Abstract
Background:
Socket-tunnel overlap during meniscal allograft transplantation (MAT) combined with anterior cruciate ligament reconstruction (ACLR) may compromise graft integrity and lead to impaired fixation and treatment failure.
Purpose/Hypothesis:
The purpose of this study was to determine optimal socket-tunnel drilling parameters for medial and lateral MAT with concurrent ACLR using artificial tibias and computed tomography (CT) scans for 3-dimensional (3D) modeling. It was hypothesized that clinically relevant socket tunnels could be created to allow for concurrent medial or lateral MAT and ACLR without significant risk for overlap at varying tunnel guide angles.
Study Design:
Descriptive laboratory study.
Methods:
A total of 27 artificial right tibias (3 per subgroup) were allocated to 9 experimental groups based on the inclination of the socket tunnels (55°, 60°, and 65°) created for simulating medial and lateral MAT and ACLR. Five standardized socket tunnels were created for each tibia using arthroscopic guides: one for the ACL tibial insertion and one for each meniscus root insertion. CT scans were performed for all specimens and sequentially processed using computer software to produce 3D models for quantitative assessment of socket-tunnel overlap risk. Statistical analysis was performed with Kruskal-Wallis and Mann-Whitney U tests.
Results:
No subgroup consistently presented significantly safer distances than other subgroups for all distances measured. Three cases (11%) and 24 cases (~90%) of tunnel overlap occurred between the ACL tunnel and tunnels for medial and lateral MAT, respectively. Most socket-tunnel overlap (25 of 27; 92.6%) occurred between sockets at depths ranging between 6.3 and 10 mm from the articular surface. For ACLR and posterior root of the lateral meniscus setting, the guide set at 65° increased socket-tunnel distances.
Conclusion:
When combined ACLR and MAT using socket tunnels for graft fixation is performed, the highest risk for tibial socket-tunnel overlap involves the ACLR tibial socket and the lateral meniscus anterior root socket at a depth of 6 to 10 mm from the tibial articular surface.
Clinical Relevance:
Setting tibial guides at 65° to the tibial articular surface with the tunnel entry point anteromedial and socket aperture location within the designated anatomic “footprint” will minimize the risk for socket-tunnel overlap.
Keywords
Meniscal deficiency results in altered knee biomechanics that can lead to chronic pain, instability, and joint degeneration.1,2,13,16,22,24,29 In the anterior cruciate ligament (ACL)–deficient knee, integrity and function of the menisci are accentuated based on their roles as secondary stabilizers of the joint.8,19,22 Tears of the medial meniscus and lateral meniscus can occur with ACL tears in up to 45% and 65% of cases, respectively.1,31 The occurrence of meniscal injuries, especially in the medial meniscus, is also higher in the setting of chronic ACL deficiency.1,16 The combination of meniscal and ACL deficiency severely alters knee biomechanics and function and increases the risk for joint degeneration, with some studies reporting a rate of associated knee osteoarthritis of >80%.2,14,16,23
Therefore, for select patients with symptomatic ACL tear and meniscal deficiency, meniscal allograft transplantation (MAT) performed in combination with ACL reconstruction (ACLR) can be an effective treatment option.3,11,16,18,20 The procedure can be technically demanding, however, as meniscal allograft root fixation and ACL graft tibial fixation require transtibial tunnels or socket tunnels centered on precise anatomic insertion sites that may overlap and could lead to impaired fixation and potential treatment failure.3,32 This may also be a consideration in patients with prior ACLR undergoing subsequent MAT. As such, tibial guide angles and socket-tunnel positioning that minimize risk for socket-tunnel overlap for arthroscopic all-inside ACLR and double-bone plug MAT must be optimized. However, optimal positioning parameters have not been well characterized to date.
The purpose of this study was to determine optimal socket-tunnel guide angulation for medial and lateral MAT combined with concurrent ACLR. We hypothesized that clinically relevant socket tunnels could be created to allow for concurrent medial MAT, lateral MAT, and ACLR without significant risk for overlap.
