Abstract
Background:
It has been demonstrated that an athlete’s psychological readiness contributes to one’s ability to successfully return to sport (RTS) after anterior cruciate ligament (ACL) reconstruction. However, the effect of graft choice on psychological readiness is not yet understood.
Purpose/Hypothesis:
The purpose of this study was to evaluate the association between graft choice and an athlete’s psychological readiness to RTS. It was hypothesized that similar ACL-Return to Sport after Injury (ACL-RSI) scores would be achieved among patients who underwent ACL reconstruction with quadriceps autograft (QA), hamstring tendon autograft (HA), and bone–patellar tendon–bone (BTB) autograft.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Patients who underwent primary ACL reconstruction at a single institution between January 2017 and December 2018 were placed into separate cohorts depending on graft type (HA, BTB, or QA; n = 30 patients in each group), and the ACL-RSI, International Knee Documentation Committee subjective form, and Lysholm scores were compared between the different graft cohorts at 6 months postoperatively, at RTS, and at 2 years postoperatively. Also, the rate of patients who achieved an ACL-RSI score of ≥65 (predictive of return to preinjury sport at 2 years postoperatively) was calculated, as was the RTS rate and time.
Results:
The QA group demonstrated significantly higher ACL-RSI scores than both the HA and BTB groups at 6 months postoperatively (P < .0001) and RTS (P = .011). The QA group also had a higher rate of achieving ≥65 on ACL-RSI than the other groups at 6 months postoperatively (P = .002) and RTS (P = .024). There was no significant difference in the RTS rate between the 3 groups, although the QA group demonstrated a significantly quicker return (QA, 8.1 months; BTB, 9.6 months; HA, 10.5 months; P < .001).
Conclusion:
Athletes undergoing primary ACL reconstruction with QA achieved a higher mean ACL-RSI score and more often achieved a score of ≥65 at the 6-month and RTS time points compared with the HA and BTB groups. QA may afford an advantage over other grafts in terms of improved psychological readiness to RTS.
Keywords
Anterior cruciate ligament (ACL) rupture is a common and debilitating athletic knee injury.11,22 The common goal of managing athletes with ACL reconstruction is to afford a return to preinjury level of function. However, despite advances in technique and the ability to achieve satisfactory knee function after ACL reconstruction, the rate of return to preinjury level of sport among athletes remains low.18,20,31 Thus, considerable research has been focused on factors that contribute to an athlete’s successful return to sport (RTS) after ACL reconstruction. 45
An increasingly recognized factor that may contribute to an athlete’s RTS is one’s psychological readiness. 49 Injuries, particularly ACL tears, are known to take a significant psychological toll on athletes. 44 Consequently, an athlete’s mental readiness may play a role, among other factors, in an athlete’s safe RTS. It has been specifically demonstrated that a high mental readiness predicts successful RTS and that a low mental readiness is associated with reinjury after ACL reconstruction.3,15,32,37 The ACL-Return to Sport after Injury (ACL-RSI) scale was developed to measure an athlete’s psychological readiness to return to play.46,47 Many factors have been shown to influence an athlete’s ACL-RSI score, such as confidence in rehabilitation, length of injury, sex, age, and sport.15,17,27,32 However, there is limited understanding of the effect of graft choice on an athlete’s psychological readiness after ACL reconstruction.
The purpose of this study was to evaluate the association between graft choice and an athlete’s psychological readiness to RTS. We hypothesized that similar ACL-RSI scores would be achieved among patients who underwent ACL reconstruction with quadriceps autograft (QA), hamstring tendon autograft (HA), and bone–patellar tendon–bone autograft (BTB).
Methods
Study Design and Participants
A retrospective review was performed for varsity-level high school, collegiate, or professional athletes who underwent primary ACL reconstruction with or without meniscal surgery by a single sports fellowship–trained surgeon (K.J.E.) at a high-volume tertiary center between January 2017 and December 2018. Patients were included in the study if they were ≥14 years of age and had undergone reconstruction with all-soft tissue QA, BTB, or HA with ≥2 years of follow-up. Exclusion criteria were additional interventions outside of meniscal repair or debridement, multiligamentous injuries, extra-articular tenodesis, revision ACL ruptures, grade 3 to 4 chondromalacia, infection, and those without complete outcomes of interest. Over the study period, 90 athletes (30 athletes in each of the 3 autograft groups) were included for retrospective analysis after meeting inclusion and exclusion criteria.
