Abstract
Background:
The Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis (KOOS-PF) is a condition-specific patient-reported outcome measure that is used to evaluate patients with patellofemoral pain.
Purpose:
To translate the KOOS-PF into the Thai language, to make necessary adaptations to a Thai cultural context, and to assess the measurement properties of the Thai version of the KOOS-PF (TH-KOOS-PF) using the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) checklist.
Study Design:
Cohort study (diagnosis); Level of evidence, 3.
Methods:
This study was conducted between December 2021 and May 2023. Language translation of the KOOS-PF was performed according to guidelines for the cross-cultural adaptation of self-report measures. Eligible participants aged 18 to 50 years were asked to complete various patient-reported outcome measures including the TH-KOOS-PF at 3 time points: baseline, 2 weeks after baseline for test-retest reliability analysis, and 2 months after baseline for responsiveness assessment.
Results:
The KOOS-PF was successfully translated and culturally adapted to create the TH-KOOS-PF. A total of 86 participants (mean age, 37.0 ± 8.1 years; 59.3% female) with patellofemoral pain participated in the validation process. Construct validity of the TH-KOOS-PF was demonstrated by confirmation of all a priori hypotheses. The standardized Cronbach alpha coefficient was 0.88 (95% CI, 0.83-0.91), which indicated good internal consistency. Test-retest reliability was high, with an intraclass correlation coefficient of 0.87 (95% CI, 0.77-0.92). The TH-KOOS-PF demonstrated good responsiveness, as evidenced by all predefined hypotheses’ being confirmed, and there were no floor or ceiling effects. Using an anchor-based approach, the minimal clinically important difference was 11.6 points.
Conclusion:
The results of this study revealed the TH-KOOS-PF to be a valid, reliable, and responsive tool for assessing Thai patients with patellofemoral pain. The TH-KOOS-PF will be useful in both clinical and research settings.
Keywords
Patellofemoral pain is characterized by anterior knee pain that is aggravated by the patellofemoral joint-loading activities with weightbearing on a flexed knee, such as squatting, stair ambulation, running, and jumping.7,29 Patellofemoral pain is a common condition, with a reported annual prevalence of 22.7% in the general population. 26 Patellofemoral pain is also a presenting symptom of patellofemoral osteoarthritis. 14
Patient-reported outcome measures (PROMs), which describe a patient's perception of his or her condition without additional interpretation of the patient's response by clinicians or others, 20 are routinely used by clinicians and researchers to evaluate the clinical course of patellofemoral disorders and to assess treatment outcomes. Two condition-specific PROMs that have been suggested for reporting patellofemoral pain research are the Anterior Knee Pain Scale (AKPS) and the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis (KOOS-PF).3,6,13 The AKPS, which was developed by Kujala et al 13 in 1993, is widely used and has been translated into many languages, including Thai. 1 The KOOS-PF was developed by Crossley et al 6 in 2018 using the COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) checklist. A recent systematic review found the KOOS-PF to be the most appropriate PROM for measuring pain and function in adults with patellofemoral pain because it had the highest overall rating among all components of content validity, and it also demonstrated sufficient feasibility for clinical use. 9 The KOOS-PF has been translated into Arabic, 2 Spanish, 16 and Gujarati. 19
The aim of this study was to translate the KOOS-PF assessment tool into the Thai language, to make necessary adaptations to a Thai cultural context, and to assess the measurement properties of the Thai version of the KOOS-PF (TH-KOOS-PF) according to the COSMIN checklist. We hypothesized that the TH-KOOS-PF would be a valid, reliable, and responsive PROM for assessing Thai patients with patellofemoral pain.
Methods
This prospective cohort study was conducted at a single institution between December 2021 and May 2023. The study comprised the following 2 parts: (1) translation and cross-cultural adaptation of the KOOS-PF from the original English version into the Thai version and (2) evaluation of the validity, reliability, and responsiveness of the TH-KOOS-PF in young to middle-aged adults with patellofemoral pain. The study protocol received institutional review board approval, and written informed consent was obtained from all participating patients.
