Abstract
Background:
There are limited evidence-based guidelines to predict which osteochondritis dissecans (OCD) lesions will heal with nonoperative treatment.
Purpose:
To train a set of classification algorithms to predict nonoperative OCD healing while identifying new clinically meaningful predictors.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
Patients with OCD of the knee with open physes undergoing nonoperative management were prospectively queried from the Research on OCD of the Knee (ROCK) cohort (https://kneeocd.org) in April 2022. Patients were included if they met the study criteria for nonoperative treatment success or failure. Nonoperative treatment success was defined as complete healing on magnetic resonance imaging (MRI) and total return to sports participation. Failure was defined as the crossover from nonoperative management to surgery at any point at or beyond the 3-month follow-up. If a patient did not meet one of these criteria, they were not included. Normalized lesion size, lesion location, patient characteristics, and symptoms were used as clinically relevant predictors.
Results:
A total of 64 patients were included, of whom 24 (37.5%) patients successfully healed with nonoperative management. Multivariate logistic regression revealed that a 1% increase in normalized lesion width was associated with an increase in the likelihood of nonoperative failure (odds ratio [OR], 1.41 [95% CI, 1.17-1.81]; P < .01). By contrast, lesions in the posterior sagittal zone (OR, 0.08 [95% CI, 0.009-0.43]; P < .01) or the medial-most coronal zone (for lesions of the medial femoral) and lateral-most coronal zone (for lesions of the lateral femoral condyle) on MRI (OR, 0.05 [95% CI, 0.004-0.44]; P < .01) were associated with a decrease in the likelihood of nonoperative treatment failure. Support vector machines had a cross-validated area under the receiver operating characteristic curve of 0.89 and a classification accuracy of 83.3%.
Conclusion:
Lesion location in the posterior aspect of the condyle on sagittal MRI and lesion location in the medial-most or lateral-most locations on coronal MRI were identified as statistically significant predictors of increased nonoperative treatment success on multivariate analysis. Machine learning models can predict which OCD lesions will heal with nonoperative management with superior accuracy compared with previously published models.
Osteochondritis dissecans (OCD) of the knee is a focal idiopathic alteration of subchondral bone and/or its precursors with a risk for instability and disruption of adjacent articular cartilage that may result in premature osteoarthritis. 14 OCD has an incidence of approximately 9.5 per 100,000 patients. 10 Common symptoms of OCD include joint discomfort, pain with exercise, and swelling. 6 The exact etiology of OCD remains uncertain, with various theories suggesting that microtrauma from activity, endocrine disturbances, ischemia, or genetic predisposition could contribute to disease pathogenesis.2,10
So far, few guidelines define which OCD lesion types are amenable to nonoperative treatment. 19 Previous studies have investigated the influence of demographics and lesion characteristics on the likelihood of healing with nonoperative management, determining that variables such as normalized lesion area and cyst-like lesion occurrence significantly differed between healing and nonhealing lesions. 11 Some symptoms—giving way, swelling, locking, and clicking—have also been noted as predictors of nonoperative treatment failure.5,16,19 However, no study has attempted to quantify the nonoperative healing potential of OCD lesions with machine-learning models and assess its predictive accuracy.
In this study, we aimed to construct a machine learning classifier to estimate the probability that an OCD knee lesion would heal with nonoperative treatment. To accomplish this, prospective cohort data were obtained from the Research on OCD of the Knee (ROCK) cohort (https://kneeocd.org) to train a series of classification algorithms. We hypothesized that a machine learning classifier equipped with ROCK data would be able to determine whether OCD lesions would heal nonoperatively after a patient’s initial encounter with their treating clinician and would serve as a valuable tool for recommending operative or nonoperative treatment.