Graft Selection
All 3 autograft options were utilized by the senior surgeon. The benefits and risks of these graft options were made clear to the patient and family, and graft decision was ultimately made by the patient and family.
Surgical Procedure
General anesthesia was used for all procedures with a femoral nerve block performed by a board-certified anesthesiologist before surgery. The patient was placed supine, and a torniquet was inflated to 250 mm Hg. Initial diagnostic arthroscopy was performed through standard anteromedial and anterolateral portals to confirm a complete tear of the ACL. The integrity of the ACL was evaluated and confirmed to be torn using an arthroscopic probe. Evaluation of the patellofemoral joint followed by medial and lateral compartments was then completed, and any meniscal pathology was addressed. Arthroscopic equipment was subsequently removed from the knee before graft harvesting.
Graft Passage and Fixation
For the QA, an all-inside technique was used. Passing sutures were placed in both the femoral and tibial sockets. The graft was then passed through the anteromedial portal and up into the femur, with the Tightrope (Arthrex) button deployed on the lateral cortex of the femur. The graft was then brought antegrade through the tibial socket using the previously placed tibial passing suture, and the Tightrope suture was pulled through the anterior cortex of the tibia. A 20-mm attachable button system (Arthrex) button was then attached to the Tightrope suture and deployed with the knee in full extension, stabilizing the graft in the sockets with suspension fixation on both the femoral and tibial sides. For the BTB and HA, the graft was brought into the joint through the tibial tunnel and then into the femoral socket. The Tightrope button was then deployed on the lateral cortex of the femur. With the knee in full extension, an interference screw was used with a diameter 1 mm less than the graft diameter to create interference fixation.
Postoperative Protocol
Patients were allowed to bear weight as tolerated in a hinged knee brace locked in extension with the use of crutches regardless of whether they had a meniscal repair. Crutches were weaned at 10 to 14 days, and each patient started physical therapy. The brace was unlocked at 2 weeks, and range of motion was initiated but limited to 0° to 90° of motion if meniscal repair was performed. At 6 weeks, the brace was discontinued altogether. Jogging in-line was initiated at 3 months with gradual increase to in-line sprinting. Once longitudinal sprinting was tolerated without issues, cutting and pivoting exercises were initiated. RTS was allowed based on appropriate progression through the rehabilitation program, quadriceps strength 90% that of the nonoperated leg, stability on Lachman and pivot-shift maneuvers, and triple-hop test 90% that of the nonoperative leg. The number and percentage of athletes who were able to RTS as well as RTS at the same level (defined as release to full sport by the senior surgeon) were collected.
Patient Characteristics and Clinical Outcomes
Patient characteristics including age, body mass index, sex, and level of play were collected. The participants were asked to complete the ACL-RSI, International Knee Documentation Committee (IKDC), and Lysholm surveys at 6 months postoperatively, the time of release to full RTS, and 2 years postoperatively. Regarding ACL-RSI, Sadeqi et al 37 reported that an ACL-RSI score of ≥65 was predictive of return to preinjury sport at 2 years postoperatively. Therefore, in addition to comparing the ACL-RSI raw scores between groups, the percentage of patients who reached an ACL-RSI score of ≥65 was also evaluated. Additional clinical outcomes evaluated included RTS at the same or higher level, time to RTS, and graft ruptures.
Statistical Analysis
All continuous variables were compared using an analysis of variance, whereas categorical variables were analyzed using the chi-square test. Statistical analysis was performed using Microsoft Excel. Differences between groups were considered statistically significant at P < .05.
A post hoc power analysis regarding ACL-RSI scores at 6 months postoperatively revealed a 100% power to detect a difference between the QA and HA cohorts and 98.4% power for the QA and BTB cohorts with alpha set to .05.
Results
The overall study cohort included 90 total patients, with 30 patients in each cohort. The overall mean age of the patients was 19.1 years. There was no significant difference between the 3 study groups with respect to collected patient characteristics (Table 1).
