Abstract
Background:
The rate of failed anterior cruciate ligament reconstruction (ACLR) remains high in the younger and more active patient populations. Suture tape augmentation (STA) in addition to ACLR may reduce the risk for revision surgery.
Purpose/Hypothesis:
The purpose of this study was to compare patient outcomes between patients who underwent primary all-inside quadrupled semitendinosus hamstring tendon autograft (QST-HTA) ACLR with and without STA. It was hypothesized that the STA cohort would demonstrate a lower incidence of subsequent revision ACLR while maintaining comparable patient-reported outcomes.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
All patients ≤40 years of age who received primary all-inside QST-HTA ACLR with and without independent STA augmentation were identified. The following validated patient-reported outcome measures (PROMs) were collected: visual analog scale for pain, Single Assessment Numeric Evaluation, Knee injury and Osteoarthritis Outcome Score subscales, and Tegner activity scale. KT-1000 arthrometer measurements were collected pre- and postoperatively. Cox proportional hazards model and nominal logistic regression analysis were used to assess additional variables associated with revision ACLR.
Results:
A total of 104 patients with a mean age of <22 years were included in the final data analysis (STA: 36 patients; control: 68 patients). Significantly fewer patients in the STA group sustained a graft failure necessitating revision surgery at the final follow-up (5.6% vs 24%; relative risk, 0.24 [95% CI, 0.06-0.97]; P = .017). Four-year graft survival was significantly higher in the STA group (97.2% vs 82.4%; P = .031). All PROMs significantly improved postoperatively except for Tegner levels, which decreased in both groups compared with their preinjury levels (P < .001). Return to sports was similar in both groups with >70% of patients returning to their previous level of competition. Regression analysis demonstrated increased risk for revision ACLR in younger patients, high school athletes, and those with higher postoperative activity levels.
Conclusion:
QST-HTA ACLR with STA was associated with reduced risk for revision ACLR compared with nonaugmented QST-HTA ACLR in this young patient population. Furthermore, the addition of suture tape did not appear to affect postoperative patient-reported and return-to-sports outcomes.
The anterior cruciate ligament (ACL) is the most common knee ligament to undergo surgical intervention. 25 Additionally, the incidence of ACL tears has increased in recent years as participation in high-risk pivoting sports (soccer, basketball, football, etc) has increased.7,17 Despite technological advancements in ACL reconstruction (ACLR), the incidence of subsequent graft failure remains high in the younger and more active patient population—ranging from 6% to as high as 39%—with a majority occurring within the first 2 years.12,23,32,39,40,51,53 There are many variables associated with subsequent graft failure, including patient sex, higher activity levels and participation in high-risk pivoting sports, increased tibial slope, joint hyperlaxity, and graft choice.6,28,49
The hamstring tendon autograft (HTA) is one of the most popular graft choices for ACLR worldwide.26,43 However, compared with the bone–patellar tendon–bone (BPTB) autograft and quadriceps tendon autograft (QTA), the HTA has been shown to have an increased risk for subsequent graft failure, and ultimately revision ACLR, 49 with failure rate exceeding 17% at postoperative year 2 and beyond.12,16,24,39,40 Additional ACLR augmentation techniques—both extra-articular and intra-articular—have been proposed and implemented to decrease this risk of subsequent revision ACLR. ‡
Suture tape augmentation (STA) is an intra-articular technique that may decrease the risk of graft failure.10,11,24,30,35 The biomechanical benefit of independent STA augmentation is well documented, as is the intra-articular safety of the additional suture material evidenced by translational animal studies.1,2,45,50,52 Recent biomechanical studies have demonstrated the potential benefit of load sharing when grafts were augmented with independent STA compared with nonaugmented grafts.1,2 However, the 2 studies comparing HTA ACLR with and without independent STA were unable to conclude that STA resulted in a significantly lower risk for revision ACLR at 2 years, although the graft failure rate trended in favor of STA in both studies.23,34
The purpose of this study was to compare patient outcomes between patients who underwent primary all-inside quadrupled semitendinosus HTA (QST-HTA) ACLR with and without STA. We hypothesized that the STA cohort would demonstrate a lower incidence of subsequent revision ACLR while maintaining comparable patient-reported outcomes.
Methods
Study Design and Patient Population
Institutional review board approval was obtained before commencement of this study. This was a single-center, retrospective cohort study examining all available skeletally mature patients ≤40 years of age who received primary QST-HTA ACLR with and without independent STA between 2011 and 2020 by the senior author (P.A.S.). The senior author has been performing this technique exclusively for QST-HTA ACLRs since 2009. Patients were excluded until 2011, though, to allow for adequate initial follow-up to ensure the efficacy of this new technique. All skeletally mature patients who received ACLR with QST-HTA after July 12, 2016, received independent STA, and those who received the same procedure before this date did not receive such augmentation. Furthermore, there were no special criteria for those who received STA and those who did not because the senior author has augmented all grafts (BPTB autograft, QTA, QST-HTA, and allografts) since July 2016 except for in 3 patients who had a previous BPTB autograft and opted not to have the augmentation done to match their other side. Those with a minimum 2-year follow-up were included in this study. Those who received revision QST-HTA ACLR, were >40 years of age, had a history of prior surgery on the ipsilateral knee, had concomitant extra-articular stabilization (ie, anterolateral ligament [ALL] reconstruction and lateral extra-articular tenodesis [LET]), underwent remnant-preserving single-bundle ACLR, underwent primary QST-HTA ACLR with STA where the suture tape was not independent from the graft, and were lost to follow-up were not included in the final data analysis.
Data Collection
After identification of all eligible patients, the following pre- and postoperative patient-reported outcome measures (PROMs) were collected using our institution's registry: visual analog scale, 20 Single Assessment Numeric Evaluation, 42 Knee injury and Osteoarthritis Outcome Score subscales, 36 and Tegner activity levels (Briggs). 8
Operative details were collected from operative notes, which included the date of surgery, concomitant procedures, and graft details. The need for subsequent surgical intervention on the ipsilateral knee or the need for contralateral ACL surgery was obtained from the patients’ electronic medical records, through telephone calls, or through office visits. Patients were deemed lost to follow-up after 3 consecutive failed attempts to reach them by either telephone or email. Patients received KT-1000 arthrometer (MEDmetric) testing preoperatively and at their 1-year follow-up by qualified research personnel who were blinded to pertinent physical examination findings. The side-to-side 30-lb (13.6-kg) and manual maximum KT-1000 arthrometer differences were recorded. The posterior tibial slope (PTS) was measured on the preoperative lateral radiograph using the posterior tibial cortex as a reference. 21 For those patients who sustained their injury during a sporting activity, return-to-sports (RTS) details were documented.
