Abstract
Background:
Allograft tendons are perceived to have a high ACL graft failure rate in primary anterior cruciate ligament (ACL) reconstruction (ACLR). Historical series may be biased by graft processing methods that degrade the biomechanical properties of donor tendons such as irradiation. Supercritical carbon dioxide (SCCO2) is a validated method of terminally sterilizing biomaterials at physiological temperatures without irradiation, but in vivo use of SCCO2-processed tendon allografts for primary ACLR has not been reported to date.
Hypothesis:
ACLR with SCCO2 allografts would result in acceptable failure rates, subjective knee scores, and clinical evaluation at 2 years postoperatively.
Study Design:
Case series; Level of evidence, 4.
Methods:
Patients underwent primary ACLR with terminally sterilized SCCO2-processed human gracilis, peroneus longus, semitendinosus, tibialis anterior, and tibialis posterior tendon allografts. Patient demographics were collected, along with tendon donor age and sex. At 1 year postoperatively, subjective International Knee Documentation Committee (IKDC) and ACL–Return to Sport After Injury (ACL-RSI) scores were collected, as well as clinical evaluation. At 2 years postoperatively, the IKDC and ACL-RSI scores were repeated, and return to sports and further knee injuries were recorded.
Results:
A total of 144 patients with a medianage of 26 (IQR 14) years formed the study group. Patients were predominately male (58%). The loss to follow-up rate was 8% (n = 12). The mean age of allograft tendon donors was 37 (range 17-58) years, and the majority were male (83%). The mean allograft diameter was 8.9 ± 1.0 mm. At 2 years, ACL graft failureoccurred in 5% (n = 7). All graft failureswere in patients aged ≤25 years (P = .007). Neither donor age (≤40 or >40 years) nor donor sex was associated with graft failure (P > .05). The median IKDC subjective score was 95 and ACL-RSI score was 75. There were no revisions for sepsis within the first 2 years postoperatively.
Conclusion:
SCCO2 processing of allograft tendons demonstrated satisfactory clinical and patient-reported outcomes at 24 months postoperatively in a consecutive series of patients with primary ACLR, with similar ACL graft failure rates and subjective knee scores compared with those reported in published series of hamstring tendon autograft and fresh frozen nonirradiated allograft.
The anterior cruciate ligament (ACL) is the most common knee ligamentous injury requiring surgery, and the global demand for ACL reconstruction (ACLR) is increasing in all age groups.14,18,48,53 Choice of graft to reconstruct the ACL is a critical decision for the surgeon and is typically based on patient factors, patient expectations, surgical experience, and graft properties.11,25,28 Practice guidelines demonstrate significant disagreement regarding allograft use in primary ACLR due to concerns of high failure rates.1,4,9 As a result, significant international practice variation is evident with use of allografts ranging between 1% and 42% of primary ACLR procedures.10,13,14,18,19,46
Benefits of allograft include absence of donor-site morbidity and reduced postoperative pain compared with autografts, as well as predictable graft size compared with hamstring tendon (HT) autografts. 11 HT autograft harvest is associated with persistent hamstring atrophy and decreased isokinetic torque at high flexion angles at long-term follow-up that may impair athletic performance in high-demand patients.11,23 Bone-patellar tendon-bone autograft harvest is associated with persistent anterior knee pain and, rarely, patellar fracture, patellar tendon rupture, and patellofemoral chondral degeneration.24,31 However, while allografts may avoid these potential complications, observational studies report a higher time-dependent risk of ACL graft failure compared with autografts, especially for irradiation-based sterilization techniques.7,20,26,48 Clinical studies and meta-analyses support these findings but suggest that heterogeneous graft-processing methods may be influencing the incidence of allograft failures. 27 Similar rerupture rates between autografts and unprocessed allografts have been reported, for example, once adjustment is performed for age, sex, and ethnicity. 26 Alternative allograft processing techniques that maintain the structural integrity of collagen without compromising sterility or immunogenicity of donor tissues are therefore desirable.