Characteristics According to Study Group a

Data are presented as mean ± SD or n (%). BMI, body mass index; BTB, bone–patellar tendon–bone; HA, hamstring tendon autograft; QA, quadriceps autograft.
RTS data are demonstrated in Table 2. The overall RTS rate did not significantly differ when compared between the QA group (86.7%), HA (76.7%), or BTB (83.3%) groups. Similarly, rate of return to the same level was not significantly different between the groups. Time to return was significantly different, with the QA returning a mean of 1.5 months before the BTB group and 2.4 months before the HA group (P < .001).
Return-to-Sport Outcomes a

Data are presented as mean ± SD or n (%). Boldface P value indicates statistically significant difference between groups (P < .05). BTB, bone–patellar tendon–bone; HA, hamstring tendon autograft; QA, quadriceps autograft.
In terms of psychological readiness, the QA group had significantly higher mean ACL-RSI scores than both the HA and BTB groups at 6 months postoperatively (QA, 65.3 ± 19.4; BTB, 46.7 ± 15.6; HA, 46.0 ± 4.22; P < .0001) and RTS (QA, 70.7 ± 17.5; BTB, 61.6 ± 7.8; HA, 60.6 ± 10.4; P = .011) (Table 3). Also, at 6 months postoperatively, the QA group had a significantly higher percentage of patients who reached the predictive threshold for RTS at 2 years postoperatively (ACL-RSI score of ≥65) (QA, 53.3%; HA, 23.3%; BTB, 13.3%; P = .002). At the RTS time point, the QA group again had a significantly higher percentage of patients who reached the ACL-RSI threshold score than the other 2 groups (QA, 69.2%; HA, 33.3%; BTB, 36%; P = .024).
Psychological Readiness Outcomes a

Data are presented as mean ± SD or n (%). Boldface P values indicate a statistically significant difference between groups (P < .05). ACL-RSI, Anterior Cruciate Ligament–Return to Sport after Injury; BTB, bone–patellar tendon–bone; HA, hamstring tendon autograft; QA, quadriceps autograft; RTS, return to sport.
Threshold reported by Sadeqi et al 37 as predictive of return to preinjury sport at 2 years postoperatively.
The remaining patient-reported outcomes are shown in Table 4. Although the BTB group demonstrated higher scores on average when compared with the HA cohort, and the QA demonstrated higher scores on average when compared with the other 2 cohorts, these differences were not statistically significant. There were 5 (17%) ruptures in the HT group, 3 (10%) in the BTB group, and 0 (0%) in the QT group at 2-year follow-up.
Patient-Reported Outcomes a
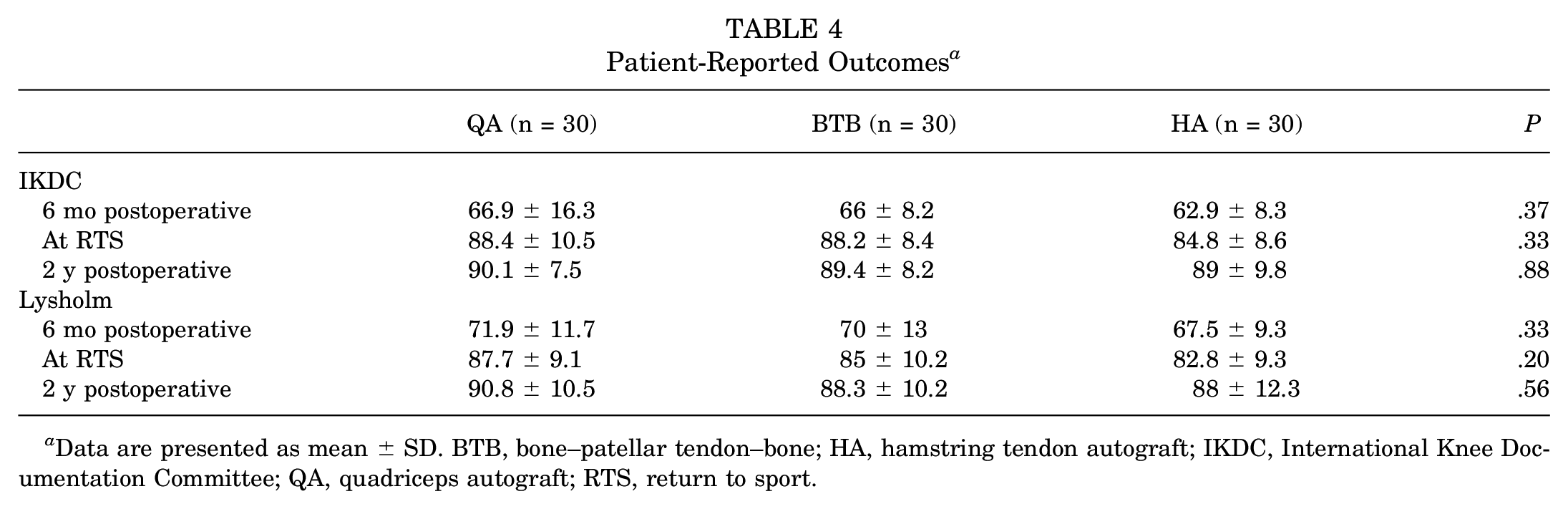
Data are presented as mean ± SD. BTB, bone–patellar tendon–bone; HA, hamstring tendon autograft; IKDC, International Knee Documentation Committee; QA, quadriceps autograft; RTS, return to sport.
Discussion
This study investigated the association between autograft choice and psychological readiness after ACL reconstruction. Our results showed that the QA group had superior ACL-RSI scores and a higher percentage of patients who scored ≥65 on the ACL-RSI at 6 months versus the BTB and HA groups. The QA cohort also demonstrated a quicker RTS when compared with the other groups. These results suggest that the QA may outperform the HA and BTB in terms of psychological readiness after ACL reconstruction, which may translate into a quicker RTS.
