Abstract
Background:
The Victorian Institute of Sport Assessment-Achilles (VISA-A) questionnaire is a validated instrument for assessing symptoms of Achilles tendinopathy (AT). However, there is a need to validate the Arabic version of the VISA-A (VISA-A-AR) in Arabic-speaking patients with AT.
Purpose:
To validate the VISA-A-AR in Arabic patients with AT and evaluate its reliability and validity.
Study Design:
Cohort study (diagnosis); Level of evidence, 3.
Methods:
The translation and cultural adaptation of the VISA-A questionnaire into Arabic followed international guidelines. A total of 81 participants were recruited, including 45 patients diagnosed with AT and 36 healthy individuals. The AT group comprised male and female native Arabic speakers aged ≥18 years who were diagnosed with and had symptoms of AT. The inclusion criteria for the healthy group were the same, except that they must not have had AT at the time of the study or previously. The exclusion criteria were individuals with a partial or complete Achilles tendon rupture or prior Achilles tendon surgery. The internal consistency of the VISA-A-AR was assessed using the Cronbach α coefficient. Test-retest reliability was evaluated using the intraclass correlation coefficient (ICC[3,1]). Construct validity was assessed through correlation analysis between VISA-A-AR scores and the Arabic versions of the Short Form-36 Health Survey (SF-36-AR) and the Numeric Pain Rating Scale (ANPRS). Differences in VISA-A-AR scores between patients with AT and healthy controls were analyzed using appropriate statistical tests.
Results:
The VISA-A-AR demonstrated a high level of internal consistency (Cronbach α = 0.935) and excellent test-retest reliability (ICC[3,1] = 0.985). Significant positive correlations were observed between VISA-A-AR scores and SF-36-AR (r(43) = 0.838, P < .001), indicating good construct validity. In addition, VISA-A-AR scores showed a significant negative correlation with ANPRS (rS(43) = -0.835, P < .001). Furthermore, VISA-A-AR scores exhibited a significant difference between patients with AT (mean, 45.82 ± 16.65) and healthy controls (mean, 99.94 ± 0.33) (P < .001).
Conclusion:
The findings of this study validate the VISA-A-AR as a reliable and valid tool for assessing symptoms of AT in Arabic-speaking patients.
Achilles tendinopathy (AT) is a common injury to the Achilles tendon characterized by pain and swelling in and around the tendon region. 25 AT is a common cause of impairment among athletes and nonathletes. 1 AT exerts a notable economic impact, particularly in sports such as long-distance running. Repetitive mechanical overload of the Achilles tendon has the potential to cause pathology over time. 23 The Victorian Institute of Sport Assessment-Achilles questionnaire (VISA-A) is a self-administered questionnaire for assessing Achilles tendon symptoms in patients with AT. 30 The VISA-A is a valid and reliable instrument used for clinical and research purposes. The VISA-A consists of 8 questions grouped into 3 subscales: pain (questions 1-3); function (questions 4-6); and activity (questions 7 and 8). The highest possible total score for all 8 questions is 100, and the results are recorded so that the higher the score, the more favorable the outcome. 30
The VISA-A questionnaire displays a high degree of responsiveness and sensitivity to clinically significant gains over the course of treatment. 22 Furthermore, it is simple for patients to complete, and the results can guide management effectively. 32 The majority of questionnaires used for lower limb injuries were created in English, which is advantageous for English-speaking communities but restricts the use of these questionnaires by non-English speakers. Therefore, an equivalent questionnaire in multiple languages is required for international comparison of research results. In 2021, 29.7% of Saudi Arabia's resident population engaged in at least 150 minutes per week of physical activity, compared to 20% in 2019. 15 We anticipate that the prevalence of AT will increase as the number of Saudi Arabians who participate in sports increases. 36 This is what prompted us to translate the VISA-A into Arabic.
The objective of this study was to cross-culturally adapt and translate the VISA-A into Arabic in order to create the Arabic language version of the VISA-A (VISA-A-AR) for the Saudi Arabian population. We hypothesized that the VISA-A-AR will be valid and reliable among the Saudi Arabian population with AT.
Methods
Study Procedure
Institutional review board approval for the study was granted, and all participants gave written informed consent before participating in this study. For this cross-sectional study, we recruited a total of 81 participants, comprising 45 individuals diagnosed with AT and 36 healthy controls. The AT group comprised male and female patients who were diagnosed with and had symptoms of AT, ≥18 years old, and native Arabic speakers. The inclusion criteria for the healthy group were the same, except that they must not have had AT at the time of the study or previously. The exclusion criteria encompassed individuals with a history of Achilles tendon surgery, as well as those with a partial or complete rupture of the Achilles tendon.