Supercritical carbon dioxide (SCCO2) has emerged as a versatile technique for terminally sterilizing biomaterials using readily available CO2 gas at physiological temperatures, preserving the microarchitecture of heat- and radiation-sensitive biomaterials, such as collagen.6,33,36,42 CO2 is in a supercritical state when it demonstrates intermediate gaseous and fluid phase characteristics at a low temperature (≤40°C) and relatively low pressure (≤7600 kPA), allowing it to effuse through biomaterials like a gas and dissolve lipids like a liquid, inactivating bacterial or viral contaminants. 39 An in vitro study has demonstrated that it preserves the biomechanical properties of collagen-based biomaterials, such as tendon and bone, while achieving adequate sterilization for implantation. 16 SCCO2-processed tendon xenografts have been used in a rabbit model of ACLR, but no in vivo clinical studies of SCCO2-processed human allografts for ACLR have been published to date. 37
The purpose of this study was to report ACL graft reinjury rates of patients undergoing primary ACLR using human allografts processed with chlorhexidine disinfectant and terminal SCCO2 sterilization, in a prospective cohort at 1 and 2 years postoperatively, as well as to report validated knee scores and clinical assessments. We hypothesized that processed human allografts with this technique would have acceptable clinical outcomes at 2 years after ACLR.
Methods
Patient Selection
Patients who underwent ACLR with SCCO2 tendon allograft between May 2019 and December 2020 under the care of 2 senior surgeons (L.A.P., J.P.R.) were eligible for inclusion (n = 213 patients). Inclusion criteria were isolated ACLR, no injury to the contralateral knee, consent to use of SCCO2-sterilized tendon, not receiving compensation for injury, and consent to participate in a prospective research database. Of the 213, 69 patients were excluded for the following reasons: more than one-third medial or lateral meniscectomy (n = 42); other significant ligamentous injury to the knee (n = 18); moderate or severe osteoarthritis evident at arthroscopy, defined as grades 3 or 4 on Outerbridge classification 30 (n = 8); and concurrent traumatic cartilage lesion requiring microfracture (n = 1), in an effort to reduce the impact of confounding factors. The remaining study group consisted of 144 patients.
The primary outcome was ACL graft reinjury rates at 1 and 2 years postoperatively in patients receiving SCCO2-sterilized allografts, defined as clinical or radiologic evidence of graft failure. The secondary outcomes were patient-reported outcome measures at 1 and 2 years postoperatively.
Ethical approval was obtained from a local independent human research ethics committee (St Vincent's Human Research Ethics Committee; ref No. IS/305). Reconstruction was performed after the knee had recovered from the acute trauma of the ACL injury and patients had a pain-free, mobile joint.
Graft Processing
The tendon allografts were retrieved from donor tissue obtained from accredited organ procurement organizations. Donors were screened for transmissible diseases, including human immunodeficiency virus 1 and 2, hepatitis B and C, syphilis, and human T-lymphotropic virus 1 and 2. Donor history and next of kin interviews were obtained to identify any history of malignancy or intravenous substance use. The tendons included within the study included gracilis, tibialis anterior, tibialis posterior, peroneus longus, and semitendinosus.
Graft processing was performed by Australian Biotechnologies, listed under the Australian Register of Therapeutic Goods and regulated by the Therapeutic Goods Administration to ensure that good manufacturing practice standards of human musculoskeletal tissues are met.43,44 The tissue was inspected to ensure that tendons met the required specifications for further processing.
The process employed by tissue banks to disinfect allografts can be subdivided into “disinfection methods” and “terminal sterilization methods.” 21 Disinfection methods include chemical and antibiotic treatments that target microorganisms, whereas terminal sterilization methods traditionally include irradiation, ethylene oxide, or heat treatments and eliminate living microorganisms to a particular sterility assurance level. SCCO2 processing is considered a terminal sterilization method. 21
The disinfection method employed an aqueous 0.5% chlorhexidine soak for 15 minutes to reduce potential bioburden in the graft and a warm saline lavage employed to rinse the sample. Low-speed centrifugation was used to remove excess moisture, and the grafts were further inspected with calibrated instruments to record the specifications of each unique graft. Microbiological sampling was performed on each donor batch to confirm the aseptic process of the graft before packaging. The tendons were individually packaged in a vapor-permeable barrier and stored at −80°C (–112°F) in preparation for terminal sterilization.