Surgical Technique
All patients received a primary arthroscopic all-inside QST-HTA ACLR utilizing a QST tendon. This graft was harvested through a small popliteal incision.33,47,52 The construct was prepared utilizing suspensory adjustable loop device (ALD) fixation (TightRope RT and TightRope Attachable Button System; Arthrex) both proximally and distally as previously described.28,46 The graft was passed through the 2 loops twice, and the 2 free ends were sutured together with No. 0 suture (FiberWire; Arthrex). Under 75 N of tension, the 4 limbs of the graft were sutured together on each side of the ALD loops proximally and distally with 2 No. 0 wraparound sutures (FiberWire). Based on intra-articular measurements, marks were made on the graft corresponding with the expected femoral and tibial socket depths, as well as the intra-articular ACL graft length measured after creating both sockets. Graft lengths were between 62 and 66 mm with a minimum length in the femoral socket of 20 mm with the intra-articular measurement between 20 and 22 mm and a minimum length in the tibia of 22 mm.
Suture tape (FiberTape; Arthrex) was used for the patients who received independent STA to internally augment the construct (InternalBrace; Arthrex). 40 In all cases, the suture tape was passed through the femoral suspensory button to remain independent of the graft (Figure 1).
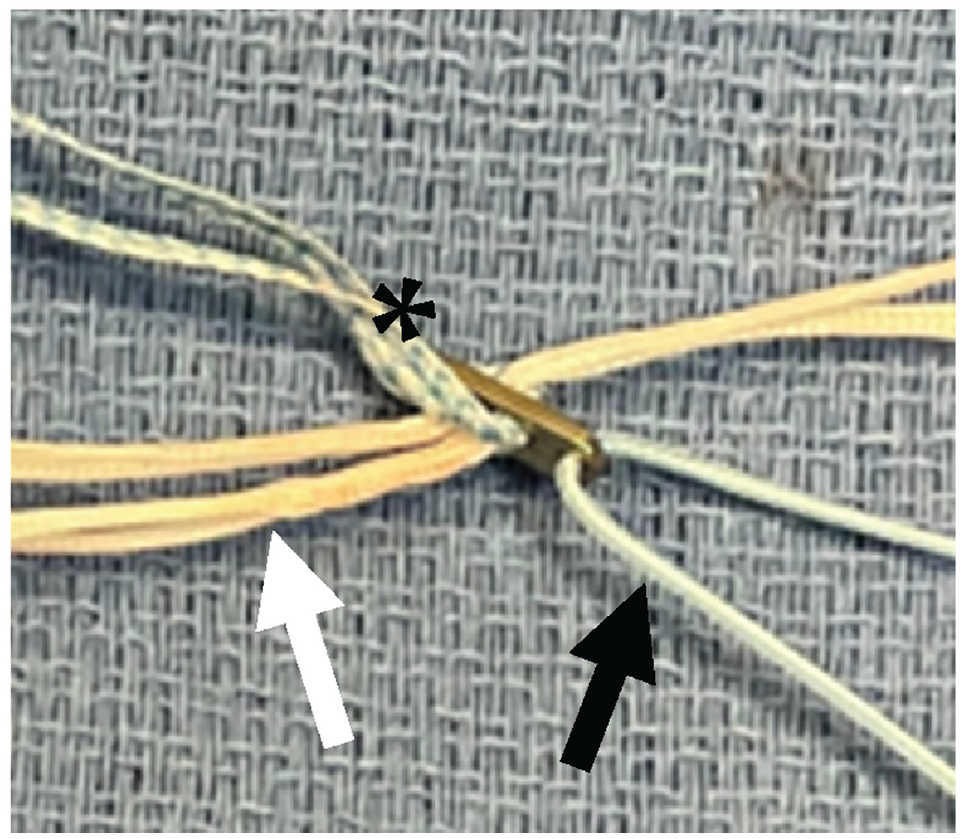
Overhead view of the femoral adjustable loop device demonstrating the suture tape (asterisk) looped through the femoral button to remain separate from the adjustable loop device (white arrow) through which the graft was passed. Black arrow, blue passing suture.
In all cases, the femoral socket was created through anteromedial portal drilling. First, with the knee at 120°, a measuring pin was drilled at the anteromedial bundle footprint on the femur until the outer cortex was breached, and this femoral intraosseous distance was measured off the pin. A low-profile reamer was used to ream to a depth of approximately 25 mm to ensure at least 20 mm of graft would be in the femoral socket, with 3 to 4 mm available for retensioning. For the tibial socket, a tibial aiming device was positioned within the native ACL footprint fibers just adjacent to the attachment of the anterior horn of the lateral meniscus. A guide sleeve was placed through a small tibial incision down to bone, a FlipCutter (Arthrex) was drilled into the footprint and opened to the appropriate graft diameter, and the tibial socket was retrocut to a depth of 28 to 30 mm based on graft length, leaving 5 to 6 mm of freedom to prevent the graft from “bottoming out” during initial graft placement and retensioning. Importantly, the intra-articular distance was measured with a special device (Arthrex) in all cases and typically was 20 to 22 mm. So, a simple calculation was done to be sure the depth of the femoral socket + the intra-articular distance + the tibial socket depth was 7 to 8 mm greater than the graft length to avoid bottoming out and a resultant lax graft. In no case did a graft bottom out.
