Abstract
Background:
Several approaches to surgical techniques and graft types exist in posterolateral corner (PLC) reconstruction. The literature lacks knowledge regarding outcomes after autograft versus allograft reconstruction for PLC injuries.
Purpose:
To comprehensively review the current literature on PLC reconstruction and compare outcomes between autograft and allograft tissues.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
The PubMed and Scopus online databases were searched with the terms “PLC,”“posterolateral knee,”“posterolateral corner,” and “reconstruction” in varying combinations. Patient characteristics, graft type, graft failure, surgical techniques, functional outcome scores, and varus laxity on stress radiographs were reviewed and compared between PLC reconstruction with autografts versus allografts.
Results:
Included were 22 studies comprising 33 cohorts: 16 autografts (n = 280 knees) and 17 allografts (336 knees). There were 69 isolated PLC reconstructions (58 allografts and 11 autografts) and 493 multiligament reconstructions (269 autografts and 224 allografts). There was no difference in the mean patient age (30.5 vs 33.5 years, respectively; P = .11) or mean follow-up (39.5 vs 37.7 months, respectively; P = .68) between the autograft and allograft groups. There was no evidence to suggest a difference in graft failures between graft types (pooled mean autograft vs allograft: 0.44 vs 0.41 failures; P = .95). There was a significant difference in the mean postoperative Lysholm scores for autografts versus allografts (89.6 vs 85.5, respectively; P = .04). There was no difference between the cohorts in preoperative or postoperative International Knee Documentation Committee (IKDC) scores or postoperative varus laxity.
Conclusion:
Our review and meta-analysis indicated no significant differences in graft failure rates or objective outcomes after PLC reconstruction based on graft type alone. There was a significant difference in postoperative Lysholm scores in favor of the autograft group and no significant difference in IKDC subjective scores.
The posterolateral corner (PLC) of the knee is primarily composed of the fibular collateral ligament, popliteus tendon, and popliteofibular ligament. These structures resist varus stress, external tibial rotation, and posterolateral tibial translation. 15 While PLC injuries may occur in isolation, they are more commonly associated with anterior cruciate ligament (ACL) and/or posterior cruciate ligament (PCL) disruptions. Identifying PLC tears is important to restore knee stability and optimize cruciate ligament reconstruction outcomes. Both biomechanical and clinical studies demonstrate that untreated PLC injuries lead to increased stress and, subsequently, higher failure rates for ACL and PCL reconstruction.10,22,23
Numerous studies, including a recent systematic review on treating acute PLC injuries, have reported superior outcomes after PLC reconstruction compared with PLC repair.8,33 With reconstruction considered the preferred surgical treatment over repair in the literature, a narrowed focus on specific reconstruction techniques or variables is now warranted.
The 2 most common surgical techniques for PLC reconstruction include tibiofibular-based techniques (ie, the LaPrade technique)4,19 and fibular-based techniques (ie, the Larson technique and Arciero technique).5,27 One biomechanical study demonstrated improved graft strength and stability with the anatomic technique. 17 However, further biomechanical and clinical evidence has supported either technique with no difference in graft strength, stability, or patient-reported outcomes.31,34,36,38,39,40
Different graft options should be considered in addition to surgical technique when planning ligament reconstruction in the knee. There is currently no consensus in the literature regarding using autografts versus allografts for PLC reconstruction, and no previous study directly compared outcomes between these options. This study aimed to provide a comprehensive systematic review and meta-analysis of the current literature on autograft versus allograft PLC reconstruction outcomes.
Methods
Literature Search and Screening
This systematic literature review and meta-analysis adhered to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. Two reviewers (K.K. and R.S.) independently searched online PubMed and Scopus databases between January 2000 and March 2022 for the literature on the PLC reconstruction. The key terms “PLC,”“posterolateral knee,”“posterolateral corner,” and “reconstruction” were used, and the search results were reduced to studies written in English and performed with living human patients.
