Abstract
Background:
The anatomy of the trochlea plays a significant role in patellar stability. The developmental anatomy of the trochlea and its relationship to patellar stability remains poorly understood.
Purpose:
To describe the developmental changes of the osseous and cartilaginous trochlear morphology in skeletally immature specimens.
Study Design:
Descriptive laboratory study.
Methods:
A total of 65 skeletally immature cadaveric knees between the ages of 2 months and 11 years were evaluated using computed tomography scans. The measurements in the axial plane of both cartilage and bone include medial, central, and lateral trochlear height; sulcus height; medial and later trochlear facet length; trochlear sulcus angle; patellar sulcus angle; condylar height asymmetry; and trochlear facet asymmetry. Additional measurements included trochlear depth and lateral trochlear inclination angle. In the sagittal plane, measurements included curvilinear trochlear length, direct trochlear length, condylar height, and patellar sulcus angle.
Results:
Analysis of trochlear morphology using condylar height, condylar height asymmetry, and trochlear depth all increased with increasing age. The osseous and cartilaginous sulcus angles became deeper with age until age 8 and then plateaued. This corresponded with an increase in trochlear depth that also plateaued around age 8. Osseous condylar asymmetry increased with age but flipped from a larger medial condyle to a larger lateral condyle around age 8. The continued growth of the trochlea with age was further demonstrated in all measures in the sagittal view.
Conclusion:
This cadaveric analysis demonstrated that there is an increase in condylar height as age increased by all measurements analyzed. These changes in condylar height continued to be seen through age 11, suggesting a still-developing trochlea past this age. By age 8, a plateau in sulcus angle, and sulcus depth suggests more proportionate growth after this point. Similar changes in trochlear and patellar shape with age suggests that the 2 structures may affect each other during development.
Clinical Relevance:
This information can help design, develop, and determine timing of procedures that may alter the anatomy and stabilize the trochlear and patellofemoral joint.
The anatomy of the patellofemoral joint plays a significant role in patellofemoral disorders. Dysplasia of the femoral trochlea is characterized by the abnormal shape and loss of depth of the trochlear groove. 23 The presence of trochlear dysplasia has been identified as a primary anatomic risk factor for patellofemoral instability2,3,7,26 and arthritis. 10 Trochlear dysplasia is also a prognostic factor used to predict recurrent patellar instability in children and adolescents.7,13,14 With good outcomes in surgical procedures such as trochleoplasty in patients with trochlear dysplasia,6,15,16 an improved understanding of trochlear development may alter timing and type of intervention in skeletally immature patients with trochlear dysplasia and related patellofemoral disorders.
Unlike the development of the pediatric hip joint, which has been well studied for decades, the developmental anatomy of the trochlea and its relationship to patellar stability has yet to be fully described. There is, however, a growing understanding of abnormal trochlear morphology.8,22 This has been accomplished with historically used measures for patellar instability obtained using an axial view on magnetic resonance imaging (MRI).1,3,18 Parikh et al 20 also used these measures on MRI to describe the growth pattern of trochlear dysplasia in adolescents. Despite this knowledge of the dysplastic trochlea, normal development of the trochlea remains to be fully understood.
In 2020, Richmond et al 21 published a study of 31 pediatric knee specimens in which they reported a relationship between the development of the trochlea and the patella using new measuring techniques in the sagittal plane on computed tomography (CT), including one that uses the anterior femoral cortex, to add to established measurements. That study was limited, however, by a small sample size. The purpose of the current study was to describe the changes of the osseous and cartilaginous trochlear morphology in skeletally immature specimens and their relationship to the development of the patella, by using a larger sample size of pediatric CT scans. Additionally, we aimed to evaluate morphologic development using the recently described sagittal plane measurements by Richmond et al.
Methods
The cadaveric tissue used in this study did not include any identifying patient information or genetic information, so no institutional review board (IRB) approval was required. The pediatric cadaveric tissue used in the study was provided by Allosource, an allograft-harvesting facility. In accordance with the waiver for IRB approval, prior consent to use this tissue for research purposes was obtained from families; there was no subsequent contact with families that made these tissue donations, and no genetic tests were performed. Age and sex were defined for each cadaveric specimen; the cause of death was not available. Specimens were screened and found absent of any gross evidence of musculoskeletal chronic disease or dysplasia.