Methods
Sample Allocation and Tunnel Creation
A total of 27 artificial right tibias (Sawbones) were allocated into 9 groups (3 in each group) for comprehensive assessment of socket-tunnel configurations. Testing groups were defined based on the angle for tibial guide inclination for meniscus root transtibial tunnel drilling and socket reaming at 55°, 60°, and 65° and then subdivided based on angle for tibial guide inclination for ACLR transtibial tunnel drilling and socket reaming at 55°, 60°, and 65° (Figure 1). Angles represented the relative inclination of the tunnel in relation to the plane of the articular surface of the proximal region of the tibia. For the same tibia, the angulation used for creating the lateral and medial meniscus roots was identical. Therefore, a sample labeled as ACL55RT60, for instance, indicates that the socket tunnel for the ACL was created at an angulation of 55° while the angulation for the socket tunnels of the medial and lateral menisci (for each root) was set at 60°.
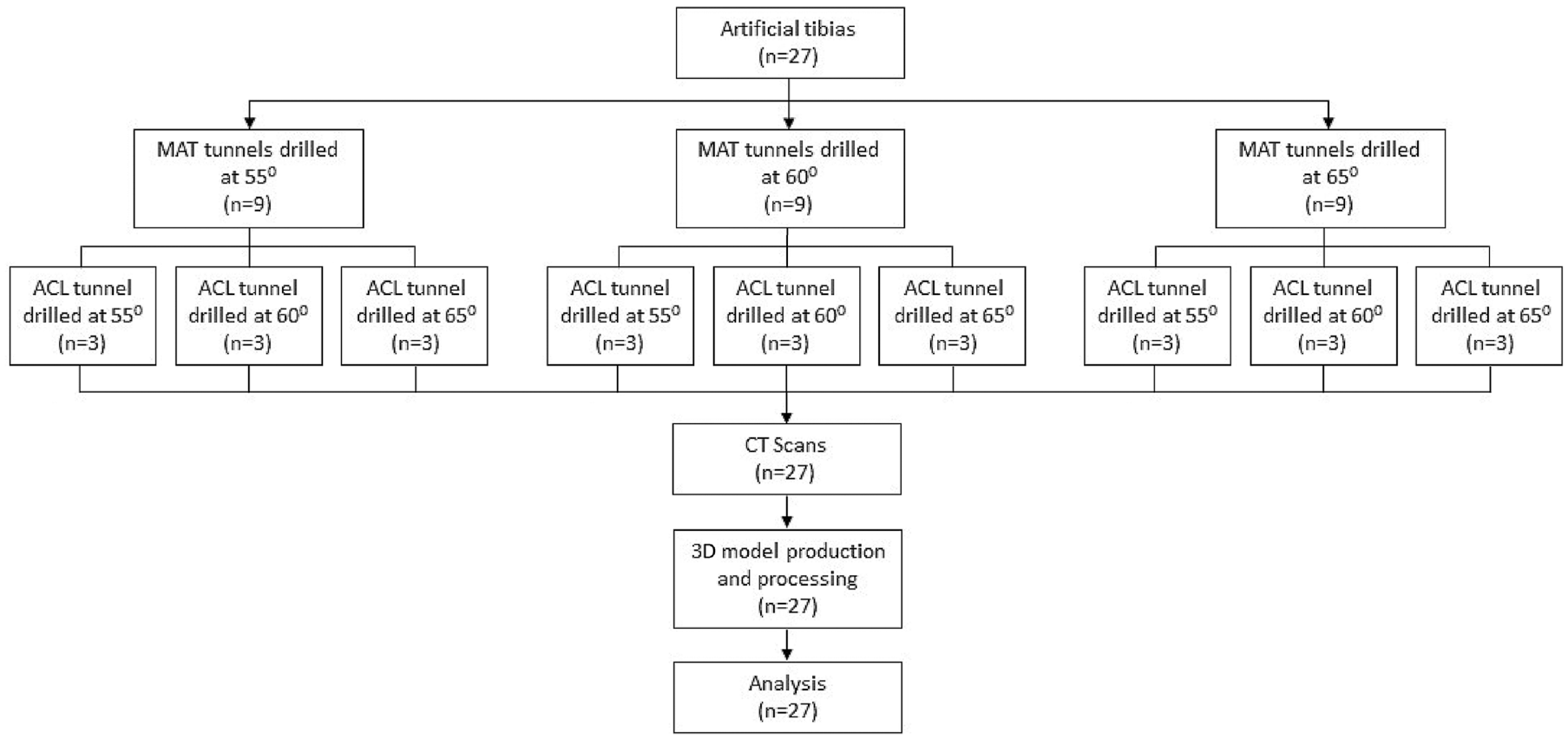
Flowchart for study sample allocation. ACL, anterior cruciate ligament; CT, computed tomography; MAT, meniscal allograft transplant; 3D, 3-dimensional.