Methods
Data and Variables
Patients with OCD of the knee with open physes who were undergoing nonoperative management were prospectively queried from the ROCK cohort as of April 2022. The ROCK prospective cohort is the largest repository related to OCD cases, with data collected on over 1400 patients with OCD knee lesions. It is a longitudinal study at 23 participating institutions, and it is registered with ClinicalTrials.gov (NCT02771496). The cohort size and fine level of detail permit optimal algorithm development and allow us to consider whether anatomic, image-based, or demographic factors play a role in a lesion’s nonoperative healing capacity. Institutional review board approval was obtained at each participating institution before patient enrollment.
The ROCK cohort includes all patients seeking care for OCD lesions at any of the participating institutions, except those specified by the following exclusion criteria: (1) diagnosis of a focal chondral defect; (2) patients aged ≥26 years at the time of enrollment; (3) patient records missing data regarding OCD lesion location; (4) patient records with an incomplete or unverified screening form; and (5) patient records with an incomplete or unverified initial visit form. For this study, additional exclusion criteria were used: patients with a closed or closing physis at baseline magnetic resonance imaging (MRI); patients with a breach of the articular cartilage overlying their OCD lesions, because such lesions have a low probability of healing with nonoperative management1,15; and patients with data missing for any feature required for model development. Patients were included if and only if they met the necessary definitions for failure or success of nonoperative management.
Failure of nonoperative management was defined as the crossover from nonoperative management to surgery at any point at or beyond a 3-month follow-up. Failure of nonoperative management was used as a surrogate for failure to heal with nonoperative management. A patient could fail nonoperative treatment due to multiple reasons—including a lack of healing based on image evaluations, persistent symptoms, and inability to comply with nonoperative treatment. Successful nonoperative treatment was defined as complete healing on radiograph or MRI and full return to sports participation documented by providers on the ROCK follow-up study form.
Demographic data and each patient’s symptoms at their initial encounter were identified from ROCK cohort screening and baseline forms. Each patient had a standardized MRI protocol at their baseline encounter, and lesion length and width were measured on sagittal and coronal T1-weighted MRI according to previous reports.11,19 Lesion length and width were then divided by the maximum length and width of the femoral condyle on coronal and sagittal T1-weighted images to produce the ratio of the condyle each lesion occupied, referred to throughout this report as “normalized lesion” length or width. We also classified lesions by their anteroposterior and mediolateral positions on sagittal and coronal MRI, according to the classification system presented by Cahill and Berg. 3 Importantly, lesions in a given zone (eg, posterior zone on sagittal MRI or medial-most and lateral-most on coronal MRI) could also extend into other zones. In these cases, the lesion was recorded as being present in both zones. For example, during analysis, a lesion extending into both the central and posterior zones on sagittal MRI would be recorded as individually present in both the posterior and central sagittal zones. Mechanical symptoms were defined as the presence of any of the following: problems bending the knee, problems straightening the knee, catching or locking, and any noise or grinding in the joint.
Statistical Analysis
First, summary statistics describing demographic information, lesion characteristics, and imaging data were produced for the cohort. Details regarding each patient’s nonoperative management protocol—casting, bracing, and nonweightbearing durations—were identified. Similarly, tables were produced that stratified nonoperative healing success by lesion zone on coronal and sagittal MRI per Cahill and Berg’s classification scheme, as well as by whether they affected the medial or lateral femoral condyle. 3 Additionally, a survival analysis was conducted to depict the time points at which patients who failed nonoperative treatment crossed over to surgery.
