Abstract
Background:
There is a lack of procedures that adequately address the subchondral bone structure and function for reconstructing osteochondral defects in the femoral condyles.
Purpose:
To biomechanically evaluate the tibiofemoral joint contact characteristics before and after reconstruction of femoral condylar osteochondral defects using a novel hybrid reconstructive procedure, which was hypothesized to restore the contact characteristics to the intact condition.
Study Design:
Controlled laboratory study.
Methods:
Tibiofemoral contact areas, contact forces, and mean contact pressures were measured in 8 cadaveric knees (mean age 52 ± 11 years; 6 women, 2 men) using a custom testing system and pressure mapping sensors. Five conditions were tested for each condyle: intact, 8-mm defect, 8-mm repair, 10-mm defect, and 10-mm repair. Medial femoral condylar defects were evaluated at 30° of knee flexion and lateral condylar defects were evaluated at 60° of knee flexion, with compressive loads of 50, 100, and 150 N. The defects were reconstructed with a titanium fenestrated threaded implant countersunk in the subchondral bone and an acellular dermal matrix allograft. Repeated-measures analysis of variance with Bonferroni correction for multiple comparisons was used to compare the results between the 5 testing conditions at each load.
Results:
Medial condylar defects significantly increased mean contact pressure on the lateral side (P < .042), which was restored to the intact levels with repair. The lateral condylar defect decreased the mean contact pressure laterally while increasing the mean pressure medially. The lateral and medial mean contact pressures were restored to intact levels with the 8-mm lateral condylar defect repair. The medial mean contact pressure was restored to intact levels with the 10-mm lateral condylar defect repair. The lateral mean contact pressure decreased compared with the intact state with the lateral condylar 10-mm defect repair.
Conclusion:
Tibiofemoral joint contact pressure was restored to the intact condition after reconstruction of osteochondral defects with dermal allograft matrix and subchondral implants for the repair of both 8- and 10-mm lateral condylar defects as well as 8-mm medial condylar defects but not completely for 10-mm medial condylar defects.
Clinical Relevance:
The novel hybrid procedure for osteochondral defect repair restored tibiofemoral joint contact characteristics to normal in a cadaveric model.
Keywords
Osteochondral injuries represent a spectrum of disease, ranging from acute osteochondral fracture defects to chronic and progressive osteochondral damage or osteochondrosis, that can eventually lead to diffuse osteoarthritis. 27 Within the osteochondral interface, the subchondral bone plays an integral role with the metabolism, function, and structure of the overlying articular cartilage.5,32,35,39 The subchondral bone and articular cartilage act as a functional unit in both healthy and diseased bone. As much as 50% of the metabolic load of the articular cartilage is sourced from the penetration of vessels into the baseplate from the underlying bone.21,26 The subchondral bone is also a modulator for pain, where the degree of pain is correlated with the extent of bone damage in osteoarthritis. 44 Joint force attenuation in healthy bone occurs through a delicate transfer of shear and tangential stresses in the form of compressive forces from the surface through the articular cartilage, the cartilage baseplate, and finally through the subchondral bone.18,35 Ultimately, as much as 80% of the joint forces can be absorbed by the summation of these knee joint shock-absorbing components. 18 In osteochondral disease and damage, this interplay between the metabolism, function, and structure of these components is disrupted. 35
Because the healing potential of osteochondral defects is limited, there is a decreasing ability to heal with increasing size of the defect. 19 Subchondral bone damage manifests in many forms, resulting in cartilage defects that occur in as much as 12% of the population, most commonly in the form of chondral or osteochondral lesions in the tibiofemoral joint.1,4,17