Socket-tunnel creation was performed by a single fellowship-trained orthopaedic surgeon (S.F.D.) using a commercially available retrograde reamer (FlipCutter 3; Arthrex) through a tibial ACL drill guide (Arthrex). Socket tunnels for the posterior and anterior roots of the lateral and medial menisci were created based on a double-bone plug suspensory-fixation MAT technique, 30 and for anatomic single-bundle ACLR, on an all-inside suspensory-fixation technique,4,15 totaling 5 socket tunnels in each tibia (Figure 2). The entry point for all tunnels was located on the anteromedial tibia in accordance with the designated guide angle and socket location with the tibial guide placed to exit within the anatomic “footprint” of the respective meniscus root or ACL insertion, respectively. The “tunnel” was defined as the initial drilling with the unflipped retrograde reamer, while the “socket” was the depth drilled retrograde from the articular surface to afford ACL graft or MAT bone plug placement. Tunnel diameters were all 3.5 mm in accordance with the reamer shaft diameter, and lengths were determined by entry point, guide angle, and required socket location and depth. The sockets were created by retrograde drilling along the tunnel axis using the adjustable reamer blade set to clinically relevant socket diameters and depths for concurrent ACLR and MAT, as follows: ACL, 9 mm (diameter) and 25 mm (depth); anterior meniscus roots, 9.5 mm (diameter) and 10 mm (depth); and posterior meniscus roots, 6 mm (diameter) and 10 mm (depth). These were done in accordance with the senior author's (S.F.D.) current clinical practice.
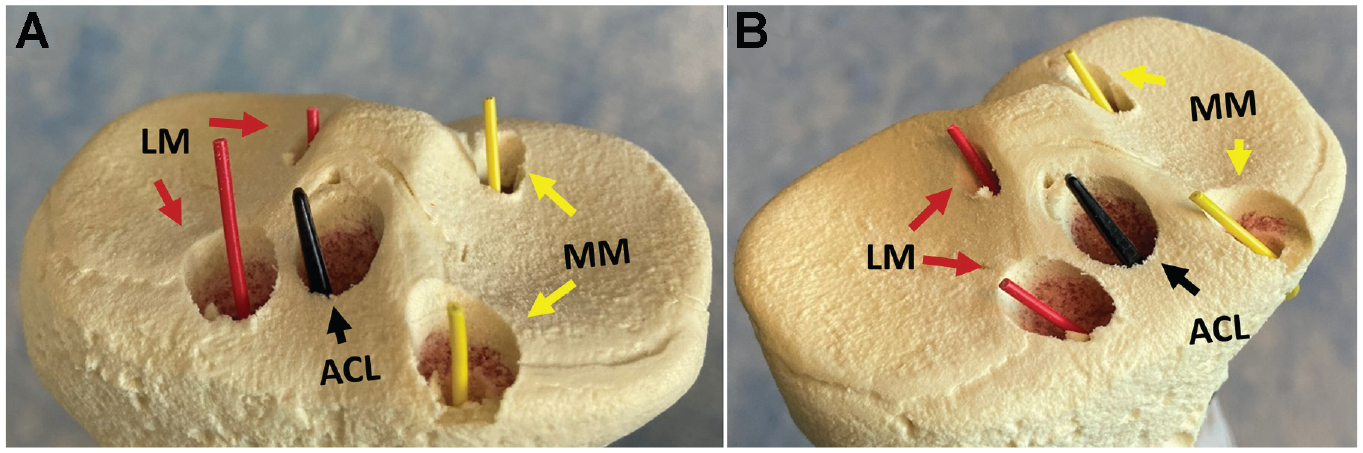
(A) Anterior and (B) oblique views of the proximal aspect of an artificial tibia after drilling the 5 socket tunnels. ACL, anterior cruciate ligament; LM, lateral meniscus; MM, medial meniscus.