KOOS for Patellofemoral Pain and Osteoarthritis
The KOOS-PF is a condition-specific PROM that was developed to evaluate patients with patellofemoral-related symptoms. 6 The questionnaire consists of 11 questions that can be divided into the following 3 parts: stiffness, pain, and quality of life. Each question has 5 response options in a Likert scale format. The score can be transformed into a scale from 0 to 100, with a higher score indicating fewer symptoms and better function.
Translation and Cross-Cultural Adaptation
After receiving permission from the developers of the original version, 6 the KOOS-PF was translated into the Thai language following the guidelines for cross-cultural adaptation of health status measures. 4 Forward translation was independently performed by 2 translators—an orthopaedic surgeon (P.L.) and an uninformed professional translator. The 2 translations were compared, and differences were resolved in a consensus meeting to facilitate synthesis of a preliminary TH-KOOS-PF questionnaire. The preliminary TH-KOOS-PF questionnaire was then independently back-translated into English by 2 professional translators who were unaware of the study concepts and the original version of the KOOS-PF. An expert committee was then convened to consolidate all translated versions into a prefinal version of the TH-KOOS-PF.
The prefinal version of the TH-KOOS-PF questionnaire was then administered to 20 patients with patellofemoral pain to evaluate comprehensibility and to identify areas requiring further clarification. That evaluation of the questionnaire revealed no problems with patient understanding, so no additional changes to the questionnaire were required. The prefinal version of the TH-KOOS-PF was then adopted as the final version of the TH-KOOS-PF assessment tool.
Validation Process
The measurement properties of the TH-KOOS-PF were assessed by evaluating them against the recommendations in the COSMIN checklist. Patients aged 18 to 50 years with patellofemoral pain were eligible for inclusion. Patellofemoral pain was defined as pain around or behind the patella that was aggravated by ≥1 of the following activities: squatting, stair ambulation, running, and/or jumping. 7 Patients with generalized knee pain, pain in other joints or referred pain, insufficient understanding of the Thai language, and/or underlying neurological conditions that affected understanding the study questionnaires were excluded. All patient evaluations occurred in an outpatient setting.
Questionnaire Details and Scoring
In addition to the TH-KOOS-PF, the PROMs that were used in this study consisted of the Thai version of the AKPS, 1 the Thai version of the KOOS, 5 the Thai version of the European Quality of Life-5 Dimensions-5 Levels (EQ-5D-5L), 21 and the global rating of change (GROC) scale. 12
The AKPS is a condition-specific questionnaire that was developed to assess subjective symptoms and functional limitations in patients with patellofemoral disorders.1,13,24 It comprises 13 items with 3 to 5 response options. Each response has a different weighted score. The total score ranges from 0 to 100 with higher scores indicating fewer symptoms and better function.
The KOOS is used to evaluate various knee disorders and traumatic knee injuries.5,23 It consists of 42 items that are divided into the following 5 subscales: Pain, Symptoms, Function in Activities of Daily Living (ADL), Function in Sport and Recreation (Sport/Rec), and Quality of Life (QoL). Each item has 5 response options in a Likert scale format. Scores for each subscale can be transformed into a scale from 0 to 100, with higher scores representing fewer problems and better function.
The EQ-5D-5L is a health-related quality of life assessment tool that was developed by the EuroQol Group.21,22 The questionnaire evaluates the following 5 dimensions of health status: mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. The calculated utility score ranges from 0 to 1, with higher scores indicating better health-related quality of life.
The 7-level GROC scale was an anchor question for assessing change in a patient's overall perception of knee pain compared with the baseline evaluation. 12 The 7 possible response levels of the GROC scale are greatly worse (–3), somewhat worse (–2), slightly worse (–1), no change (0), slightly better (+1), somewhat better (+2), and greatly better (+3).
Questionnaire Administration
Participating patients were asked to complete the questionnaires at 3 different time points in this study: baseline, 2 weeks after baseline, and 2 months after baseline. At the baseline visit, patients were asked to complete the TH-KOOS-PF as well as the AKPS, KOOS, and EQ-5D-5L. The patients were then provided with stamped preaddressed envelopes to return the second and third sets of questionnaires.