For terminal sterilization, SCCO2 processing was employed. The tendons were thawed. A cellulose pad soaked with a mixture of peracetic acid and hydrogen peroxide (NovaKill Gen 2; NovaSterilis) was used in the bottom of the pressure vessel before loading with the tendon allografts. The tendons were then loaded in the pressure vessel and treated with a proprietary SCCO2 sterilization protocol at 9900 kPa (143 psi) and a physiological temperature of 36°C (96.8°F) for 60 minutes. The grafts were preserved within the sealed pouches and stored at –80°C (–112°F) until surgery.
Operative Technique and Rehabilitation
A standardized surgical technique and postoperative rehabilitation protocol were followed for all patients and has been described previously. 34 The tendon allograft was prepared as either a 2-, 3-, 4-, or 6-strand graft using a No. 1 Vicryl (Ethicon) whipstitch to achieve a suitable size appropriate for the patient's anatomy. Allografts were wrapped in vancomycin-soaked gauze before implantation. Anatomic femoral tunnel drilling was performed via the anteromedial portal for a target tunnel position 5 mm anterior to the posterior wall of the lateral femoral condyle in the intercondylar notch. The tibial tunnel was drilled one-third of the way between the anterior horn insertion of the lateral meniscus and medial tibial spine. Femoral and tibial graft fixation was achieved with a PEEK RCI interference screw (Smith+Nephew) in all patients.
Surgery was performed as an outpatient procedure. Patients were permitted to bear weight as tolerated using crutches immediately after surgery; a brace was not used. A rehabilitation program was instituted, supervised by a physical therapist with the aim of achieving full extension by 2 weeks and full range of motion by 6 weeks. Measurement of medial tibial posterior slope, defined as the angle between the medial tibial plateau and the anatomic tibial axis (using landmarks of center of tibial plafond and midpoint of the tibial shaft, 20% distal to the tibial plateau), on EOS imaging was performed within 6 weeks of ACLR. The rehabilitation protocol included progressive intensive plyometric, agility, and sports-specific training. Return to competitive sport involving jumping, pivoting, or side-stepping was not advised until sport-specific and physical therapist-led criteria were met 9 to 12 months after the reconstruction.
Patient-Reported Outcome Measures and Clinical and Radiological Evaluation
Patients were evaluated via clinical examination at 12 months after index surgery using the International Knee Documentation Committee (IKDC) Knee Ligament Evaluation. 17 Ligament laxity was assessed with Lachman and pivot-shift tests and the KT-1000 arthrometer (Medmetric Corp) using the side-to-side difference of manual maximum anterior displacement between knees. A 1-year assessment was performed by either an experienced physical therapist (L.J.S.) or a knee research exercise physiologist, as well as the treating surgeon (L.A.P., J.P.R.). The single-leg hop test was used for functional assessment. Lysholm Knee Score 8 and ACL–Return to Sport After Injury (ACL-RSI) score 49 were also collected. Patients were asked whether they returned to their previous level of sports after surgery, had any family history of ACL injury, and had sustained any further knee injuries.
At 24 months after surgery, patients completed patient-reported outcomes including the IKDC Knee Ligament Evaluation and ACL-RSI scale and indicated whether they had returned to their previous level of sport after surgery and sustained any further knee injuries. Clinical examination was not performed at 24 months.
An ACL graft failureor contralateral ACL injury was considered to have occurred if any of the following was present: (1) further knee ACLR performed in our unit or by another orthopaedic surgeon; (2) clinical examination and/or magnetic resonance imaging findings evaluated by our unit that confirmed ACL deficiency/instability; or (3) report of another injury characteristic of an ACL tear such as a noncontact pivoting injury associated with instability and effusion, to either the reconstructed or the native knee, that had not been evaluated by our unit. For this last group, it was assumed that an ACL graft failureor contralateral ACL injury was a worst-case scenario. Any patients reporting knee injury or instability at 2 years postoperatively were invited for clinical review at our clinic to confirm ACL stability.