Imaging and Formatting
After socket-tunnel creation, helical computed tomography (CT) imaging of each tibia was performed in a Toshiba Aquilion 64 scanner (Canon Medical Systems) using a bone acquisition protocol at 0.5-mm slice thickness and spacing, under the supervision of a single radiologist, blinded to specimen allocation. CT imaging was standardized in terms of positioning, slice number and orientation, and acquisition time.
Each CT scan was sectioned, one slice at a time, in the x-y plane using 3D Slicer Version 4.10.2 (https://www.slicer.org), which is a multiplatform, free open-source software for visualization and image computing. 7 Each slice was traced using the “level tracing” feature in 3D Slicer and then manually optimized using the “paint” and “erase” tools. Once all slices in a scan were evaluated, a 3-dimensional (3D) model was produced and then “smoothed” using the smoothing feature (Figure 3). After smoothing, the models were exported as .STL files and loaded into Autodesk Meshmixer (free software for working with triangle meshes; https://meshmixer.com) for further optimization. This process was repeated to produce the highest-quality model for each specimen based on the clarity, accuracy, and consistency of the internal geometry (Figure 4).

Computed tomography (CT) scans of a right proximal tibia after anterior cruciate ligament and meniscal allograft bone socket tunnels were created in the bone. CT scans were visualized and first processed using 3D Slicer software. Each scan was manually optimized and evaluated on 2-dimensional views—(A) axial, (B) coronal, and (C) sagittal—before (D) the 3-dimensional models were produced.

Model renderings of a right proximal tibia after socket-tunnel creation for anterior cruciate ligament and medial and lateral meniscal allograft transplantation tunnels. Autodesk Meshmixer was used for refinement of the 3-dimensional models.
Once each model was created, the proximal tibia was separated into individual .STL files using Meshmixer and decimated from ~130,000 triangles to 10,000 to 13,000 triangle faces to allow for processing in SolidWorks (SolidWorks Corp). Once loaded into SolidWorks, an .SLDPRT file was generated (which is a 3D model file format for SolidWorks). The opacity of the model was reduced to 30% to assist in internal evaluation. For each tunnel, entry and exit holes were defined by selecting a triangle face parallel to the apparent plane of the hole to create a 3-point circle on the defined plane. Once all holes were defined, reference axes were generated through the center point of each respective pair to create centerline distance references for each tunnel.
To ensure socket-tunnel diameter accuracy for analysis, a reference plane was created at the surface of the entry hole normal to the centerline distance of each socket tunnel, and a circular extruded cut was made along the axis with the previously measured socket diameter and depth. Once all socket tunnels were defined, the distance between individual socket tunnels was measured using the “evaluation” tool in SolidWorks (Figure 5). For socket tunnels whose axes crossed inside the models, the “measure” tool was used to determine the nearest point on each axis, and the radius of each tunnel was subtracted from the distance between those points to determine the shortest distance between the tunnels. For tunnels whose axes did not cross within the model, the nearest point should be at the articular surface. In those cases, points were manually selected along each respective tunnel and were carefully checked to ensure no nearer point existed before the “measure” tool was used to determine the nearest-point distance.