To replicate previous studies examining the healing potential of OCD lesions with logistic regression, a multivariate logistic regression model was developed to ascertain the effects of age, normalized lesion dimensions, and isolated pain or mechanical symptoms on the likelihood of the success of nonoperative management.11,19 Next, a logistic regression model unique to this study was built: the Boruta feature selection algorithm was applied to potential predictors in the ROCK cohort to screen for variables of importance. 12 Next, the relationship between important variables selected by the Boruta algorithm and nonoperative healing was assessed by univariate analysis. Variables selected by the Boruta algorithm that met a significance threshold of P≤ .2 on univariate analysis were included in this study’s multivariate logistic regression to classify nonoperative healing. The predictors selected by the Boruta algorithm and univariate analysis were race, normalized lesion width, normalized lesion length, lesion location on coronal MRI, and lesion location on sagittal MRI. Several variables included in the model provided by Wall et al 19 —patient age and isolated pain versus mechanical symptoms—were not included in the models specific to this study, as the Boruta algorithm and univariate analysis did not select these variables. Next, a suite of hyperparameter-tuned machine learning algorithms, including a random-forest classifier, a neural network, linear- and radial-kernel support vector machines (SVMs)—a k-nearest-neighbor classifier, and a generalized boosted classifier—was modeled on this final feature set. Thereafter, the model specified by Wall et al, this study’s unique logistic regression model, and each previously mentioned machine learning classifier was trained on a random 80% of the cohort and tested on the remaining 20% of the cohort 5 times in a process termed “cross-validation.” Every patient was, therefore, a part of the test set 1 time and the training set 4 times. This entire cross-validation process was repeated 3 times for each model, and the mean 5-fold cross-validation accuracy across all iterations was computed for each model. Additionally, each model’s area under the receiver operating characteristic (ROC) curve (AUC), sensitivity, specificity, negative predictive value (NPV), and positive predictive value (PPV) were recorded. 18
No imputation was conducted throughout the investigation, and all patient records with missing data required for model development were excluded. All statistical analyses were conducted in the R Environment for Statistical Computing (R version 4.4.2). Summary statistics for the study cohort were computed and reported via the TableOne package. Model training, cross-validation, and hyperparameter tuning of various machine-learning models were performed using the Caret package and its dependencies. ROC curves were created with the MLeval package, which was also used to compute metrics of sensitivity, specificity, NPV, PPV, and their corresponding 95% CIs. The sensitivity and specificity reported for each ROC curve are those that produced the greatest Youden index (sensitivity+specificity– 1). Survival analysis was conducted with the survival package. Figures were created with the ggplot2 package and its dependencies. All statistical tests employed in univariate analysis were 2-sided. Odds ratios (ORs) with their 95% CIs were computed where possible for the coefficients of predictor variables in each regression model. The threshold for statistical significance in all regression analyses was P≤ .05.
Results
Cohort Characteristics
The study population included 64 patients, of whom 24 (37.5%) healed successfully with nonoperative management. The mean age of the study cohort was 11.7 years, of whom 40.6% were women; 75% of lesions occurred in the medial femoral condyle, 18.8% in the lateral femoral condyle, and 6.2% in the trochlea. A full description of relevant cohort characteristics is included in Table 1.
Characteristics of the Study Cohort (N = 64) a

Data are presented as mean ± SD or No. of patients (%). LFC, lateral femoral condyle; MFC, medial femoral condyle; MRI, magnetic resonance imaging.
Defined as lesion length or width divided by the maximum length or width of the femoral condyle on coronal or sagittal T1-weighted MRI to produce the ratio of the condyle each lesion occupied.
Rates of successful healing according to different nonoperative management strategies are presented in Figure 1. Also, 7, 6, and 11 patients were found to have healed successfully at the 3-, 6-, and 9-month follow-up time points, respectively (Figure 2). The time between baseline visit and operative management for those failing nonoperative treatment is shown in Figure 3.
