table 3 summarizes the surgical procedures performed on each group. Associated meniscal lesions and their respective treatments had a homogeneous distribution among the groups.
Intraoperative Data a

Data are presented as n (%). ALLR, anterolateral ligament reconstruction; LM, lateral meniscus; MM, medial meniscus; PM, partial meniscectomy.
Pearson chi-square or likelihood ratio chi-square test.
ACL Graft Failure
The overall graft failure rate was 12 of 260 (4.6%) (6.9% in the no-ALLR group and 2.3% in the ALLR group; P = .076). Within the age categories, this cohort showed 3 of 18 (17%) ACL graft failures for patients aged <20 years, 8 of 114 (7%) for patients aged between 20 and 30 years, and 1 of 113 (0.9%) for patients aged >30 years. The mean age for patients with an ACL graft failure was 30.3 years (SD, 9.1 years) in the ALLR group and 20.1 years (SD, 4.3 years) in the no-ALLR group.
Multivariable Analysis
The Cox model was performed to estimate the risk of graft rupture, per unit of exposure time, and explore relationships with potential explanatory variables (Table 4). For the purposes of multivariable analysis, sex,37,42 age,31,46,57 sports type 3 and whether a combined ALLR was performed or not21,22,24,26,35,36,53 were variables included in the Cox model. The graft failure rate was also nonsignificantly different between the groups (adjusted HR, 0.36 [95% CI, 0.096-1.364]; P = .133). Patient age was the only factor associated with ACL graft failure in this analysis (Table 4).
Adjusted Cox Regression Between ACL Graft Failure and ALLR a

ACL, anterior cruciate ligament; ALL, anterolateral ligament; ALLR, anterolateral ligament reconstruction; HR, hazard ratio; SE, standard error.
Age at the time of surgery categorized as ≤20, 20 to 30, and >30 years.
Type of sport categorized as no pivot, pivot: no contact, and pivot: contact.
Complications
This combined procedure was found to have more complications and a higher reoperation rate related to hardware removal, but this difference could not be statistically tested given the lack of events in the no-ALLR group. The rate of other complications, including contralateral ACL rupture, was not significantly different between groups (Table 5).
Complications a
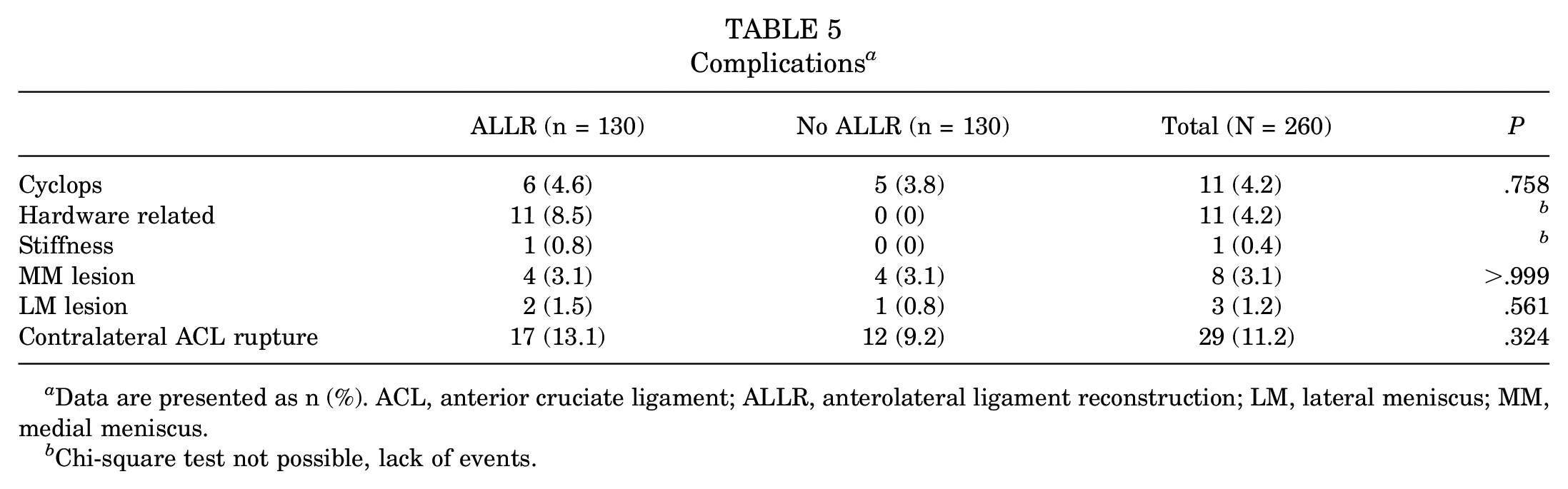
Data are presented as n (%). ACL, anterior cruciate ligament; ALLR, anterolateral ligament reconstruction; LM, lateral meniscus; MM, medial meniscus.
