Abstract
Background:
Extracorporeal shockwave therapy (ESWT) is a noninvasive treatment modality that is used in the treatment of chronic Achilles tendinopathy (AT).
Purposes:
To (1) retrospectively assess outcomes after ESWT for both noninsertional AT (NAT) and insertional AT (IAT) at >1-year follow-up and (2) identify potential predictors of outcomes.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
Chart review was conducted to identify patients who underwent ESWT for AT with a minimum of 1-year follow-up. Data collected and assessed included patient demographic characteristics, pathological characteristics including the location of AT (NAT or IAT), presence of a Haglund deformity, and severity of tendon degeneration on magnetic resonance imaging (MRI), in addition to treatment characteristics including number of sessions and intensity of ESWT. The Victorian Institute of Sports Assessment–Achilles (VISA-A) and visual analog scale (VAS) pain scores were obtained before ESWT, 6 months after ESWT, and at final follow-up. Failures were also recorded, which were defined as no improvement in VISA-A or VAS scores or need for surgical intervention. Linear regression was performed to identify potential predictors of inferior subjective clinical outcomes and failures. Survival analysis was conducted using Kaplan-Meier curves.
Results:
The study included 52 patients with IAT and 34 patients with NAT. The mean follow-up in the NAT cohort was 22.3 ± 10.2 months and the mean follow-up in the IAT cohort was 26.8 ± 15.8 months. Improvements in VISA-A and VAS scores were observed in the NAT cohort at 6-month follow-up and at final follow-up (P < .05). Improvements in VISA-A and VAS scores were recorded in the IAT cohort at 6-month follow-up, which subsequently deteriorated at final follow-up. In the NAT cohort, the failure rate at 6-month follow-up was 11.8%, which increased to 29.4% at final follow-up. In the IAT cohort, the failure rate at 6-month follow-up was 32.7%, which increased to 59.6% at final follow-up. Predictors of inferior subjective clinical outcomes and failures in the NAT cohort included pre-ESWT subjective clinical score, male sex, presence of a cardiovascular risk factor, and more severe MRI grading of tendinopathy. Predictors of inferior subjective clinical outcomes and failures in the IAT cohort included pre-ESWT subjective clinical score and more severe MRI grading of tendinopathy.
Conclusion:
Superior subjective clinical outcomes together with a lower failure rate were maintained for >1 year in the NAT cohort compared with the IAT cohort, calling into question the long-term benefit of ESWT for patients with IAT.
Keywords
Achilles tendinopathy (AT) is a common musculoskeletal injury and accounts for up to 18% of all injuries in runners. 2 AT can be subcategorized anatomically into insertional AT (IAT) and noninsertional AT (NAT). NAT is defined as degeneration of the Achilles tendon in a watershed area 2 to 6 cm from the insertion of the tendon into the calcaneus. 23 Conversely, IAT occurs 2 cm proximal to the attachment of the tendon on the calcaneal tuberosity and can be associated with retrocalcaneal bursitis, insertional calcific tendinosis, and bony protuberance from the posterosuperior aspect of the calcaneus. 15 AT, whether insertional or noninsertional, is a clinical diagnosis that can be further evaluated with ultrasound and magnetic resonance imaging (MRI) to assess the severity of the degenerated tendon. 25
Nonoperative management is the mainstay treatment for AT. Achilles eccentric strengthening and stretching exercises have produced satisfactory results at short-term to midterm follow-up. 16 Exercise rehabilitation provides mechanical loading to the tendon and the surrounding muscle belly of the gastrocnemius-soleus complex, which promotes tendon remodeling, improvement in gastrocnemius-soleus endurance and strength, and a reduction in tendon pain.21,22 Other first-line management strategies include nonsteroidal anti-inflammatory drugs, exercise modification, hyperthermia, taping, and splintage. 13 Regardless of the management used, prolonged loss of time from sporting activities is often frustrating for athletes and physicians alike.
