Abstract
Background:
The growing popularity of glucagon-like peptide-1 receptor agonists (GLP-1-RAs) for weight loss could significantly impact joint preservation and arthroplasty. While this will in part be driven by the association between obesity, osteoarthritis (OA), and total joint arthroplasty (TJA), recent evidence also indicates that GLP-1-RAs may have direct joint-protective, anti-inflammatory effects.
Purpose:
To evaluate the association between GLP-1-RA use and the onset and progression of hip and knee OA in an obese population.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
A national health network was queried for patients with an index visit between June 1, 2021, and January 1, 2023, and a body mass index (BMI) ≥30. Patients were stratified into groups without (n = 1,092,225) and with(n = 237,043) preexisting hip and/or knee OA. One-to-one propensity score matching was used to balance GLP-1-RA use based on age, sex, race, BMI, and comorbid type 2 diabetes mellitus. Primary outcomes were incidence of hip OA, knee OA, major joint injections, total hip arthroplasty (THA), and total knee arthroplasty (TKA) within 1 year. Cox proportional hazards models were used to estimate hazard ratios (HRs) between cohorts prescribed and not prescribed GLP-1-RAs.
Results:
In patients with preexisting OA, GLP-1-RA use correlated with reduced odds of conversion to THA (1.1% vs 2.2%; HR, 0.6; 95% CI, 0.5 to 0.8) and TKA (1.4% vs 2.1%; HR, 0.8; 95% CI, 0.6 to 0.9) within 1 year. In patients without preexisting OA, GLP-1-RA use was associated with an increased incidence of hip OA (0.9% vs 0.7%; HR, 1.4; 95% CI, 1.2 to 1.6), knee OA (2.1% vs 1.9%; HR, 1.3; 95% CI, 1.2 to 3.1), major joint injections (2.2% vs 1.8%; HR, 1.4; 95% CI, 1.3 to 1.5), and TKA (0.09% vs 0.04%; HR, 2.6; 95% CI, 1.6 to 4.3). Comparing cohorts without prior OA, patients who were prescribed a GLP-1-RA demonstrated slightly greater decreases in BMI (–1.00; 95% CI, –1.06 to −0.96) at 1-year after the index visit compared with patients not prescribed a GLP-1-RA (–0.90; 95% CI, –0.94 to −0.84). However, in patients with a prior diagnosis of hip or knee OA, there was no difference noted in BMI change.
Conclusion:
GLP-1-RAs may provide direct disease-modifying behaviors in patients with preexisting OA diagnosis, per a reduced risk of conversion to TJA not attributable to weight loss. Further investigation is also needed to elucidate the association between GLP-1-RA use and the increased incidence of OA diagnosis and conversion to TKA in patients with no preexisting OA diagnosis.
Keywords
Obesity has remained a growing epidemic in the United States, now afflicting roughly 40% to 45% of adults, with 9% classified as morbidly obese.10,30 This rising prevalence of obesity has had a significant impact on the fields of joint preservation, sports medicine, and arthroplasty, as it has played a notable role in the increased demand for total joint arthroplasty (TJA), particularly among younger patients. 6 By 2029, nearly half of Americans are projected to be obese, with obese patients expected to comprise approximately 70% of all total knee arthroplasties (TKAs) and 55% of all total hip arthroplasties (THAs).3,4 This is largely attributable to the association between obesity and osteoarthritis (OA), as adults with obesity are at a 4- to 5-fold increased risk of developing OA of the knee. 1 Moreover, individuals with a body mass index (BMI) exceeding 40 are 8.5 times more likely to require THA compared with their nonobese counterparts. 2 Thus, the recent enthusiasm surrounding glucagon-like peptide-1 receptor agonists (GLP-1-RAs) for medically supervised weight loss can be expected to generate downstream effects for orthopaedic surgeons.17,19 Despite being initially indicated for type 2 diabetes mellitus (T2DM) management, GLP-1-RAs have quickly garnered national attention for their ability to promote substantial weight loss among obese patients. 11
In 2014, liraglutide became the first GLP-1-RA to receive US Food and Drug Administration (FDA) approval for weight loss under the brand name Saxenda (Novo Nordisk), a more potent form of the original T2DM-indicated brand, Victoza (Novo Nordisk). 8 Then, in 2021, semaglutide was approved under the brand name Wegovy (Novo Nordisk), a higher dose of its popular predecessor, Ozempic (Novo Nordisk). The most recent of these next-generation GLP-1-RA drugs to be introduced was tirzepatide, a GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) receptor dual agonist, which received FDA approval in November 2023 under the brand name Mounjaro (Eli Lilly and Company). The therapeutic effects of these drugs may not be limited to T2DM and obesity management, however, as recent evidence suggests a potential for GLP-1-RAs to provide direct protection of joint tissue.27,31 Studies involving osteoarthritic mouse models have shown that liraglutide has potent anti-inflammatory effects by reducing several proinflammatory cytokines implicated in OA and inhibiting apoptosis in chondrocytes.5,24,36 As GLP-1 receptors are also present on articular cartilage and synovial membrane in humans, these drug properties may provide a means of preventing or limiting the progression and symptoms of OA.20,21 This possibility has recently begun to be explored, as the Shanghai Osteoarthritis Cohort study followed 1800 patients with osteoarthritis and found that GLP-1-RA use for at least 2 years translated to moderately decreased Western Ontario and McMaster Universities Osteoarthritis Index scores, lower analgesic consumption, decreased cartilage loss, and a lower incidence of knee surgery. 36 However, there is a paucity of additional literature, and, as this prior study collected data between 2011 and 2017, it did not capture the potential therapeutic effects of the more potent, next-generation GLP-1-RA and GIP dual agonists, nor did it include patients without preexisting OA.