CT air arthrogram scans (GE Litespeed Scanner) with 0.625-mm cuts were taken on 65 fresh-frozen cadaveric knees from skeletally immature specimens ranging in age from 2 months to 11 years. Air arthrogram improves visualization of cartilage surfaces on cadaveric CT scans and is possible due to the dissected state of these cadaveric specimens. This study uses a data set from a previously published paper on this topic (31 specimens) and expands this set by the inclusion of a larger number of specimens (65 specimens), including older but still skeletally immature specimens. 21
Axial plane measurements included central, medial, and lateral condylar height of both cartilage and bone, sulcus height of both cartilage and bone, medial and lateral trochlear facet length, osseous sulcus angle (OSA), cartilaginous sulcus angle (CSA), patellar sulcus angle (PSA), osseous and cartilaginous condylar height asymmetry, osseous and cartilaginous trochlear facet asymmetry, trochlear depth, and lateral trochlear inclination angle (Table 1). The PSA was measured using the slice with the greatest width of the patellar cartilage. Additionally, measurements of the trochlea were taken from the slice most proximally, where the trochlear groove was entirely covered by articular cartilage. A reference line across the posterior condyles at the first axial slice with full posterior articular cartilage was used for all condylar height measurements and lateral inclination angle.
Axial Computed Tomography Measurements of the Trochlea and Patella

Measurements taken of both osseous and cartilaginous structures.
In the sagittal plane, measurements described by Richmond et al 21 were evaluated. These included curvilinear trochlear length, direct trochlear length, condylar height, and PSA as previously described but in the sagittal plane (Table 2).
Sagittal Computed Tomography Measurements of the Trochlea and Patella as Described by Richmond et al 21
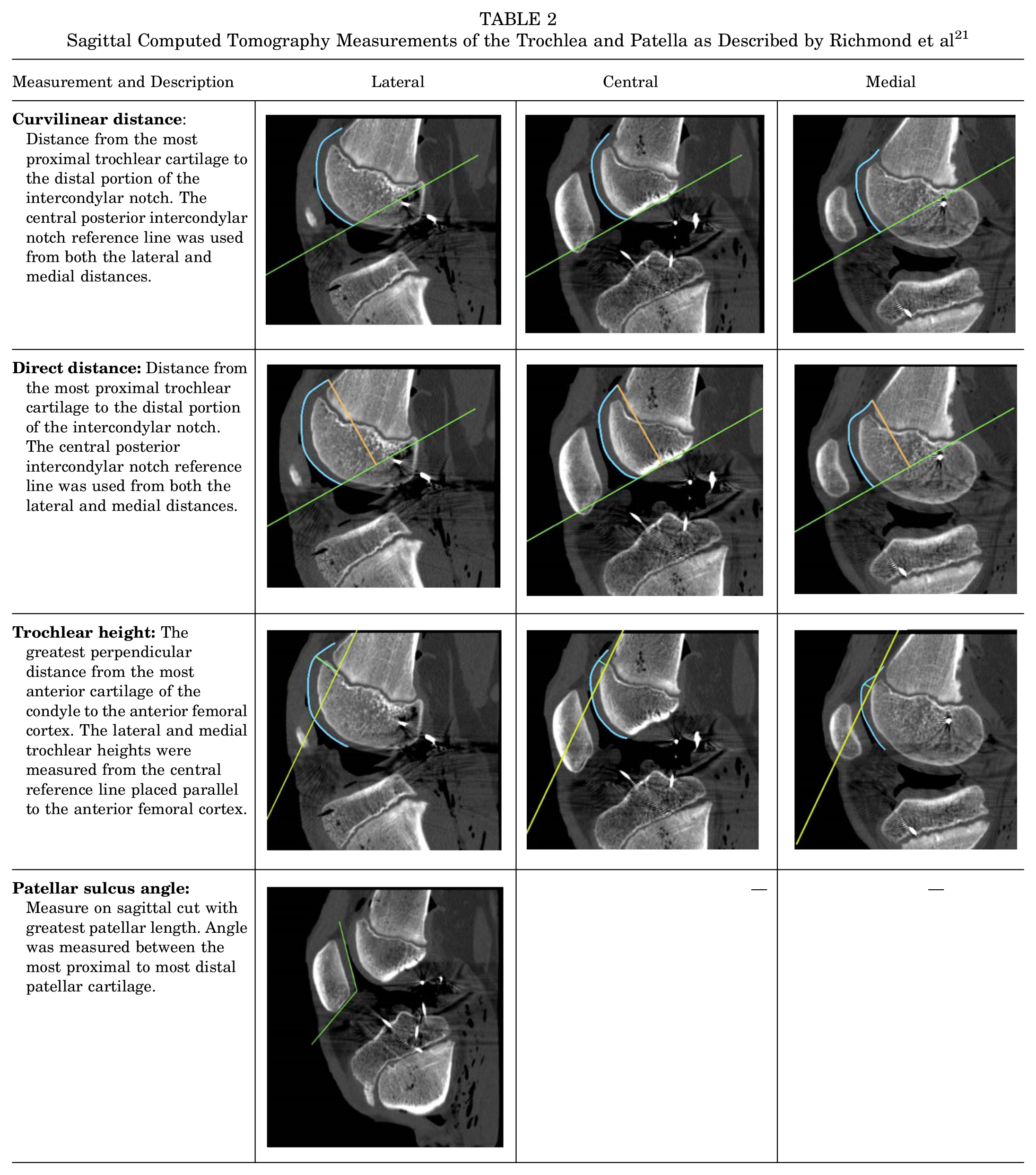
Not all specimens had a patella present due to prior tissue harvest (n = 13), so patellar measurements in both the axial and sagittal planes were not recorded in these specimens. The 3 specimens <1 year old did not have sagittal images. Additionally, their trochleae were primarily cartilage; thus, only axial cartilage measurements were made on these specimens.