Given the limitations of nonoperative management, other noninvasive modalities have gained interest. Extracorporeal shockwave therapy (ESWT) is a noninvasive treatment modality, not approved by the US Food and Drug Administration, that is often indicated after failure of first-line treatment modalities for AT. 13 ESWT produces focused longitudinal sound waves that create a biological cascade via mechanotransduction, inducing local neovascularization, cell proliferation, calcific resorption, downregulation of metalloproteinases, and inhibition of substance P.5,28 ESWT has been used in various sports medicine pathologies including plantar fasciitis, peroneal tendinopathy, lateral epicondylitis, patellar tendinopathy, and adductor tendinopathy. 20 In recent years, studies have been conducted to investigate the role of ESWT in AT, with good outcomes reported at final follow-up. 24 However, many of these studies are limited by follow-up times of <6 months together with small patient cohorts. 24
The purpose of this retrospective review was to evaluate clinical outcomes after ESWT for AT at a minimum 1-year follow-up. Furthermore, the current study subdivided IAT and NAT with regard to outcomes and predictors of success of treatment over 1 year. We hypothesized that ESWT would lead to improved clinical outcomes in patients with AT.
Methods
Patient Recruitment
This retrospective cohort study used chart review for retrieval of data on individual patients after approval by the institutional review board (i21-01478). All patients who underwent ESWT for AT by the senior author (J.G.K.) as part of standard clinical care between June 21, 2017, and December 28, 2020, were identified. The inclusion criteria were age >18 years at the time of ESWT, a minimum 12 months of follow-up, and the presence of a pre-ESWT MRI scan. Exclusion criteria included a history of diabetes, treatment with platelet-rich plasma (PRP), lack of baseline MRI, a history of prior Achilles tendon surgery, and inflammatory arthritic conditions such as rheumatoid arthritis, psoriatic arthritis, and gout. Figure 1 illustrates the patient selection process. A total of 112 patients (152 ankles) who underwent ESWT for the treatment of AT were identified. A total of 25 patients were excluded from the study due to a history of prior Achilles tendon surgery (n = 11) or peritendinous PRP injection (n = 14). In addition, 1 patient was lost to follow-up due to death from an unrelated illness. In total, 86 patients (105 ankles) were included in the study.
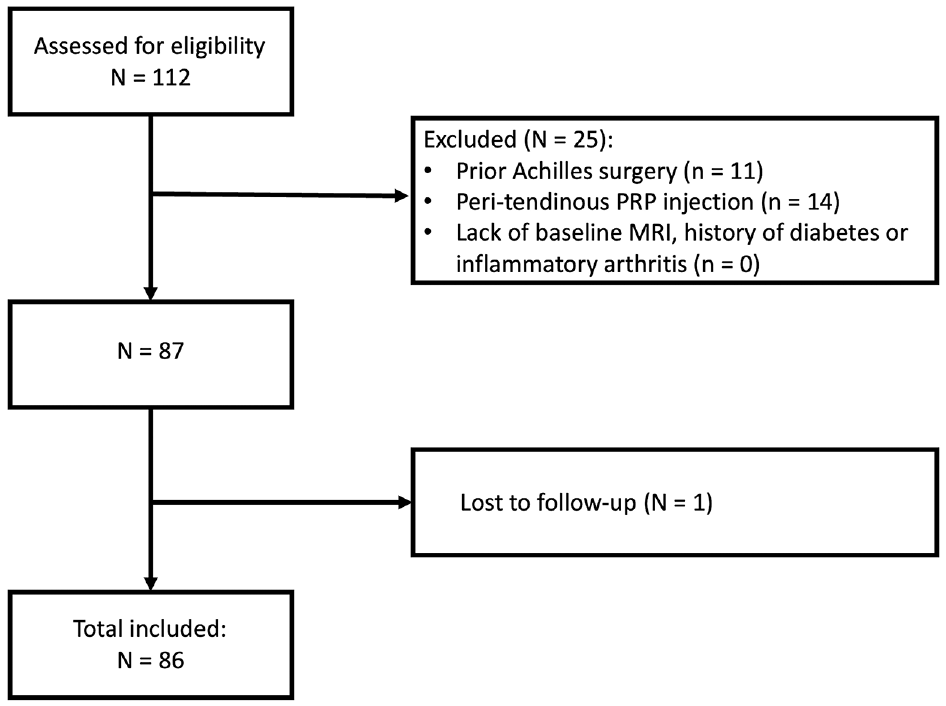
Flowchart demonstrating the patient selection process. MRI, magnetic resonance imaging; PRP, platelet-rich plasma.