Therefore, this national database study aimed to evaluate the impact of current GLP-1-RAs and GIP receptor dual agonists in obese patients with and without preexisting hip and/or knee OA. Primary outcomes assessed were the incidence of symptomatic OA in patients without a prior diagnosis, rates of major joint injections (as a surrogate for arthritis severity), and conversion to TJA within a 1-year period after initiation of GLP-1-RA treatment, as compared with endpoints for those patients not prescribed a GLP-1-RA.
Methods
Data Source
The US collaborative network within the TriNetX analytics platform was utilized by the present analysis to access real-time data from the deidentified electronic health records of >89 million adult patients from 61 health care organizations within the United States. 22 TriNetX has partnered with health care organizations across 30 countries and thousands of sites, as well as data providers covering 99% of US health plans, to create a linked and continually updated global health research network. By aggregating data directly from electronic medical records on an ongoing basis, TriNetX offers up-to-date information that better reflects the current patient population. Its partner health care organizations include a diverse range of hospitals and primary care and specialty providers, covering various geographies, age groups, and income levels. However, while detailed data are available on diagnoses and procedures, other factors such as socioeconomic and lifetime variables are not comprehensively captured, which may limit the generalizability of this otherwise robust data set. Current Procedural Terminology (CPT) codes; International Classification of Diseases (ICD) Clinical Modification, 9th and 10th Revision (ICD-9 and -10, respectively), codes; RxNorm codes; and Logical Observation Identifiers Names and Codes can be used to query diagnoses, procedures, medications, and laboratory values, respectively. Patient details are also available within the platform. The TriNetX analytics platform has been deemed exempt from the Case Western Reserve University institutional review board by a qualified expert as defined in Section §164.514(b)(1) of the Health Insurance Portability and Accountability Act Privacy Rule, as all patient records are deidentified.
Patient Selection
The study design and results reporting followed the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 32 All queries of the network were conducted on January 28, 2024. Primarily, we identified 1,329,268 adult patients who presented with a BMI of ≥30 between June 1, 2021, and January 1, 2023. As a requirement, these patients had their BMI recorded at least 1 year after their primary visit (index visit). This time period was selected to reflect the period in which newer-generation GLP-1-RAs with greater potency have received FDA approval. 29 This yielded 2 major subgroups: (1) obese patients without an existing diagnosis of hip and/or knee OA (n = 1,092,225) and (2) obese patients with an existing diagnosis of hip and/or knee OA (n = 237,043). Subgroup 2 was further segregated as (2a) patients with an existing diagnosis of hip OA (n = 81,973) and (2b) patients with an existing diagnosis of knee OA (n = 194,186). All subgroups were then further subdivided into cohorts that were prescribed a GLP-1-RA and cohorts that were not prescribed a GLP-1-RA within the study period. RxNorm Concept Unique Identifiers were used to identify the use of semaglutide (1991302), albiglutide (1534763), dulaglutide (1551291), liraglutide (4755968), lixisenatide (1440051), exenatide (60548), or tirzepatide (2601723) (Figure 1, Appendix Table A1). We excluded all patients who received their first GLP-1-RA prescription at any time other than the index event.

STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) flowchart depicting patient selection. BMI, body mass index; GLP-1, glucagon-like peptide-1; OA, osteoarthritis.
Outcomes Evaluated
For the OA-free cohorts, we assessed the rates of diagnosis for hip OA (ICD-10 code M16), knee OA (ICD-10 code M17), THA (CPT code 27130), TKA (CPT code 27447), and major joint injections (CPT code 1022217; used as a proxy for OA progression/severity) within 1 year of the index event. Notably, TriNetX automatically maps ICD-10 codes to ICD-9 codes in periods before the adoption of ICD-10. For the cohorts with prior OA, we assessed conversion to THA for the hip OA cohort, conversion to TKA for the knee OA cohort, and occurrence of major joint injections for both cohorts within 1 year of the index event. Secondarily, we assessed the change in BMI from the date of index presentation to at least 1 year later.