At the 2-week time point, patients were asked to complete the TH-KOOS-PF and the GROC scale to evaluate test-retest reliability. A time interval of 2 weeks is often accepted as long enough to sufficiently prevent recall of previous responses.17,27
At the 2-month time point, patients were asked to complete the TH-KOOS-PF, AKPS, and KOOS, as well as the GROC scale, to evaluate responsiveness.
Construct Validity
The Spearman rank correlation coefficient (rS) was used to assess associations between the TH-KOOS-PF and the other PROMs, in which rS values were interpreted as negligible (0 to <0.3), low (0.3 to <0.5), moderate (0.5 to <0.7), strong (0.7 to <0.9), or very strong (0.9 to 1.0). 18 Construct validity of the TH-KOOS-PF was evaluated by testing the following a priori hypotheses specific to these associations:
Correlation of ≥0.7 in the same direction between the TH-KOOS-PF and the AKPS scores
Correlation of ≥0.7 in the same direction between the TH-KOOS-PF and the KOOS–Pain scores
Correlation of ≥0.5 in the same direction between the TH-KOOS-PF and the KOOS–Symptoms scores
Correlation of ≥0.5 in the same direction between the TH-KOOS-PF and the KOOS-ADL scores
Correlation of ≥0.7 in the same direction between the TH-KOOS-PF and the KOOS–Sport/Rec scores
Correlation of ≥0.5 in the same direction between the TH-KOOS-PF and the KOOS-QoL scores
Correlation of ≥0.5 in the same direction between the TH-KOOS-PF and the EQ-5D-5L utility scores
Good construct validity is defined as confirmation of ≥75% of the a priori hypotheses. 27
Reliability
Reliability of the TH-KOOS-PF was assessed for the following 3 parameters: internal consistency, test-retest reliability, and measurement error.
Internal Consistency
The Cronbach alpha was used to determine the homogeneity of the items in the TH-KOOS-PF questionnaire. A Cronbach alpha value within the range of 0.70 to 0.95 is generally considered to demonstrate good internal consistency. 27
Test-Retest Reliability
Test-retest reliability of the TH-KOOS-PF was assessed among a group of patients with stable condition, as evidenced by a GROC score of −1, 0, or +1 at the 2-week time point. The test-retest reliability of the TH-KOOS-PF was evaluated by calculating the intraclass correlation coefficient (ICC; 2-way random-effects model) with 95% CI. An ICC value of ≥0.70 is considered to indicate high reliability in a sample size of ≥50 patients. 27
Measurement Error
Measurement error was assessed by the standard error of measurement (SEM), which was calculated using the following formula: SD ×√(1 – ICC). The collective standard deviation obtained from among all patients at the baseline assessment was used for this calculation. The SEM was then converted to the smallest detectable change (SDC). Any observed changes that are smaller than the SDC are likely to be the measurement error. 17 The SDC at the individual level (SDCind) was calculated using the following formula: SEM × 1.96 ×√2. The SDC at the group level (SDCgrp) was calculated by dividing the SDCind value by √n.27,28 Bland-Altman analysis was used to evaluate agreement between the first (baseline) and second (2-week time point) TH-KOOS-PF scores among patients with stable condition.
Responsiveness
Responsiveness of the TH-KOOS-PF was assessed by evaluating the Spearman rank correlations of the following a priori hypotheses between the TH-KOOS-PF change score and the change scores of the other PROMs.
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the AKPS score
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the KOOS–Pain score
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the KOOS–Symptoms score
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the KOOS-ADL score
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the KOOS–Sport/Rec score
Correlation of ≥0.5 in the same direction between the change in the TH-KOOS-PF score and the change in the KOOS-QoL score
Good instrument responsiveness is defined as confirmation of ≥75% of the a priori hypotheses. 27
Responsiveness of the TH-KOOS-PF was also evaluated via a criterion approach. The GROC scale at 2 months after the initial assessment was used as the criterion measure of change to create 2 dichotomized groups (improved vs unimproved). The improved group consisted of patients who reported their change to be greatly or somewhat better, whereas the unimproved group included patients who reported their change to be slightly better or less.8,11 The ability of the TH-KOOS-PF change score to distinguish between an improved and an unimproved patient was evaluated by generating a receiver operating characteristic (ROC) curve with the sensitivity and specificity of each possible change in score. The area under the ROC curve (AUC) demonstrated the ability of the TH-KOOS-PF to correctly classify a patient as either improved or unimproved according to an external criterion. An AUC of >0.70 was considered to be acceptable. 27
Interpretability
Interpretability is defined as the ability of a PROM to be translated from quantitative scores or a change in scores to a qualitative meaning.17,27 Although interpretability is not considered a measurement property, it is an important characteristic of a PROM. 17 Interpretability of the TH-KOOS-PF was demonstrated in the following 2 aspects: floor and ceiling effects and minimal clinically important difference (MCID).