To obtain interrater reliability, 2 medical student researchers (S.G.A. and N.T.) made the measurements independently on the CT scans. Previously, 31 of the scans were analyzed. Measurements were performed using OsiriX Lite Imaging Software (Version 12.0.3; Pixme), following training and confirmation of methodology by the senior authors (K.G.S., M.T.). To obtain intrarater reliability, the same 2 medical students remeasured 40 scans 4 weeks after making the initial measurements.
Statistical Analysis
Intraclass correlation coefficients (ICCs) and 95% CIs were calculated for the measurements. The ICC(2,1) was used to analyze the interrater reliability between the 2 independent raters, and the ICC(3,1) was used to analyze the intrarater reliability for 1 rater.
The mean and standard deviation of the measurements was reported for every age group (<1, 2, 3, 4, 5, 7, 8, 9, 10, and 11 years). All analyses were completed in RStudio (Version 2021.09.1; Integrated Developement for R. RStudio, PBC).
Results
The study cohort included 65 CT scans of skeletally immature knee specimens aged from 2 months to 11 years (46 men and 19 women, mean age, 6.42 years). Mean values of the osseous, cartilaginous, and patellofemoral measurements stratified by age are displayed in Tables 3, 4, and 5, respectively. Most measurements yielded an excellent level of reliability, with some good and moderate ratings.
Osseous Measurements Taken in the Axial Plane a

F, female; M, male; OSA, osseous sulcus angle. The table does not include the 3 specimens in the <1 age group as they did not have osseous measurements.
Cartilaginous Measurements Taken in the Axial Plane a

CSA, cartilaginous sulcus angle; F, female; M, male; PSA, patellar sulcus angle.
Patellofemoral Measurements Taken in the Sagittal Plane a

PSA, patellar sulcus angle.
Axial Measurements
Analysis of trochlear morphology of osseous and cartilaginous measurements of condylar height, condylar height asymmetry, and trochlear depth demonstrated a significant increase in the size of the medial and lateral trochlea as age increased (P < .001 for all) (Figure 1). The osseous and cartilaginous sulcus angles decreased (became deeper) with age until around age 8 and then plateaued. This corresponded with an increase in trochlear depth that also plateaued around age 8. In contrast, medial and lateral condylar height continued to increase up to age 11. Cartilaginous condylar asymmetry rose slightly with age with the lateral condyle consistently larger than the medial condyle. Osseous condylar asymmetry increased with age but flipped from a larger medial condyle to a larger lateral condyle around age 8 (Figure 2).

The mean medial and lateral cartilaginous condylar height with respect to age.