Statistical Analysis
All statistical analyses were conducted within the TriNetX platform. One-to-one propensity score matching was utilized to balance the following covariates between cohorts: age, race, sex, BMI, and T2DM. A greedy nearest-neighbor algorithm with a caliper of 0.1 pooled standard deviation of the logit of the propensity score was utilized to accomplish this. Cohorts were considered well matched if the standardized mean difference was ≤0.1. Independent-samples t tests and chi-square tests for continuous and categorical variables, respectively, were used to assess baseline differences between the cohorts. Hazard ratios (HRs) and corresponding 95% confidence intervals were calculated with a Cox proportional hazards model. A P value of <.05 was considered significant.
Results
Newly Diagnosed OA and Treatments After Starting GLP-1-RA
On propensity matching, the 2 obese, OA-free patient cohorts each had 61,069 patients (Table 1). There were no differences in age, race, sex, or comorbid T2DM between cohorts; however, after propensity matching, the cohort of patients who were not prescribed GLP-1-RAs had a slightly higher mean initial BMI compared with patients who were prescribed GLP-1-RAs (36.5 ± 5.6 vs 36.2 ± 5.7; P < .001). Between OA-free cohorts, patients who were prescribed a GLP-1-RA had a greater incidence of a new diagnosis of hip OA (0.9% vs 0.7%; HR, 1.41; 95% CI, 1.24-1.60), knee OA (2.1% vs 1.9%; HR, 1.25; 95% CI, 1.15-1.35), and major joint injections (2.2% vs 1.8%; HR, 1.35; 95% CI, 1.25-1.47) within 1 year of the initial prescription (Table 2, Figure 2). Although there was an increased incidence in TKA among patients prescribed GLP-1-RAs (0.09% vs 0.04%; HR, 2.61; 95% CI, 1.60-4.25), there was no difference in the 1-year incidence of THA between patients who were versus were not prescribed GLP-1-RAs (0.05% vs 0.05%; HR, 1.13; 95% CI, 0.69-1.85).
Characteristics of Hip and Knee OA-Free Cohorts Prescribed and Not Prescribed GLP-1 Agonists a

Data are presented as mean ± SD or n (%) unless otherwise indicated. BMI, body mass index; GLP-1, glucagon-like peptide-1; OA, osteoarthritis; PSM, propensity score matching; St. Diff, standardized difference; T2DM, type 2 diabetes mellitus.
Odds of New OA Diagnosis and/or Total Joint Arthroplasty a

Data are presented as n (%) unless otherwise indicated. In obese cohorts, data are presented within 1 year of index event. Patient counts of ≤10 are reported by TriNetX as 10 to maintain patient confidentiality. Values with P < .05 are shown in bold. GLP-1, glucagon-like peptide-1; HR, hazard ratio; OA, osteoarthritis; THA, total hip arthroplasty; TKA, total knee arthroplasty.

Survival curves for osteoarthritis (OA) and total joint arthroplasty in obese OA-free patients given and not given glucagon-like peptide-1 receptor agonists (GLP-1-RAs). THA, total hip arthroplasty; TKA, total knee arthroplasty.
Outcomes for Preexisting Hip or Knee OA After Starting GLP-1-RA
After propensity matching, the 2 cohorts with preexisting hip OA each had 6714 patients (Table 3). There were no differences in age, race, sex, or diagnosis of T2DM between cohorts, though there was a statistically signficant difference in BMI. Within 1 year of being prescribed a GLP-1-RA, obese patients with prior hip OA had a largely reduced incidence of THA compared with patients who were never prescribed GLP-1-RAs (1.1% vs 2.2%; HR, 0.60; 95% CI, 0.45-0.79) (Table 4, Figure 3). However, there was no difference in the percentage of patients who received an injection into a major joint between the GLP-1-RA and no–GLP-1-RA cohorts (12.0% and 12.7%, respectively; HR, 1.07; 95% CI, 0.97-1.18).