Floor and ceiling effects of a PROM were considered to be present when >15% of respondents had the lowest or highest possible score, respectively.17,27 The presence of floor or ceiling effect may adversely influence content validity, reliability, and responsiveness of a PROM. 27
The MCID or the optimal change score cutoff value to distinguish between the improved and unimproved groups was determined by analyzing the ROC curve from the responsiveness testing. Using an anchor-based approach, the MCID is the change score value that corresponds with the point closest to the left-top corner of the curve. 30 This value is a change score that yields the lowest likelihood of misclassification.
Statistical Analysis
Descriptive statistics were used to summarize patient demographic and clinical characteristics. Categorical data are presented as frequency and percentage. Normally and nonnormally distributed continuous data are reported as mean with standard deviation and median with range, respectively. A Bland-Altman plot was generated using MedCalc for Windows software Version 16.4.3 (MedCalc Software). The AUC from the ROC curve was calculated using R Version 4.3.1 (R Project for Statistical Computing). Other statistical analyses were performed using PASW Statistics for Windows Version 18.0 (SPSS, Inc). P < .05 was considered to indicate statistical significance for all tests.
Results
Translation and Cross-Cultural Adaptation
The KOOS-PF was successfully translated from the original English-language version into the Thai-language version. None of the items or responses in the KOOS-PF required cross-cultural adaptation in this study. The final version of the TH-KOOS-PF is available separately as Supplemental Material.
Patient Characteristics
Between December 2021 and May 2023, a total of 86 patients with patellofemoral pain were enrolled in this study. Demographic and clinical characteristics of the study patients are presented in Table 1. The mean age of patients was 37.0 ± 8.1 years, and 59.3% were female. The mean TH-KOOS-PF score was 47.0 ± 16.6 at baseline. All 86 patients completed and submitted the second set of study questionnaires at the 2-week time point, and 82 patients (95.3%) completed and submitted the third set of study questionnaires at the 2-month time point.
Patient Demographic and Clinical Characteristics (N = 86) a

Data are presented as mean ± SD or n (%).
Construct Validity
The Spearman rank correlation coefficients for the hypothesized associations between the TH-KOOS-PF and the AKPS, KOOS, and EQ-5D-5L are shown in Table 2. All of the a priori hypotheses were confirmed, which demonstrated good construct validity of the TH-KOOS-PF.
Correlations for the A Priori Hypotheses Concerning the TH-KOOS-PF Score and the AKPS, KOOS, and EQ-5D-5L Scores a

ADL, Activities of Daily Living; AKPS, Anterior Knee Pain Scale; EQ-5D-5L, European Quality of Life–5 Dimensions–5 Levels; KOOS, Knee injury and Osteoarthritis Outcome Score; QoL, Quality of Life; Sport/Rec, Sport and Recreation; TH-KOOS-PF, Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis.
Reliability
Internal consistency for the TH-KOOS-PF was good, with a standardized Cronbach alpha coefficient of 0.88 (95% CI, 0.83-0.91). A total of 61 patients with stable condition who reported a GROC scale score of −1, 0, or +1 at 2 weeks after the baseline assessment were included in the test-retest reliability analysis. Results of the test-retest reliability analysis for the TH-KOOS-PF assessment tool are shown in Table 3. The ICC value for the TH-KOOS-PF was 0.87 (95% CI, 0.77-0.92), which indicated high test-retest reliability. The SEM was 3.9 points. The SDCind and SDCgrp were 10.9 and 1.4 points, respectively. A Bland-Altman plot showing the difference between the first and second measurements of the TH-KOOS-PF is presented in Figure 1.