The high incidence of chondral or osteochondral lesions sparked the development of many cartilage repair techniques, which currently include microfracture, mosaicplasty or osteochondral autograft transfer system (OATS); osteochondral allograft (OCA) transplantation; particulated articular cartilage implantation (PACI); autologous chondrocyte implantation (ACI); and matrix-induced autologous chondrocyte implantation (MACI). 22 In long-term studies of patients who underwent mosaicplasty or microfracture procedures, those aged >40 years were found to have lower patient-reported outcome scores.13,24 Patients who underwent ACI continued to have poorer knee strength than control patients at minimum 2-year follow-up as well as an inability to return to sport and a reported complication rate of nearly 50%.20,29,31,34 Notable flaws in these existing procedures include the possibility of morbidity and failure to effectively address or protect the subchondral bone.2,41,42 Current osteochondral defect repair techniques also lack an adequate delivery vehicle for biologics and scaffolds.3,14 -16,36
In this study, we presented a versatile, novel, hybrid reconstruction procedure for femoral condylar osteochondral defects utilizing titanium fenestrated threaded implants in the subchondral bone and human dermal allograft. It creates a functional reconstruction that structurally repairs the subchondral bone damage at time zero and provides a fixation platform for the delivery of biologics through the design of the fenestrated threaded implant, which allows the potential of biologic integration into the subchondral bone. The objective of this study was to biomechanically evaluate the tibiofemoral joint contact characteristics before and after reconstruction of femoral condylar osteochondral defects using this novel hybrid procedure. We hypothesized that the reconstruction of osteochondral defects of the femoral condyle would restore the contact characteristics to the intact condition.
Methods
Specimen Preparation and Testing Setup
Eight cadaveric knee specimens (6 female and 2 male specimens; mean age, 52 ± 11 years) were obtained for testing (Science Care, Phoenix, AZ). The specimens were dissected of all soft tissue, leaving only the bony articulation of the femur and tibia. The articular surfaces of the specimens were examined and determined to be free of any signs of arthritis or osteochondral defects. Before removal of the joint capsule and ligamentous structures, the femur and tibia were marked to reproduce the native rotational position of the knee when mounted on the testing system. The tibia and femur were potted with the tibial and femoral shaft centered in a 2-inch PVC pipe with plaster of Paris.
To measure contact pressure across the tibiofemoral joint, an Instron materials testing machine was used to apply a compressive load across the joint at 30° or 60° of knee flexion, and a Tekscan pressure measurement system was used. Medial and lateral femoral condylar defects were analyzed: medial femoral condylar defects were analyzed at 30° of flexion, and lateral condylar defects were analyzed at 60° of flexion. These have been reported as the flexion angles at which significant tibiofemoral compression occurs, corresponding to the most common locations of osteochondral defects.4,10,17,28,37,40,43
The specimens were mounted onto the materials testing machine in an inverted fashion (Figure 1). The femoral mounting cylinder was attached to an arc that allowed for the adjustment and fixation of the knee flexion angle. This arc was attached to an x-y translator that allowed for anterior-posterior and medial-lateral translation. The femur was mounted in the cylinder with the epicondylar axis aligned with the medial-lateral translation of the translator. The femur was positioned at 30° of flexion. The tibia was mounted in a cylinder that attached to the crosshead of the Instron. The tibia was locked in a rotation position based on the marker alignment with the femur before disarticulating the knee. The tibial cylinder also allowed for varus-valgus angulation and fixation.

Knee specimen mounted on an Instron materials testing system at 30° of flexion.
Once the femur was locked in flexion, the tibia was compressed onto the femur to simultaneously load both condyles. The translational position of the femur and valgus position of the tibia was then locked into place. At this point, only the tibia could be moved upward (distracted) with the crosshead to allow for condylar defect creation and repair. This position was maintained throughout testing for the flexion angle being tested.