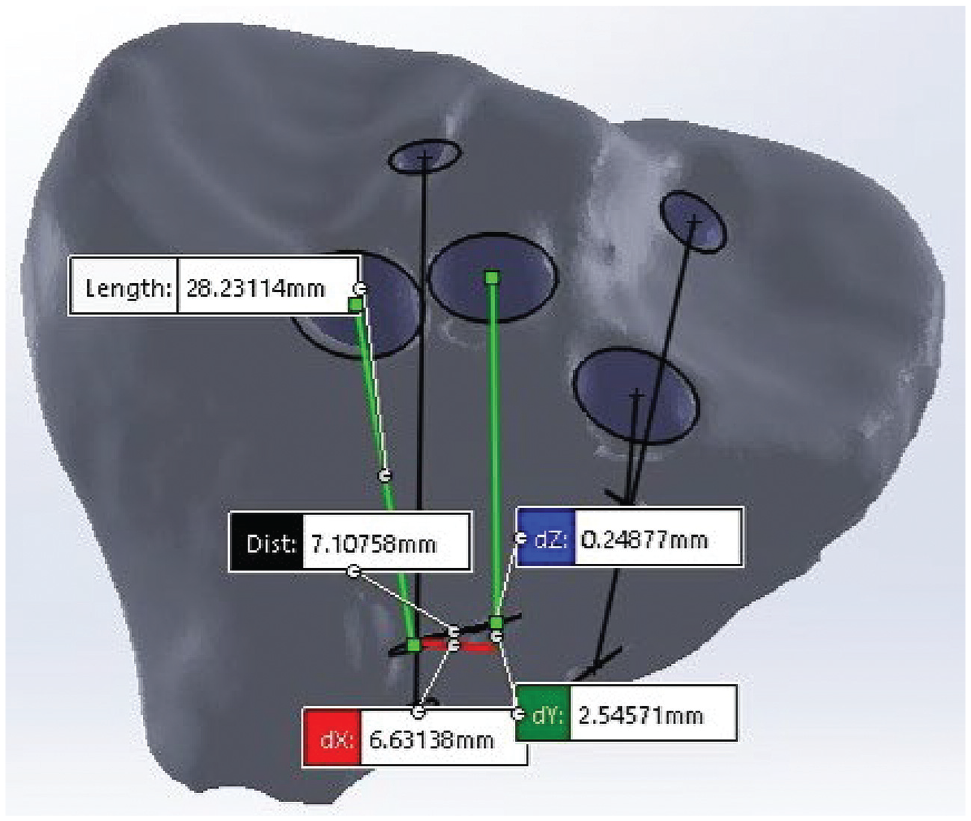
Three-dimensional computed tomography model rendering and evaluation of a right proximal tibia after socket-tunnel creation for anterior cruciate ligament and medial and lateral meniscal allograft transplantation tunnels. All socket tunnels and axes were defined and the distances between individual socket tunnels were measured using the evaluation tools in SolidWorks. The centerline distance is demonstrated by the black line.
Data Analysis
To test the study hypothesis, distances (mean ± 95% CI in millimeters) between the ACL tibial socket tunnel and each of the meniscus root socket tunnels were characterized. The variables considered were the distances between the central axes of the tunnels (centerline distance) and the calculated distance for the closest edges between the tunnels (nearest-edge distance). Overlap was defined as a distance <2 mm between socket tunnels, as previously reported.9,12 As there were several data sets that failed tests for normality, nonparametric statistical analyses were performed using the Kruskal-Wallis test. The Mann-Whitney U test was used to compare differences between each of 2 independent groups. A P value <.05 was considered statistically significant. Significance values were adjusted by Bonferroni correction for multiple tests.
Results
ACL and Medial Meniscus Roots
The mean centerline and nearest-edge distances between tibial socket tunnels for the ACLR and medial meniscus roots are shown in Table 1. Regarding socket-tunnel distance between the ACLR and anterior root of the medial meniscus, the mean centerline and nearest-edge distances increased with increasing tibial guide inclination angle for the ACLR socket tunnel; however, no statistically significant differences were noted. There were no significant differences in centerline or nearest-edge distance with regard to socket-tunnel distance between the ACLR and the posterior root of the medial meniscus.
Centerline and Nearest-Edge Distances Between ACL Tibial Tunnels and Tunnels of the Medial Meniscus a

Data are reported as mean ± 95% CI. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction.
ACL and Lateral Meniscus Roots
The mean centerline and nearest-edge distances between tibial socket tunnels for the ACLR and lateral meniscus roots are shown in Table 2. No statistically significant differences for centerline or nearest-edge distance between tibial guide inclination angle groups were noted for ACLR and anterior root of the lateral meniscus. However, for the ACLR and posterior root of the lateral meniscus socket-tunnel distances, the ACL60RT65 subgroup (ACL tunnel drilled at 60° and posterior root of lateral meniscus drilled at 65° of angulation) was associated with significantly greater distances when compared with the ACL60RT55 (P = .016) and ACL65RT55 (P = .006) groups. In addition, the ACL55RT55 group was associated with significantly greater distances when compared with the ACL60RT55 (P = .012) and ACL65RT55 (P = .016) groups.