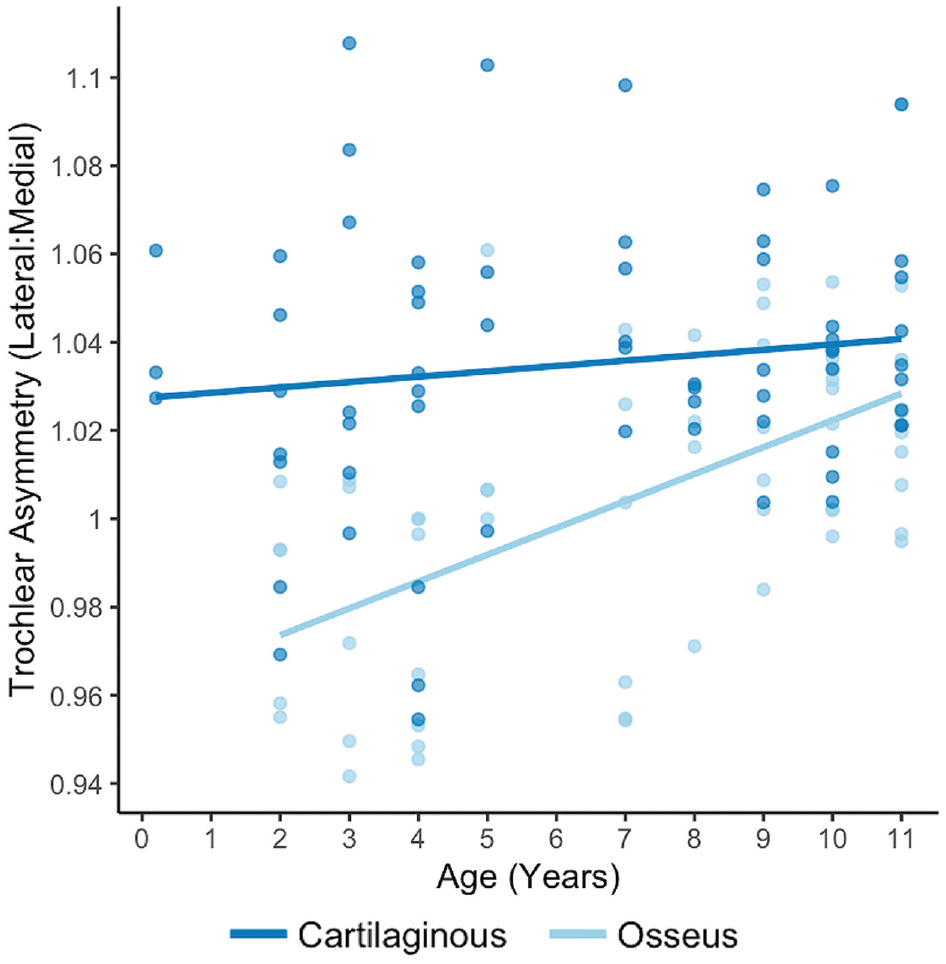
The osseous and cartilaginous trochlear condylar asymmetries with respect to age. Asymmetry values <1 signify a larger medial trochlear condyle and values >1 signify a larger lateral trochlear condyle.
PSA, OSA, and CSA all decreased with age until about the age of 8. OSA changed the most (range, 165.14°-149.04°), CSA changed the least (range, 150.8°-147.06°), and PSA showed an amount of change between the 2 (range, 133.48°-128.41°). A linear regression looking at the slope change over all ages showed a significant decrease of 2.2° (P < .001) for each 1-year increase in age for OSA. PSA and CSA also showed a negative slope observationally, with P values above the statistically significant cutoff of P < .05 at .051 and .076, respectively. Lateral inclination angle and patellar tilt appear to remain within a constant range across all ages.
Sagittal Measurements
With respect to height measures, the lateral trochlea was consistently higher than the medial trochlea: on average, 4.9 mm higher across age groups. Length in the sagittal plane followed the same pattern with a consistently longer lateral trochlea: on average, 7.6 mm longer. Measures of direct and curved distances showed significantly higher variability of lateral trochlear height (P < .001; Levene test).