Biomechanical Testing
Five conditions were tested for each condyle: intact, 8-mm defect, 8-mm repair, 10-mm defect, and 10-mm repair. Medial condylar defects were evaluated at 30° of knee flexion and lateral condylar defects were evaluated at 60° of knee flexion, with compressive loads of 50, 100, and 150 N. Osteochondral defects (Outerbridge grade 4 with removal of any osteochondral remnants) were created and then reconstructed with a titanium fenestrated threaded implant countersunk in the subchondral bone and an acellular dermal matrix allograft. To start, the intact, native medial and lateral tibiofemoral joint contact pressure was measured using Tekscan pressure mapping sensors (model 4000). The sensors were first calibrated in the Tekscan software after cycling for 10 cycles from 25 to 150 N to precondition the sensor. Calibration was performed using a 2-point calibration curve for each specimen with a compressive force of 50 and 150 N along with a sensitivity of 35 out of the 40 levels of settings in the Tekscan software. This sensitivity was chosen based on pilot testing to be as sensitive as possible but not to saturate the sensor reading. This calibration resulted in a mean saturation pressure of 2604 ± 676 kPa across all specimens.
Contact pressure measurements were then recorded initially for the intact, native joint for the 3 levels of cyclic compression. Before data collection, the specimen was loaded for the complete cycles to precondition the specimen. Data were then collected on the second trial of loading. Five cycles each from 25 to 50 N, 25 to 100 N, and 25 to 150 N were performed at a rate of 10 mm of displacement per minute.
After testing the intact specimens, the osteochondral defects and repairs were performed. To create the osteochondral defect near the center of the contact area, the middle of the defect was first measured from the Tekscan software relative to the corner of the Tekscan pad aligned with the specimen. Next, acrylic paint was applied to the tibial plateau, and the specimen was compressed to leave an outline of the contact on the femoral condyle. The center of the paint outline was then confirmed to be near the center measured by the Tekscan outline. Once this center was defined, a 1.1-mm K-wire guide pin was drilled normal to the surface of the condyle. This guide pin was then reamed with an 8-mm reamer to create an 8-mm defect that was 13 mm deep. The defect condition was then tested in the exact manner as the intact knee.
Once the 8-mm defect was tested, it was then reconstructed with a 7-mm S-CORE implant (Subchondral Solutions) and 10-mm human dermal allograft (Figure 2). The human dermal allograft was punched with a biopsy punch to create a 10-mm circular graft. The graft thickness was then measured. The S-CORE implant was then inserted to the level of the subchondral bone, approximately 2 mm deep for all specimens. The human dermal allograft was then inserted and secured in the remaining 2 mm of void in the defect to align with the articular surface in a press-fit fashion. Care was taken to smooth the graft to the level of the articular surface such that the graft was not proud. The reconstructed defect was then tested to measure tibiofemoral contact pressure.

Photographs of a knee specimen showing the hybrid procedure for osteochondral defect repair. (A) First, the defect was made using a planing reamer inserted over a guide pin. (B) Next, a coring reamer was used to remove bone for the implant insertion. (C) The titanium fenestrated threaded implant was inserted and countersunk below the articular surface, and (D) an acellular dermal matrix allograft was placed on top of the implant.
After testing the 8-mm defect, the 10-mm defect was created. First, the graft and implant were removed. The 8-mm reamer was reinserted in the void, and the guide pin was inserted to find the original alignment of the guide pin with the articular surface. A 10-mm reamer was then used to ream a 10-mm hole, 13 mm deep. The 10-mm defect condition was then tested. After this test, the 10-mm defect was repaired in the same manner as the 8-mm using a 9-mm S-CORE implant and a 12 mm–diameter human dermal allograft. The 10-mm defect repair was then tested.
After all medial condylar defects and repairs were tested at 30° of flexion, the knee flexion was changed to 60° for evaluating lateral condylar defects. The tibiofemoral medial-lateral and anterior-posterior translation as well as the tibial varus-valgus degree of freedom were unlocked to find the native position of the knee before testing at 60° of flexion. The Tekscan pad was also recalibrated at the 60° position using the manner previously described.
The medial and lateral contact force, area, and mean pressure were measured at the peak of the contact force for each cycle and averaged across the 5 cycles. These values were then averaged across all specimens, and a repeated-measures analysis of variance with a Bonferroni correction for multiple comparisons was used to compare the results between the 5 testing conditions at each load. A paired Student t test was used to compare data between medial and lateral defects. P < .05 was used to determine statistical significance.