Test-Retest Reliability of the TH-KOOS-PF a

In total, 61 patients with stable condition who reported a global rating of change scale score of −1, 0, or +1 at 2 weeks after the initial assessment were included in the test-retest reliability analysis. ICC, intraclass correlation coefficient; MD, mean difference; SDCgrp, smallest detectable change at the group level; SDCind, smallest detectable change at the individual level; SEM, standard error of measurement; TH-KOOS-PF, Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis.

Bland-Altman plot to evaluate the test-retest reliability of the Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis (TH-KOOS-PF). The first measurement occurred at baseline, and the second measurement occurred at the 2-week time point.
Responsiveness
The scores of the TH-KOOS-PF and other questionnaires at baseline and 2 months are presented in Table 4. Spearman rank correlation coefficients for the hypothesized associations between the change score of the TH-KOOS-PF and the change scores of the AKPS and KOOS are presented in Table 5. All of the a priori hypotheses were confirmed, which demonstrated good responsiveness of the TH-KOOS-PF.
Mean Baseline and 2-Month PROM Scores and the Mean PROM Change Scores Between Baseline and 2 Months a

ADL, Activities of Daily Living; AKPS, Anterior Knee Pain Scale; KOOS, Knee injury and Osteoarthritis Outcome Score; PROM, patient-reported outcome measure; QoL, Quality of Life; Sport/Rec, Sport and Recreation; TH-KOOS-PF, Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis.
Correlations for the A Priori Hypotheses Concerning the TH-KOOS-PF Change Score and the Change Scores for the AKPS and KOOS a

ADL, Activities of Daily Living; AKPS, Anterior Knee Pain Scale; KOOS, Knee injury and Osteoarthritis Outcome Score; QoL, Quality of Life; Sport/Rec, Sport and Recreation; TH-KOOS-PF, Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis.
Mean baseline and 2-month TH-KOOS-PF scores and the TH-KOOS-PF change score relative to the 7 levels of the GROC scale are shown in Table 6. Patients in the improved group had a mean TH-KOOS-PF change score of 18.8 points, which was significantly higher than the mean TH-KOOS-PF change score in the unimproved group (P < .001). The ROC curve to demonstrate the sensitivity and specificity of the TH-KOOS-PF change score for distinguishing improved from unimproved patients is presented in Figure 2. The AUC was 0.73 (95% CI, 0.62-0.85), which was considered acceptable (>0.70).
Mean Baseline and 2-Month TH-KOOS-PF Scores and the TH-KOOS-PF Change Score Relative to the GROC Scale a

GROC, global rating of change; TH-KOOS-PF, Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis. Dashes indicate no patients reporting this level.
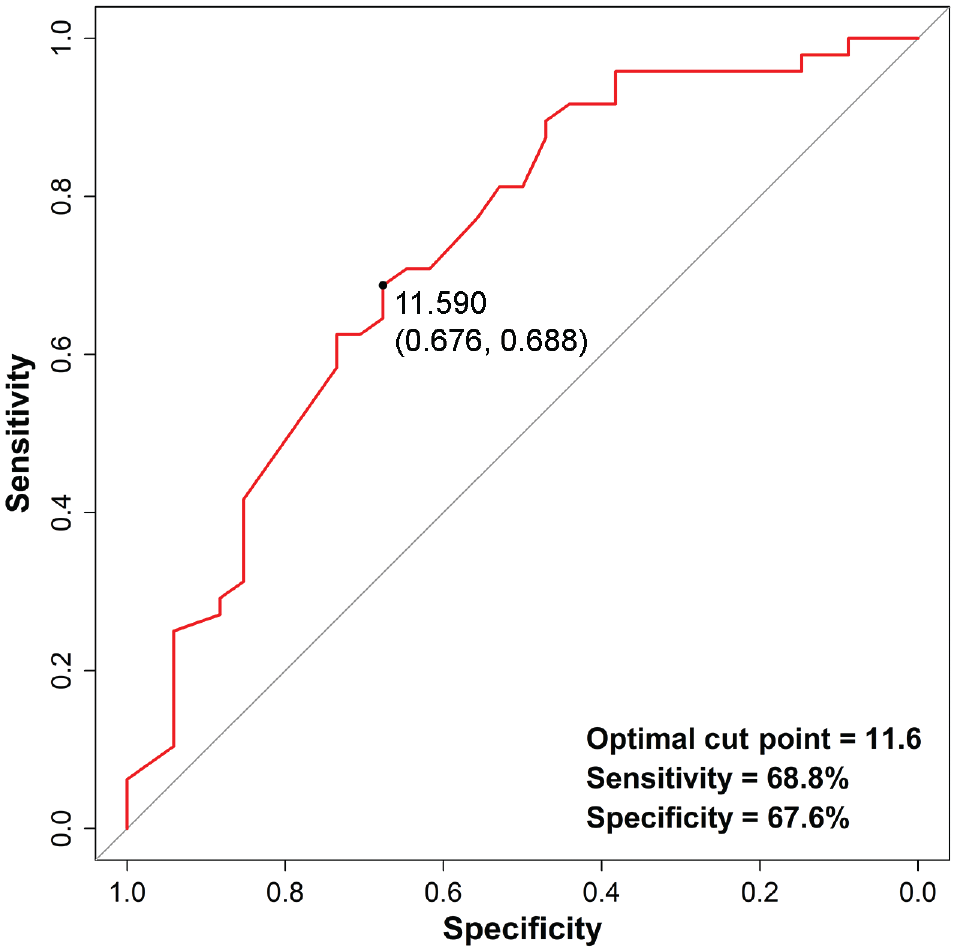
Receiver operating characteristic curve to demonstrate the sensitivity and specificity of the Thai version of the Knee injury and Osteoarthritis Outcome Score for patellofemoral pain and osteoarthritis (TH-KOOS-PF) for distinguishing improved from unimproved patients. The area under the curve was 0.73 (95% CI, 0.62-0.85), which was considered acceptable. Using an anchor-based approach, the minimal clinically important difference for the TH-KOOS-PF was a change score of 11.6 points, with a sensitivity of 68.8% and a specificity of 67.6%.
Interpretability
There were no patients who scored the lowest or highest possible score in this study, which indicates that the TH-KOOS-PF assessment tool has no floor or ceiling effects. Using an anchor-based approach, the MCID for the TH-KOOS-PF was a change score of 11.6 points with a sensitivity of 68.8% and a specificity of 67.6% (Figure 2).