Discussion
Using CT scans of pediatric cadaveric knee specimens aged 2 months to 11 years, we were able to describe the osseous and cartilaginous trochlear morphology over early childhood development. A pattern of growth of the trochlea was shown through increased medial, central, and lateral trochlear height in both osseous and cartilaginous measurements with age. One of the main findings of this study was that this growth appeared to continue past the age of 8, which was previously suspected by Richmond et al, 21 and through the age of 11. The findings also suggest that by age 8, the growth of the medial, central, and lateral trochlea are more symmetrical based on a plateau in sulcus angle and sulcus depth at this age. The condylar height and trochlear length measurements in the sagittal plane corroborated this pattern of development, establishing them as potentially useful in evaluating trochlear morphology.
A few studies have observed morphologic features of the trochlea in skeletally immature patients and described how they change with age.17,21,24 These studies have found a linear increase in lateral, central, and medial trochlear heights. A study of normal pediatric knee MRIs measured these changes in both articular cartilage and subchondral bone in the axial plane but did not explore the relationship of the trochlea to the patella and had a mean patient age of 11.4 years. 24 One of the primary findings of the study by Richmond et al 21 was that OSA, CSA and PSA all deepened continuously until the age of 8. The current study replicated these findings whereby all 3 measurements decreased with age until 8 years and then plateaued. In our study, however, OSA and PSA decreased significantly more than CSA. The similar results between PSA and OSA may support the findings by Richmond et al that there could be a relationship between the development of the trochlea and that of the patella.
Our findings of a more subtle change in CSA demonstrates some agreement with studies by Glard et al 9 and Pagliazzi et al 19 that reported very little change in CSA from an early age. Using biometric evaluation previously described by Wanner, 25 Glard et al determined that sulcus angle was largely determined by genetics. This was based on the findings of similar shape, including sulcus angle, in fetuses and adults. These studies, however, examined the skeletally immature and mature trochlea separately and not over the course of its development. The relationship between the trochlea and patella throughout development remains to be fully understood. Our study provides a better insight into this interplay throughout the process of development and postulates that patellar anatomy could perhaps develop secondarily and progressively in correspondence with the shape of the developing trochlea.
Most clinical measurements of the trochlea are obtained from axial cross-sectional imaging, but additional sequences are important for describing the complete morphology. The sagittal view provides additional visualization of the proximal trochlea, including proximal cartilage extension, and relative lateral and medial trochlear heights. The radiographic correlates of trochlear dysplasia, such as the crossing sign and trochlear bump in the Dejour classification, utilize the sagittal or true lateral view to categorize these features. 7 More recently, new measures and views have been proposed to describe the morphology of the trochlea more completely. One study has even found an oblique view of the trochlea in the sagittal plane that can more accurately evaluate trochlear morphology than axial or traditional sagittal views using previously described measurements. 1 Using the measurements of Richmond et al, 21 we found similar results of a much larger lateral trochlear height and length that increased with age.
In contrast to the results by Richmond et al, 21 our sagittal measurements of direct and curved trochlear distance demonstrated significantly more variability of the lateral condyle than the medial condyle. Using MRIs in the sagittal plane, Biedert et al 4 found that the lateral trochlea has a high clinical relevance based on their finding that a (too) short lateral articular trochlea is likely another factor in lateral patellar instability. Our findings of more variable development of the lateral trochlea may help to explain how a (too) short lateral trochlea and other dysplastic features of the lateral trochlea could develop. The lateral trochlea is used to calculate several of the established measures of trochlear dysplasia, including trochlear depth, sulcus angle, facet asymmetry, and lateral trochlear inclination. As these parameters have been shown to have the strongest association with lateral patellar dislocation, 3 understanding the development of the lateral trochlea is useful in potentially detecting earlier signs of trochlear dysplasia during development.
A study by Trivellas et al 24 described trochlear morphology development in 246 normal pediatric knee MRIs from patients aged 3 to 16 years old. Their results also demonstrated a similar consistent pattern of development and growth when looking at measures of trochlear height in subchondral bone as well as articular cartilage. Using an older cohort than our study, they concluded that final trochlear development is nearly complete around age 11. In agreement with this study, we found that trochlear development continued through age 11. Understanding at what age the trochlea is still developing provides valuable information that can aid clinical decision-making about possible surgical interventions and timing of surgery in patients with patellofemoral instability.