The determination of the sample size for this study was guided by 2 main considerations. First, the 8-item questionnaire necessitated at least 80 respondents, drawing from the recommended respondent-to-item ratio of 10 to 1. 28 In addition, to determine the appropriate sample size for test-retest reliability, we conducted a power analysis. We employed an online sample size calculator tailored for reliability studies (http://wnarifin.github.io) and drew from previously published data on translations of the VISA-A, where the intraclass correlation coefficient (ICC) was in the range of 0.84 to 0.99.8,12,17,19,24,31 With an anticipated ICC of 0.91, a precision of ±0.07, and a 95% CI, the calculator recommended a sample size of 25 participants. By adopting these methodologies, we ensured that the sample size was adequate for producing reliable results.
Participants were recruited from physical therapy clinics in Saudi Arabia. Among these, patients diagnosed with AT were selected, while healthy controls were chosen from individuals accompanying the patients, such as family members. Both groups were subjected to a thorough screening process to ascertain their eligibility based on the inclusion and exclusion criteria.
Upon meeting the eligibility criteria, participants provided demographic information, including age and sex, and completed the VISA-A-AR questionnaire, the Arabic version of the 36-Item Short Form Health Survey (SF-36-AR), 2 and the Arabic version of the Numeric Pain Rating Scale (ANPRS). 4 For participants diagnosed with AT, a follow-up assessment was conducted 1 week after the initial testing using the VISA-A-AR questionnaire.
Translation Process
The translation process was conducted in accordance with the instructions of Beaton et al. 9 The first stage was performing the initial translation or forward translation. Two translators translated the VISA-A from its original language (English) into Arabic. The first was a medically trained translator, and the second was a translator with no medical training. Both translators were native Arabic speakers with English proficiency. Each translator provided a translated copy of the VISA-A (T-1 and T-2) and a written report of issues and suggestions encountered during the translation of the terms and phrases. The second stage involved combining the 2 initial or forward translations (T-1 and T-2) into a single translation of VISA-A (T-12). In the third stage, 2 native English-speaking bilingual translators performed a back translation of the T-12 translation, translating the questionnaire back into its original language (English) from the T-12 version without reviewing the primary questionnaire. This was done to eliminate any potential bias during translation. This stage resulted in 2 back-translated versions of the VISA-A (BT-1 and BT-2). In the fourth stage, a committee of experts reviewed all translated versions in order to approve and produce a draft of an Arabic version of the VISA-A. The expert committee consisted of 8 individuals, including 2 forward translators, 2 back translators, and the 4 authors of this study, all of whom are licensed physical therapists. The physical therapists hold at least a bachelor's and master's degree in physical therapy and possess experience in the field ranging from 5 to 13 years. The fifth stage involved testing the translated VISA-A-AR. The translation guidelines of Beaton et al 9 suggested a sample of 30 to 40 individuals to complete the translated version for the fifth stage. A testing sample of 36 healthy Saudi participants completed the VISA-A-AR. This was done to assess the clarity of the VISA-A-AR and to collect feedback.
Outcome Measures
Arabic Version of the VISA-A
The VISA-A-AR is a self-reported questionnaire containing 8 questions that cover 3 domains: pain in the Achilles tendon region (questions 1-3); function (questions 4-6); and activity (questions 7 and 8). The scoring range for the first 7 questions is between 0 and 10, while question 8 has a range of 0 to 30. 30 In the first 6 questions of the VISA-A, the severity of a subjective continuum of symptoms is reported. The last 2 questions are rated using a categorical scale. The score ranges from 0 to 100, with a lower score representing more severe AT symptoms.
36-Item Short Form Health Survey
The SF-36 is a commonly used questionnaire that assesses health-related quality of life in 8 different dimensions: physical functioning; body pain; role limitations due to physical health problems; role limitations due to emotional problems; emotional well-being; social functioning; energy/fatigue; and perceptions of overall physical health. 16 Typically, respondents answer questions based on their experiences over the past 4 weeks. The SF-36 score limits provide valuable information about an individual's health status and well-being. A score of 100 indicates the best possible health outcome, with optimal functioning and well-being in the assessed aspect. In contrast, a score of 0 indicates the worst possible health outcome, with significant limitations or impairments in that particular health aspect.