Studies were included if they involved reconstruction of the PLC utilizing tibiofibular-based or fibular-based reconstruction techniques, reported outcomes, specified graft type, were published in English, and only involved living human patients. Cadaveric studies, technique papers, and review articles were excluded. In addition, studies that did not define graft type, had <2 years of follow-up, and were not limited to patients ≥16 years old were excluded.
The titles, abstracts, and full texts were screened and reviewed by the 2 reviewers (K.K. and R.S.). Any disagreements between reviewers were resolved through discussion.
Quality Assessment
The quality assessments of the studies were performed separately by the same 2 reviewers (K.K. and R.S.) using the Methodological Index for Nonrandomized Studies criteria.
Data Extraction
Studies were reviewed, and data were extracted by the same 2 reviewers independently and recorded into spreadsheets. Data included study title, year of publication, author, journal, study design, level of evidence, sample size, mean age, mean follow-up, surgical technique, graft type, graft failure, concurrent ligament injury, the most commonly reported pre- and postoperative patient-reported outcome scores (Lysholm score, International Knee Documentation Committee [IKDC] subjective score), and side-to-side-difference in varus laxity on stress radiographs.
Statistical Analysis
For our review, the cohorts within the included studies were compared based on the type of graft received: autograft versus allograft PLC reconstruction. All analyses were run in the software SAS (SAS Enterprise Guide software, Version 7.1; SAS Institute). Data compared included demographic characteristics, graft failure rates, pre- and postoperative Lysholm and IKDC scores, and postoperative varus laxity. Normally distributed numeric data were expressed as a pooled mean and 95% CI, and where there were 2 groups based on graft type, these groups were analyzed using 2-samples independent t tests. The following assumption was made for the meta-analyses: when only the range was available, the standard deviation was estimated by dividing the range by 4. The threshold for significance was set at P < .05 for all comparisons.
Results
Eligibility
A total of 70 titles were identified in adherence to the PRISMA guidelines. After complete screening of the abstracts and manuscripts, 48 additional titles were eliminated, leaving 22 studies in the systematic review (Figure 1). ‡ An agreement of 100% was obtained between the independent reviewers regarding which titles to include for analysis.
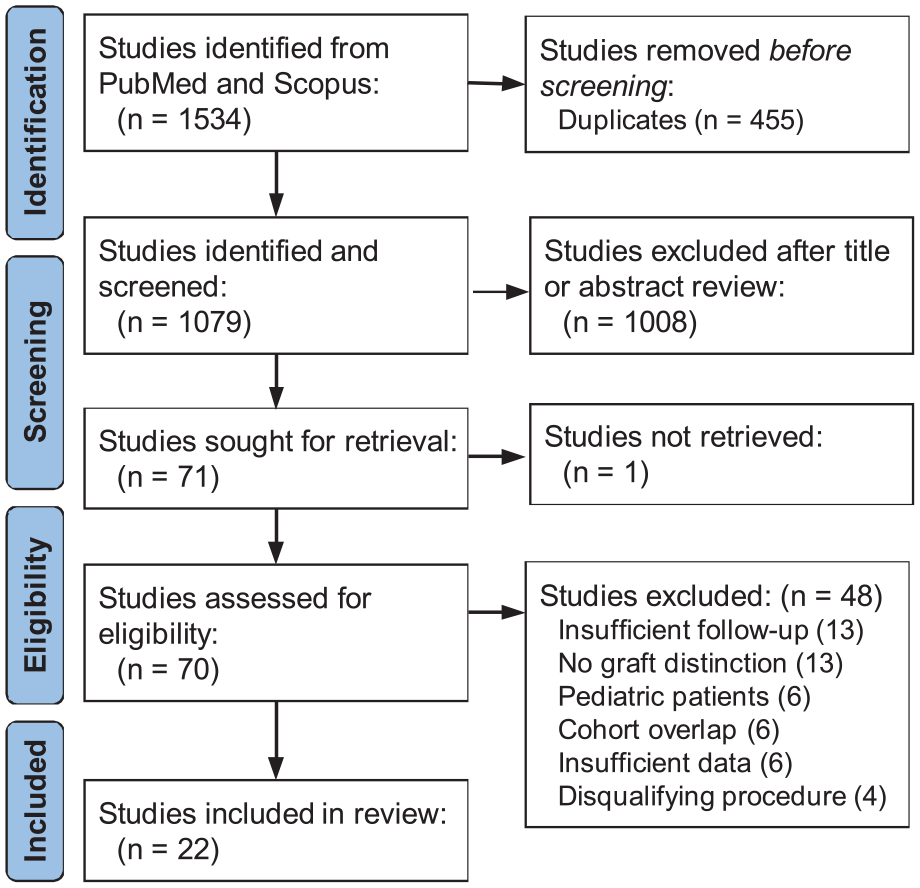