Despite abundant attention and research focused on optimizing ACL reconstruction, RTS after the injury remains far from guaranteed. Meta-analyses have demonstrated that return to preinjury level of sport after ACL reconstruction ranges between 63% and 83%.4,5,29 Accordingly, there is considerable interest in understanding the factors associated with successful RTS after ACL reconstruction. More recently, psychological readiness has emerged as an important factor when considering RTS. 3 A 2019 study evaluating 135 patients found that limb symmetry on single and crossover triple hop, subjective knee scores, and higher activity levels were associated with return to preinjury performance. 48 However, psychological readiness during rehabilitation was the only factor that remained significant in a multivariate model. In a separate series evaluating 795 patients who did not RTS after ACL reconstruction, Nwachukwu et al 33 reported that 64.7% of patients cited a psychological reason for not returning. The specific factors noted in this series included fear of reinjury, depression, lack of interest, and lack of motivation.
The ACL-RSI has specifically been evaluated in several studies. In 2018, a prospective series by Sadeqi et al 37 first reported the significant association between the ACL-RSI score and successful RTS. In a separate investigation, the ACL-RSI was shown to predict RTS better than functional tests at 9 months postoperatively. 15 In the context of our reported findings, the QA may afford an advantage for successful rehabilitation and eventual RTS, as it demonstrated superior ACL-RSI scores when compared with the BTB and HA cohorts. Further long-term studies will inform the implications of these differences. However, it remains possible that this difference may contribute to fewer retears in the QA population, as lower psychological readiness has been associated with higher risk of retears. 32
It remains speculative as to why the QA may afford improved psychological readiness when compared with other autograft options. In biomechanical studies, the QA has demonstrated more favorable structural properties, more collagen, and a cross-sectional area that is nearly twice that of the BTB.19,21,41 It has also shown greater stiffness when compared with the HA. 21 Together, these could contribute to the athlete’s sense of stability and subsequent confidence in his or her knee, or more effective progression through physical therapy. Further, while the BTB has historically been associated with postoperative knee pain as well as pain with kneeling, the QA has been found to have low rates of anterior knee pain, less donor-site morbidity, and improved cosmesis.1,6,28,40 It has also been associated with less pain and analgesic consumption in the postoperative period when compared with the HA. 10 These factors may also contribute to an athlete’s sense of “trusting” one’s knee and subsequently improved psychological readiness.