Results
Representative contact patterns for medial and lateral defects are shown in Figure 3. The medial condylar defects significantly decreased tibiofemoral contact force on the medial side and increased force on the lateral side at all loads (P≤ .037 for all) (Figure 4). Contact force was restored to intact levels with 8-mm defects at all loads but was not restored to the intact levels with repair of the 10-mm defect. Similarly, lateral condylar defects significantly decreased tibiofemoral contact force on the lateral side and increased contact force on the medial side across all loads (P≤ .008 for all) (Figure 4). Lateral condylar defect repairs restored both medial and lateral contact forces to the intact levels.
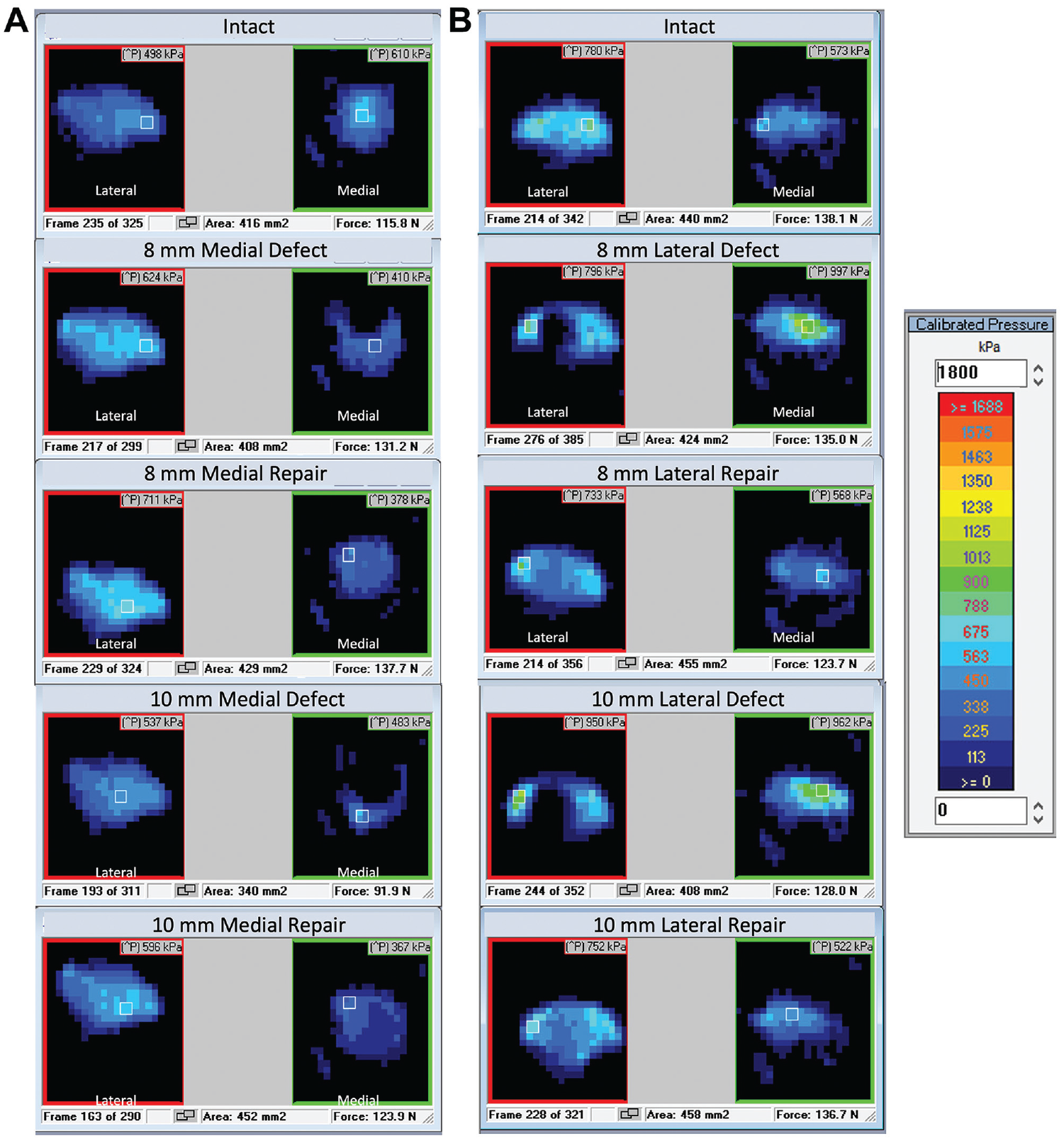
Representative Tekscan contact patterns for (A) medial condylar defects and (B) lateral condylar defects.