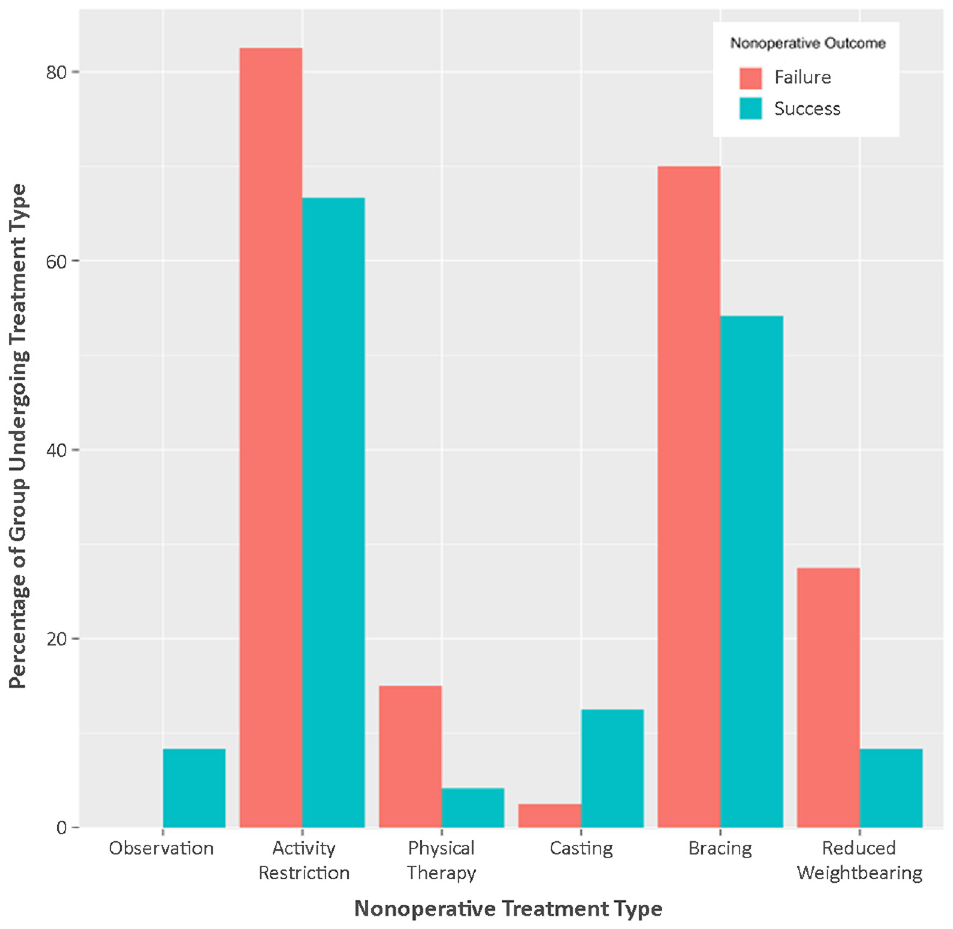
Nonoperative management strategies. All percentages are based on the entire group of 64 nonoperative patients.

Time points at which patients successfully healed (n = 24).

Time to surgery for patients who failed nonoperative management (n = 40).
Inference From Logistic Regression
When the model developed by Wall et al 19 was applied to the study data, no independent variable was found to have a statistically significant association with successful nonoperative healing. However, normalized lesion width approached significance (OR, 1.12 [95% CI, 1-1.29]; P = .06). Additionally, the model had a cross-validated accuracy of 58.2% and an AUC of 0.60, which is slightly better than a model with no discriminative ability. 13 In the logistic regression model developed by the present study, a 1% increase in lesion width relative to the width of the condyle was associated with a statistically significant increase in the likelihood of nonoperative failure (OR, 1.41 [95% CI, 1.17-1.81]; P < .01). By contrast, lesion presence in the posterior aspect of the condyle (OR, 0.08 [95% CI, 0.009-0.43]; P < .01), as well as the medial-most (for medial femoral condyle lesions) or lateral-most (for lateral femoral condyle lesions) coronal zones on MRI (OR, 0.05 [95% CI, 0.004-0.44]; P < .01), were associated with a significant decrease in the likelihood of a patient undergoing surgical management after starting on a nonoperative course. Table 2 compares the effect sizes, ORs, and significance for predictors in the 2 logistic regression models.
Comparison of Logistic Regression Coefficients Between the Present Study and Wall et al a

Dashes indicate areas not applicable. Bold P values indicate statistical significance (P≤ .05). MRI, magnetic resonance imaging; OR, odds ratio.
The regression in the present study was adjusted for race, although no coefficient for an individual race neared statistical significance.