Diagnostic and Treatment Characteristics
All patients obtained an ankle MRI at the time of presentation at the same radiology facility, which was reported by a single board-certified musculoskeletal radiologist. A senior orthopaedic attending surgeon (J.G.K) performed the same assessment and established intra- and interobserver reliability. The second MRI evaluation was conducted for all patients by both the board-certified musculoskeletal radiologist and the senior orthopaedic attending surgeon (J.G.K) at the time of data collection for this study in June 2022. The degree of tendinopathy was subdivided into 3 grades: (1) fusiform thickening involving less than one-third of the cross-sectional area (CSA) of the tendon, (2) fusiform thickening involving one-third to two-thirds of the CSA of the tendon with or without interstitial tears, and (3) fusiform thickening involving more than two-thirds of the CSA of the tendon with interstitial tears. The presence of a Haglund deformity, insertional calcific tendinosis, and/or retrocalcaneal bursitis was also recorded. No ultrasound evaluation of the Achilles tendon was obtained for any patients at our institution.
All patients underwent ESWT using the same focal ESWT machine (Duolith SD1; Storz Medical). The applicator was placed over the site of maximal tenderness without the concomitant use of local or topical anesthesia. Initially, patients received 500 introductory pulses starting at an energy flux density per shock of 0.07 mJ/mm2, gradually increasing to 0.1 mJ/mm2. After the introductory pulses, the energy flux density per shock was subsequently increased to and maintained at a moderate intensity of 0.12 to 0.17 mJ/mm2 for a total of 2000 pulses per session. Each patient underwent 1 ESWT session per week for a total of 3 weeks.
Although all patients in this review had experienced failure of at least 1 treatment protocol of at least 3 to 6 weeks of physical therapy before ESWT, the protocols were heterogeneous and included eccentric regimens, functional isometric regimes, and other less formal therapy strategies. Once patients were enrolled for ESWT, a standard eccentric physical therapy program was prescribed and performed on all patients starting after the second ESWT treatment and lasting for 3 weeks thereafter. If significant improvement in clinical symptoms after the third session of ESWT was noted, patients were advised to exercise within the confines of their symptoms and present for reevaluation at 6 months after ESWT. If no improvement and/or a deterioration in clinical symptoms after the third session of ESWT was noted, patients were recommended to undergo surgical intervention via tendoscopic Achilles debridement.
Data Collection
Date of birth, medical record number, date of initial ESWT session, prior treatments including physical therapy and corticosteroid injections, pre-ESWT MRI, and patient demographic characteristics such as age, sex, body mass index (BMI), past medical history, laterality, follow-up period, and time from ESWT to operative intervention were collected. Victorian Institute of Sports Assessment–Achilles (VISA-A) 18 and visual analog scale (VAS) scores 9 were collected before the first ESWT session, at the routine 6-month follow-up appointment, and at final follow-up. Patients who did not present for an in-person assessment at 12 months after ESWT were contacted by telephone to assess their clinical status and potential failure. VISA-A and VAS questionnaires were provided to these patients by email and were returned to the office. Final follow-up was defined as the most recent in-person or telephone assessment after ESWT or before surgical intervention after failure of ESWT. Failure after ESWT was also recorded, which was defined as no improvement in VISA-A or VAS scores or a need for surgical intervention.