Centerline and Nearest-Edge Distances Between ACL Tibial Tunnels and Tunnels of the Lateral Meniscus a
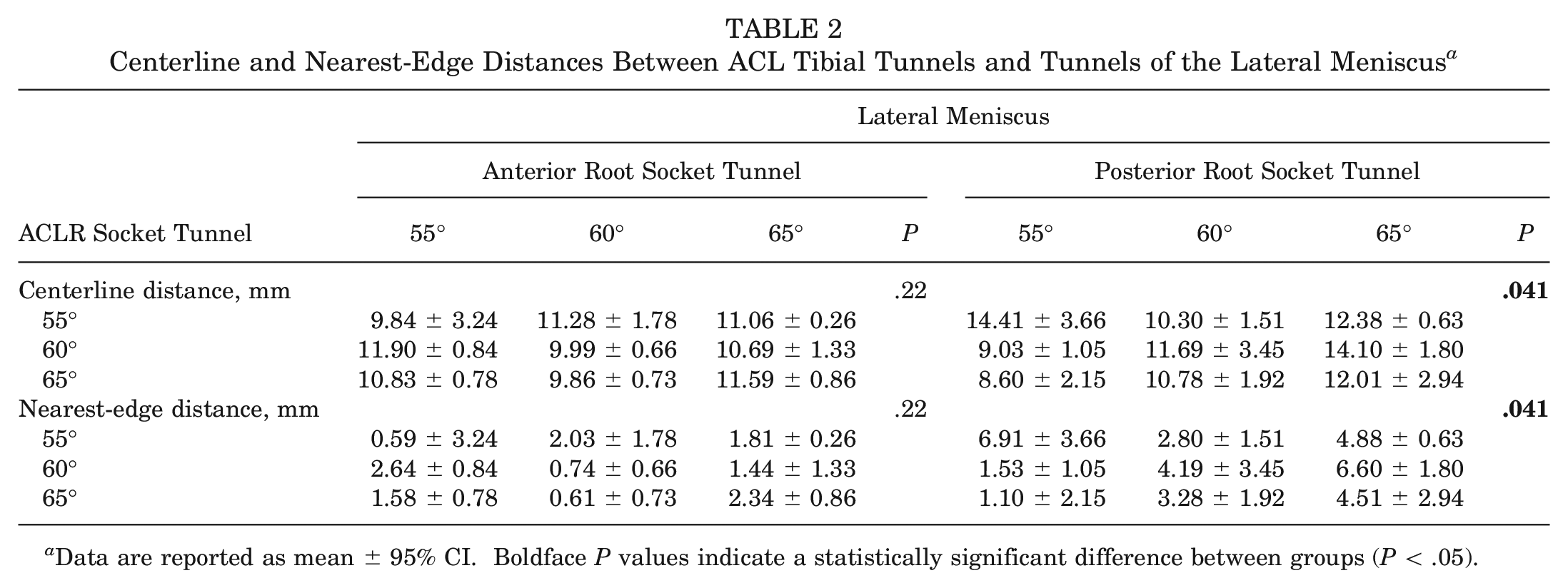
Data are reported as mean ± 95% CI. Boldface P values indicate a statistically significant difference between groups (P < .05).
Socket-Tunnel Overlap
Using the a priori definition for socket-tunnel overlap of nearest-edge distance of <2 mm, we found that overlap occurred in 27 of 108 possible combinations (25%), as follows: ACLR and anterior root of lateral meniscus, 17 cases (63%); ACLR and posterior root of lateral meniscus, 7 cases (26%); and ACLR and medial meniscus anterior root, 3 cases (11%) (Table 3). No cases of socket-tunnel overlap were found for ACLR and medial meniscus posterior root.
Subgroups With Overlap (Distance ≤2 mm) Between Socket Tunnels a

There were 3 specimens per subgroup. Dashes indicate that overlaps did not occur in any specimens of a subgroup. ACL, anterior cruciate ligament; ACLR, anterior cruciate ligament reconstruction; RT, meniscus root.
The vast majority of socket-tunnel overlap (25 of 27 cases; 92.6%) occurred between sockets at depths ranging between 6.3 and 10 mm from the articular surface. In 2 cases (7.4%), specimens from subgroups ACL60RT60 and ACL65RT55, the socket overlap occurred at the articular surface of the tibia. For all other sockets, no overlap occurred at the tibial articular surface, and nearest-edge distance or “residual bone bridge thickness” ranged from 2.1 to 10.2 mm. Every combination of ACL and root angulation had at least one episode of overlap when performing combined ACLR and lateral meniscus root repair as would be done in a meniscal transplant. Concomitant ACLR and medial meniscus root repairs only had overlap with ACL55 combinations.
