Abstract
Background:
Reduced graft failure rates have been reported after anterior cruciate ligament (ACL) reconstruction combined with anterolateral complex (ALC) augmentation. However, the preoperative diagnosis of concomitant ALC injury remains a clinical challenge.
Purpose:
To identify the altered rotational tibiofemoral position on magnetic resonance imaging (MRI) in ACL-injured patients with concomitant ALC injury.
Study Design:
Cross-sectional study; Level of evidence, 3.
Methods:
Based on the evaluation of ALC abnormalities on MRI scans by experienced surgeons, 123 patients with nonchronic (<3 months) ACL injury confirmed by arthroscopy were included. The patients were divided into 2 groups—an ALC-injured group (n = 57) and an ALC-intact group (n = 66). The altered rotational tibiofemoral position was evaluated and compared by quantitatively measuring internal rotational tibial subluxation (IRTS) and axial internal tibial rotation (ITRa) on MRI. Multivariate logistic regression and receiver operating characteristic (ROC) analyses were performed to identify the factors associated with concomitant MRI-determined ALC injury.
Results:
The ALC-injured group showed significantly increased IRTS (P < .001), ITRa (P < .001), lateral anterior tibial subluxation (ATS) (P < .001), and global ATS (GATS) (P = .002) compared with the ALC-intact group, while no significant difference in medial ATS (P = .810) was observed. A strong positive correlation was identified between IRTS and ITRa (rP = 0.809; P < .001). Multivariate analyses revealed that IRTS (P < .001) and GATS (P = .016) were associated factors for the presence of concomitant MRI-determined ALC injury. IRTS (area under the curve [AUC] = 0.734) was more strongly associated with the outcome than GATS (AUC = 0.658) in ROC analyses, suggesting a more significant internal rotational subluxation than anterior subluxation of the tibia. An IRTS threshold of 3.1 mm demonstrated a specificity of 84.2% for indicating the presence of concomitant MRI-determined ALC injury.
Conclusion:
The presence of concomitant MRI-determined ALC injury in ACL-injured patients was associated with a significant increase in IRTS and ITRa compared with those with intact ALC, indicating that these MRI measurements of the altered rotational tibiofemoral position could serve as potential quantifiable indicators for identifying concomitant ALC injury in clinical practice.
Keywords
The anterolateral complex (ALC) of the knee consists of the distal iliotibial band with its Kaplan fiber, the anterolateral ligament (ALL), and the anterolateral capsule of the knee joint, contributing to internal rotational stability as a secondary stabilizer to the anterior cruciate ligament (ACL). 4,9,21 Recently, the clinical relevance of ALC has drawn much attention, with studies reporting the reduction in the graft failure rate of conventional ACL reconstruction (ACLR) combined with an augmentation of ALC.2,5,16 While there is a lack of evidence, appropriate indications for ALC augmentation may include failed ACLR, high-grade pivot shift, generalized ligamentous laxity, young patients, and a desire to return to high-risk pivoting sports.4,5
A lack of clear indications for ALC augmentation can be attributed to the clinical challenge in the preoperative diagnosis of concomitant ALC injury. A high-grade pivot shift under anesthesia is regarded by some as an indicator of internal rotational knee instability and may reflect an ALC injury17,19; nonetheless, the grading results rely on the examiner's perception and experience. 11 To identify additional features of concomitant ALC injury, some researchers have introduced magnetic resonance imaging (MRI) characteristics to assess injuries and abnormalities of the ALL. 7 However, others have claimed that routinely performed 3 mm-thick MRI scans are insufficient to make a clinical decision.1,18 Furthermore, a study reported that the MRI-determined ALC injury was not associated with a positive pivot-shift test in ACL-injured knees. 15 Therefore, alternative approaches are still needed to develop an objective and accurate method for preoperatively identifying the concomitant ALC injury.