Statistical Analysis
All statistical analyses were performed using RStudio software (Version 3.3.0). Mann-Whitney U test was performed to compare pre- and post-ESWT VISA-A and VAS scores. The Welch t test was performed to compare post-ESWT VISA-A and VAS scores between the IAT and NAT cohorts. Chi-square analysis was conducted to assess whether differences in failure rates between the IAT and NAT cohorts existed. Univariate linear regression was used to assess the independent factors predictive of the post-ESWT VISA-A scores, post-ESWT VAS scores, and failure after ESWT. The independent variables included in the model were pre-ESWT VISA-A, pre-ESWT VAS, age, sex, BMI, history of smoking, presence of a cardiovascular risk factor (hypertension, hypercholesterolemia, obesity, current smoker), prior corticosteroid injection, severity of tendinopathy based on MRI findings, and location of tendinopathy. A P value of <.05 was considered significant. Kaplan-Meier analysis was performed to evaluate survivorship at final follow-up. The intraobserver reliability for the senior musculoskeletal radiologist's repeated evaluation of the severity of AT on MRI for all patients was assessed using the intraclass correlation coefficient (ICC). Interobserver reliability between the senior musculoskeletal radiologist and senior orthopaedic resident regarding MRI assessment of the severity of AT was also evaluated via ICC.
Results
Patient Demographic Characteristics
Patient demographic and pathological characteristic data are listed in Table 1. A total of 52 patients (67 ankles) presented with IAT, and 34 patients (38 ankles) presented with NAT. In the NAT cohort, 23 patients (67.6%) were male and 11 patients (32.4%) were female. In the IAT cohort, 33 patients (63.5%) were male and 19 patients (36.5%) were female. The mean age of the NAT cohort was 54.2 ± 15.0 years (range, 18-83 years), and the mean age of the IAT cohort was 53.0 ± 14.4 years (range, 19-79 years). The mean BMI in the NAT cohort was 25.9 ± 5.3 kg/m2 (range, 19.6-39.5 kg/m2), and the mean BMI in the IAT cohort was 27.7 ± 4.4 kg/m2 (range, 20.1-38.5 kg/m2). The mean follow-up time in the NAT cohort was 22.3 ± 10.2 months (range, 12.0-68.0 months), and the mean follow-up time in the IAT cohort was 26.8 ± 15.8 months (range, 12.0-53.0 months). In the NAT cohort, 30 patients (88.2%) presented with unilateral AT (17 right-sided, 13 left-sided) and 4 patients (11.8%) presented with bilateral AT. In the IAT cohort, 37 patients (71.2%) presented with unilateral AT (21 right-sided, 16 left-sided) and 15 patients (28.8%) presented with bilateral AT. In the NAT cohort, 13 patients (38.2%) had a cardiovascular risk factor and 1 patient (2.9%) had a prior intratendinous corticosteroid injection. In the IAT cohort, 23 patients (44.2%) had a cardiovascular risk factor and 7 patients (13.5%) had a prior peritendinous corticosteroid injection.
Patient Demographic and Pathological Characteristics a

Data are expressed as mean ± SD or n (%) unless otherwise noted. AT, Achilles tendinopathy; MRI, magnetic resonance imaging; NA, not applicable.
Pathological Characteristics
In the IAT cohort, 20 patients (38.4%) had a Haglund deformity and 11 patients (21.2%) had retrocalcaneal bursitis. In the NAT cohort, MRI demonstrated that 23 patients (67.7%) had grade 1 AT, 6 patients (17.7%) had grade 2 AT, and 5 patients (14.7%) had grade 3 AT. In the IAT cohort, MRI demonstrated that 17 patients (32.7%) had grade 1 AT, 14 patients (26.9%) had grade 2 AT, and 21 patients (40.4%) had grade 3 AT. The intraobserver reliability for MRI assessment of severity of AT was 0.973 (95% CI, 0.925-0.981; P < .001). The interobserver reliability for MRI assessment of severity of AT between the senior musculoskeletal radiologist and senior orthopaedic resident was 0.968 (95% CI, 0.938-0.981; P < .001). The mean duration of symptoms before ESWT in the NAT cohort was 12.5 ± 14.3 months (range, 3-60 months), and the mean duration of symptoms before ESWT in the IAT cohort was 17.9 ± 15.1 months (range, 3-60 months).