The graft was first fixed on the femoral side with the ALD. It was then passed into the tibial socket with the ALD and a fixation button (Arthrex) was applied. For the control group, the tibial graft was then fixed via the ALD with the knee in full hyperextension. The knee was then cycled through a full range of motion (ROM) 15 to 20 times, and ALD retensioning was done on the femoral side and then on the tibial side, with the knee always held in hyperextension. For the STA cohort, the tape was first fixed distally with a bioabsorbable anchor (SwiveLock; Arthrex) with the knee fully hyperextended, 40 and then the graft was tensioned on the tibial side via the ALD with the knee in full hyperextension. As with the control group, the knee was cycled followed by graft retensioning on both the femoral side and the tibial side via the corresponding ALD with the knee hyperextended.
Postoperative Protocol
All patients underwent an accelerated rehabilitation protocol. Each patient received a continuous passive motion machine (KinexCONNECT; Kinex Medical Company) to be used at home for the first 2 weeks after surgery. Immediate partial weightbearing was allowed after surgery as tolerated while in the long-leg brace and on crutches if meniscal repair was not performed. If meniscus root or radial repair was performed, then the patient was to be nonweightbearing for the first 4 to 6 weeks after surgery to allow for adequate protection of the meniscal repair. Full weightbearing was typically achieved by postoperative week 2, when patients demonstrated adequate leg control, or immediately after the conclusion of the 4- to 6-week nonweightbearing period for those who underwent concomitant meniscal repair. Cycling was started at 2 weeks with progression to closed-chain exercises once patients achieved full weightbearing status. Jogging in place in a functional ACL brace was initiated during postoperative weeks 8 to 11 dependent on return of full ROM (0° to ≥130°), as well as adequate proprioception/balance as assessed by the physical therapist (PT). Jogging on the treadmill in the functional ACL brace was allowed by postoperative weeks 11 to 15 as recommended by the PT after progression through lunges/multidirectional steps (with sports cord resistance) and isotonic exercises. Initial functional testing was performed in postoperative week 16. Agility exercises were initiated in postoperative month 5. Leg strength was assessed clinically using a dynamometer, Y-balance testing, jump-landing posture, and hop testing by the treating PT. Release to RTS depended on passage of functional testing by the patient's PT and an office visit confirming full ROM with good objective ACL stability and good clinical muscle strength. Six months was the earliest release time to RTS, but commonly it was closer to 8 to 9 months. Patients were routinely seen in the clinic at 2 weeks, 12 weeks, 6 months, and 1 year after their surgery. KT-1000 arthrometer testing was performed in all patients at their 1-year office visit by qualified research personnel with ≥2 years of experience.
Statistical Analysis
The Shapiro-Wilk test was used to assess the normality of the distribution for all continuous variables. Based on the normality of the distribution, either the Student t test or Mann-Whitney U test was used to assess continuous variables, which are represented as either mean with 95% confidence interval or median with interquartile range. Either the chi-square test or Fisher exact test was used to assess categorical variables, which are reported as number with percentage. Cox proportional hazards model and nominal logistic regression analysis were used to assess if additional variables were associated with revision ACLR. Odds ratio (OR) and relative risk (RR) are presented with the 95% confidence interval (Wald test). The 2-year and 4-year graft survivals were assessed using the Kaplan-Meier approach. Data analyses were performed using JMP (Version 17; SAS Institute Inc). For statistical analysis in this study, P values of <.05 were considered statistically significant.
Results
There were 186 potentially eligible patients who received QST-HTA ACLR within the described timeline. There were 123 eligible patients (STA: 39 patients; control: 84 patients) after the exclusion criteria were applied. Of the remaining patients, 19 patients (15%) were deemed lost to follow-up; however, only 3 patients were lost to follow-up in the STA group (P = .12). In the final data analysis, 36 patients with independent STA and 68 controls were included. Figure 2 is a flowchart detailing the total number of patients included in final data analysis and the reason for patient exclusion.
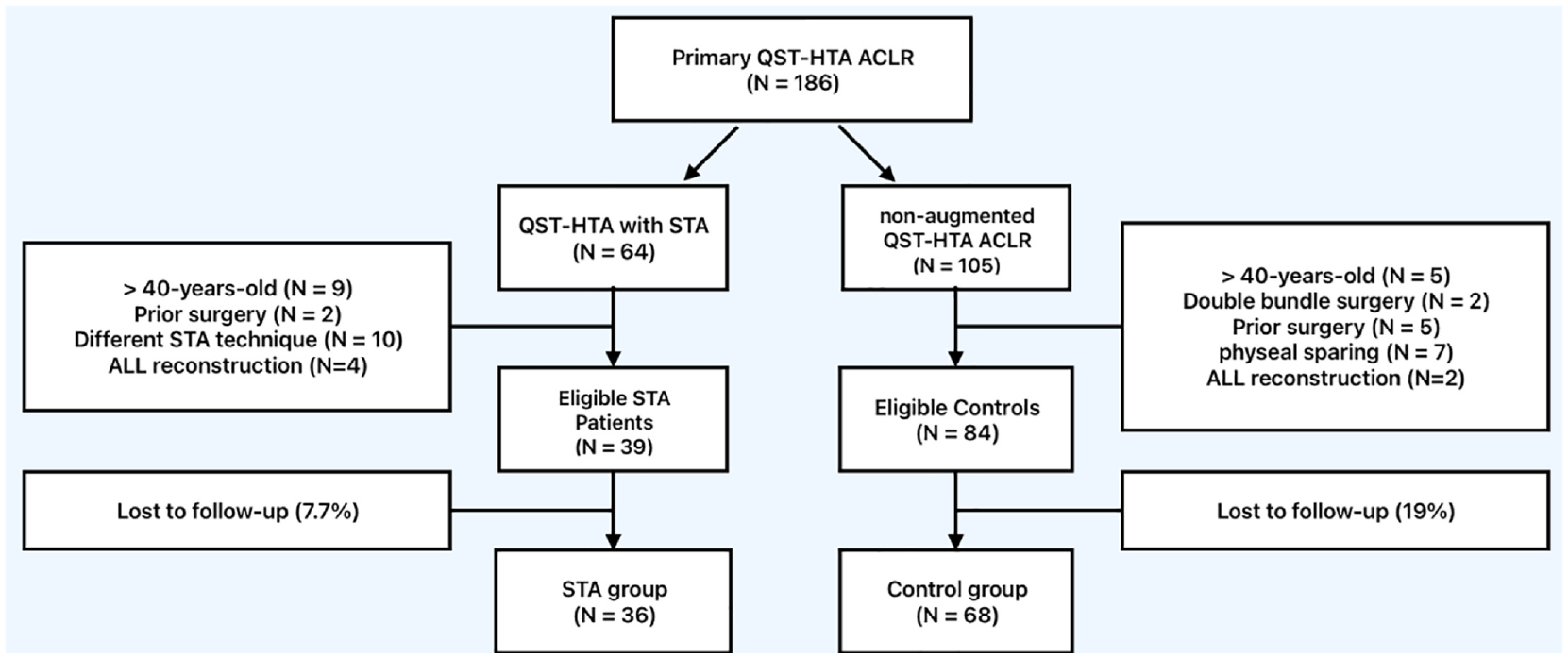
Flowchart detailing included and excluded patients. ACLR, anterior cruciate ligament reconstruction; ALL, anterolateral ligament; HTA, hamstring tendon autograft; QST, quadrupled semitendinosus; STA, suture tape augmentation.