Chi-square test not possible, lack of events.
All postoperative patient-reported outcome measures (SKV and Lysholm, IKDC, Tegner Activity Scale, ACL–Return to Sport After Injury, and Marx scores) were similar in both groups (Table 6).
Postoperative Scores a

ACL-RSI, Anterior Cruciate Ligament–Return to Sport After Injury; IKDC, International Knee Documentation Committee; SKV, Self Knee Value; TAS, Tegner Activity Scale.
t test for 2-paired samples/2-tailed test.
Survivorship and Risk Analysis
The Kaplan-Meier analysis demonstrated an equivalent ACL graft survivorship between the groups at every time point assessed (log-rank test, P = .1255) (Figure 4).
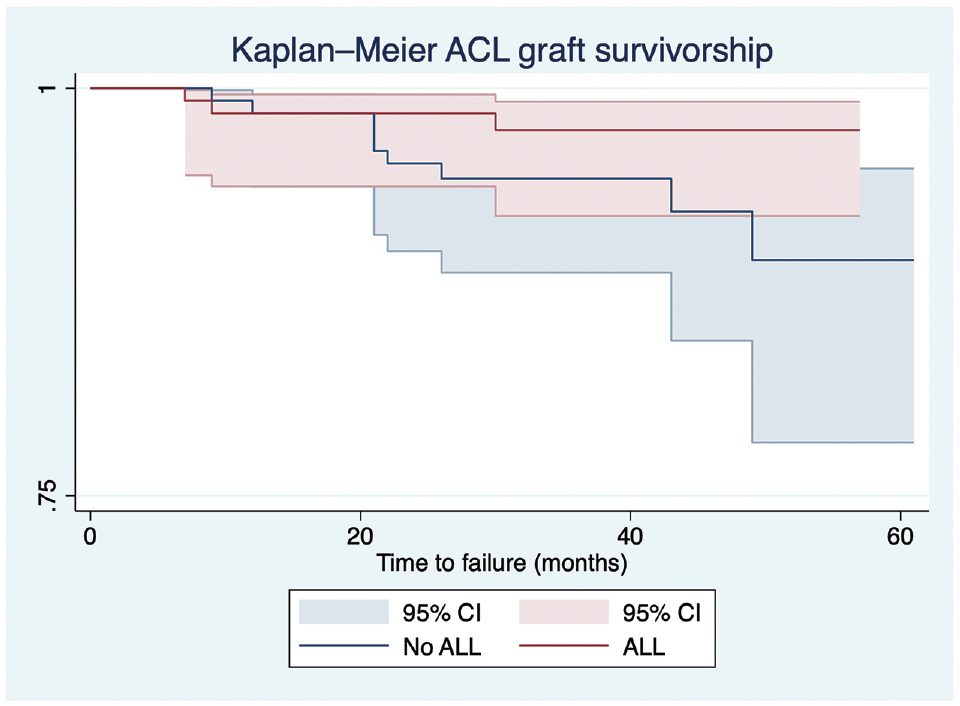
Kaplan-Meier analysis of anterior cruciate ligament (ACL) graft survivorship for each group. Log-rank test, P = .1255. ALL, anterolateral ligament.
Discussion
The main finding of this study was that patients who underwent isolated ACL reconstruction and patients who underwent combined ACL and ALL reconstruction conditioned to intraoperative US-guided diagnosis of the ALL lesion had equivalent ACL graft failure rates. Another important finding was that there were significant differences between groups with respect to reoperation and complication rates.
Combined lateral extra-articular procedures have regained popularity after years of ostracism after the Snowmass consensus meeting in 1989. 16 At the time, the clinical improvements of lateral extra-articular procedures were not yet fully studied and reported, and there was a major concern about possible complications. 16
Despite the reported improvements of combined ACL and ALL reconstruction in ACL graft survivorship reported in the literature,14,33,35,43,49,50 currently, based on various expert meetings, the indications for combined reconstruction do not include diagnosis of the ALL lesion itself.2,20,47,48 When we draw a patient profile to which we will indicate a combined ACL and ALL reconstruction, we are establishing a one-size-fits-all technique. Whereas when the ALLR is conditioned to its lesion diagnosis, the previously demonstrated clinical benefits can be achieved with an equivalent ACL graft failure rate, as the present study demonstrates. Although our study supports reconstruction of the ALL after confirmation of its injury, there are studies that suggest a combined reconstruction of the ACL and ALL as a preventative measure for the knee, for example, when suturing the medial meniscus ramp, in which reconstruction of the ALL has a protective effect. 51 At the time of this expert consensus,2,20,48 clinical studies based on MRI and US of the ALL had not yet come to light. Helito et al 28 recently reported a clinical study in which ALL abnormalities were the only variable with significant correlation to the pivot-shift grade. Although MRI is the gold standard for diagnosing most ligament injuries in orthopaedics, studies of ALL identification on MRI show discrepancy regarding sensitivity; the visualization of this ligament in its entirety on MRI scans varies in the literature between 11% and 96%.8,13,25,56 This inconsistency can be mainly due to orientation and thinness, as well as the proximity of the ALL to its neighboring ligament structures. 6 Among the diagnostic tests, US is a dynamic, low-cost, nonirradiating, and widely available option in the context of ALL lesions, 8 reaching 100% of sensitivity in the literature, 12 and is also statistically associated with high-grade pivot shift. 8 The latter is also observed in our population, in which the pivot-shift variable showed a statistical variance between the groups.