A PRISMA flowchart outlining the study selection process. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
The 22 included studies comprised 33 cohorts—16 autografts (n = 280 knees) and 17 allografts (336 knees). The original studies divided these cohorts based on surgical techniques or concomitant ligamentous injuries, which were not included for analysis in this review. Overall, there were 69 isolated PLC reconstructions (58 allografts and 11 autografts), 493 multiligament (2- to 4-ligament) injury reconstructions (269 autografts and 224 allografts), and 54 unspecified reconstructions (all allograft). For multiligamentous injuries in the autograft group (n = 269), there were 116 PLC + ACL reconstructions, 94 PLC + PCL reconstructions, 58 PLC + ACL + PCL reconstructions, 1 PLC + medial collateral ligament (MCL) reconstruction, and 1 PLC + ACL + MCL reconstruction. For multiligamentous injury in the allograft group (n = 224), there were 56 PLC + ACL reconstructions, 127 PLC + PCL reconstructions, 22 PLC + ACL + PCL reconstructions, 3 PLC + ACL + PCL + MCL reconstructions, and 1 PLC + ACL + MCL reconstruction. Studies did not consistently delineate outcomes based on concurrent ligament injuries; thus, no subgroup analysis was performed to evaluate outcomes based on different combinations of ligamentous injuries.
Patient Characteristics
The patient inclusion and exclusion criteria varied with each study. All studies included patients >16 years old who underwent PLC reconstruction utilizing either tibiofibular- or fibular-based reconstruction techniques. There was significant variability throughout the studies regarding the mechanism of injury, concomitant ligamentous injuries, and previous procedures.
A total of 22 studies (n = 592 patients) reported age, with a pooled mean of 32 years old. The mean age of patients in the autograft cohorts (n = 280) and allograft cohorts (n = 312) was 30.5 years (95% CI, 27.2-33.9 years old) and 33.48 (95% CI, 31.7-35.3 years old), respectively, with no significant difference between the cohorts (P = .11). A total of 22 (n = 552 patients) studies reported a follow-up time with a pooled mean of 38.5 months. The mean follow-up time of those in the autograft cohorts (n = 239) and allograft cohorts (n = 313) was 39.5 (95% CI, 32.4-46.5) and 37.7 (95% CI, 32.0-43.4) months, respectively, with no significant difference between the cohorts (P = .68). Patient demographic characteristics by study are provided in Table 1.
Summary of Patient Characteristics for the Included Studies a

ACL, anterior cruciate ligament; F, fibular surgical technique; NR, not reported; PCL, posterior cruciate ligament; PLC, posterolateral corner; TF, tibial-fibular surgical technique.
Data are reported as mean (range).