Mean tibiofemoral contact force at 50-, 100-, and 150-N compressive loads for medial condylar defects (left column) and lateral condylar defects (right column). Error bars indicate standard errors of the means. Statistically significant difference (P < .05): *versus intact; +versus 8-mm defect; #versus 8-mm repair; $versus 10-mm defect.
When compared with the intact condition across all loads, lateral condylar defects at the tibiofemoral joint had a mean increase in medial contact force of 52.1% and 54.2% for the 8-mm and 10-mm defects, respectively, and a mean decrease in lateral contact force of 54.6% and 47.1% for the 8-mm and 10-mm defects, respectively (P≤ .037 for all). For medial condylar defects at the tibiofemoral joint, there was a mean decrease in medial contact force across all loads of 29.9% and 27.9% for the 8-mm and 10-mm defects, respectively, and a mean increase in lateral contact force across all loads of 38.4% and 56.9% for the 8-mm and 10-mm defects, respectively, when compared with the intact conditions (P≤ .008 for all).
During the intact condition at 30° of knee flexion, there was a trend of lower lateral contact force compared with medial contact force, which was statistically significant at the higher loads (P = .0184 at 100 N and P =.0385 at 150 N). For the intact condition at 60° of knee flexion, there was a trend of higher lateral contact force compared with medial contact force, which was statistically significant at 100 N (P = .0205; 2-tailed Student t test).
The 10-mm medial condylar defects decreased medial contact area, which increased with repair to above the intact levels (P = .036 for 100 N and P = .018 for 150 N). The 10-mm lateral condylar defects decreased lateral contact area (P≤ .042), which increased with the 10-mm lateral condylar defect repairs to above intact levels (P≤ .001) (Figure 5).