Recent studies have suggested an approach for evaluating knee rotation through quantitative MRI measurement of tibiofemoral position.12,25 The altered rotational tibiofemoral position can be described by internal rotational tibial subluxation (IRTS) measured on sagittal MRI 25 and by the internal tibial rotation measured on axial MRI (ITRa). 12 The IRTS was found to be slightly increased in ACL-injured patients compared with normal knees, 25 while ITRa was initially utilized in patients with patellar instability. 25 Given that a concomitant ALC injury can lead to internal rotational knee instability,4,9,21 it is plausible to suggest that an increased IRTS and ITRa may be associated with such injuries.
This study aimed to evaluate and compare the difference in IRTS and ITRa between concomitant MRI-determined ALC-injured patients (ALC-injured group) and MRI-determined ALC-intact patients (ALC-intact group). We hypothesized that increased IRTS and ITRa would be observed in the ALC-injured group compared with the ALC-intact group.
Methods
Patients
Patients who underwent primary single-bundle ACLR with autologous hamstring tendon grafts between 2012 and 2018 by 1 of the senior authors (C.W.) were retrospectively reviewed and included in the study after meeting the following inclusion criteria: (1) available MRI scans at our institute; (2) no other concomitant ligament injuries or fractures; (3) no history of patellar dislocation; (4) no history of knee surgery; and (5) injury-to-MRI time of <3 months. A total of 310 patients were initially enrolled, and their descriptive data, arthroscopic findings, and preoperative knee MRI scans were obtained and analyzed. The study protocol received institutional review board approval. Given the retrospective nature of this study, we sought and obtained a waiver for informed consent from our ethics committee. As a result, participants did not provide informed consent.
MRI Determination of Concomitant ALC Injury
Concomitant MRI-determined ALC injury was evaluated on preoperative MRI by 3 experienced surgeons (Y.Y, C.W., and J.W.) based on the characterized abnormalities reported by previous studies.7,20,22 Each examiner was blinded to the other examiners’ results. As mentioned in previous studies, there were challenges in identifying the ALL and other ALC structures on the MRI scans,1,18 and a poor interobserver reliability in diagnosing ALL injuries in the setting of an ACL tear was reported. 22 To ensure accurate categorization of the ALC condition for further analyses, we excluded 187 patients with indistinct ALC structures and those with inconsistent diagnoses from 3 examiners. A total of 57 patients with confirmed concomitant MRI-determined ALC injury were included in the ALC-injured group, and 66 patients with identifiable intact ALC were included in the ALC-intact group.
MRI Measurements
The MRI scans were performed using a GE Discovery MR750 3.0 T System (PrizMed Imaging, Unit A Willowick) according to the standardized institutional technique. Slice thicknesses were 3 mm for the sagittal T1-weighted images (T1WIs) and fat-saturated proton density-weighted images. Patients were arranged to maintain the supine position, and the knee was placed in the knee coil with neutral rotation. In addition, patients were required to maintain their knee extended with no hyperextension. A slight knee flexion of within approximately 5° was acceptable, if inevitable. All MRI measurements were performed in Adobe Illustrator CC 2021 (Adobe Inc). The original image, including a measuring scale, was imported into the software. The distance was measured by tracking the movement of the reference line and recorded in pixels on the image. Subsequently, the measuring scale was measured to determine the conversion factor from pixels to mm.
To ensure the reliability of the measurements, 30 knees were randomly selected for intra- and interobserver reliability testing of the measurements using the intraclass correlation coefficient (ICC) with 95% CI. 10 The intraobserver reliability test involved repeating the measurements at a 6-week interval by a single observer (Z.Z.) using the 2-way mixed effects, absolute agreement, and single rater/measurement model. The results showed good to excellent reliability for IRTS (ICC, 0.94 [95% CI, 0.88-0.97]) and excellent reliability for ITRa (ICC, 0.96 [95% CI, 0.93-0.98]) measurements. The interobserver reliability was assessed based on measurements performed by 3 observers (Z.Z., W.B., and C.W.) blinded to each other's results using the 2-way random effects, absolute agreement, and single rater/measurement model. The results demonstrated good to excellent reliability for IRTS (ICC, 0.86; 95% CI, 0.75-0.92) and ITRa (ICC, 0.88 [95% CI, 0.80-0.94]) measurements. The first observer's first observation was used for further analysis.