Characteristics of Cohorts With Prior Hip OA Prescribed and Not Prescribed GLP-1 Agonists a
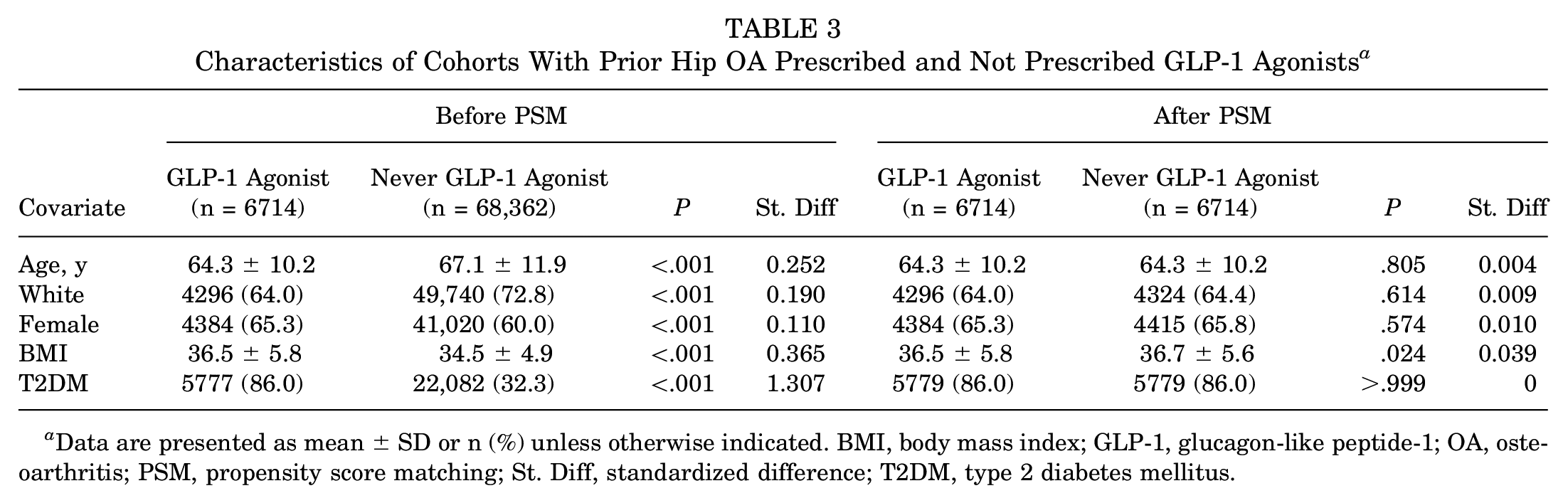
Data are presented as mean ± SD or n (%) unless otherwise indicated. BMI, body mass index; GLP-1, glucagon-like peptide-1; OA, osteoarthritis; PSM, propensity score matching; St. Diff, standardized difference; T2DM, type 2 diabetes mellitus.
Odds of THA and Injection Procedures Within 1 Year of Index Event in Obese Cohorts With Prior Hip OA a
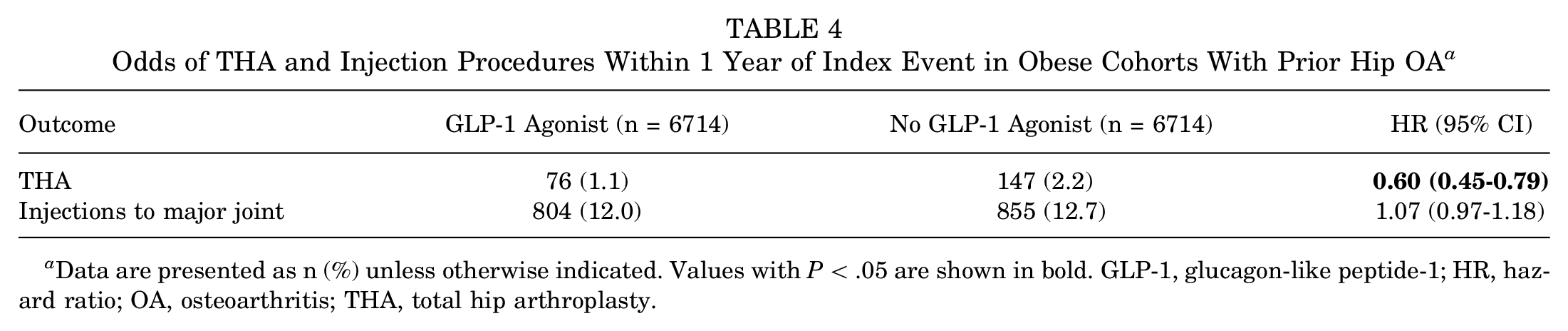
Data are presented as n (%) unless otherwise indicated. Values with P < .05 are shown in bold. GLP-1, glucagon-like peptide-1; HR, hazard ratio; OA, osteoarthritis; THA, total hip arthroplasty.

Survival curves for total joint arthroplasty in obese patients with prior hip and/or knee osteoarthritis (OA) given and not given glucagon-like peptide-1 receptor agonists (GLP-1-RAs). THA, total hip arthroplasty; TKA, total knee arthroplasty.
Similarly, after propensity matching, the 2 cohorts with preexisting knee OA each had 16,124 patients (Table 5). There were no differences in age, race, sex, or diagnosis of T2DM; however, the cohort prescribed GLP-1-RAs had a slightly lower initial mean BMI than the cohort that was never prescribed GLP-1-RAs (37.2 ± 5.8 vs 37.5 ± 5.7; P < .001). Within 1 year of initially being prescribed a GLP-1-RA, patients with prior knee OA had moderately decreased odds of having a TKA compared with patients not prescribed GLP-1-RAs (1.4% vs 2.1%; HR, 0.75; 95% CI, 0.63-0.89) (Table 6). However, there was no difference in the percentage of patients who received an injection into a major joint between the GLP-1-RA and no–GLP-1-RA cohorts (12.8% and 13.9%, respectively; HR, 1.03; 95% CI, 0.97-1.10).