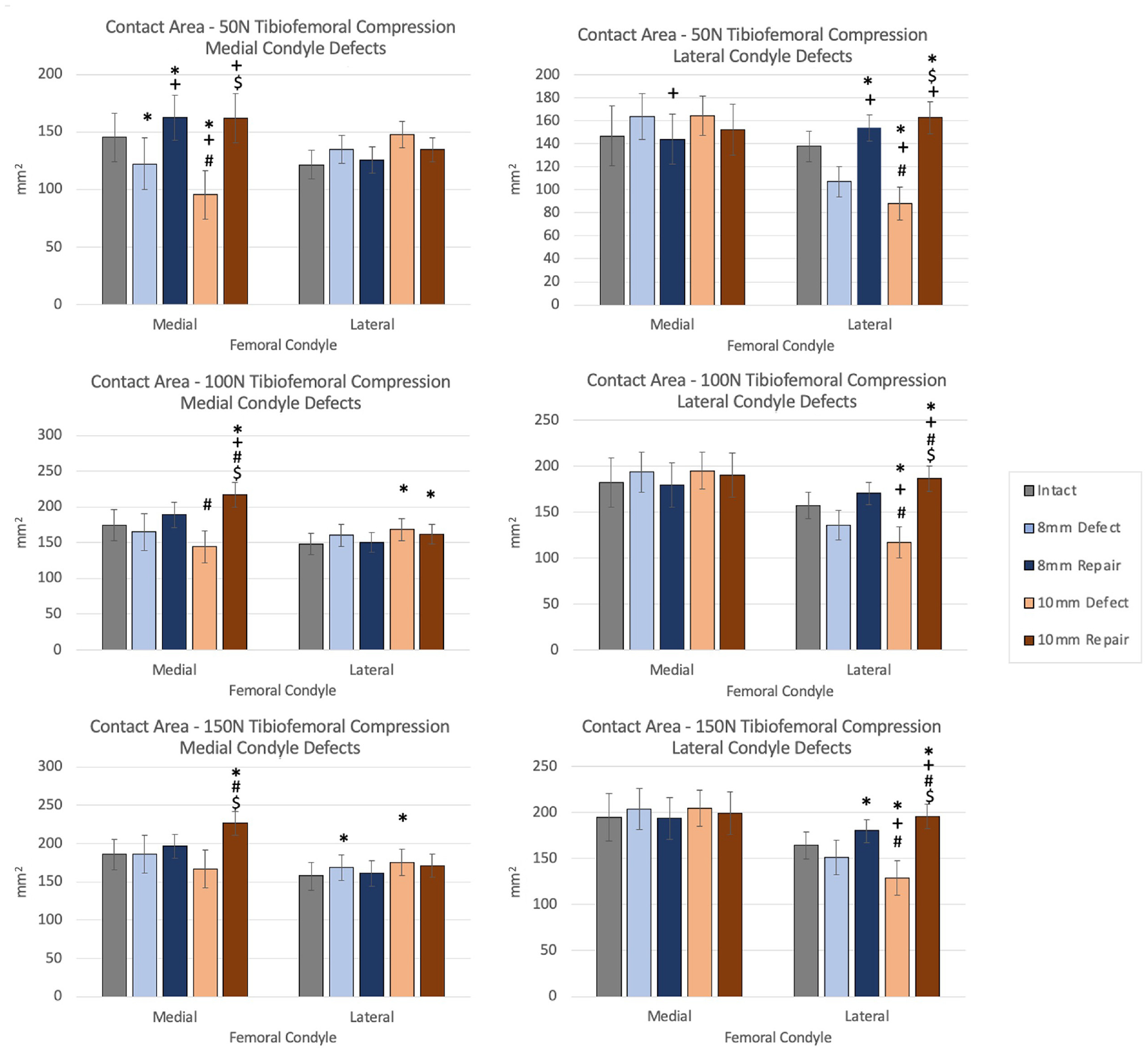
Mean tibiofemoral contact area at 50-, 100-, and 150-N compressive loads for medial condylar defects (left column) and lateral condylar defects (right column). Error bars indicate standard errors of the means. Statistically significant difference (P < .05): *versus intact; +versus 8-mm defect; #versus 8-mm repair; $versus 10-mm defect.
Medial condylar defects increased mean contact pressure on the lateral side at 50 N for the 8-mm defect (P = .04) and at 100 N and 150 N compared with intact for both 8-mm and 10-mm defects (P≤ .042). Medial condylar defect repairs restored lateral mean contact pressures to intact conditions (Figure 6).

Mean tibiofemoral contact area at 50-, 100-, and 150-N compressive loads for medial condylar defects (left column) and lateral condylar defects (right column). Error bars indicate standard errors of the means. Statistically significant difference (P < .05): *versus intact; +versus 8-mm defect; #versus 8-mm repair; $versus 10-mm defect.
Lateral condylar defects significantly decreased tibiofemoral lateral contact pressure (P = .026 for the 8-mm defect at 100 N; P≤ .034 for both 8-mm and 10-mm defects at 150 N) and increased medial mean contact pressure (both defects at 100 N and 150 N, 8-mm defect at 50 N; P≤ .04). Lateral condylar defect repair restored both medial and lateral mean contact pressure to intact levels across all loads for 8-mm defect repairs, while restoring only the medial mean contact pressure to intact levels across all loads for the 10-mm repairs.
With repair of 10-mm lateral condylar defects, there was a statistically significant decrease in lateral contact pressure across all loads when compared with the intact state (P < .05 for all) (Figure 6).