Comparison of Model Classification Performance
An SVM with a linear kernel had the highest cross-validated classification accuracy of any model trained (83.9%). The logistic regression model developed in this study had an accuracy of 79.6%. In comparison, the same metrics for a radial-kernel SVM and the model developed by Wall et al 19 were 78.1% and 58.2%, respectively.
An SVM with a radial kernel had the highest cross-validated AUC of any model developed at 0.89 (95% CI, 0.81-0.97), with optimal sensitivity and sensitivity metrics of 82.5% (95% CI, 68%-91%) and 87.5% (95% CI, 69%-96%). The PPV and NPV for this model were 91.7% (95% CI, 78%-97%) and 75% (95% CI, 57-87%), respectively. The logistic regression developed in this study (AUC, 0.86 [95% CI, 0.77-0.95]) and the SVM with a linear kernel (AUC, 0.85 [95% CI, 0.76-0.94]) had comparable metrics, while logistic regression models from previous studies performed worse. Table 3 compares the performance characteristics of the 3 models developed in this study to the 3 model specifications reported from previous literature. Figure 4 shows the ROC curves for each available model.
Comparison of Classification Algorithm Performance During 5-Fold Cross Validation a

AUC, area under the receiver operating characteristic curve; NR, not reported; NPV, negative predictive value; PPV, positive predictive value; SVM, support vector machines.
These values are taken directly from Krause et al 11 ; the impact of cyst-like lesions could not be analyzed in the present study.
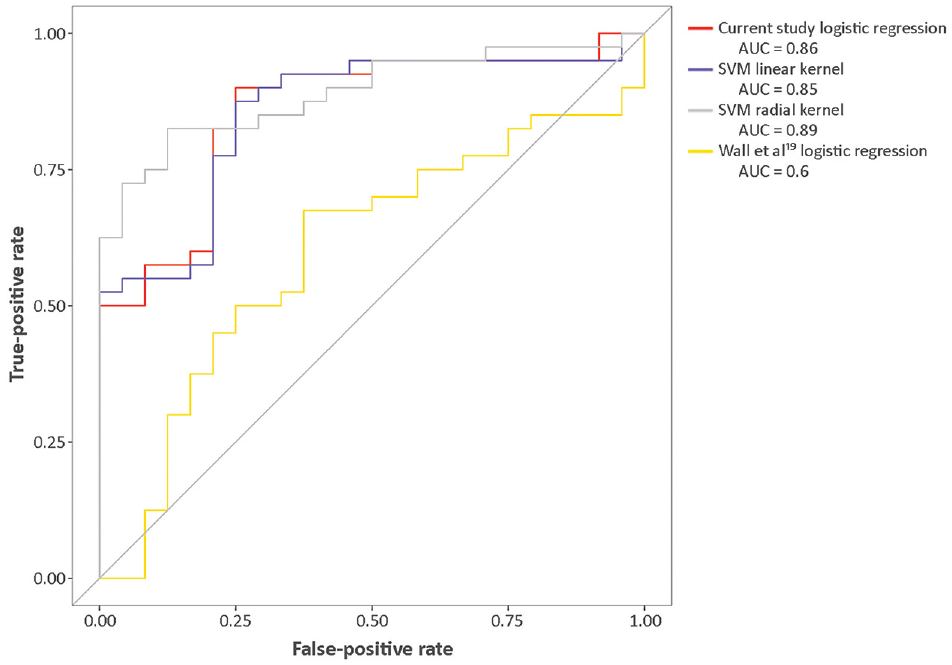
ROC curves and their areas under the curve (AUCs) for each available model. AUC, area under the curve; ROC, receiver operating characteristic; SVM, support vector machine.
Nonoperative Healing Differences by Anatomical Zone on MRI
In the overall cohort, 50% of lesions were present in the posterior zone of the condyle. However, lesions extending into this zone were more common within the group of patients who successfully healed with nonoperative management: 70.8% of the patients who successfully healed with nonoperative management had lesions in this zone. By contrast, lesions extending into this radiographic zone were less common in the group of patients who failed nonoperative management, with 37.5% of patients having lesions in this zone.