Statistical Analysis
Statistical analysis was performed with SPSS Version 29 (IBM). Means were compared between groups with independent t test. Proportions were compared between groups with chi-square test or the Fisher exact test if cell counts were <5. Statistical significance was set at P < .05. Univariate binary regression analysis was performed to assess the influence of graft donor age, graft donor sex, patient age, patient sex, posterior medial tibial slope, and family history of ACL injury on ACL graft failure. Odds ratios and 95% confidence intervals (CIs) are reported.
Results
Study Group
In total, 144 patients formed the study group, of which 126 (88%) were evaluated at 1 year and 132 (92%) were evaluated at 2 years and included in the analysis. The loss to follow-up rate was 8% (12/144 patients). There was no difference in the median age (P = .310) or sex distribution (P = .624) between those evaluated at 2 years and those lost to follow-up. Details of the patients at surgery are provided in Table 1. Radiographic posterior tibial slope was assessed in 121 of 132 patients and was missing in 11 patients.
Patient Characteristics a

ACL, anterior cruciate ligament; IQR, interquartile range.
Allograft Characteristics
Similar proportions of peroneus longus (25%), semitendinosus (18%), tibialis anterior (28%), and tibialis posterior (23%) tendon allografts were used (Figure 1). A higher proportion of allografts were sourced from male donors (n = 109; 83%), with 50% aged <40 or less years (66/132). The mean allograft diameter was 8.9 ± 1.0 mm. The mean donor age was 37 years (range, 17-58 years). The distribution is shown in Figure 2. The allograft tendon was formed into 2 strands in 57 (43%), 3 strands in 28(21%), 4 strands in 45(34%), 6 strands in 2 (2%).

Distribution of allograft type.

Distribution of allograft donor age.
ACL Graft Failure
ACL graft failure occurred in 7 (5%) patients at 2 years. Three patients (2%) sustained a contralateral ACL rupture at 22, 33, and 35 months. The mechanism of injury and characteristics of those who sustained an ACL graft failureare shown in Table 2. In 6 of the 7 patients ACL graft rupture was confirmed by clinical examination and MRI scan at our institution. One 17 year old female was classified as graft failure without a history of trauma on the basis of clinical laxity to examination. She had 6mm side to side difference on KT1000 manual maximum testing at 12 months with a positive pivot shift test and MRI scan confirmed an intact ACL graft. The mean diameter of failed grafts was 9.5 mm and was not significantly different from those with intact ACL grafts (8.9 mm; P = .10). Three ACL graft failureswere to peroneus longus tendons. The mean posterior medial tibial slope was 12° in those with failed grafts and 11° in those with intact ACL grafts (P = .44). The median age of those who sustained an ACL graft failurewas significantly younger than of those with intact ACL grafts (19 vs 27 years; P = .02). Prevalence and univariate binary regression analysis of selected factors (sex, age, posterior tibial slope, family history of ACL rupture, allograft donor age, and allograft donor sex) on ACL graft failureis shown in Table 3. All patients with ACL graft failure were aged ≤25 years, and a significant difference in ACL graft failure rates between patients ≤25 years old (11%) compared with those >25 years old (0%) was observed (P = .007, Fishers exact test). ACL graft failureoccurred in 5 (10%) of those with a posterior tibial slope ≥12° and 2 (3%) of those with a posterior tibial slope <12° (P = .094).
Characteristics of Patients Sustaining ACL Graft Failure a

ACL, anterior cruciate ligament; F, female; M, male.
Prevalence and Binary Logistic Regression of ACL Graft Failure a

ACL, anterior cruciate ligament.
Posterior tibial slope measures were missing in 11 patients.
Odds ratio not calculated as cell count = 0.
The patients who were excluded because of concurrent injuries (n = 69) were also sent the 2-year survey regarding further knee injuries, and 65 (94%) responded. No further ACL graft injuries were reported. The rate of ACL graft failurein the consecutive series with 2 year follow up was 4% (7/197).
Patient-Reported Outcomes
Patient-reported outcomes were completed by the 125 patients with intact ACL graft at 24 months. The median IKDC subjective score was 95 (interquartile range [IQR] = 14). The median ACL-RSI score was 75 (IQR = 36), and 85 (68%) had an ACL-RSI score of ≥60, indicating greater psychological readiness to return to sports. The distribution of the ACL-RSI score is shown in Figure 3. At 12 months, the median Lysholm Knee Score was 95 (IQR = 11), and the mean was 92 ± 10.