Characteristics of Cohorts With Prior Knee OA Prescribed and Not Prescribed GLP-1 Agonists a
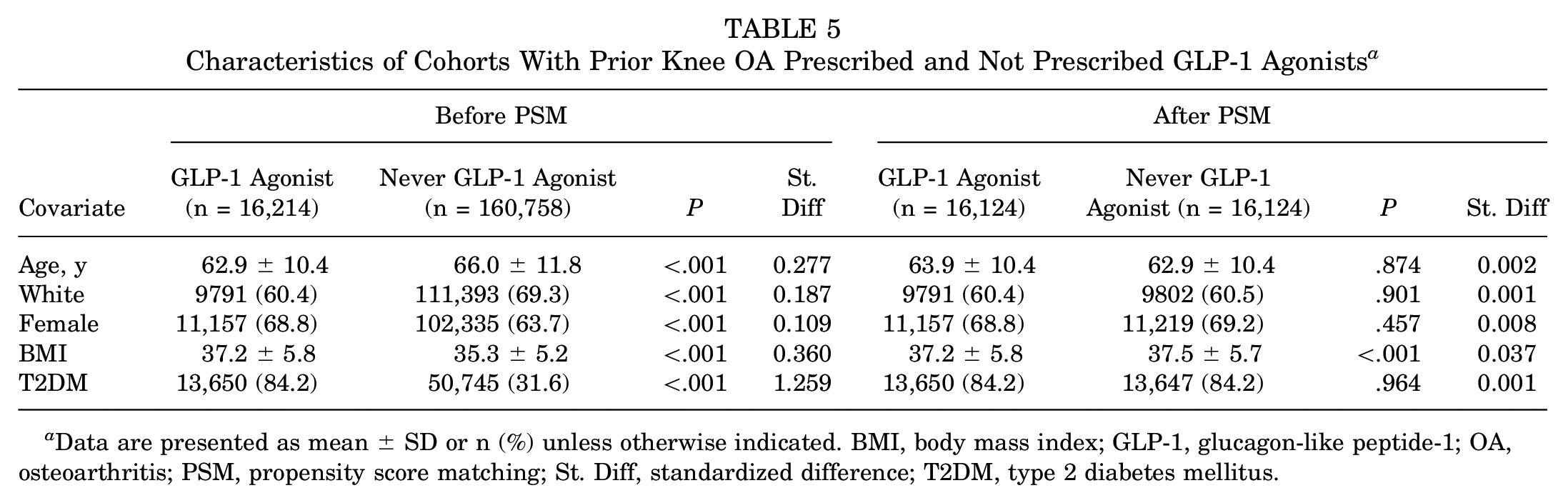
Data are presented as mean ± SD or n (%) unless otherwise indicated. BMI, body mass index; GLP-1, glucagon-like peptide-1; OA, osteoarthritis; PSM, propensity score matching; St. Diff, standardized difference; T2DM, type 2 diabetes mellitus.
Odds of TKA and Injection Procedures Within 1 Year of Index Event in Obese Cohorts With Prior Knee OA a

Data are presented as n (%) unless otherwise indicated. GLP-1, glucagon-like peptide-1; HR, hazard ratio; OA, osteoarthritis; TKA, total knee arthroplasty.
BMI Outcomes
Comparing cohorts without prior OA, patients who were prescribed a GLP-1-RA demonstrated slightly greater decreases in BMI (–1.00; 95% CI, –1.06 to −0.96) at 1-year after index visit than patients who were not prescribed a GLP-1-RA (–0.90; 95% CI, –0.94 to −0.84) (Table 7). However, in patients with a prior diagnosis of hip OA, there was no difference in BMI change between patients who were (–0.90; 95% CI, –1.10 to −0.71) and were not (–1.00; 95% CI, –1.19 to −0.81) prescribed a GLP-1-RA. Similarly, in patients with prior knee OA, there was no difference in BMI change between patients who were (–1.20; 95% CI, –1.33 to −1.07) and were not (–1.20; 95% CI, –1.32 to −1.08) prescribed a GLP-1-RA.