Discussion
The results of the present study suggest that the highest risk for overlap involves the ACLR tibial socket and the lateral meniscus anterior root socket at a depth of 6 to 10 mm from the tibial articular surface. The data also highlighted some risk for overlap when creating ACLR sockets and lateral meniscus posterior root or medial meniscus anterior root sockets, although to a much smaller degree. Based on the socket and tunnel diameters used, overlap of sockets at the tibial articular surface (aperture) is a relatively minor concern, as it only occurred in 2 of the combinations, and the residual bone bridge between sockets was >2 mm for all other combinations. When considering a combined ACLR and meniscal transplant situation, every combination of ACLR and root angulation had at least 1 episode of overlap when performing combined ACLR and lateral meniscus root repairs (anterior and posterior), while concomitant ACLR and medial meniscus root repairs only had overlap with ACL55 combinations. Taken together, these data suggest that tibial guides set at 65° to the tibial articular surface and placed using anteromedial entry points and socket aperture locations within the designated anatomic footprints minimize risk for socket-tunnel overlap such that ACLR–lateral meniscus anterior root socket overlap is the only high-risk combination. Given all these data, our hypothesis was confirmed that clinically relevant socket tunnels could be created for concomitant ACLR and medial or lateral MAT with minimal risk of overlap.
Gursoy et al 9 reported similar findings from their study examining the risk for tunnel overlap when performing ACL and posterior cruciate ligament reconstructions in conjunction with posterior medial and lateral menisci root repairs. Those investigators used an image processing software to create tunnels in 3D models produced from CT scans of 20 cadaveric knees. They reported 100% overlap between ACLR and posterior root of the lateral meniscus tunnels when using standard techniques, with overlap completely eliminated when a more distal entry point for the lateral meniscus tunnel and parallel orientation with the ACLR tunnel were implemented. Bozkurt et al 3 evaluated a surgical technique for simultaneous combined transplantation of the lateral and medial meniscus with ACLR in cadaveric specimens using a keyhole technique for the lateral meniscus and a modified technique for the posterior root of the medial meniscus. The shortest distances between the tunnels on the tibia were 1.9 ± 0.4 cm and 1.8 ± 0.9 cm on the tibial crest, with no overlap between tunnels on CT scan. Other techniques may be associated with greater risk for overlap.17,32 Methods to mitigate overlap concerns include the use of a bone bridge technique for MAT11,18,20,32 or staging ACLR and MAT10,18; however, bone bridge MATs are associated with other risks and complications, and the current recommendation for meniscus- and ACL-deficient knees is to address them concurrently in order to better restore knee biomechanics and provide a combined protective effect with regard to knee stability.§
When using socket tunnels for double–bone plug suspensory-fixation MAT and anatomic single-bundle ACLR all-inside suspensory-fixation techniques, the following steps are recommended for reducing overlap-related complications: (1) temporarily placing a metal rod in the socket or tunnel when drilling at-risk combinations 5 so that the surgeon can use it as a reference, better direct the tunnels that will still be created, and avoid communication between them; (2) creating all the tunnels using step 1 and only after that proceed with the passage and fixation of the grafts in order not to compromise the integrity of the grafts during the creation of the tunnels (even if communication occurs between the tunnels when using this technique, this will not necessarily result in treatment failure if the graft or fixation devices are not damaged during tunnel creation); (3) directing the tunnels for the lateral meniscus to a site lateral to the tibial anterior tuberosity (but this may require an additional incision and more morbidity related to muscle dissection); (4) better distributing the tunnels’ entry points in the anteromedial tibia; and (5) using a modified anatomic plug technique for the lateral meniscus in which a 4-mm spherical anterior bone plug is used (therefore smaller than the plug size currently used and with a lesser risk of tunnel overlap), or consider using soft tissue–only fixation for the anterior horn of the lateral meniscus. Regarding step 5, using a keyhole technique as opposed to transosseous tibial drilling could also address this, although this technique may have its own pitfalls. 17 This is explained by the proximity between the anatomic insertion of the anterior root of the lateral meniscus and the tibial insertion of the ACL, which are only 5 to 7 mm apart.10,17 There is also the possibility of using a nonanatomic technique for the anterior horn of the lateral meniscus to artificially create a greater distance between the lateral meniscus anterior root and ACL tibial insertion sites. However, the literature shows that this has only been used for salvage procedures. 6 Hence, there is not enough evidence to support the use of a nonanatomic technique for lateral meniscal transplantation since an anatomic technique is the optimal treatment and a nonanatomic technique may lead to incongruency and degenerative changes. 28 The findings of this study agree with those in prior literature 26 and clearly demonstrate the greatest risk of overlap when drilling the lateral meniscus anterior horn, making it necessary for surgeons to have alternative fixation strategies if they encounter this issue. Ultimately, tunnel overlap may not be clinically significant, although it can potentially lead to compromised fixation of 1 or both grafts depending on fixation strategy.