Discussion
In this study, the KOOS-PF assessment tool was successfully translated into Thai after following the international guidelines for cross-cultural adaptation of health status measures. The measurement properties of the TH-KOOS-PF were assessed by evaluating them against the COSMIN checklist. The TH-KOOS-PF was shown to be valid, reliable, and responsive for assessing young to middle-aged Thai patients with patellofemoral pain.
The International Patellofemoral Research Network recently published standards for the REPORTing of quantitative PatelloFemoral Pain (REPORT-PFP) research. 3 The REPORT-PFP checklist suggests the AKPS or KOOS-PF assessment measures as condition-specific PROMs that should be used evaluate patients with patellofemoral pain.
The AKPS is an established PROM that has been and continues to be widely used to assess patients with patellofemoral disorders. The AKPS was published in 1993, and the COSMIN checklist was published in 2010.13,17 Despite its recognized good measurement properties, limitations of the AKPS include (1) unspecified duration of symptoms; (2) terminology that some patients find difficult to understand, such as “subluxation,”“atrophy,” and “flexion deficiency”; (3) arbitrary weighting for response options; and (4) concerns about its content validity.6,10
The KOOS-PF assessment measure was developed according to the COSMIN checklist to ensure the efficacy of its measurement properties. The KOOS-PF demonstrated good content validity because initial item generation included input from 50 patients and 14 health care professionals. 6 The KOOS-PF was also found to be sufficiently feasible for use in both clinical and research settings. The KOOS-PF asks patients to evaluate their knee symptoms over the past week. It requires only 5 minutes to complete, and it is easy to calculate the patient's score. Moreover, it is available at no cost. 9
The TH-KOOS-PF was developed using the guidelines for the cross-cultural adaptation process of health status measures. 4 For translation into Thai language, no terms or items in the original questionnaire required cross-cultural adaptation. In the Arabic language version of the KOOS-PF, which was validated among Saudi Arabians, the authors described cross-cultural adaptation of the questionnaire by integrating prayer or religious-cultural activity into 8 of 11 items. 2
The construct validity of the TH-KOOS-PF was also confirmed to be good because all a priori hypotheses were confirmed. The TH-KOOS-PF score was found to strongly correlate with the AKPS, KOOS–Pain, and KOOS–Sport/Rec (rS≥0.7 for all). A strong correlation between the KOOS-PF and the AKPS was also demonstrated in a validation study of the Spanish version of the KOOS-PF. 16
The TH-KOOS-PF showed good internal consistency, with a Cronbach alpha of 0.88. Cronbach alpha values of the original and previously translated versions ranged from 0.81 to 0.93, except for the Gujarati version, which had a low Cronbach alpha of 0.12.2,6,16,19 The TH-KOOS-PF also demonstrated high test-retest reliability with an ICC of 0.87, which is comparable with the original version (ICC, 0.86). 6 A 2-week interval was reported to be a sufficiently long duration to prevent recall when assessing test-retest reliability.17,27 Moreover, we used the GROC scale to identify patients with stable clinical condition to analyze test-retest reliability.