BMI in Cohorts Given and Not Given GLP-1 Agonists a
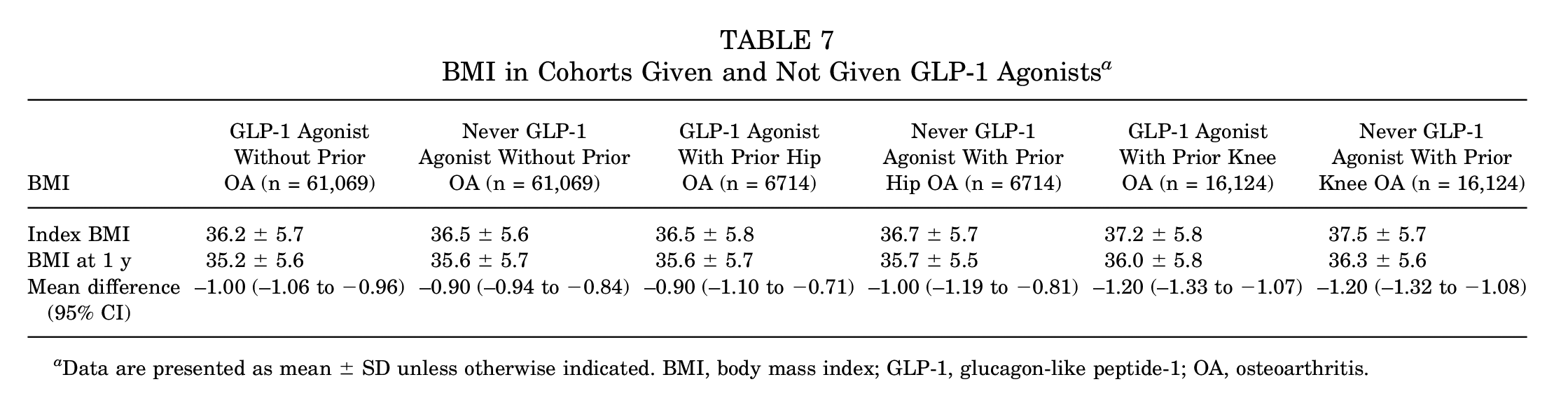
Data are presented as mean ± SD unless otherwise indicated. BMI, body mass index; GLP-1, glucagon-like peptide-1; OA, osteoarthritis.
Discussion
While much of the enthusiasm surrounding the GLP-1-RA drug class has stemmed from the ability to promote substantial weight loss, this may ultimately account for only a small portion of the influence that these drugs can have on the fields of joint preservation, sports medicine, and arthroplasty. In addition to the benefits conferred through weight loss, GLP-1-RAs may provide a direct anti-inflammatory and analgesic effect to joint tissue. Therefore, this large database study aimed to explore the impact of contemporary GLP-1-RAs on the development and progression of OA in obese patients. Our results showed that patients without a preexisting diagnosis of OA who were prescribed a GLP-1-RA had a greater incidence of hip and knee OA within 1 year of the initial prescription, with a concurrent increase in incidence of TKA but not THA. However, patients with a prior diagnosis of hip and knee OA who were prescribed a GLP-1-RA had largely reduced odds of THA and moderately reduced odds of TKA, respectively, within 1 year although no difference in major joint injections was noted.
The increased incidence of OA diagnosis in patients prescribed a GLP-1-RA is contrary to what would be expected, particularly when considering the BMI data for the initially OA-free cohort. Within this cohort, patients prescribed a GLP-1-RA demonstrated greater reductions in BMI over a 1-year period than their non–GLP-1-RA counterparts. While this would theoretically confer greater reprieve of weightbearing forces applied to the hip and knee, it should be noted that despite the difference being statically significant, the change differed by only 1 point, which may not be clinically significant. Nevertheless, this unexpected increase in the incidence of OA was accompanied by an increase in major joint injections and a particularly marked increase in odds of conversion to TKA, although not THA, for GLP-1-RA users. These findings are even more paradoxical in the context of the recent literature, as Meurot et al 21 found that liraglutide administration in an OA mouse model led to a decrease in several proinflammatory cytokines implicated in the progression and pathogenesis of OA, including nitrite, prostaglandin E2, and interleukin-6, while also providing an analgesic effect. Chen et al 5 similarly found that liraglutide provided in vivo antiapoptotic effects on chondrocytes in an OA mouse model, resulting in reduced degradation of the extracellular cartilage matrix. While it is notable that these models had preexisting OA on GLP-1-RA administration, there is no existing literature to the authors’ knowledge that would suggest a mechanism for GLP-1-RAs to directly promote the onset of symptomatic OA. Therefore, we suspect that these results may instead be explained by a mediating variable not captured by this study, such as a potential for increased physical activity to help with weight loss and/or glucose control associated with GLP-1-RA use, thereby increasing mechanical stress. It is also unclear why there was only an increased risk for TKA but not THA, despite similarly increased risks of OA in both joints. This may be because of joint-specific factors that might influence the rate of OA progression in the setting of GLP-1-RA use. It is also possible that differences in medication adherence between groups may explain some of the observed findings. Further investigations involving patients without preexisting OA are needed to explain the increased risk of OA and TKA associated with GLP-1-RA use, as well as the differing outcomes observed for the hip and knee. Notably, these investigations should be prospective in nature in order to broaden the scope of variables that can be accounted for, which may clarify some of the unexpected findings in the present analysis.