Clinical Outcomes: NAT Versus IAT
Clinical outcomes after ESWT for chronic AT are listed in Table 2. In the NAT cohort, VISA-A scores improved after treatment with ESWT, from a pre-ESWT score of 49.6 ± 14.9 (range, 18-82) to a post-ESWT score of 76.2 ± 21.6 (range, 25-96) at 6-month follow-up to a post-ESWT score of 74.7 ± 23.3 (range, 21-96) at final follow-up (P < .0001). In the IAT cohort, VISA-A scores improved after treatment with ESWT, from a pre-ESWT score of 46.6 ± 14.9 (range, 21-79) to a post-ESWT score of 63.9 ± 23.2 (range, 19-98) (P < .0001) at 6 months of follow-up. However, VISA-A scores subsequently deteriorated in the IAT cohort to a post-ESWT score of 54.7 ± 26.5 (range, 19-98) at final follow-up (P < .001). There was no statistically significant difference between pre-ESWT VISA-A scores in the NAT and IAT cohorts (P = .4831). There were statistically significant, higher post-ESWT VISA-A scores in the NAT cohort compared with the IAT cohort at 6-month (P = .014) and at final (P = .001) follow-up.
Summary of Clinical Outcomes a

Data are expressed as mean ± SD or n (%). AT, Achilles tendinopathy; ESWT, extracorporeal shockwave therapy; VISA-A, Victorian Institute of Sports Assessment–Achilles; VAS, visual analog scale.
In the NAT cohort, VAS scores significantly decreased after treatment with ESWT, from a pre-ESWT score of 5.9 ± 1.6 (range, 3-8) to a post-ESWT score of 2.4 ± 2.3 (range, 0-6) at 6-month follow-up to a post-ESWT score of 2.8 ± 2.5 (range, 0-7) at final follow-up (P < .0001). In the IAT cohort, VAS scores significantly decreased after treatment with ESWT at 6-month follow-up, from a pre-ESWT score of 6.1 ± 1.5 (range, 3-10) to a post-ESWT score of 3.8 ± 3.1 (range, 1-8) (P < .0001). However, VAS scores subsequently deteriorated in the IAT cohort to a post-ESWT score of 4.9 ± 2.9 (range, 2-8) at final follow-up (P = .024). No statistically significant difference was seen between pre-ESWT VAS scores in NAT and IAT cohorts (P = .5913). A statistically significant, lower post-ESWT VAS score was seen in the NAT cohort compared with the IAT cohort (P = .0009).
Linear Regression Analysis: NAT Cohort
Linear regression analysis demonstrated that pre-ESWT VISA-A score (P = .0008; R2 = 0.2926), male sex (P = .028; R2 = 0.138), presence of a cardiovascular risk factor (P = .0098; R2 = 0.1855), and increasing MRI grading of tendinopathy (P = .0002; R2 = 0.354) had a statistically significant negative correlation with post-ESWT VISA-A scores (Table 3). Pre-ESWT VAS score (P = .0075; R2 = 0.1952), presence of a cardiovascular risk factor (P = .0037; R2 = 0.2287), and increasing MRI grading of tendinopathy (P = .0008; R2 = 0.29) had a statistically significant negative correlation with post-ESWT VAS scores (Table 3). Male sex (P = .0064; R2 = 0.2045) had a statistically significant correlation with failure (Table 3). The strongest correlator for post-ESWT VISA-A scores and post-ESWT VAS scores in the NAT cohort was increasing MRI grading of tendinopathy.
Correlation Analysis for Noninsertional AT a

AT, Achilles tendinopathy; ESWT, extracorporeal shockwave therapy; MRI, magnetic resonance imaging; NA, not applicable; VAS, visual analog scale; VISA-A, Victorian Institute of Sports Assessment-Achilles.