The association between self-reported knee function and RTS remains unclear. A 2014 systematic review found a poor correlation between functional variables and RTS. 13 However, separate studies have contrasted this and report a significant association between RTS and higher IKDC scores.4,30,50 It remains possible that psychological readiness may be an independent contributor that explains this discordance. This is supported by the lack of correlation between the ACL-RSI score and an athlete’s strength or power. 34 In our series, there was no significant difference noted in the IKDC or Lysholm scores. However, we did find that the RTS time was faster in the QA group by a mean of 1.5 months compared with the BTB cohort and 2.4 months with the HA group. Although few studies have formally compared RTS rates, the QA has shown quicker return than the BTB in the setting of revision ACL reconstruction. 9 Further investigations will assist in understanding the complex and multifactorial contributors to successful RTS and its timeline.
The optimal graft choice in ACL reconstruction remains under debate. The HA is an appealing option due to its low risk of patellar fracture, minimal anterior knee pain, and less osteoarthritis risk when compared with the BTB autograft.36,38,43 However, several drawbacks including weakness in hip extension and terminal knee flexion, graft creep, and tendon harvesting concerns represent potential disadvantages, particularly in the athlete.2,23,26,39 BTB affords bone-to-bone healing and has been shown to result in superior knee stability and less failure than HA.7,12 However, the BTB graft has been associated with significant and prolonged anterior knee pain, particularly with kneeling.25,35 Recently, the QA has increased in utilization with encouraging results in both the primary and the revision settings.8,9,14,16,42 There remain distinct disadvantages of the QA use in ACL reconstruction—namely, the deficit in quadriceps strength when compared with other graft options. 24 However, our data add to these early promising studies regarding use of the QA and suggest that the QA may offer superior psychological results as well.
Limitations
Several limitations should be considered. Surgeon bias with graft options may have been present, as this was not a randomized series. This makes the data prone to selection bias, although our comparison between the cohorts did not identify any significant demographic differences. A separate limitation is lack of collection of meniscal repair versus debridement data in this retrospective data set. Despite a post hoc power analysis that indicated adequate power, our sample size is relatively small and thus increases the likelihood of a type II error. All reconstructions were performed by a single surgeon. Despite the consistency, it remains possible that other surgeons or centers may experience different results. We also did not perform a comparison between patients with and without meniscal pathology, which may be a potential confounding variable. RTS is also a subjective endpoint and depends on surgeon decision making. Last, clinical examination and biomechanical data were not reported, and it remains unknown how these factors contribute to psychological readiness after ACL reconstruction.
Conclusion
Athletes who underwent primary ACL reconstruction with QA achieved a higher mean ACL-RSI score and more often achieved a score of ≥65 at the 6-month and RTS time points compared with those who underwent reconstruction with HA or BTB. QA may afford an advantage over other grafts in terms of improved psychological readiness to RTS.
Footnotes
Final revision submitted April 13, 2024; accepted April 23, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.C.B. has received education payments from ImpactOrtho and hospitality payments from Stryker and Medtronic. J.D.H. has received grant support from Arthrex and DJO; education payments from Smith & Nephew, Gemini Mountain Medical, ImpactOrtho, Medical Device Business Services, and Goode Surgical; and hospitality payments from Stryker, Zimmer Biomet, Avanos Medical, Heraeus Medical, and DePuy Synthes. S.V.T. has received education payments from ImpactOrtho and hospitality payments from Stryker and Avanos Medical. J.M.H. has received education payments from ImpactOrtho and hospitality payments from Stryker, Medtronic, and NuVasive. J.L.M. has received education payments from Arthrex, Smith & Nephew, ImpactOrtho, Davol, Stryker, Vericel, DePuy Synthes, and Goode Surgical. K.J.E. has received education payments from Goode Surgical; nonconsulting fees from Goode Surgical and Arthrex; and hospitality payments from Stryker, Zimmer Biomet, Smith & Nephew, Desert Mountain Medical, and Trice Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Western Institutional Review Board–Copernicus Group (reference No. 20216101).