Conversely, the results demonstrated in the cohort of patients with preexisting hip and/or knee OA diagnosis supported the existing literature. Despite comparable reductions in BMI after a 1-year period among GLP-1-RA users and nonusers alike, patients with preexisting OA who were prescribed a GLP-1-RA displayed markedly reduced odds of conversion to THA, as well as moderately reduced odds of TKA. With the BMI outcome data being comparable among GLP-1-RA users and nonusers, these findings cannot be reasonably attributed to weight loss, therefore supporting the potential for direct therapeutic effects on OA by GLP-1-RAs. Similarly, while the Shanghai Osteoarthritis Cohort study reported significantly decreased Western Ontario and McMaster Universities Osteoarthritis Index scores, cartilage loss, analgesic consumption, and incidence of TKA in obese patients taking a GLP-1-RA for at least 2 years, the authors noted that the reduced cartilage loss could not be fully attributed to weight loss. 36 If additional studies can corroborate these findings and confirm the suggested disease-modifying behaviors of GLP-1-RAs in OA, the future of orthopaedics may involve the routine administration of these drugs to obese patients presenting with symptomatic OA. Future investigations may also wish to explore dose-dependent effects and compare GLP-1-RAs with GLP-1/GIP receptor dual agonists.
Finally, it is notable that the BMI outcome data reported in this study were also rather unanticipated, as only the cohort of patients without preexisting OA diagnosis demonstrated more significant reductions in BMI after 1 year of GLP-1-RA use compared with nonusers. While the GLP-1-RA users who had a preexisting OA diagnosis did still achieve a reduced BMI at the 1-year follow-up, it was not significantly different from the BMI of patients who were not prescribed a GLP-1-RA. This is a surprising finding given recent phase 3 weight loss data from liraglutide, semaglutide, and tirzepatide, which showed a mean weight loss of 8% to 20.9% with consistent adherence for roughly 1 year, depending on the drug and dose.12,13,23,26,34 Previous studies28,33 have reported an inherent rate of failure for GLP-1-RAs and poor long-term adherence, with Weiss et al 33 reporting 50.9% adherence at 12 months and 47.4% at 24 months. Furthermore, there has been an observed rebound effect with discontinuation of the drug, as patients often regain the majority of weight loss achieved while taking it.25,35 So, while GLP-1-RAs have proven efficacious in clinical trials, the findings of this study call into question the real-world effectiveness and clinical application of these drugs.
Limitations
The retrospective nature of this study limited controllable factors such as dosing of GLP-1-RAs and concurrent medications administered. Notably, a dosage-based subanalysis was not feasible given the small cohort sizes and low incidence of outcomes, and we therefore included GLP-1-RAs and doses that are not classified as next-generation GLP-1 agonists. Patient BMI may also have been measured or self-reported, introducing potential for inaccuracies with the latter. Furthermore, the TriNetX platform does not document medication adherence, and the findings of this analysis should be interpreted with that in mind. Previous studies have reported a large degree of variability in the 1-year adherence of GLP-1 RAs, which has been limited because of the cost and availability of the drugs, side effects, and general poor adherence to injectable medications.7,9,32 Similarly, the TriNetX platform documents medications based on a prescription being filled, although the actual fill percentage is unknown. Large joint injections may not be a good surrogate for OA diagnosis/treatment. Many large joint injections could have been for shoulder pathology. Additionally, hip injections are not as commonly performed given the need for ultrasound or radiographic guidance. It is also notable that many patients with radiographic OA are asymptomatic. Nevertheless, the present study comprises an intention-to-treat analysis that evaluates contemporary GLP-1-RA use and associated outcomes within a 1-year period. Additionally, not all patients had BMI values recorded after the index event. Finally, as we used a deidentified patient database, results may also have been skewed because of inaccuracies in recording events within the electronic health record, although the large sample size (1,329,268 adult patients) should reduce the impact of these errors in the data set. While many of these potential limitations are inherent to the nature of this retrospective large database analysis, the TriNetX platform has been utilized by several publications in high-impact orthopaedic journals.14-16,18
Conclusion
Despite recent evidence to suggest the potential for GLP-1-RAs to provide direct joint-preserving effects, this national cohort study found an increased incidence of hip OA, knee OA, major joint injections, and TKA within 1 year of starting a GLP-1-RA for obese patients without preexisting OA. However, this study also found reduced odds of conversion to both THA and TKA associated with GLP-1-RA use in patients without preexisting OA, results that were notably not attributable to weight loss. There is a need for more studies to elucidate the possible disease-modifying behaviors of GLP-1-RAs in symptomatic OA. Notably, these investigations should also include patients without preexisting OA of the hip and knee to elucidate the paradoxical effects of GLP-1-RAs with respect to increased OA development in obese populations.