The percentage of lesions occurring in the medial-most zone (for lesions of the medial femoral condyle) and lateral-most zone (for lesions of the lateral femoral condyle) on coronal imaging was similarly different between patients who healed nonoperatively and those who did not. In the overall cohort, 18.8% of patients had lateral-most or medial-most coronal zone lesions. Lesions in these zones were more common among patients who healed with nonoperative management. Also, 29.2% of patients who healed successfully with nonoperative management had lesions in these zones, while those who went on to require surgery had lesions in these zones only 12.5% of the time. Tables 4 to 6 show the frequencies and percentages of lesions that failed nonoperative treatment by anatomical location and location on MRI.
Nonoperative Healing Rates Stratified by Anatomical Lesion and Sagittal MRI Zone a

LFC, lateral femoral condyle; MFC, medial femoral condyle; MRI, magnetic resonance imaging.
Nonoperative Healing Rates Stratified by Anatomical Lesion and Coronal MRI Zone a

LFC, lateral femoral condyle; MFC, medial femoral condyle; MRI, magnetic resonance imaging.
Nonoperative Healing Rates Stratified by Anatomical Lesion Location, Sagittal MRI Zone, and Coronal MRI Zone a

LFC, lateral femoral condyle; MFC, medial femoral condyle; MRI, magnetic resonance imaging.
Discussion
In this study, we identified and validated several clinically relevant predictors of nonoperative treatment success while showing that machine learning models can predict which OCD lesions will heal with nonoperative treatment with superior accuracy. First, lesion location in the posterior aspect of the condyle on sagittal MRI and lesion location in the medial-most or lateral-most locations on coronal MRI were identified as statistically significant predictors of increased nonoperative treatment success on multivariate analysis, adding to inferences drawn from prior studies.5,11,16 This study also validated results reported by Wall et al, 19 indicating that increased lesion width relative to condyle width is an important predictor of a lesion’s healing potential. However, the presence of mechanical symptoms versus isolated pain was not found to be significantly associated with nonoperative healing, in contrast with previous reports. Regarding model performance, the models developed in this report represent a clinically meaningful improvement on previous efforts reported in the literature, with considerable improvements in sensitivity, specificity, NPV, and PPV. 13 Last, this study was the first to use definitive OCD healing defined as complete lesion healing on imaging and a full return to sports participation as determined by the patient’s provider—as the primary outcome. This outcome represents an improvement on previous studies, which evaluated progression toward healing at different follow-up time points. As a result, the models presented in this study can be used as clinically meaningful screening tools to determine whether a patient needs surgery after their baseline OCD visit.
Several risk factors that increase the likelihood of nonoperative treatment failure have been reported throughout the literature. First, De Smet et al 5 reported in 1997 that instability, larger lesion size, and closed growth plates predicted worse nonoperative healing outcomes on univariate analysis. Next, Pill et al 16 (2003) employed similar univariate analysis to confirm that unstable, larger lesions were less likely to heal nonoperatively while adding that lesions not in the weightbearing regions of the distal femur were more likely to heal. Given these findings, Wall et al 19 limited their 2008 study to stable, juvenile OCD lesions. They also determined that increased lesion length and width relative to the size of the femoral condyle, as well as the presence of mechanical symptoms (eg, giving-way, swelling, locking, or clicking) compared with isolated pain, was predictive of decreased progression toward healing on radiographs at 6 months on multivariate analysis adjusted for patient demographics. With a similar multivariate analysis, Krause et al 11 could not verify that increased lesion size or the presence of mechanical symptoms predicted reduced healing potential. However, their 2013 report added that the presence and increased size of cyst-like lesions were associated with reduced odds of nonoperative treatment success.