Patient characteristics are summarized in Table 1. The mean final follow-up was >4 years and was comparable between both groups. The mean patient age was <22 years (range, 13-39 years) and was comparable between groups. There were proportionately more male patients in the STA group compared with the control group (69% vs 47%; P = .039). There were no significant differences seen regarding body mass index or procedural side. PTS and knee recurvatum were both comparable between groups. Additionally, there were no differences seen in the proportion of adolescent patients included in each group.
Patient Characteristics a

Data are presented as mean (95% CI) or n (%). AP, anteroposterior; STA, suture tape augmentation.
Intraoperative characteristics are summarized in Table 2. The mean graft diameter was comparable between the STA and control groups (9.0 mm [95% CI, 8.8-9.2 mm] vs 8.9 mm [95% CI, 8.7-9.0 mm], P = .23). Compared with the control group, the STA group demonstrated a significantly higher odds of necessitating a lateral meniscus procedure (OR, 3.53 [95% CI, 1.48-8.43]; P = .007) secondary to a larger number of concomitant lateral meniscus repairs (OR, 6.58 [95% CI, 2.25-19.2]; P < .001) at the time of ACLR. The overall incidences of meniscal tears, medial meniscus procedures, arthroscopic partial lateral meniscectomies, and chondral procedures were comparable between groups.
Intraoperative Characteristics and Concomitant Procedures a

Data are presented as mean (95% CI) or n (%) unless otherwise indicated. APM, arthroscopic partial meniscectomy; LM, lateral meniscus; MM, medial meniscus; OR, odds ratio; STA, suture tape augmentation. Dashes indicate not applicable.
One patient may have sustained a medial and a lateral meniscus tear.
One patient may have undergone a combination of meniscal repair and partial meniscectomy.
Subsequent procedural characteristics are summarized in Table 3. Overall, there was a 69% reduced risk of undergoing a subsequent procedure for the STA group (RR, 0.31 [95% CI, 0.12-0.84]; P = .010) secondary to a 76% reduced risk of necessitating revision ACLR (RR, 0.24 [95% CI, 0.06-0.97]; P = .017). The remaining procedural parameters including the necessitation for subsequent partial meniscectomy, meniscal repair, meniscal allograft transplantation, arthrolysis, and contralateral ACLR were comparable between groups. Furthermore, the median times to the subsequent procedures—on both the ipsilateral and contralateral knees—were comparable between groups. Additionally, a majority of the ACL reinjuries necessitating surgeries were sports related, with only 3 patients in the control group sustaining a non–sports-related reinjury (P > .99).
Subsequent Procedural Details a
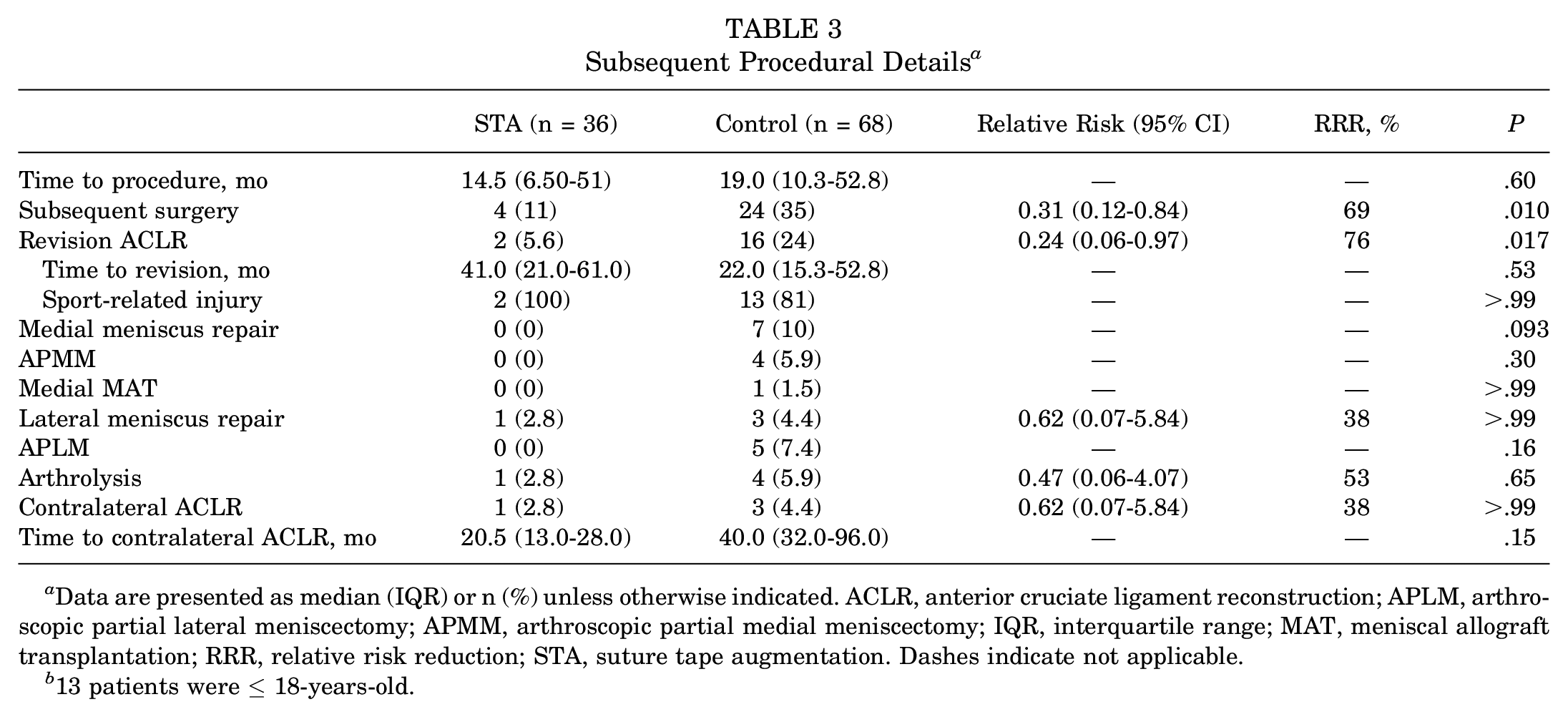
Data are presented as median (IQR) or n (%) unless otherwise indicated. ACLR, anterior cruciate ligament reconstruction; APLM, arthroscopic partial lateral meniscectomy; APMM, arthroscopic partial medial meniscectomy; IQR, interquartile range; MAT, meniscal allograft transplantation; RRR, relative risk reduction; STA, suture tape augmentation. Dashes indicate not applicable.