US, MRI, and the pivot-shift test have been proven to be capable of clinically identifying the presence of an ALL injury,8,28 but their efficacy is contingent on the proficiency of the examiner. Variations in experience and expertise among different medical centers exist; however, the key takeaway is that diagnosing an ALL injury may serve as a valuable indicator for determining the necessity of ALLR.
To the best of our knowledge, this is the first study evaluating a technique for diagnosing ALL lesions with this many patients and longer follow-up. Most diagnostic studies are snapshot studies, assessing the presence of injuries associated with ACL insufficiency at one point in time.15,25,28,56 Long-term follow-up clinical studies are needed for the validation and incorporation of these diagnostic tests in the decision tree of ALLR indications.
In addition to visualizing the ALL in its entirety, as previously described, US allows the marking of landmarks for the reconstruction of this ligament. In cadaveric models, the ALL has been found to originate on the femur 4.7 mm from the lateral collateral ligament insertion in the posterior and proximal directions. 32 This holds significant importance, as research indicates that the positioning of the femoral tunnel of the ALL plays a crucial role in achieving favorable isometry. 41 The tibial attachment of the ALL was described to be approximately midway between the center of the Gerdy tubercle and the anterior margin of the fibular head. 32 Although these data help in localizing the tunnels during reconstruction of the ALL, palpation of these anatomic points can be inaccurate in percutaneous surgery, 5 which will guide the fixation of the graft, independently of the technique chosen for ACL reconstruction. This leaves it up to the surgeons to choose the technique, to graft the ALL either continuously with the ACL graft or independently, as it has been shown that cortical attachment provides satisfactory graft incorporation.11,55
The present study showed a greater reoperation rate regarding hardware removal for the group with combined ACL and ALL reconstruction. This is because of the author’s (E.C.) previous technique of using a staple to fix ALL. Nowadays, with the knotless anchor at both edges of the ALL graft (Knotless FiberTak; Arthrex), no reoperation regarding the removal of material has been observed.
Despite the survival of the ACL graft being equivalent between the 2 populations at 2 years of follow-up, indicating the relevance of the indication guided by US, diagnosis of the ALL lesion should not be the only criterion for the indication of ALLR. We need to separate curative indications linked to the identification of an ALL lesion from preventive indications related to other parameters. 7 Other preventive measures such as meniscal sutures should still be considered in the decision tree, given the protective effect of ALL on these indications in the long term, especially over the present study’s follow-up. The strength of this study is that it brings to light the importance of including ALL lesion imaging diagnosis among ALLR indications.
Limitations
The limitations of this study include its retrospective nature and nonrandomized design; also, a priori sample size calculation was not performed because we included all patients eligible in this cohort. However, the 2 groups were matched by a propensity score with regard to age at the time of surgery, meniscal status, BMI, and Tegner Activity Scale score, many factors that could render the data analysis more accurate with fewer confounding variables. Furthermore, in this study, there was no surgical correlation of US findings with the anatomy of ALL injuries, as the reconstruction was carried out using minimally invasive techniques. Another notable methodological constraint is the lack of a cohort with ALL injuries diagnosed using US who did not undergo ALLR. However, such a scenario would raise ethical concerns, as diagnosing a lesion without subsequent treatment would be deemed unacceptable. 47 Also, although clinically similar, the follow-up was statistically different between the groups in the univariate analysis; however, this was accounted for in the multivariable analysis as the exposure time was the metric used to perform graft survivorship analyses in the Cox model.
Conclusion
Combined ACL and ALL reconstruction conditioned to intraoperative US-guided diagnosis of the ALL lesion has an equivalent ACL graft failure rate to isolated ACL. Intraoperative US diagnosis of an ALL injury may be an indication for the addition of an ALLR.
Footnotes
Final revision submitted May 4, 2024; accepted May 20, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: E.C. is a consultant for Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Toulouse University Hospital (RnIPH 2021-64).