Discussion
Tibiofemoral joint contact forces were restored to the intact condition after reconstruction of osteochondral fracture defects with dermal allograft matrix and subchondral implants when under 50- and 100-N loads for repair of lateral condylar defects in all conditions, and for repair of 8-mm medial condylar defects. Repair of 10-mm medial condylar defects restored the lateral contact forces at 50- and 100-N loads; however, 10-mm medial condylar defect repairs failed to completely restore the medial contact force to intact levels across all loads and the 8-mm medial condylar defect repairs failed to restore medial contact force to the intact level for a 150-N load. This is demonstrated by decreases in contact force compared with the intact level.
Our findings of specific contact characteristics being dependent on defect size and location are in line with a study performed on bovine knees, which demonstrated that subchondral bone contact is dependent on the defect size and intra-articular location. The lateral condylar defects showed significant subchondral bone contact at a smaller defect size compared with medial condylar defects. 9 In another study performed on cadavers measuring contact forces on large femoral osteochondral allografts, with the knee at full extension position, their data showed a trend for lateral condylar defect plugs having higher contact force for each given extension moment applied to the knee when compared with medial condylar defect plugs. 8 A difference in load distribution among the femoral condyles depending on knee flexion angle was also evident in our data with the contact characteristics observed in both the intact and the defect conditions (Figures 4 and 5). This would suggest that contact force distribution among the femoral condyles has a dependence on flexion angle, with full-thickness osteochondral defects redistributing the contact force to the unaffected condyle. Hence, it is important for repair of femoral osteochondral defects to adequately restore contact forces to avoid osteochondral degeneration in the contralateral condyle due to the dispersion of contact.
Guettler et al 12 had similar findings of redistribution of peak contact pressure for larger osteochondral defects of >10 mm in diameter in fresh-frozen cadaveric knees. They also found that 8-mm defects had distinctly different patterns of peak pressure distribution, suggesting a threshold for the size of the osteochondral defect on the tibial condyles at which the distribution of compressive stress is altered. At the threshold defect size, the peak contact pressures start to increase disproportionately. In our study, there was a decrease in mean contact pressures laterally for lateral condylar defects and medially for medial condylar defects. Our findings similarly suggest that there is a threshold size for osteochondral defects after which the joint contact characteristics could not be restored consistently. The increase in contact area and decrease in mean contact pressures with repairs, especially with the 10-mm defect repairs, is likely due to the higher material compliance of the dermal allograft redistributing the contact stresses to a larger area compared with the intact subchondral bone along with the anatomical geometry of the contacting surfaces. This would allow for more contact area of the dermal allograft to the articulating joint surface, while also further dissipating and distributing contact stresses throughout the allograft to decrease contact pressures at the joint. This effect would be attenuated with the larger surface areas of dermal allograft for the repair of the larger defects in this study. Hence, there was a failure to restore contact characteristics to the intact state for medial condylar 10-mm defects.
Particularly in the cases of medial condylar defects, studies performed on cadaveric knees have found lower stiffness and contact force distribution in the medial condyle compared with the lateral condyle.8,33 Clinically, the medial condyle of the femur seems to be more sensitive to altered biomechanics compared with the lateral condyle, which suggests that the natural shape of the medial condyle can generally affect the contact characteristics of the tibiofemoral joint. 4 The medial condyle has a rounder surface when examining the anatomic geometry compared with that of the lateral condyle, which is flatter, allowing for more dissipation of contact stresses. Along with a threshold size and intra-articular location of the defects determining the contact characteristics of the tibiofemoral joint, the morphology of the medial condyle could explain why it is difficult to repair larger defects. This is seen with repair of the 10-mm medial condylar defects, which restored joint contact forces toward the intact state although not completely in this current study.