13 patients were ≤ 18-years-old.
The 4-year graft survival including all patients is depicted in Figure 3. Graft survival at 4 years was significantly higher for the STA group compared with the control group (97.2% vs 82.4%; P = .031). Of note, there was 1 patient (2.8%) in the STA group and 4 patients (5.9%) in the control group who sustained a graft failure after the 4-year period (P > .05). When stratifying our cohorts based on age, of the patients <18 years, graft survival was 73% (22/30) for the control group and 100% for the STA group. When extending to five years, the graft survivability among adolescent patients in the control group further decreased to 70%. The 1 patient in the STA group who underwent a revision ACLR during this time was a 20-year-old collegiate basketball player. Of note, there was no difference for graft survival at 2 years between the STA and control groups (2.8% vs 11.8%; P = .11).
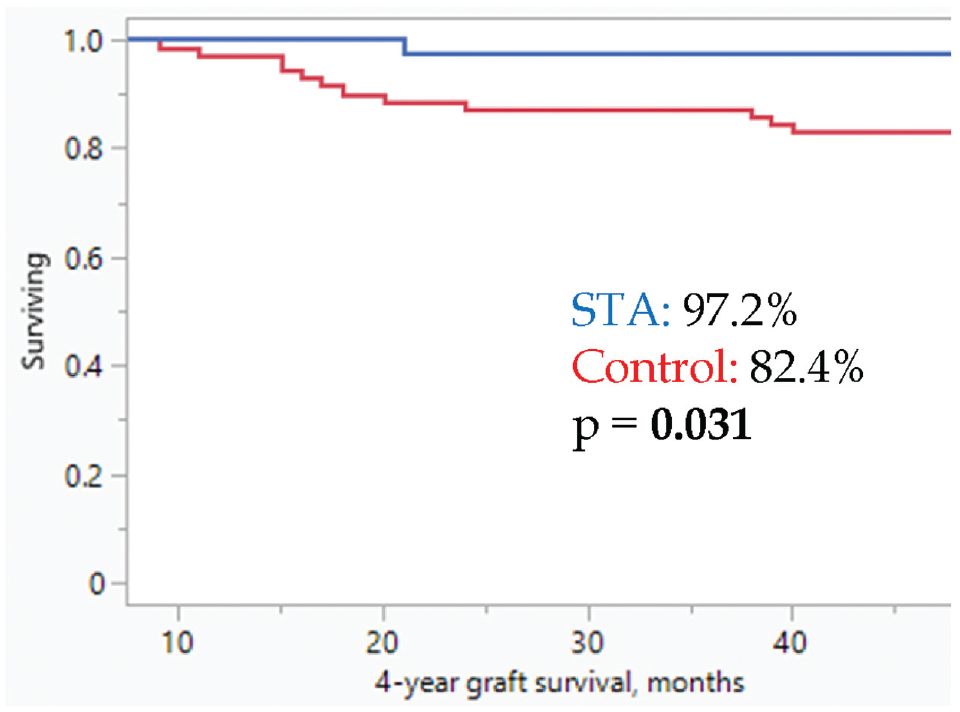
Kaplan-Meier survival plot depicting a 4-year graft survival between the suture tape augmentation (STA) group (blue line) and the control group (red line). Each step-off represents a graft failure. Comparison between groups was performed using the Wald test.
Preoperative and postoperative (final follow-up) PROMs and preoperative and 1-year postoperative KT-1000 arthrometer measurements are summarized in Table 4. All preoperative PROMs were comparable between both groups. For both groups, all PROMs significantly improved at the final follow-up, except for the Tegner activity levels, which significantly decreased postoperatively compared with their preinjury levels (P < .001). Activity levels were comparable between groups both before injury and at the final follow-up, as were the KT-1000 arthrometer measurements.