Numeric Pain Rating Scale
The NPRS is a commonly used self-assessment instrument for measuring pain intensity. It consists of a numeric point scale ranging from 0 to 10, with 0 indicating no pain and 10 indicating severe pain. Individuals are asked to select the number on the scale that corresponds most closely to their current level of pain. 14 The NPRS provides a straightforward and simple method for individuals to quantify and communicate their pain experience, allowing healthcare professionals to assess and monitor pain levels over time.
Statistical Analysis
The data were analyzed using SPSS Version 28 (IBM Corp). Using the mean, standard deviation, and frequencies, we summarized the characteristics and observations of the outcome variables. The Shapiro-Wilk test was used to test the normality of the variables. The internal consistency of the VISA-A-AR was determined using Cronbach α, with values between 0.70 and 0.95 being acceptable. 34 Test-retest reliability was estimated using the ICC described by McGraw and Wong. 27 For this study, a 2-way mixed effects with absolute agreement and single rater/measurement approach (ICC[3,1]) was chosen. ICC values of >0.90, 0.75 to 0.90, 0.5 to 0.75, and <0.5 are indicative of excellent, good, moderate, and poor reliability, respectively. 29 Independent samples of the Mann-Whitney U test and chi-square test were used to compare characteristics and outcome measures between the groups. Using Pearson product-moment correlation and the Spearman rank-order correlation, we calculated the degree of correlation between the VISA-A-AR and SF-36-AR and ANPRS. Correlation values of 0.1 to 0.3, 0.3 to 0.5, and 0.5 to 1.0 are indicative of small, medium, and large association, respectively. The significance level was set at P < .05.
Results
Participants
A total of 45 participants with AT (mean age, 31.96 ± 7.63 years) and 36 healthy controls (mean age, 31.75 ± 8.74 years) were recruited. All participants completed the VISA-A-AR, SF-36-AR, and ANPRS. Of the participants with AT, 28 (62.22%) also completed the VISA-A-AR questionnaire again after 7 days (Figure 1). Participant characteristics and results on the outcome measures as well as group comparisons are reported in Table 1.
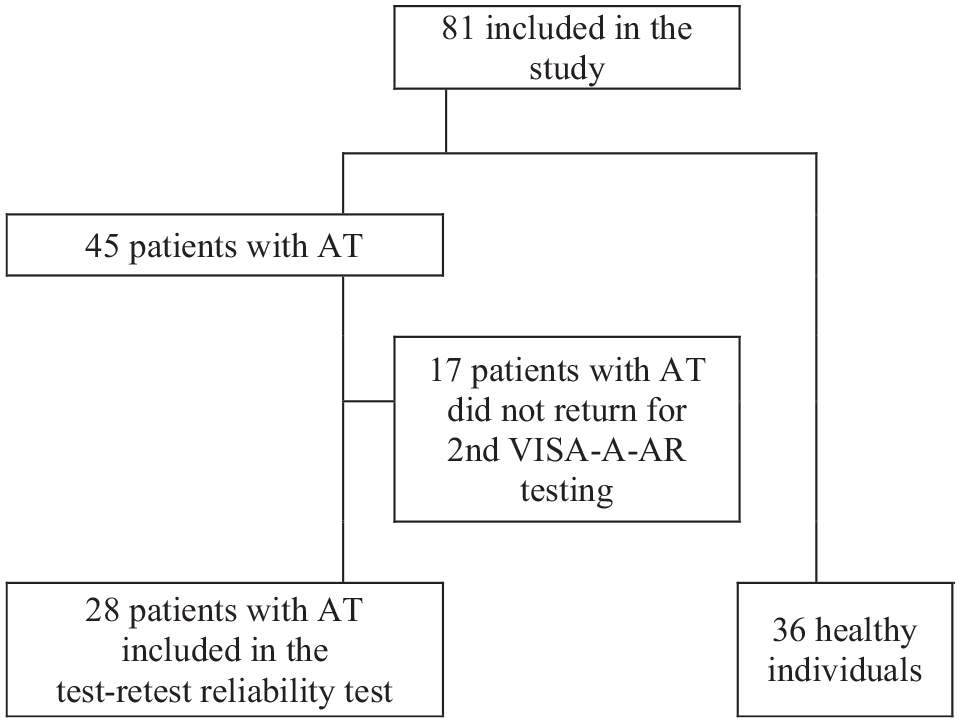
Flowchart of participant enrollment. AT, Achilles tendinopathy; VISA-A-AR, Arabic version of the Victorian Institute of Sport Assessment-Achilles questionnaire.