Trivellas et al 24 also found that the anatomy of the cartilage in developing specimens more accurately represents the shape of a mature trochlea at an earlier age than that of the subchondral bone. This is in line with our findings, especially when looking at trochlear condylar asymmetry. Cartilaginous condylar asymmetry remained relatively constant across all age groups with the lateral condyle being larger than the medial condyle. Osseous condylar asymmetry, on the other hand, had a larger range of values and, more interestingly, showed a flip from a larger medial condyle to a larger lateral condyle at the age of 8. The change in condylar asymmetry is likely explained by the continued ossification of the distal femoral epiphysis. Boeyer and Ousley 5 found that by 3.7 weeks of life, 95% of all individuals will exhibit an ossified distal femoral epiphysis. The distal femoral epiphysis will continue to grow, forming both femoral condyles, until it completely fuses with the metaphysis between 14 and 16 years of age in women and 16 and 18 years of age in men. 11 The change in osseous condylar asymmetry in our study appears to demonstrate 2 things. Measurements of the subchondral bone show a larger medial condyle before age 8, which may indicate that the medial condyle ossifies earlier. For the lateral condylar subchondral bone to catch up and eventually become larger than the medial condyle after age 8, there also appears to be a point where there is a greater amount of ossification of the lateral trochlea until it more closely resembles the growth of the articular cartilage. This knowledge may inform clinicians in use of measures of the articular cartilage rather more confidently than subchondral bone in skeletally immature patients to predict final trochlear shape and potential risk of patellofemoral instability.
Strengths and Limitations
The strengths of this study include the use of a skeletally immature population in early development (mean age, 6.42 years; range, 2 months–11 years) and the access to pediatric cadaveric specimens, which is incredibly rare. This pediatric specimen database provides a unique perspective on the anatomic development of the trochlea using a CT scan database. The CT scan does have excellent resolution for visualization of bone structure. Although CT scans have limited cartilage resolution in clinical settings, CT air arthrograms on cadavers have very high resolution for cartilage due to increased differentiation between the cartilage and synovial fluid or bone. 12 The use of pediatric cadaveric specimens allowed for the joints to be open and the presence of air arthrograms to better be appreciated. The use of cadaveric tissue CT scans is also among of the strengths of the study because CT scan availability in young patients is limited, in part due to concerns about the use of ionizing radiation in patients. While the 65 analyzed specimens are more than double the amount used in a previous CT-based study by Richmond et al, 21 a larger sample size still could help strengthen this study. The large increase in size at age 8, seen in Figure 1, may highlight this limitation as this age group had some of the least number of specimens (n = 4). This makes it possible for a couple of large specimens to skew an entire age group. The evaluation of when trochlear development is completed was limited by a population with a maximum age of 11. The addition of specimens beyond this age could help to more definitively describe when trochlear development is complete. Stratification by sex was limited by a low number of female specimens overall and an uneven distribution across most age groups.
Future studies on the development of the trochlea may improve our understanding of this joint and assist with the design and implementation of procedures that aim to stabilize and/or normalize the patellofemoral joint. As these procedures become more common, understanding the development of the trochlea will become increasingly important. Future studies on the relationship between trochlear and patellar development and morphology may also improve our understanding of the normal and pathologic function of this joint in terms of stability and osteoarthritis.
Conclusion
This cadaveric CT scan analysis demonstrated that there is an increase in condylar height with increasing age from 2 to 11 years. These changes in condylar height continue to be seen through age 11, suggesting that the trochlea continues to develop past this age. By age 8, sulcus angle and sulcus depth values plateau, which suggests more proportionate growth of the lateral and medial condyles after 8 years of age. This information can help with the design, development, and implementation of procedures that may alter (and perhaps biomechanically improve) the anatomy of the patellofemoral joint.
Footnotes
Acknowledgements
The authors acknowledge Nicole Pham, MPH.
Final revision submitted October 28, 2023; accepted November 13, 2023.
One or more of the authors has declared the following conflict of interest or source of funding: The pediatric cadaveric tissue used in the study was provided by Allosource Inc. M.T. has received hospitality from Aesculap Biologics. S.N.P. has received education payments from CDC Medical. H.B.E. has received education payments from Pylant Medical and hospitality payments from Stryker. D.W.G. has received royalties from Arthrex, consulting fees from OrthoPediatrics, and nonconsulting fees from Arthrex and DePuy Synthes. P.D.F. has received consulting fees from WishBone Medical. P.L.W. has received education payments from Pylant Medical. K.G.S. has received education payments from Evolution Surgical and hospitality payments from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