Graft Choice
Eleven studies § reported PLC reconstruction with autografts, and 13 studies reported PLC reconstruction with allografts. ‖ The tendons utilized for autografts were as follows: unspecified hamstring (n = 5); semitendinosus (n = 6); semitendinosus and gracilis (n = 3); long head of biceps femoris (n = 1); and unspecified (n = 1). The tendons utilized for allografts were as follows: Achilles (n = 9); tibialis posterior (n = 3); semitendinosus (n = 2); semitendinosus and gracilis (n = 1); tibialis anterior (n = 1); and unspecified tibialis (anterior or posterior) (n = 1). Concurrent procedures were performed according to the needs of each patient.
Procedures
PLC reconstruction was performed in all knees. The most commonly reported concomitant procedures were ACL reconstruction, PCL reconstruction, and combined ACL/PCL reconstruction, which were reported consistently throughout autograft and allograft cohorts. A total of 10 studies ¶ reported procedures other than concurrent ligament reconstructions. These reported procedures included 21 high tibial osteotomies (5 autografts and 16 allografts), 34 partial meniscectomies (all allograft), 3 meniscal repairs (all allograft), 3 cartilage debridements (all allograft), and 1 cartilage microfracture (allograft). Studies did not consistently delineate patient outcomes based on primary versus staged procedures, acute versus chronic injuries, or concomitant procedures. However, 41 procedures were reported as staged (18 autografts and 23 allografts) and 19 as revision procedures (2 autografts and 17 allografts), with all other surgeries reported as primary procedures. PLC reconstruction was performed utilizing variations of a tibiofibular-based or fibular-based reconstruction technique.4,21,27 Data for the autograft and allograft cohorts were not analyzed or compared based on the surgical technique.
Graft Survivorship and Objective Outcomes