Preoperative and Postoperative PROMs and KT-1000 Arthrometer Measurements a
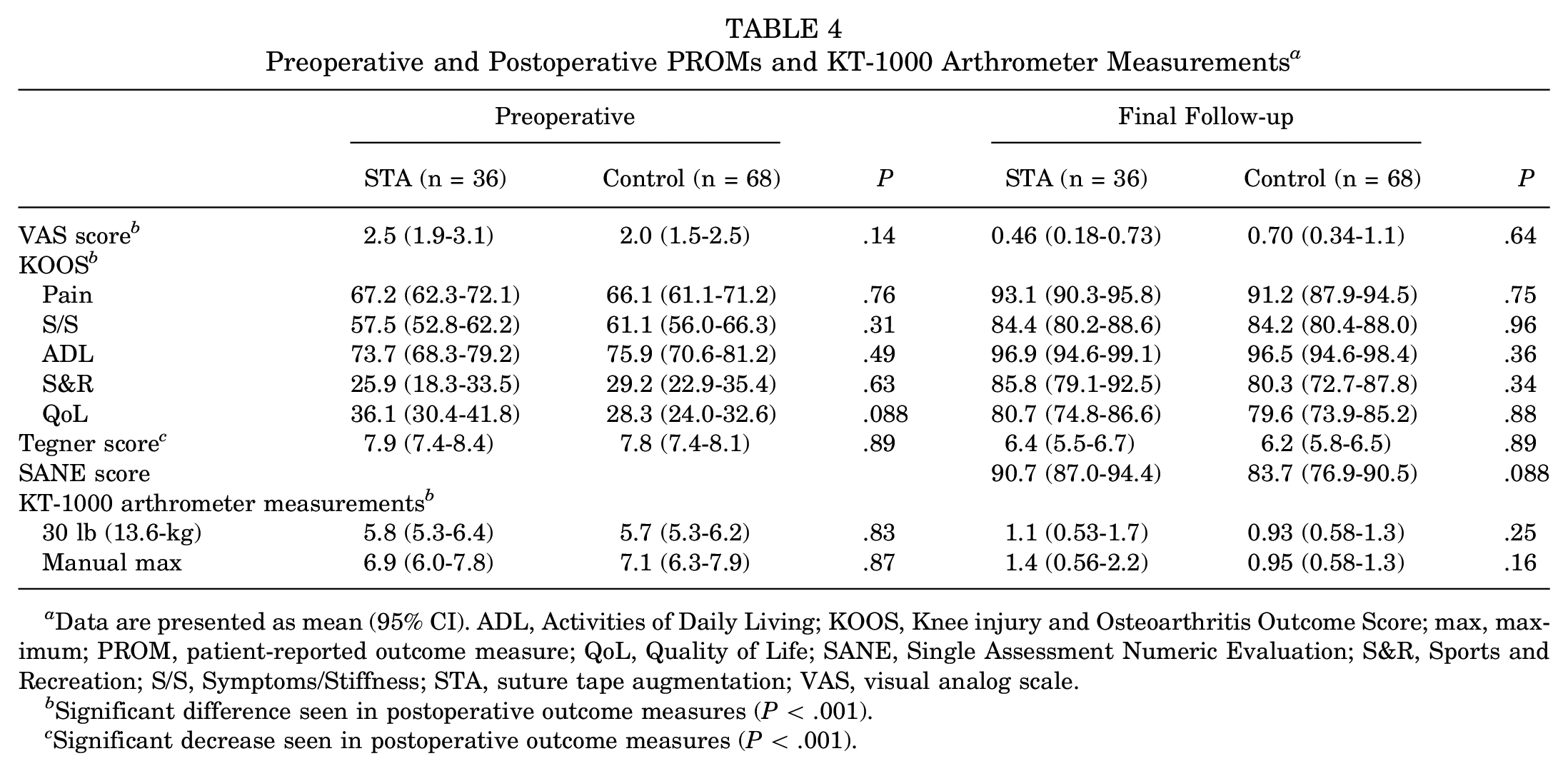
Data are presented as mean (95% CI). ADL, Activities of Daily Living; KOOS, Knee injury and Osteoarthritis Outcome Score; max, maximum; PROM, patient-reported outcome measure; QoL, Quality of Life; SANE, Single Assessment Numeric Evaluation; S&R, Sports and Recreation; S/S, Symptoms/Stiffness; STA, suture tape augmentation; VAS, visual analog scale.
Significant difference seen in postoperative outcome measures (P < .001).
Significant decrease seen in postoperative outcome measures (P < .001).
RTS data detailing immediate return are summarized in Table 5. The level of competition and type of sport played were comparable between groups. Both groups had a comparable number of patients participating in high-risk pivoting sports (eg, basketball, football, soccer, volleyball, and lacrosse). There were no differences seen in the RTS rate or time to RTS. Additionally, there were no differences seen in the number of patients who did not return to the same level of play as before their injury. The most common reason for a lower postoperative activity level was graduation from high school or college and not continuing to play competitively at the next level. There were five patients in the overall population that did not return to sport due to disinterest, and three patients in the control group that could not functionally return.
Return to Sports a

Data are presented as n (%) or mean (95% CI). RTS, return to sports; STA, suture tape augmentation.
Other sports include skiing, volleyball, softball, cheer/tumbling, running, tennis, wrestling, wakeboarding, lacrosse, kickball, martial arts, and mountain biking.
Secondary regression analyses were performed between pre- and postoperative patient variables and the need for revision ACLR. For this study cohort, risk of revision ACLR was higher for younger patients (hazard ratio [HR], 2.46 [95% CI, 1.47-4.12]; P < .001) and patients with higher postoperative Tegner levels (HR, 1.83 [95% CI, 1.11-3.04]; P = .018). In addition, high school athletes also demonstrated a significantly higher odds of necessitating revision ACLR (OR, 4.36 [95% CI, 1.32-14.4]; P = .016), whereas participation in only recreational sports at the time of injury demonstrated decreased odds for revision ACLR (OR, 0.15 [95% CI, 0.03-0.70]; P = .007). Other parameters including preinjury Tegner levels, graft diameter, and the remaining intraoperative characteristics, as well as patient characteristics, were not associated with the increased need for revision ACLR in this patient population.
Discussion
The most important finding of this study is that a QST-HTA ACLR with STA was associated with a reduced risk of subsequent revision ACLR compared with a nonaugmented control in patients with similar ages, activity levels, and final follow-up times. Additionally, a significant difference in revision ACLR at 4 years after the primary procedure was found (2.8% vs 17.6%)—in favor of the STA group—despite there not being a significant difference in 2-year revision ACLR (2.8% vs 11.8%). This is the first study to our knowledge to show that QST-HTA ACLR with independent STA led to a lower subsequent revision ACLR rate. Moreover, this is also the first study to demonstrate continued graft protection after the 2-year postoperative period, suggesting that STA significantly attenuates the incremental decline of graft survivability in younger patients who sustained their initial injury during competitive athletics.