Linear Regression Analysis: IAT Cohort
Linear regression analysis demonstrated that pre-ESWT VISA-A score (P < .0001; R2 = 0.4105) and increasing MRI grading of tendinopathy (P < .0001; R2 = 0.4599) had a statistically significant negative correlation with post-ESWT VISA-A scores (Table 4). Pre-ESWT VAS score (P = .0246; R2 = 0.09701) and increasing MRI grading of tendinopathy (P < .0001; R2 = 0.4278) had a statistically significant negative correlation with post-ESWT VAS scores (Table 4). Increasing MRI grading of tendinopathy (P < .0001; R2 = 0.5825) had a statistically significant correlation with failure (Table 4). The strongest correlator for post-ESWT VISA-A scores, post-ESWT VAS scores, and failure in the IAT cohort was increasing MRI grading of tendinopathy.
Correlation Analysis for Insertional AT a

AT, Achilles tendinopathy; ESWT, extracorporeal shockwave therapy; MRI, magnetic resonance imaging; NA, not applicable; VAS, visual analog scale; VISA-A, Victorian Institute of Sports Assessment–Achilles.
Failures and Survivorship
Data regarding failures are listed in Table 2. Failure was defined as no improvement in VISA-A or VAS scores or the need for surgical intervention. At 6-month follow-up, 4 failures (11.8%) were recorded in the NAT cohort and 17 failures (32.7%) were recorded in the IAT cohort. At final follow-up, 10 failures (29.4%) were recorded in the NAT cohort and 31 failures (59.6%) were recorded in the IAT cohort. A statistically significant, higher failure rate in the IAT cohort (59.6%) compared with the NAT cohort (29.4%) (P = .0081) was seen. Five patients (14.7%) with refractory NAT underwent surgical intervention, and 19 patients (36.5%) with refractory IAT underwent surgical intervention. All 24 patients underwent arthroscopic Achilles tenosynovectomy and debridement. Of the 20 patients with Haglund deformity, 13 patients (65.0%) required resection of a Haglund deformity. The mean time from ESWT to surgical intervention in the NAT cohort was 5.4 ± 3.1 months (range, 2-10 months) and the mean time from ESWT to surgical intervention in the IAT cohort was 3.6 ± 1.8 months (range, 1-7 months) (P = .2783).
Survival analysis was conducted using Kaplan-Meier survival curves (Figure 2). The Kaplan-Meier survival curve showed a statistically significant difference between the NAT and IAT cohorts with regard to time to failure after ESWT (log-rank test: P = .0035).
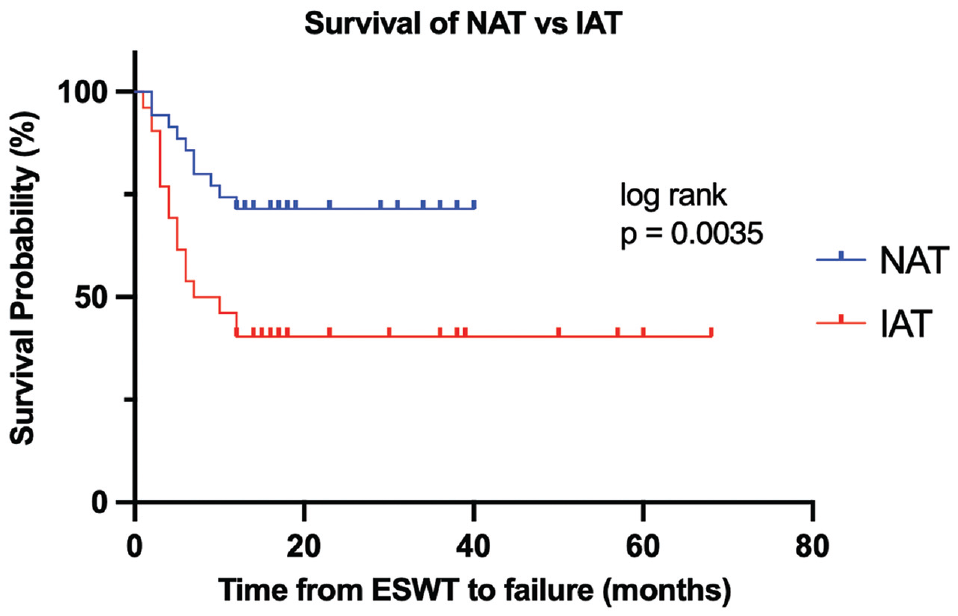
Survivorship as demonstrated via Kaplan-Meier curve. ESWT, extracorporeal shockwave therapy; IAT, insertional Achilles tendinopathy; NAT, noninsertional Achilles tendinopathy.