The findings of this study support the reported association of Wall et al 19 between increased normalized lesion width and an increased likelihood of failing nonoperative treatment. However, these findings fail to support Wall et al’s reported associations between normalized lesion length, mechanical symptoms, and patient outcomes. In addition, the present study represents an application of the Wall et al model to external data, indicating the generalizability of their model. Last, our study results support the finding of Pill et al 16 that lesions in nonweightbearing regions of the femoral condyle have increased healing potential. Including lesion location in the sagittal and coronal MRI planes, corresponding to the zones of reduced weightbearing as defined by Cahill and Berg, 3 is the crucial addition that allowed this study’s predictive models to have marked improvements in classification accuracy and AUC compared with previous efforts.3,4,16
Few evidence-based guidelines define which OCD lesions are amenable to nonoperative treatment. For example, Cahill et al 4 found that 50% of patients with OCD fail nonoperative treatment and need surgical intervention. Other reports have documented nonoperative healing rates between 49% and 100% at various follow-up time points.7-9,17,20 Without guidelines, each surgeon uses their clinical intuition and experience with OCD to decide whether to operate. Wall et al 19 and Krause et al 11 presented an evidence-based solution to this problem. However, their regressions, with validated AUCs of 0.66 and 0.60, respectively, would be considered worse than “acceptable” by current guidelines for model evaluation. 18 By contrast, the logistic regression model and SVMs developed by this study would be classified as “excellent.” These subjective descriptions are accompanied by objective differences in performance as measured by the Youden index: the model of Krause et al 11 was between 19.5% and 27% less sensitive and specific than the models produced in this study, and the model of Wall et al 19 was up to 22.5% and 25% less sensitive and specific. This specificity, sensitivity, NPV, and PPV improvements across most ROC thresholds imply that the models of the current study represent a superior prognostic tool for nonoperative healing success. However, these differences in model performance could be due to differences in the patient populations in each study or outcomes used for model development. For instance, Wall et al 19 and Krause et al 11 constructed their models with a different outcome than the one used in this study. While Wall et al and Krause et al predicted progression toward healing on imaging, this study predicted a full return to sports and complete healing. Similarly, the patients in each study could have variations in their baseline characteristics that cause different variables to predict nonoperative treatment success. Because of these possibilities, the models and risk factors presented in this study must be evaluated on new data to determine whether they are generalizable to all OCD patients.
Limitations
Despite its clinical relevance and potential benefit for patients with OCD and clinicians, this study has several notable limitations. First, the models presented were developed on ROCK cohort data alone. As a result, these patients were managed by subspecialist clinicians with specific research interests in OCD management, which may have jeopardized the ability of these predictive models to generalize to new OCD lesions in different practice scenarios. Despite these sources of bias that may harm model generalizability, we attempted to limit subjectivity by ensuring that every model input was objective and easily measurable with standard MRI equipment. Therefore, if the models presented in this study do not generalize well to new data, it can be inferred that the lack of generalization is likely due to unidentified individual-level differences between members of this study cohort and new patients. Another limitation of this study was its small sample size. Although this cohort represents the second-largest study of OCD patients treated with nonoperative management, it does not have enough data to identify statistically significant differences in the healing potential of OCD lesions based on their anatomical sites (eg, lateral femoral condyle, medial femoral condyle). Summary statistics in this report show that lesions of the lateral femoral condyle healed nonoperatively at a lower frequency than those at the trochlea or medial femoral condyle. However, these frequency differences were not statistically significant, as there were relatively small numbers of lesions in the lateral femoral condyle and trochlea.
Similarly, sample size constraints restricted this study’s ability to assess many covariates while producing valid model coefficients. As more data are inputted into the ROCK prospective cohort and more patients are added to the cohort on which these models can be trained, these predictive models will continue to improve. More covariates will be able to be reliably added to current models without jeopardizing accuracy or clinical inference. Another limitation of this analysis was that it could not directly construct the model developed by Krause et al, 11 which incorporated the presence and size of cyst-like lesions as covariates, as these data were unavailable for members of the ROCK cohort. However, their report states that their predictive models had similar capabilities as the model developed by Wall et al. 19 In the future, we intend to update the models presented continually, eventually testing their performance on new, prospective patient data. This performance will be compared with clinician decision-making to validate their performance externally. This external validation step and widespread model deployment will occur by making the models publicly available for use. This will allow for additional data collection, amplifying the number of records with lesions in lower-frequency anatomical locations, such as the trochlea and lateral femoral condyle. Eventually, models may be constructed for OCD sites other than the knee joint.