The overall graft survival for the control group in our study was 76% at a mean final follow-up of 5 years. This decreased from 88% at 2 years and 82% at 4 years. In a case-control study examining 200 patients who received primary nonaugmented HTA ACLR, Salmon et al 39 found that graft survival at 5 years in patients <18 years of age at the time of surgery was 77%, while their overall graft survivability at this time point was only 89%. This was similar to the survival rate of our adolescent patients at 4 years within the control cohort (73%), which decreased from 80% at 2 years—when a majority of the adolescent graft failures occurred. When extending to 5 years, adolescent graft survivability further decreased to 70% in our control cohort, as 1 additional patient sustained a graft failure during this time. Interestingly, none of the adolescent patients in the STA group sustained a graft failure during these time points. The graft survival in the younger cohort of the Salmon et al 39 study, at the final follow-up of 20 years, decreased further to 61%, which was 25% lower than in those who were ≥18 years of age at the time of surgery. Their work highlights the fact that graft failure not only still occurs after the 2-year postoperative period, during which most graft failures are believed to occur, but also continues to occur even 20 years postoperatively, just at a significantly decreased rate. Consistent with the study performed by Salmon et al, 39 of the 16 patients in the control group in our study who underwent a subsequent revision ACLR, 13 patients (81%) were <18 years of age at the time of their initial surgery. It is worth noting that <20% of the total cohort of the study by Salmon et al 39 were adolescent patients, whereas >40% of our cohorts consisted of patients ≤18 years of age.
Our secondary regression analyses confirmed that younger patient age, higher postoperative Tegner activity levels, and participation in high school sports all demonstrated an increased risk for subsequent revision ACLR. Graft size, preoperative KT-1000 arthrometer measurements, knee recurvatum, PTS, sex, preoperative activity level, concomitant pathology at the time of primary ACLR (ie, meniscal and/or chondral procedures), type of sport played at the time of injury, and time to RTS were not found to be risk factors for subsequent revision surgery in this study cohort.
In a case series examining 862 Australian soccer players of all ages after ACLR with semitendinosus and gracilis autografts with proximal and distal interference screw fixation, Manara et al 30 found that graft failure occurred in 10% of all patients. However, graft failure occurred in 24% of adolescent patients, with significantly more males being affected than females (30% vs 8%). Even patients aged 19 to 25 years appeared to be at a higher risk of sustaining a graft rupture in 5 years compared with those aged >25 years, particularly for their male population. This suggests that even in a population that regularly participates in pivoting sports, younger patient age and possibly male sex lead to an increased risk for subsequent graft rupture. Although our cohort was not composed exclusively of soccer players, our data showed that younger patients who regularly participate in sports at a competitive level appear to experience the consequences of further injury, especially if they had not received intra-articular augmentation with suture tape.
In a retrospective registry study comparing primary ACLR with and without independent STA, Daniel et al 10 found that ACLR with STA led to a decreased risk of subsequent revision ACLR compared with nonaugmented ACLR. However, there was heterogeneity among the groups pertaining to type of autograft used, as there were significantly more BPTB autografts in the control group and more all–soft tissue QTAs in the STA group.
There have been 2 other studies examining patient outcomes after primary all-inside QST-HTA ACLR with and without independent STA.23,34 Kitchen et al 23 found that those with suture tape augmentation demonstrated significantly higher postoperative activity levels. Although they were unable to find a significant difference in retear rate due to underpowering, they found a trend toward a lower graft rupture rate in their STA group (5% vs 18%). Notably, their mean patient age for both groups was <16 years (STA: 15.7 years; no STA: 14.9 years). Similarly, Parkes et al 34 found that patients with a mean age of 25 years who had suture tape augmentation demonstrated higher activity levels postoperatively; however, they were unable to find any differences in other parameters, including graft failure rate (STA: 3%; control: 6%). Given that the mean patient age of our cohorts was <22 years, our finding of a 76% decreased risk for revision ACLR at a mean final follow-up of >4 years is encouraging.
Bodendorfer et al 5 found that patients treated with all-inside quadrupled semitendinosis tendon with suture tape augmentation in addition to ACLR exhibited significantly higher patient-reported outcomes (PROs) as well as RTS outcomes. Notably, they did not independently fix the suture tape in their STA group relative to the graft but instead ran the suture tape within the loop of the ALD with the graft, similar to the technique initially described by Smith and Bley. 44 Additionally, they also combined allografts and HTAs. Nonetheless, despite the promising results of significantly higher PROs in their study, we were unable to elicit similar findings. Notably, in their study the graft retear rate was 6.7% for both the augmented and nonaugmented groups.
Both cohorts in this study demonstrated relatively high preinjury activity levels with mean Tegner levels exceeding 7 in both groups. Additionally, each group consisted predominantly of patients who participated in high-risk pivoting sports either competitively or recreationally on a regular basis. Furthermore, >60% of the patients in each group participated in either high school or collegiate athletics. Given that both groups consisted of mostly high-risk individuals,6,27,48 a significant decrease in revision surgery with the addition of suture tape to augment the final construct is promising. Regarding activity levels, it was found that Tegner levels for both groups significantly decreased at the final follow-up compared with their preinjury levels. Activity levels tend to decrease after ACL surgery, especially if the patients do not continue with athletics at the next level of competition. 41
In this study, there were proportionately more male patients in the STA group and more female patients in the control group. In a systematic review of 20 studies (male: 35,935; female: 21,455) examining sex-specific outcomes after ACLR, Mok et al 32 demonstrated that 7 of the included studies found a significantly higher odds of graft rerupture in men compared with women. They also found that males reported a significantly higher RTS rate compared with their female counterparts. Despite these findings, we were unable to elicit any significant associations between sex and revision ACLR or higher RTS rate in our cohort.