Individual study outcome data are provided in Table 2, and the meta-analysis results are available in Table 3. Graft failures, defined as revision PLC reconstruction, were reported for all studies. In the autograft cohort, the failure rate ranged from 0% to 9.1%, with a mean rate of 2.5%. In the allograft group, this rate was 0% to 7.4%, with a mean rate of 2.1%. There was no statistically significant evidence to suggest a difference in graft failure rates according to graft type, with 7 of 280 failures (2.5%) in the autograft group and 7 of 336 failures (2.1%) in the allograft group and a pooled mean of 0.44 graft failures in the autograft cohort and 0.41 graft failures in the allograft cohort (P = .95).
Summary of Outcome Data for the Included Studies a

ACL, anterior cruciate ligament; F, fibular surgical technique; IKDC, International Knee Documentation Committee; MCL, medial collateral ligament; NR, not reported; PCL, posterior cruciate ligament; PLC, posterolateral corner; TF, tibial-fibular surgical technique.
Postoperative side-to-side differences on varus stress radiographs.
Results of Meta-analysis a
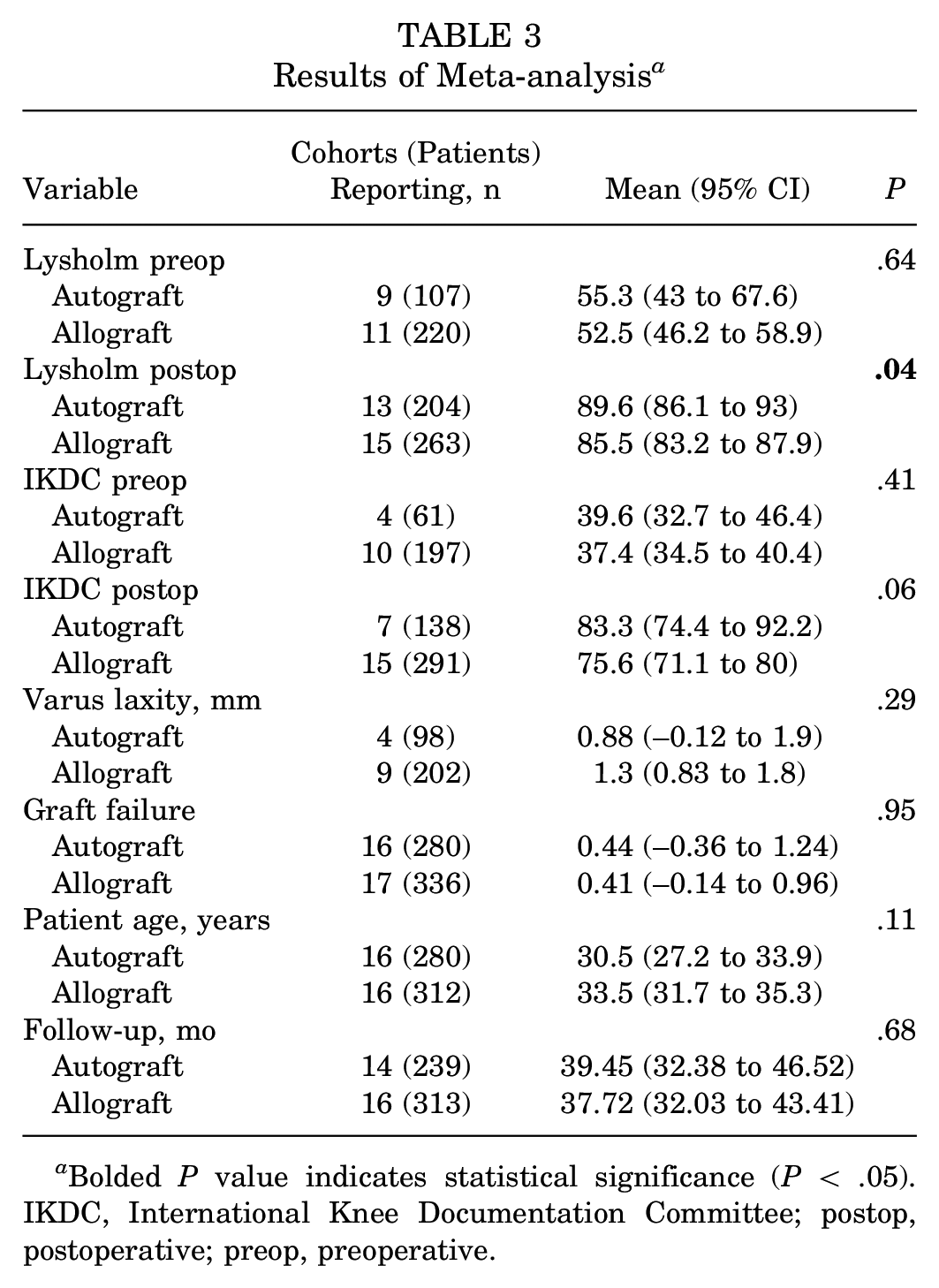
Bolded P value indicates statistical significance (P < .05). IKDC, International Knee Documentation Committee; postop, postoperative; preop, preoperative.
Thirteen cohorts (n = 300) reported postoperative varus laxity: 4 cohorts (n = 98) were autografts, and 9 cohorts (n = 202) were allografts. There was no significant difference between the cohorts, with opening measurements equaling 0.88 mm and 1.3 mm for autografts and allografts, respectively (P = .29).
Patient-Reported Outcomes
Across the 22 included studies, 20 patient cohorts (n = 327) included the preoperative Lysholm score: 9 autograft cohorts (n = 107) and 11 allograft cohorts (n = 220). There were 28 patient cohorts (n = 467) with a postoperative Lysholm score—13 autografts (n = 204) and 15 allografts (n = 263). There was no significant difference in the preoperative Lysholm score (P = .64); however, a significant difference was detected in the postoperative Lysholm score (P = .04). The results showed that, on average, patients who received an autograft had a postoperative Lysholm score that was 4 points greater than those who had an allograft procedure (Table 3).