Distribution of Anterior Cruciate Ligament–Return to Sport After Injury (ACL-RSI) score at 2 years.
In total, 85 (68%) of the 125 patients reported that they had returned to their preinjury level of activity. Of the 40 patients who had not returned to sports, 29 (73%) reported that this was due to their knee. The distribution of current regular activity level reported is shown in Figure 4. A total of 86 (69%) patients reported that their knee did not restrict their activity level. Kneeling difficulty was reported as none in 78 (62%) patients, mild in 32 (26%) patients, moderate in 14 (11%) patients, and severe in 1 (<1%) patient.
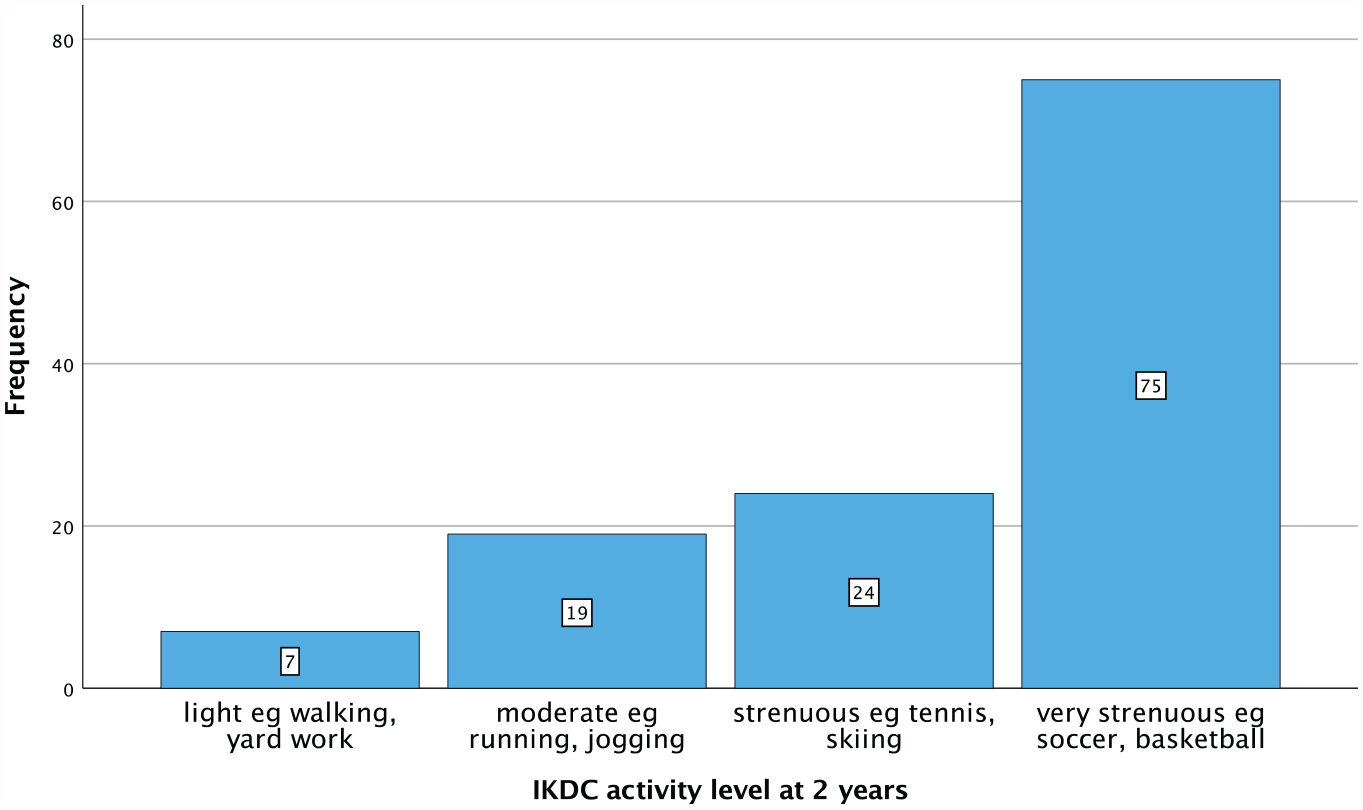
Distribution of reported activity level at 2 years. IKDC, International Knee Documentation Committee.