Participant Characteristics and Outcome Measures a

Data are shown as mean ± SD. ANPRS, Arabic version of the Numeric Pain Rating Scale; SF-36-AR, Arabic version of the 36-Item Short Form Health Survey; VISA-A-AR, Arabic version of the Victorian Institute of Sport Assessment-Achilles questionnaire.
Mann-Whitney U test.
Translation
The comparison between the back-translated version and the original version revealed that the translation was semantically identical, confirming that no further changes were required (Table 2).
Comparison of Original and Translated Versions of the VISA-A

Comparison between the original and translated versions of the VISA-A and the translated and back-translated versions. VISA-A, Victorian Institute of Sport Assessment-Achilles questionnaire.
The back translations (B1 and B2) were very similar to the original version and did not indicate any further modification to the Arabic version. None of the testing sample reported issues with comprehension of the context of the questions in the VISA-A-AR. After expressing their satisfaction with the final version, the various members of the expert committee gave their approval for the VISA-A-AR (Appendix Figure A1).
VISA-A-AR Validation
Difference Between Groups
The known groups validation was performed by comparing the total score of the VISA-A-AR between individuals with AT and healthy controls using the Mann-Whitney U test. The VISA-A-AR score was statistically significantly lower in individuals with AT (mean, 45.82 ± 16.65) than in healthy controls (mean, 99.94 ± 0.33) (P < .001) (Table 1).
Construct Validity
The construct validity of the VISA-A-AR was examined using data from participants with AT by assessing the relationship of the total score on the VISA-A-AR with the total score on the SF-36-AR and the ANPRS separately using Pearson product-moment correlation and Spearman rank-order correlation, respectively. For Pearson product-moment correlation, there was a statistically significant, strong positive correlation between VISA-A-AR and SF-36-AR (r[43] = 0.838, P < .001), with the VISA-A-AR explaining 70.2% of the variation in the SF-36-AR. For Spearman rank-order correlation, there was a statistically significant, strong negative correlation between the VISA-A-AR and the ANPRS (rs[43] = –0.835, P < .001), with the VISA-A-AR explaining 69.7% of the variation in the ANPRS.
Reliability of the VISA-A-AR
Test-retest Reliability
Participants with AT completed the VISA-A-AR a second time 1 week after the first VISA-A-AR assessment. ICC(3,1) estimates and their 95% CIs were calculated using SPSS Version 28 (IBM Corp) based on a single rating, absolute-agreement, 2-way mixed-effects model. The test-retest reliability showed an ICC(3,1) of 0.985, indicating excellent reliability, 21 with a 95% CI of 0.951 to 0.994. The mean scores of the first and the second tests of the VISA-A-AR were 45.82 ± 16.65 and 47.36 ± 16.40, respectively.
Internal Consistency
The internal consistency of the VISA-A-AR was evaluated using Cronbach α. The internal consistency analysis was used to assess the first 6 items of the VISA-A-AR. The VISA-A-AR had a high level of internal consistency, as determined by a Cronbach α of 0.935.
Discussion
This study outlined the steps taken during the translation and cultural adaptation of the VISA-A into Arabic. The most important findings of the present study were that the constructed Arabic version of the VISA-A scale was reliable (ICC, 0.985), valid, consistent (Cronbach α, 0.935), and comparable to the English version. The VISA-A-AR questionnaire can be used among the Arabic-speaking population to evaluate the clinical condition after AT. The advantage of completing the questionnaire in the language with which the patient is most comfortable will surely help clinicians to understand the patient's functional problem effectively.
Translation
The VISA-A has been translated into 12 other languages: Chilean, German, Swedish, Italian, Brazilian Portuguese, Chinese, French, Turkish, Dutch, Spanish, Persian, and Danish. ‡ Various studies have discussed the benefits of adopting the Arabic version of medical questionnaires among the Saudi Arabian population in clinical settings.3,5,6 A previous study on Saudi Arabian patients with knee injuries demonstrated that questionnaire translation is a beneficial method for obtaining accurate self-reported symptoms. 7 This type of research emphasizes the usefulness of symptom reporting when the questionnaire is administered in the patient's native language.