Discussion
This retrospective study demonstrated improvement in subjective clinical outcomes after ESWT for treatment of both NAT and IAT with no complications observed at 6 months of follow-up. At >1 year of follow-up after ESWT, a statistically significant higher failure rate was present in the IAT cohort (59.6%) compared with the NAT cohort (29.4%), calling into question the long-term benefit of ESWT for patients with IAT.
The Achilles tendon is composed of well-organized tenocytes and tenoblasts surrounded by predominantly type 1 collagen tightly packed in parallel bundles. 11 Excessive loading of the tendon during vigorous physical exercise precipitates tendinopathic degeneration. The failed, erratic healing response produces a haphazard proliferation of tenocytes together with disruption of the collagen fiber network, collagen microtears, and increase in noncollagenous matrix. 13 This process occurs in both NAT and IAT, and therefore any therapies targeting restoration of the normal ultrastructure of the tendon are beneficial in regaining normal function. Eccentric Achilles tendon strengthening exercises are commonly used to treat both types of AT and lead to tenocyte hypertrophy, suppression of neurovascular ingrowth, and reduction in glycosaminoglycan content. Despite these beneficial changes, there is no consensus as to the effectiveness of this modality nor is there a robust relationship between the histological changes and clinical outcomes provided by eccentric exercises. 17 Therefore, in patients with more severe pathology, biological augmentation via ESWT may be warranted to improve the biological milieu of the tendon structure to facilitate accelerated healing and sustain histological as well as clinical improvements. ESWT promotes tissue regeneration by stimulating tenocytes to produce type 1 collagen, reducing the expression of destructive matrix metalloproteinases and decreasing the synthesis of catabolic interleukins.7,27 In addition, ESWT has a profound influence on pain transmission by interfering with substance P, neurovascular sprouting, and calcitonin gene-related peptide expression in the dorsal root ganglion.8,14,26 These effects should have a positive effect on histological and clinical outcomes in both types of AT.
This retrospective review found improvements in VISA-A and VAS scores at >1-year follow-up after treatment with ESWT for AT, highlighting the functional and analgesic benefits provided by ESWT. Previous studies have demonstrated similar improvements in subjective clinical outcomes after ESWT for AT compared with physical therapy alone. A double-blind randomized control trial by Abdelkader et al 1 found that ESWT potentiated the effects of calf eccentric loading exercises in patients with NAT. At 16 months of follow-up, a statistically significant higher VISA-A score was observed in the ESWT cohort (80 ± 5.3) compared with the control group (67 ± 5.6). Furthermore, Yan et al 30 conducted a comparative study between active and nonactive patients with IAT who underwent ESWT and found improvements in VISA-A and VAS scores in both cohorts at final follow-up.
In the current study, subgroup analysis illuminated significant discrepancies in outcomes between the NAT and IAT cohorts. The NAT cohort had statistically significant, higher VISA-A and lower VAS scores at final follow-up compared with the IAT cohort. In addition, a higher failure rate was found in the IAT cohort compared with the NAT cohort. These findings must be interpreted in the context of the mechanical, biological, and clinical differences that exist between the 2 patient populations. A total of 38.4% of the IAT cohort presented with a Haglund deformity. The mechanical impingement of the Achilles tendon due to these bony prominences leads to profound degeneration of the tendon, as represented by the high levels of grades 2 and 3 AT in this cohort. As a result, in these cases of severe tendinopathy, ESWT is inadequate to address both the diseased tendon and mechanical bony impingement, and more aggressive surgical intervention is required. The results from our study are consistent with the current literature, as evidenced by Wu et al, 29 who demonstrated inferior VISA-A and Likert scores after ESWT for IAT in the Haglund deformity cohort compared to the non–Haglund deformity cohort in addition to a 3.45 times higher risk of treatment failure. The current study shows that ESWT may provide short-term clinical benefit in patients with IAT for up to 6 months. This can be beneficial with regard to surgical planning in the athletic population, as treatment with ESWT may facilitate athletes to participate in training and competition during the in-season and undergo surgical intervention in the off-season.