A summary of outcomes by patient age (≤25 years vs >25 years) are provided in Table 4. Younger patients (≤25 years old) had a significantly higher IKDC median score (P = .024), ACL-RSI median score (P = .034), rate of return to sports (P = .002), activity levels (P = .002), and ACL graft failurerate (P = .007) compared with older patients (>25 years old). We did not observe any differences in clinical outcomes between older donors (>40 years old) and younger donors (≤40 years old) (P > .05) or female donors and male donors (P > .05).
Outcomes at 2 Years in Patients by Age a

Values are presented as n (%) unless otherwise indicated. ACL-RSI, Anterior Cruciate Ligament–Return to Sport After Injury; IKDC, International Knee Documentation Committee; IQR, interquartile range.
Clinical Assessment
Clinical IKDC evaluation was performed at 1 year on 108 patients with intact ACL grafts and 4 patients with ACL graft failure. The remaining 14 patients were unable to attend and completed patient-reported outcomes only. The IKDC evaluation includes subcategories of effusion, range of motion, ligament evaluation, and overall IKDC grade, with grade A representing a “normal” knee, grade B a “nearly normal” knee, grade C an “abnormal” knee, and grade D a “severely abnormal” knee. The number of patients with each IKDC grade for each IKDC subcategory is shown in Figure 5. If those with an ACL graft failurewere givenan abnormal grade, then the proportion of patients with a normal overall IKDC grade at 1 year would be 70 of 112 (63%).

Distribution of International Knee Documentation Committee (IKDC) subgroup grades at 1 year.
Ligament Evaluation
The distribution of laxity on pivot-shift testing, instrumented laxity with KT-1000 arthrometer, and overall IKDC ligament grade at 1 year is shown in Figure 6. The mean manual maximal side-to-side difference in KT-1000 arthrometer testing was 1.7 ± 1.4 mm.

International Knee Documentation Committee ligament grades at 1 year. Manual Max: Manual Maximum testing on KT1000 arthrometer.
Single-Leg Hop Test
Hop testing was performed on 106 patients. Two patients did not complete hop testing due to pregnancy or injury to other joints. Of the 106 patients, 93 (88%) were able to hop within 90% distance of their contralateral limb at 1 year.
Other Subsequent Surgery and Complications
One patient underwent removal of a loose body and microfracture of the lateral femoral condyle at 9 months. One patient underwent arthroscopic removal of a cyclops lesion anterior to the ACL at 35 months. There were no infections, superficial or deep, postoperatively.
Discussion
This study investigated the clinical outcomes of terminal SCCO2-sterilized tendon allografts at 2 years postoperatively in patients undergoing primary ACLR. We observed an ACL graft failure rate of 5% at 24 months, a contralateral ACL rupture rate of 2%, a high rate of return to sports, and successful clinical outcomes, suggesting that terminal SCCO2 sterilization results in a suitable graft choice for primary ACLR.
Graft processing involves both the decellularization of biomaterials to remove immunogenic cellular components and provide a suitable extracellular matrix for host integration, as well as sterilization to remove contaminant bacteria, viruses, and fungi that may cause serious infection in the host. 36 Outcomes of allografts for primary ACLR have been reported to be determined by the method of graft processing, donor source tendon and age, and patient age and activity levels. Gamma irradiation, the most commonly used method of terminal allograft sterilization, exhibits a dose-dependent disruption to the hierarchical morphology of collagen fibers, resulting in a decrease in the ultimate tensile strength of allograft tissues, potentially explaining the higher failure rates observed in other series.12,32,51 Compared with nonirradiated allografts, even low-dose irradiation (<2.5 Mrad) of allografts produces higher revision ACLR rates, as well as inferior clinical and subjective knee scores. 32
As these factors are infrequently controlled for in experimental or observational study designs, systematic reviews and meta-analyses report a wide estimate of ACL graft failure rates for allograft, ranging from 0% to 10.5% at 24 months. 15 If graft processing methods that are known to alter the collagen ultrastructure of tendon allografts are adjusted for, such as gamma irradiation and chemical processing, equivalent failure risk of unprocessed or fresh frozen allografts is observed.26,27 The heterogeneity of included studies and outcomes from meta-analyses has therefore led to practice variation regarding allograft use for primary ACLR,1,3,4,9,28 and the recommendations of practice guidelines may be biased by evidence from large series that included heavily irradiated allografts prone to failure.20,26 It is therefore imperative to identify efficacious graft-processing methods that do not compromise the innate biomechanical properties of allograft tendon and provide equivalent or better outcomes than autologous tissue.