Limitations
This study is not without limitations. First, all surgical procedures in our study were performed by a single surgeon, which represents both a limitation and a strength. Tunnel placement was largely left to the discretion of the single surgeon, which could influence bias; however, this also recreates reproducibility, as any error or bias was likely to be replicated by this single surgeon. In order to minimize error, 3 specimens per ACL–meniscal guide combination were used. While this is a small sample size of only 3 specimens per subgroup, it was felt that this would best limit variability while remaining conscious to the overall number of specimens. Another limitation was the use of Sawbones models for the procedures when human specimens would be preferred to best mimic real anatomy and clinical data more accurately. Sawbones models have the advantage of ease of availability and less cost, as well as consistency, as all the models had the same anatomy with regard to tibial size, slope, plateau, and spines. It is also worth considering that Sawbones models are devoid of cartilage, such that the sockets made in our model were likely deeper than those in a true clinical situation, in which approximately 3 mm of cartilage would be present. Next, the angulation of the socket tunnels was standardized only in relation to one plane (tibial articular surface), and these tunnels have an obvious 3D configuration. We used a bone plug technique for both menisci; minor adjustments such as directing the lateral meniscus root tunnels toward a site that is lateral to the anterior tibial tuberosity could change the trajectory of the tunnels and therefore change the overall distances between tunnels. Placement of the guide was therefore performed using clinically relevant and routine placement on the anteromedial tibia for all specimens. While a limitation, this most replicates true surgical conditions, as this is the same guide used in the operating room.
Conclusion
When performing combined ACLR and MAT using socket tunnels for graft fixation, the highest risk for tibial socket-tunnel overlap was found to involve the ACLR tibial socket and the lateral meniscus anterior root socket at a depth of 6 to 10 mm from the tibial articular surface. Setting tibial guides at 65° to the tibial articular surface with the tunnel entry point anteromedial and socket aperture location within the designated anatomic footprint will minimize the risk for socket-tunnel overlap.
Footnotes
Final revision submitted October 18, 2023; accepted October 24, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.D. has received research support from Arthrex; education payments from Arthrex, Encore Medical, Smith+Nephew, Medical Device Business Services, and Zimmer Biomet; nonconsulting fees from DJO and Synthes; honoraria from Encore Medical; and hospitality payments from Stryker. C.R.C. has received research support from Arthrex and Zimmer; consulting fees from Arthrex and CONMED Linvatec; nonconsulting fees from Arthrex, CONMED Linvatec, and Musculoskeletal Transplant Foundation; and royalties from Arthrex, CONMED Linvatec, and Musculoskeletal Transplant Foundation. C.W.N. has received education payments from Arthrex and Encore Orthopedics, consulting fees from Guidepoint Consulting and Arthrex, nonconsulting fees from Arthrex and Vericel, hospitality payments from Synthes GmbH, and other financial or material support from the AO Foundation. J.P.S. has received research support from Arthrex; education payments from Elite Orthopedics; consulting fees from Arthrex, DePuy Synthes/Medical Device Business Services, Orthopedic Designs North America, and Smith+Nephew; nonconsulting fees from Synthes and Medical Device Business Services; and royalties from Thieme. J.L.C. has received research support from AO Trauma, Arthrex, Collagen Matrix, DePuy Synthes, Musculoskeletal Transplant Foundation, Purina, Regenosine, and Sites Medical; consulting fees from Arthrex and Trupanion; royalties from Arthrex, Musculoskeletal Transplant Foundation, and Thieme; and was a board or committee member for the Musculoskeletal Transplant Foundation. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