Fourteen cohorts reported the preoperative IKDC score (n = 258)—4 autografts (n = 61) and 10 allografts (n = 197)—and 22 cohorts (n = 429) reported the postoperative IKDC score—7 autografts (n = 138) and 15 allografts (n = 291). No significant difference was found between the cohorts regarding the pre- or postoperative IKDC scores (P = .64 and P = .06, respectively).
Discussion
The findings of this review indicated no significant differences in objective outcomes when comparing autografts and allografts for PLC reconstruction, with both graft types restoring function and stability, even in the presence of concurrent cruciate reconstructions, at a minimum 2-year follow-up. The inclusion and exclusion criteria were variable for each study, but all studies met the predetermined inclusion criteria for this meta-analysis. There was significant heterogeneity of variables throughout the studies regarding the mechanism of injury, chronicity of injury, concomitant ligamentous injuries, and previous procedures. The goal of this analysis, as with all meta-analyses, was to obtain a large enough sample size to give power to the findings observed in relation to graft outcomes, regardless of individual study variability.
The included studies reported many differing variables; however, graft failure rates and laxity on varus stress radiographs were the most commonly reported objective outcomes. The Lysholm score and the IKDC subjective score were the most commonly reported patient-reported outcomes, with the autograft and allograft groups demonstrating significant improvements after PLC reconstruction when comparing pre- and postoperative scores. No significant between-group differences were detected in postoperative IKDC scores. However, a significant difference of 4 points was noted in favor of the autograft group when comparing the postoperative Lysholm scores. Additionally, this difference is unlikely to be clinically significant. There is no previously reported minimal clinically important difference for Lysholm scores regarding PLC reconstruction; nonetheless, there has been a previously reported minimal clinically important difference of 10.1 for Lysholm scores in ACL reconstruction. 28 Both the autograft and allograft groups easily met the minimal clinically important difference of 10.1 for Lysholm scores for ACL reconstruction, although this is not entirely applicable.
There are likely other important factors in determining outcomes for PLC reconstruction, including associated cruciate ligament injuries. Most of the studies included in this review did not delineate their outcomes based on concurrent cruciate injuries; however, previous studies have demonstrated differing outcomes when PLC reconstruction is associated with ACL and/or PCL reconstruction. Lutz et al 25 performed a retrospective review comparing return to work and return to sport-related activities after combined ACL/PLC reconstruction versus combined PCL/PLC reconstruction. They found that the PCL/PLC group had a significantly longer time to return to work and a significantly lower rate of return to sports and recreational activities. Schechinger et al 30 performed a case series comparing 2-ligamentous injuries, defined as PLC + ACL or PLC, and multiligamentous knee injuries, defined as PLC plus ACL and PCL. Their study found no significant difference in the IKDC or Lysholm scores between the 2 groups. No patients in either group required revision reconstruction. Shi et al 32 retrospectively reviewed 18 patients who underwent isolated PLC or PLC reconstruction combined with another ligamentous injury. They reported mean postoperative Lysholm scores of 91.67 (±3.34) for isolated PLC reconstruction, 89.63 (±3.11) for PLC/PCL reconstructions, and 91.67 (±1.53) for PLC/ACL reconstructions. These scores were within 2 points of each another; however, the final postoperative Lysholm score for the PLC/ACL/PCL group was only 80.50 (±6.36), demonstrating a somewhat expectedly worse outcome after more severe initial injuries. Rochecongar et al 29 performed a systematic review evaluating the difference in outcomes between combined PLC/ACL injuries and combined PLC/PCL injuries. Lysholm scores increased from 77 to 92 for the ACL group and 65 to 89 for the PCL group. These studies demonstrate the variability in findings throughout the literature regarding outcomes in PLC reconstruction with associated ACL or PCL injuries.