In vitro basic science studies of SCCO2 sterilization have demonstrated that it largely preserves the load to failure, young modulus elasticity, cross-sectional area, and stiffness of a range of allograft and xenograft materials, including tendon. Six studies2,5,16,29,37,41 have examined the effect of SCCO2 sterilization on tendon properties, 4 on animal tendons,2,16,37,41 and the remainder on human cadaveric tendons. Accumulated evidence from animal studies suggests that SCCO2 sterilization preserves the collagen structure of musculoskeletal allografts. Separate comparisons of large samples of porcine and ovine extensor tendons have indicated that SCCO2 sterilization provides for equivalent ultimate tensile failure and stiffness of tendons compared with unprocessed tendons.16,41 Further, histomorphometric analyses have demonstrated no differences in the extracellular matrix composition of SCCO2-sterilized and unprocessed tendons, as well as a preserved collagen hierarchical structure without evidence of significant cross-linking or changes to the orientation of collagen fibers.2,16,37,41 By comparison, gamma-irradiated tendons exhibited reduced ultimate tensile strength, lower loads to failure, lower modulus of elasticity, and an increase in void space between collagen fibers.
Studies of SCCO2-sterilized human tendons differ in the results of biomechanical testing but have significant methodological flaws that preclude generalizing their findings. Baldini et al 5 compared 38 samples of unprocessed, low-dose irradiation and SCCO2-processed tibialis posterior and anterior allografts and reported no differences in load to failure or failure stress but decreased tendon stiffness for SCCO2 tendons. Nichols et al 29 performed a similar comparison of 9 human bone-patellar tendon-bone allografts and reported that while the biomechanical properties compared with those of unprocessed tendons were preserved, SCCO2 did not result in adequate decellularization of tendons under scanning electron microscope. Uquillas et al 47 reported that SCCO2 processing preserved the mechanical properties of human cadaveric bone-ACL-bone allografts in 9 samples. We would urge caution in extrapolation of these studies due to the small sample sizes, most likely reflecting the restrictions placed by the significant costs associated with laboratory testing of human allografts. Further, the study of Baldini et al likely suffers from selection bias, as tendon diameter was not controlled between groups and the SCCO2 group was predominately composed of postmenopausal women (mean age, 58.7 ± 4.4 years). Postmenopausal female allograft origin is a known risk factor for failure of fresh frozen allografts and potentially explains the lower stiffness observed. 37 However, the effect of donor sex and age has been found to be modest in a recent biomechanical study of fresh frozen allograft tendons, suggesting that specific tendon may be more important. 3 We agree with Sun et al 41 that the effect of SCCO2 on tissue stiffness is not yet fully elucidated and would benefit from more biomechanical studies of human tendon allograft.