Conclusion
This study identified and validated several clinically relevant predictors of nonoperative treatment success while showing that machine learning models such as SVMs can predict which OCD lesions will heal with nonoperative management with superior accuracy compared with canonical models such as logistic regression. These models can be used as clinical screening tools to help determine whether an OCD lesion will heal with nonoperative management during the patient’s baseline visit.
Authors
Thomas Johnstone, BS (Stanford University School of Medicine, Stanford, California, USA; Department of Orthopaedic Surgery, Stanford University School of Medicine, Stanford, California, USA); Joseph Espiritu, BS (Stanford University School of Medicine, Stanford, California, USA); Marc Tompkins, MD (Department of Orthopaedic Surgery, University of Minnesota Medical School, Minneapolis, Minnesota, USA); Matthew D. Milewski, MD (Department of Orthopaedic Surgery, Boston Children’s Hospital, Boston, Massachusetts, USA); Carl Nissen, MD (Department of Orthopaedics, University of Connecticut School of Medicine, Farmington, Connecticut, USA); Kevin G. Shea, MD (Stanford University School of Medicine, Stanford, California, USA; Department of Orthopaedic Surgery, Stanford University School of Medicine, Stanford, California, USA); and the ROCK Group: Bradley Nelson, MD (Department of Orthopaedic Surgery, University of Minnesota, Minneapolis, Minnesota, USA); Anthony Egger, MD (Department of Orthopaedic Surgery, Children’s Healthcare of Atlanta, Atlanta, Georgia, USA); Christian Anderson, MD (Tennessee Orthopaedic Alliance, Tennessee, USA); James Lee Pace, MD (Department of Orthopaedic Surgery, Children’s Health Andrew’s Institute, Plano, Texas, USA); John Polousky, MD (Department of Orthopaedic Surgery, Akron Children’s Hospital Department of Orthopaedics, Akron, Ohio, USA); Jutta Ellemann, MD (Department of Orthopaedic Surgery, University of Minnesota, Minneapolis, Minnesota, USA); Norbert Meenen, MD (Orthopaedic Department, Pediatric Sports Medicine, Altona Children’s Hospital, University Medical Center Hamburg-Eppendorf, Bleickenallee, Hamburg, Germany); Eric Edmonds, MD (Department of Orthopaedic Surgery, Rady Children’s Hospital, San Diego, California, USA); Henry Ellis, MD (Department of Orthopaedic Surgery, Scottish Rite for Children, Frisco, Texas, USA); Peter Fabricant, MD (Department of Orthopaedic Surgery, Hospital for Special Surgery, New York, New York, USA); Aaron Krych, MD (Department of Orthopaedic Surgery, Mayo Clinic, Rochester, Minnesota, USA); Greg Myer, MD (Department of Orthopaedic Surgery, Cincinnati Children’s Hospital Medical Center, Cincinnati, Ohio, USA); Mininder Kocher, MD (Department of Orthopaedic Surgery, Boston Children’s Hospital, Boston, Massachusetts, USA); James Carey, MD (Department of Orthopaedic Surgery, Penn Medicine, Philadelphia, Pennsylvania, USA).
Footnotes
Correction (February 2025):
This article has been updated to correct the names of authors James Carey and James Lee Pace. A correction has been published.
Final revision submitted March 24, 2024; accepted April 16, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.T. has received hospitality payments from Aesculap Biologics. M.D.M. has received education payments from Kairos Surgical. K.G.S. has received education payments from Evolution Surgical and hospitality payments from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Pennsylvania (reference No. 820147).