In an effort to decrease the risk of revision ACLR, extra-articular augmentation utilizing numerous techniques has been described and implemented with success in young, at-risk patients.13,15,35,37 In particular, the SANTI and STABILITY groups found that extra-articular stabilization with either an ALL reconstruction or an LET was effective in decreasing the risk of subsequent graft failure in relatively young patient populations.13,15,35 In the STABILITY trial, Getgood et al 15 demonstrated that patients who received isolated ACLR demonstrated a higher overall risk of clinical failure compared with those augmented with LET at 2 years (40% vs 25%). In their study, clinical failure was defined as an asymmetric pivot shift (grades 1-3) or evidence of graft rupture confirmed either on magnetic resonance imaging or intraoperatively. Of note, despite the relatively high degree of clinical failure, graft ruptures necessitating revision ACLR in the isolated ACLR and ACLR + LET groups were only noted to be 11% and 4%, respectively, still yielding statistical significance. In our study cohort, all patients with a documented reinjury and having a positive pivot shift—which would fit the above definition of a clinical failure—underwent revision ACLR.
With the negative connotation surrounding ligament augmentation techniques, there has been a concern for intra-articular side effects that may be caused by the introduction of foreign material within the knee joint. Devices such as the Kennedy Ligament Augmentation Device and Gore-Tex artificial ligaments were once used, both of which resulted in undesirable outcomes, such as tunnel osteolysis, infection, and severe joint synovitis, thus veering many orthopaedic surgeons away from the implementation of intra-articular augmentation.3,14 However, the augmentation in this study was polyethylene core braided polyester suture, which has not been shown to lead to negative intra-articular side effects, as seen in a translational canine study. 45 Furthermore, none of the prior studies relating to independent STA noted an increased risk of complications that may be attributed to the suture tape compared with the nonaugmented groups.10,11,23,29,34 Additionally, we did not note joint overconstraint in the STA group, evidenced by our 1-year KT-1000 arthrometer measurements.
Because of the increasing incidence of ACL tears and ultimately ACLR in the young active patient population, implementing means of decreasing the risk of a second ACL injury to the ipsilateral knee is crucial. Revision ACLR demonstrates poorer outcomes compared with a primary ACLR.22,26,53 Additionally, revision ACLR puts an economic strain on the health care system and, more importantly, a psychosocial burden on the patient.9,19 For these reasons, decreasing the overall revision ACLR rate in the adolescent and young adult patient population is crucial.
Biomechanically, ACLR with STA results in a reduction of graft elongation and increased graft stiffness, thus increasing overall graft failure loads compared with nonaugmented grafts through the concept of load sharing.1,2 The key principle of this concept is the complete independence of the suture tape in relation to the graft, which allows the graft to undergo the uninterrupted stresses important for healing and only assisting in graft protection when a higher load is reached. Potentially, this may ultimately protect the construct from the novel failure mechanism associated with ALDs, the “Chinese finger trap” design.4,16,18 Because the suture tape is passed through the femoral button and fixed distally with an anchor, it theoretically remains at a relatively fixed length. However, it is unclear how this mechanism of failure that is unique to ALDs affects the suture tape and vice versa, as there is a paucity of literature describing ALDs ± STA versus fixed loop devices ± STA.
Traditionally, HTAs have been composed of a quadrupled construct with both the semitendinosus and gracilis tendons.11,13,15,23,30,39 There is a paucity of literature describing the clinical and/or biomechanical outcomes between isolated semitendinosus tendon use versus semitendinosus tendon with additional gracilis tendon augmentation, particularly in the setting of comparable final graft widths when quadrupled. Because a graft diameter of <7 mm is a known risk factor for subsequent graft failure,13,15 additional augmentation with a gracilis autograft may be warranted to mitigate this risk. However, it has been shown that sufficient graft width with QST-HTAs can be achieved as documented in the current study and in the existing literature.5,10,11,15,33,34,38,46 Furthermore, given the nature of the all-inside technique with a blind tibial socket as opposed to a full tunnel, the graft length is typically shorter, approximately 65 mm, which allows for the quadrupling of the semitendinosus tendon alone to reproducibly produce a >8 mm–diameter graft.31,44
The surgical technique used for both groups consisted of the use of ALDs on both the femur and tibia, rather than rigid fixation, which has traditionally been used.4,16,18 A recent systematic review and meta-analysis performed by Elmholt et al 12 did not demonstrate an increased advantage of using an ALD compared with a fixed loop device, specifically for femoral fixation. Although determining the clinical advantage of one fixation technique over the other with and without STA is well out of the scope of the current study, we may be able to justify the cost of the additional suture tape and anchor in our STA cohort after QST-HTA ACLR with ALDs, as the cost of a revision ACLR is significantly more than that of the implementation of 2 additional implants during the primary procedure.
This study was not without limitations. Given the retrospective nature of this study, there is always a risk for selection bias. We attempted to mitigate this risk by including all available patients. Additionally, because all the procedures were performed by the senior author at his institution, the results of this study may not be generalizable. There were 19 patients lost to follow-up, with a trend of more patients being lost in the control group. Therefore, the results, particularly with subsequent revision ACLR, may be either over- or underrepresented. The time points at which these patients received surgery were 10 years apart, which brings up the concern of both selection bias and technique familiarity. This time frame was necessary as the senior author started the all-inside technique in 2009, so the decision was made to include patients who received surgery 2 years after initial technique implementation to avoid the confounding of a “learning curve” with this new surgical technique. Furthermore, the senior author performs all-inside ACLR with ALDs for all soft tissue grafts, including his most popular autograft (all-soft QTA), thus increasing his familiarity with the technique. It was necessary to collect patients as far back as 2011 to obtain an adequate number of nonaugmented QST reconstructions based on the fact that it is the senior author's least common graft choice. Moreover, he started STA for all QST autografts after July 2016, necessitating the longer time frame up to 2020 for patient collection, again, to obtain adequate numbers for the STA group. In addition, many patients who received all-inside QST-HTA ACLR were excluded based on our criteria or were lost to follow-up (n = 82), compromising our total patient numbers. Finally, this study may not be powered to determine subtle differences between groups, but only dramatic differences.
Conclusion
QST-HTA ACLR with STA was associated with reduced risk for revision ACLR compared with nonaugmented QST-HTA ACLR in this young patient population. Furthermore, the addition of suture tape did not appear to affect postoperative patient-reported and RTS outcomes.
Footnotes
Final revision submitted January 5, 2024; accepted February 26, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: P.A.S. has received royalties from Arthrex; nonconsulting fees from Arthrex and Kairos Surgical; consulting fees from Arthrex; and education payments from Arthrex and United Orthopedics. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Salus IRB.