The patient-reported outcome measures provided by patients in our study indicated that terminal SCCO2 sterilization results in acceptable clinical and subjective knee scores that are comparable with those in published literature on ACLR with fresh-frozen allograft. Park et al 32 performed a systematic review and meta-analysis of fresh-frozen allografts versus irradiated allografts and reported a pooled mean Lysholm score of 90 (95% CI, 88-92) at a mean follow-up of 50 months. At 12 months, we have reported mean Lysholm knee scores of 92 ± 10. The proportion of patients with fresh-frozen allografts reporting combined IKDC grades A and B was 86% (95% CI, 0.90-0.93). 32 The proportion of patients with SCCO2-sterilized allografts with 12-month IKDC grade A was 63% and grade B was 34%, indicating 96% of patients had a normal or nearly normal knee. By comparison with a historical cohort of fresh-frozen allograft procedures by the senior authors of this manuscript (J.P.R., L.A.P.), patients with SCCO2-sterilized allografts demonstrated a similar mean IKDC subjective score of 91 ± 11 at 24 months postoperatively. 38 In our cohort, 72% of patients demonstrated <3 mm knee laxity on KT-1000 arthrometer testing (Figure 6), a near identical proportion to the fresh-frozen allograft cohort. 38 Any difficulty kneeling was reported by 38%, so use of allograft does not completely solve the issue of kneeling discomfort; however, this rate is considerably lower than the reported prevalence of 78% with patellar tendon grafts and 68% with HT autografts from the New Zealand ACL registry. 35 Importantly, we did not detect any deep infections in this cohort, albeit the rate of septic revision is likely to be very low in patients with allografts. 52 All 7 ACL graft failures in our cohort were from the 66 patients ≤25 years old. This is a group at highrisk of both ipsilateral and contralateral ACL injury, and the rate we observed is concordant with the published estimates of rerupture rates of autograft in this population. 50 There were no ACL graft failures in patients >25 years old.
Limitations of the current study include the short-term follow-up, the lack of a comparator group, and observational study design, as well as use of an additive sterilant during graft processing. While the majority of reruptures have been demonstrated to occur within the first 2 postoperative years as patients return to sporting activities, previous registry studies have suggested that this risk may accelerate over time for allografts. 45 Therefore, the differences between autografts and allografts may become larger at longer-term follow-up. 26 We did not have a contemporaneous control group for comparison. While we have been able to provide comparisons with historical groups of patients receiving a variety of grafts treated by the same surgeons at similar time points, a methodological improvement for future studies would be to conduct a randomized controlled trial comparing terminal SCCO2-sterilized allograft with fresh-frozen allograft or, alternately, 4-strand HT autograft. A trial such as this would elucidate whether the supposed benefits of allograft in terms of reduced morbidity results in meaningful clinical differences from autograft. The population studied represents only those without significant meniscal or articular cartilage pathology at the time of ACLR, which is associated with considerably worse outcomes. Due to the self-reported nature of 2-year follow-up in this study, there is an inherent risk that the incidence of ACL graft failure is higher than reported, as ACL status was not confirmed via clinical examination beyond 12 months or systematic magnetic resonance imaging examination. We attempted to minimize this by inviting any patients who reported further injuries or instability to either knee during the study period for clinical assessment in our clinic, but it remains possible that atraumatic ACL graft failures may have occurred and may not have been recognized, especially in low-demand patients. Grafts were washed for 15 minutes in chlorhexidine before SCCO2 sterilization. Sobel et al 40 have determined that soaking human patellar tendon allografts in a 4% chlorhexidine gluconate for 30 minutes had no effect on the structural properties of 16 human patellar tendon allografts. Finally, the effect of using peracetic acid in the SCCO2 sterilization protocol to achieve a sterility assurance level of 106 is unknown but may confound outcomes. Basic science studies have reported decreased, unchanged, and increased load to failure for animal tendons treated with peracetic acid. 22
Conclusion
SCCO2 processing of allograft tendons demonstrated satisfactory clinical and patient-reported outcomes at 24 months postoperatively in a consecutive series of patients with primary ACLR, with similar ACL failure rates and subjective knee scores compared with those reported in published series of HT autograft and fresh-frozen nonirradiated allograft.
Footnotes
Acknowledgements
The authors acknowledge the assistance of Claire Monk in the recruitment and follow-up of patients. This study was supported by the Friends of the Mater Foundation and Australian Biotechnologies.
Final revision submitted November 19, 2023; accepted December 6, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: L.A.P. has received IP royalties from Australian Biotechnologies, stock and stock options from Australian Biotechnologies, research support from the Australian Orthopaedic Association, research support from the Friends of the Mater Foundation, IP royalties from Signature Orthopaedics, and research support from Smith+Nephew. J.P.R. has received research support from 360 MedCare, Smith+Nephew, and the Friends of the Mater Foundation. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from St Vincent's Hospital (ref No. 15/305).