Numerous factors are involved in determining graft choice—including patient preference, surgeon experience, availability, cost, operative time, and morbidity. One advantage of allograft selection is shorter operative time due to the lack of need for autograft harvest. Arguments have been made that this shorter operative time may offset increased costs associated with allografts. Yet, multiple studies have demonstrated increased overall costs for ACLR when allograft tissue is used compared to autograft tissue, regardless of operative time.1,3,26 A study published in 2011 by Barrera et al 1 showed a significant difference in overall cost per case. They reported a total mean cost per case of $4147 ± $943 in the allograft group compared with $3154 ± $704 in the autograft group, despite the mean operating room time being >12 minutes in autograft cases. In 2010, Nagda et al 26 found that the mean total cost was $5465 for allograft ACL reconstruction and $4872 for autograft ACL reconstruction. Also, in 2010, Cooper and Kaeding 3 found a significant cost difference of $1123.16, with no significant differences in the operating room, anesthesia, or pharmacy costs. They concluded that the additional cost of using an allograft for ACL reconstruction was not offset by the decreased operative and recovery room costs.
Although costs appear to favor the autograft group, donor site morbidity is always a determining factor that must be considered when deciding to utilize an autograft versus an allograft. In the setting of a multiligamentous knee injury, some surgeons prefer to use a combination of an autograft for the cruciate ligaments and an allograft for the PLC injury. Surgeons prefer autograft reconstruction of the ACL and PCL because this technique decreases overall donor site morbidity and total operative time. It is important to note that allograft usage has become extremely safe with very low rates of adverse outcomes with improved donor screening and sterilization processes. 13
Significance and Limitations
To our knowledge, this is the first systematic review performed to evaluate outcomes of PLC reconstruction based on autograft or allograft selection. Previous reviews and studies have demonstrated the importance of PLC injury identification in multiligamentous knee injuries,7,10,22,23 the superiority of reconstruction versus repair for an acute PLC injury,8,33 and the lack of clinically significant difference in tibiofibular-based versus fibular-based reconstruction techniques for PLC reconstruction.31,38,39,40 We feel that determining the superiority between an autograft and an allograft is important to address as the treatment of PLC injuries evolves. Based on the findings of this study, we are unable to recommend autografts over allografts or vice versa, indicating that the graft choice should be determined based on the clinical scenario and a discussion between the surgeon and the patient.
We recognize that this review is not without limitations. First, this study did not control for surgical technique between the autograft and allograft groups. Based on previous literature, it was accepted that tibiofibular-based and fibular-based reconstruction techniques for PLC reconstruction had equivalent outcomes. Second, there was considerable variability in concurrent ligament injuries throughout the allograft and autograft groups. As stated previously, no consensus exists on how this affects PLC reconstruction outcomes. No study reported information on graft processing/irradiation. Most studies did not report the mechanism of injury, concurrent procedures, previous operations, or chronicity of injury, making it difficult to account for differences in management associated with these findings. Furthermore, as listed in the results section, many different allograft and autograft tissues were utilized throughout the other studies. It is unclear how these differing specific graft choices may affect autograft and allograft outcomes for PLC reconstruction. Last, although we believe this review has an adequate number of patients (N = 616) to perform an appropriate meta-analysis, all studies except for 1 included in the review were of level 3 or 4 evidence. Further studies with level 1 or 2 evidence would be of benefit to clearly define outcomes based on graft choice alone.
Conclusion
Factors may determine graft choice—including patient preference, surgeon experience, associated injuries, operative time, morbidity, and cost. Our analysis indicated no significant differences in the objective findings, including graft failure rate and side-to-side difference in varus laxity, based on graft type alone. PLC reconstruction significantly improved patient-reported outcomes with either autograft or allograft reconstruction. To further evaluate the findings demonstrated in this review, future prospective research should be performed directly comparing the outcomes between autograft and allograft PLC reconstruction.
Footnotes
Final revision submitted September 13, 2023; accepted September 30, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.M. has received education payments and consulting fees from Arthrex, Exactech, Linvatec, Smith+Nephew, and Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