Footnotes
Appendix
GLP-1 Agonist Prescriptions to Matched Cohorts a
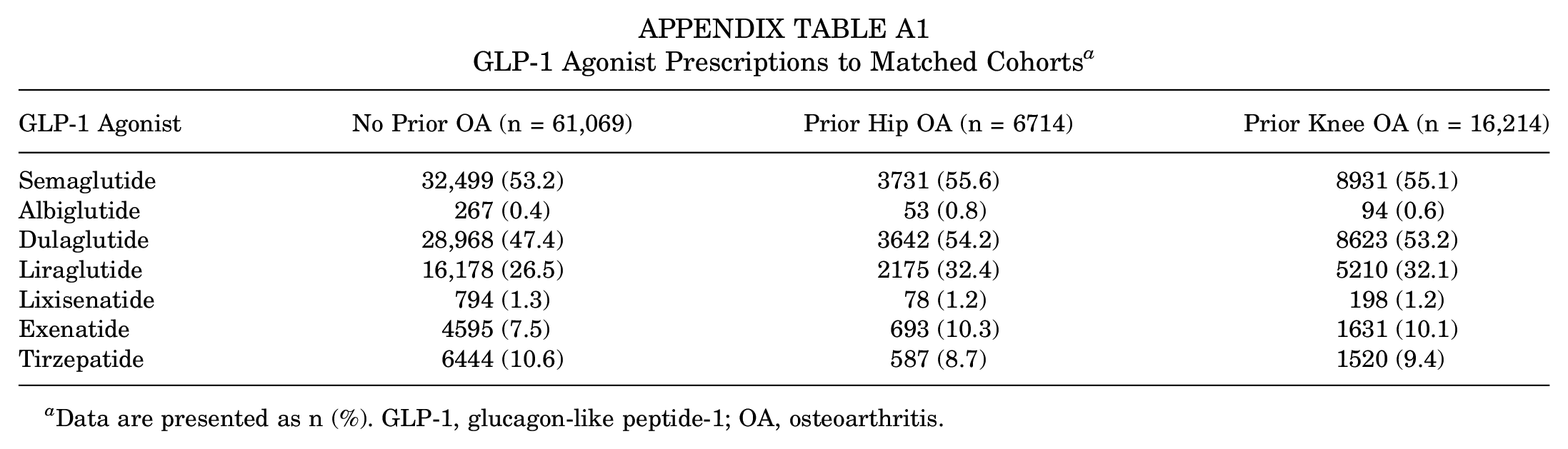
| GLP-1 Agonist | No Prior OA (n = 61,069) | Prior Hip OA (n = 6714) | Prior Knee OA (n = 16,214) |
|---|---|---|---|
| Semaglutide | 32,499 (53.2) | 3731 (55.6) | 8931 (55.1) |
| Albiglutide | 267 (0.4) | 53 (0.8) | 94 (0.6) |
| Dulaglutide | 28,968 (47.4) | 3642 (54.2) | 8623 (53.2) |
| Liraglutide | 16,178 (26.5) | 2175 (32.4) | 5210 (32.1) |
| Lixisenatide | 794 (1.3) | 78 (1.2) | 198 (1.2) |
| Exenatide | 4595 (7.5) | 693 (10.3) | 1631 (10.1) |
| Tirzepatide | 6444 (10.6) | 587 (8.7) | 1520 (9.4) |
Data are presented as n (%). GLP-1, glucagon-like peptide-1; OA, osteoarthritis.
Final revision submitted October 10, 2024; accepted October 14, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This project was supported by the Clinical and Translational Science Collaborative (CTSC) of Cleveland, which is funded by the National Institutes of Health (NIH), National Center for Advancing Translational Science (NCATS), and Clinical and Translational Science Award (CTSA) (grant UL1TR002548). D.C.K. has received consulting fees from Dynavax Technologies Corporation; Becton, Dickinson and Company; Merck Sharp & Dohme Corporation; and Pfizer. P.K.S. has received consulting fees from Intellijoint Surgical, LimaCorporate SpA, Signature Orthopaedics USA Corp, Synthes GmbH, Zimmer Biomet Holdings, DePuy Synthes Products, and Medical Device Business Services; education payments from Stryker; and acquisitions from Encore Medical. N.D.H. has received consulting fees from MicroPort Orthopedics, Encore Medical, Intellijoint Surgical, OMNIlife science, and Zimmer Biomet Holdings; hospitality payments from Globus Medical; and education payments from Smith+Nephew. A.F.K. has received royalties from Ortho Development Corporation and Zimmer Biomet Holdings; consulting fees from UOC USA, Bodycad USA Corp, Zimmer Biomet Holdings, and Ortho Development Corporation; and nonconsulting fees from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by TriNetX.