In comparison with other current surgical management techniques such as OATS or MACI for osteochondral defects, this novel hybrid reconstructive procedure eliminates the risk for donor-site morbidity and can be accomplished within a single surgery, unlike in MACI. 22 The procedure of using a dermal allograft matrix with a subchondral implant at time zero is biomechanically akin to an OCA transplantation, but with lower risk of complication from graft failure of the osseous portion of the allograft seen commonly in OCA, which could lead to subchondral collapse and deficiency in bone healing.7,11 Although PACI is a single-stage cartilage restoration technique, failure by graft lamination and failure of incorporation are still concerning complications postoperatively. 3 The novel hybrid approach in the current study provides initial structural integrity and restores the articulating surface congruency of the joint at time zero, as demonstrated in cadaveric elbow radiocapitellar osteochondral defects, where contact characteristics were successfully restored to the intact conditions. 6 In the current study, we had similar findings of contact characteristics being restored to the intact condition with reconstruction for all lateral condylar defects and for smaller defects in the medial condyle. The implant utilized in this procedure also has the potential to permit healing by marrow communication over time from its fenestrated design in addition to its initial structural support as a platform for biologics.
In this study, our biologic of choice was the dermal allograft, as it provides an effective initial lining of the defective surface after repair due to its intrinsic viscoelastic property providing a cushioning effect. Additionally, dermal allograft has been shown to vascularize both intrasubstance and on the graft periphery in patients as early as 6 months postoperatively, while maintaining function for ≥1 year in patients that underwent shoulder superior capsular reconstruction. 30 The potential of dermal allografts to become biologically functional can help to address the entire osteochondral unit in this hybrid approach. Of note, this hybrid approach can utilize other biological scaffolds or grafts and is not limited to use of only a dermal allograft. Furthermore, tibiofemoral contact characteristics can be restored to intact levels with the hybrid approach, whereas in comparison with OATS, the contact pressure was only partially restored where there was increased border contact pressure posttransplantation when compared with the intact knee in a cadaveric study. 23
Sismondo et al 33 developed and tested a similar technique using hydrogel implants, which was able to restore contact pressures in cadaveric knees at time zero. However, long-term studies in patients showed worsening outcomes with the hydrogel implants, and a sizable subset of patients developed implant failure after 12 months. 25 The worsened long-term outcomes seen in tibiofemoral osteochondral defect patients treated with the hydrogel implants would suggest the need to not only restore contact characteristics of the joint but also restore the biologic subchondral bone, which needs to heal for better outcomes given its role in pain modulation. 44 Hence, the advantage of our novel hybrid approach is to not only initially restore structural integrity but also have potential for promoting healing of the subchondral bone in the long term to improve patient outcomes.
Limitations
Limitations of this study include the following. One, the lack of soft tissue and meniscus during the testing conditions, which contributes some compression force and affects the distribution of contact stresses at the knee joint. 38 Two, the effect on contact characteristics with inclusion of a Tekscan sensor between the articular surfaces, which would be consistent throughout all testing conditions and not affect their comparisons. Three, only unidirectional loading of compressive force was applied to the tibiofemoral joint. Four, this model did not account for biochemical cartilage healing and can only represent contact characteristics at time zero; hence, this requires further long-term clinical studies where physiological healing and cyclic loading can be evaluated. Five, there are no studies on cyclic compressive load to failure of dermal allografts to date; thus, further investigation on mode of failure and deformation of dermal allografts in the setting of tibiofemoral joint articulation is warranted.
Conclusion
Tibiofemoral joint contact pressure was restored to the intact condition after reconstruction of osteochondral fracture defects with dermal allograft matrix and subchondral implants for the repair of both 8-mm and 10-mm lateral condylar defects as well as 8-mm medial condylar defects, but not completely for 10-mm medial condylar defects.
Footnotes
Final revision submitted December 26, 2023; accepted February 12, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was partially supported by Subchondral Solutions, which also provided the implants and graft material used. D.T.D. has received hospitality payments from Globus Medical, has patents related to the study content, and has stock/stock options in Subchondral Solutions. M.H.M. has stock/stock options in Subchondral Solutions. T.Q.L. has stock/stock options in Subchondral Solutions. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