Internal Consistency
The VISA-A-AR proved to be a valid and reliable method for assessing AT and comparable to the original version. The VISA-A-AR was easy to understand and implement for all the respondents. The VISA-A-AR had high internal consistency, indicating that none of the questions should be excluded, as proven by a Cronbach α of 0.935 for internal consistency for the patient population. This result was consistent with previous research findings that reported Cronbach α in the range of 0.73 to 0.97, such as in the Swedish, German, Danish, French, Italian, Spanish, Brazilian Portuguese, Chinese, Persian, and Dutch versions of the VISA-A questionnaire. §
Reliability
The scores of test-retest reliability mark the stability of the VISA-A-AR in the assessment of patients experiencing AT. Despite the importance of this measure, we acknowledge a limitation arising from a notable rate of dropouts during the retest phase. However, the test-retest reliability result, although influenced by the dropouts, was consistent with previous research findings that reported ICCs in the range of 0.79 to 0.99, such as in the Swedish, German, Danish, French, Italian, Spanish, Brazilian Portuguese, Chinese, Persian, and Dutch versions of the VISA-A questionnaire. § A similar research study published by Vascellari et al 35 , which translated the Achilles tendon Total Rupture Score into Italian, retested the patients after 5 days. 26 The advantage of performing a retest after 5 to 7 days rather than on the same day is that it decreases the likelihood that the patients remember their responses to the questions while minimizing clinical change to the patient's health condition.10,17
Validity
The VISA-A-AR score was significantly lower in individuals with AT compared to healthy controls. This result was consistent with previous research findings that reported significant differences between individuals with AT and healthy controls, such as in the Swedish, German, Danish, French, Spanish, Chinese, Persian, and Turkish versions of the VISA-A questionnaire.8,10,13,17,19,24,33
The obtained mean score of the validity of the VISA-A-AR is similar to that of the Chinese version. The mean score of the Chinese version of VISA-A indicated that it was effective in differentiating the healthy and risk groups. The mean score of the Chinese version of VISA-A was remarkably lower for the risk groups than for the healthy groups, similar to the VISA-A-AR. The Spanish version, submitted by Hernández-Sánchez et al, 17 documented that the scores of the VISA-A questionnaire were suitable to differentiate between the individuals with and without symptoms of AT.
The VISA-A-AR showed a strong statistically significant relationship with the SF-36-AR. This result was consistent with previous research findings, such as in the French, Spanish, and Chinese versions of the VISA-A questionnaire.10,17,19
The results of our study regarding the relationship with the SF-36-AR were similar to the results of research on the Chinese version of VISA-A. It showed moderate to high correlations with scores of the physical components of the SF-36 and low correlations with scores of the mental components of the SF-36. These findings support good convergent and discriminant validity of the VISA-A-AR. 10
The major advantage of the VISA-A-AR is that it explains the clinical severity for patients with AT. It can be administered easily, as patients will be more expressive in their native language. Hence, the VISA-A-AR is proven to be a reliable, valid tool comparable to the original English version. It will help physicians and physical therapists assess Arabic-speaking individuals with AT and will be a valuable tool for research studies among individuals with AT.
Limitations
During the retest of the VISA-A-AR, a considerable percentage of participants (38%) did not complete the VISA-A-AR. This incomplete response rate represents a significant weakness of our study, potentially affecting the robustness of our findings and conclusions regarding the test-retest reliability.
The method of recruiting participants had certain limitations that could introduce bias and affect the generalizability of the results. Our patient group consisted of individuals diagnosed with AT, while healthy controls were often chosen from the patient's social circle, who were most often family members. This approach opens the door to potential selection bias due to its nonrandom nature. It also has the potential to introduce biases related to factors such as age and athletic status. Consequently, this nonrandom sampling method may restrict the validity of our results when applied to wider populations. To address these concerns and enhance the clinical relevance of future studies, adopting a matched-cohort analysis based on age and athletic status could provide a more balanced comparison between patients and controls.
Conclusion
The findings of this study validate the VISA-A-AR as a reliable and valid tool for assessing AT symptoms in Arabic-speaking patients. The VISA-A-AR questionnaire can be utilized in clinical and research settings to evaluate the impact of AT and monitor treatment outcomes in this population.
Footnotes
Appendix
Acknowledgements
The authors would like to thank the Deanship of Scientific Research at Majmaah University for supporting this work under Project Number (R-2023-849).
Final revision submitted September 11, 2023; accepted September 18, 2023.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Ministry of Health in Saudi Arabia IRB (ref No. 20-192E).