NAT is believed to be in part due to a relative vascular watershed area of the tendon and an inability to regenerate tenocytes after repetitive mechanical loading. 12 It is purported that this poorly vascularized region predisposes patients to tendinopathic degeneration after repetitive traumatic insults. ESWT promotes neovascularization, 6 leading to good clinical outcomes in this study, although there was a moderate failure rate in this cohort. The current study did not evaluate the vasculature or the histology of the treated tendon, but the improved clinical outcomes tend to confer the beneficial effect of the neovascularization of the tendon affected by ESWT demonstrated in a previous study. 19 Interestingly, linear regression analysis in the current retrospective study demonstrated a statistically significant negative correlation between the presence of a cardiovascular risk factor and subjective clinical outcomes in the NAT cohort. This suggests that ESWT may not be an effective treatment modality in vasculopathic patients with NAT compromised blood supply; however, further research on the ability to promote neovascularization by ESWT is warranted.
Our study demonstrated that in the NAT cohort, male sex was an independent predictor for poorer post-ESWT VISA-A scores and post-ESWT failure. Multiple in vitro and biomechanical studies have examined the impact of biological sex on tendon pathology. Bryant et al 4 found that estrogen secretion promoted increased collagen synthesis to maintain the strain of the Achilles tendon, thus potentially reducing the incidence of AT. Furthermore, Andrew et al 3 evaluated Achilles tendon loading between 15 male and 15 female runners. Those authors found statistically significant, greater Achilles tendon loads in the male cohort compared with the female cohort, suggesting that men may be at a greater risk of AT due to the excess stress across the tendon. Intziegianni et al 10 demonstrated that women exhibited increased Achilles tendon cross-sectional area compliance compared with men, indicating that female Achilles tendons may possess a superior adaptive ability to high loads, thus reducing the risk of AT. The histological and biomechanical differences between male and female Achilles tendons may account for the discrepancies in clinical outcomes between the 2 cohorts; however, further comparative studies are necessary to determine the impact of sex on outcomes after ESWT for AT.
Limitations
This retrospective study has several limitations and inherent biases. The primary limitation is the short-term follow-up. Although this provides useful analysis of short-term outcomes, robust conclusions regarding the duration of symptom relief and duration between ESWT and potential surgery cannot be drawn. Second, no imaging at final follow-up was recorded to definitively assess whether the severity of AT had improved or deteriorated. The mean age across both cohorts was approximately 50 years with small standard deviations, which limits the generalizability of these findings to younger and older populations. Last, no control group was included in this study, which limits the true understanding of the potential benefits of ESWT versus time alone. A double-blind, randomized controlled trial with a larger patient cohort and longer follow-up period is necessary to clarify the role ESWT in the management of AT.
Conclusion
This study found that ESWT for patients with NAT and those with IAT led to improved subjective clinical outcomes at short-term follow-up. Superior subjective clinical outcomes together with a lower failure rate were maintained for >1 year in the NAT cohort compared with the IAT cohort. Therefore, ESWT may be more beneficial in the long-term treatment of NAT, whereas it may be considered a temporizing treatment for IAT. These findings will help in establishing treatment protocols as well as patient expectations for patients with chronic AT.
Footnotes
Final revision submitted January 24, 2024; accepted February 23, 2024.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.G.K. has received consulting fees from Arteriocyte Medical Systems, Arthrex, and In2Bones USA LLC; nonconsulting fees from Arthrex; education payments from Gotham Surgical Solutions & Devices and Arthrex; and honoraria from In2Bones USA LLC. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from NYU Langone Health (ref No. i21-01478).