Responsiveness of the TH-KOOS-PF was supported by confirmation of all a priori hypothesized correlations between the change score of the TH-KOOS-PF and those of the AKPS and the KOOS subscales. The strongest correlation was for the association between the change scores of the TH-KOOS-PF and the KOOS–Sport/Rec (rS = 0.70; 95% CI, 0.57-0.80). ROC curve analysis revealed an AUC value of 0.73, which indicates an acceptable ability of the TH-KOOS-PF change score to discriminate between improved and unimproved patients.
The TH-KOOS-PF had no floor or ceiling effects, which is similar to the original and other translated versions.2,6,16,19 Using an anchor-based approach, the MCID of the TH-KOOS-PF was 11.6 points. The MCID value is larger than the SDC value, which indicates the clinical importance of a change score on an individual level and confirms that the change does not reflect measurement error. 27 For presenting clinical outcomes in research, the MCID can be used to calculate the percentage of responders in each treatment group who report a change greater than the MCID value. 25
Limitations
Despite the successful development of the TH-KOOS-PF, this study has some limitations that should be mentioned. All patients in this study received nonoperative treatment for their symptoms, but no specific treatment protocol was applied. This could have influenced both clinical change in our study patients and the identified MCID value. However, the lack of a specific treatment protocol is similar to the current lack of an established protocol for treating patients with patellofemoral pain in routine clinical practice. In response, physicians usually combine different treatment options for individualized patient care. Last, using a single anchor question for MCID calculation might have increased susceptibility to recall bias, as patients may have inaccurately remembered their baseline status, and the responses could have been affected by their current status. 15
Conclusion
The results of this study revealed the TH-KOOS-PF to be a valid, reliable, and responsive tool for assessing Thai patients with patellofemoral pain. The TH-KOOS-PF will be useful in both clinical and research settings.
Supplemental Material
sj-pdf-1-ojs-10.1177_23259671241279465 – Supplemental material for Cross-Cultural Adaptation and Validation of the Thai Version of the Knee Injury and Osteoarthritis Outcome Score for Patellofemoral Pain and Osteoarthritis (KOOS-PF)
Supplemental material, sj-pdf-1-ojs-10.1177_23259671241279465 for Cross-Cultural Adaptation and Validation of the Thai Version of the Knee Injury and Osteoarthritis Outcome Score for Patellofemoral Pain and Osteoarthritis (KOOS-PF) by Kongpob Reosanguanwong, Wasaphon Suphakitchanusan and Pisit Lertwanich in Orthopaedic Journal of Sports Medicine
Footnotes
Acknowledgements
The authors gratefully acknowledge the original authors of the KOOS-PF for granting permission to translate the English version into Thai. The authors also thank Suchitphon Chanchoo, Wacharapol Tepa, and Narumol Sudjai for their assistance with data collection and statistical analysis.
Final revision submitted February 18, 2024; accepted April 3, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was funded by a research grant (R016531016) from the Faculty of Medicine Siriraj Hospital, Mahidol University. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Medicine Siriraj Hospital, Mahidol University (ref No. 749/2021).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