Determination of the Longitudinal Tibial Axis
The longitudinal tibial axis (LTA) was initially determined as a reference line on the central slice of the sagittal view, with the intercondylar eminence, anterior tibial cortex, and posterior tibial cortex appearing as a concave shape. 8 Three circles were drawn to determine the LTA. The proximal circle was tangent to the anterior, posterior, and proximal borders of the tibial cortex, while the 2 distal circles were tangent to the anterior and posterior cortical borders. The center of the 2 distal circles was positioned on the circumference of the adjacent proximal circle. The LTA was defined by a line connecting the centers of the most proximal and the most distal circle (Figure 1A). The center of the middle circle may not be exactly positioned on the longitudinal axis (Figure 1A).

MRI measurements for the evaluation of (A-C) anteroposterior and (D-F) rotational tibiofemoral positions. (A) The LTA was measured on the central slice determined by 3 circles. (B) A reference line parallel to the LTA was then drawn on the central lateral slice at the posterior margin of the hyperintense region of the posterior femoral condyle and another at the posterior margin of the hyperintense region of the tibial plateau on the image. The distance between these 2 lines was measured as LATS. The angle between the line perpendicular to the LTA and the line tangent to the tibial plateau was calculated as the LPTS. (C) The same protocol was followed for the measurements of MATS and MPTS on the central medial slice. (D) For the measurement of ITRa, the sagittal central slice showing the level of the axial slice was used. (E) The PFCL was drawn tangent to the femoral condyles. (F) The PTL was drawn tangent to the tibial plateau. The angle between the PFCL and PTL was measured as ITRa. ITRa, axial internal tibial rotation; LATS, lateral anterior tibial subluxation; LFC, lateral femoral condyle; LPTS, lateral posterior tibial slope; LTA, longitudinal tibial axis; MATS, medial anterior tibial subluxation; MFC, medial femoral condyle; MPTS, medial posterior tibial slope; PCL, posterior cruciate ligament; PFCL, posterior femoral condylar line; PTL, posterior tibial line.
We measured the angle between the LTA and the vertical line to evaluate the variation in knee flexion. Patients with large knee flexion angles were excluded from the analysis. For patients with an irregularly shaped tibia, the apex point of the best-fit curve of the anterior border was used as a landmark to determine the proximal circle. Detailed information about the measuring protocol for such patients, designed to improve the reliability of the measurements, is presented in Appendix Figure A1.
Measurement of Anteroposterior Tibiofemoral Position
On sagittal T1WIs, measurement of anterior tibial subluxation (ATS) in the lateral and medial compartments (LATS and MATS) was performed according to previous studies,23-25 using the LTA 8 as the reference line (Figure 1, B and C).
In the lateral compartment, LATS was measured on the central lateral slice that showed the most medial section of the fibula at the tibiofibular joint. A line parallel to the LTA was drawn at the posterior margin of the hyperintense region of the posterior femoral condyle and another at the posterior margin of the hyperintense region of the tibial plateau on the image. The distance between these 2 lines was measured as the LATS (Figure 1B). The thickness of the bone cortex was not taken into consideration in the measurements, as it appears hypointense on T1WIs and can be difficult to distinguish from the surrounding structures.
In the medial compartment, MATS was measured on the central medial slice that showed the insertion of the medial gastrocnemius tendon on the femur. The same protocol was performed (Figure 1C).
The global ATS (GATS) was defined as the mean value of the LATS and MATS 6 : GATS = (LATS+MATS)/2.
Measurement of Rotational Tibiofemoral Position
The rotational tibiofemoral position was evaluated on sagittal and axial MRI. On the sagittal view, the IRTS was calculated by subtracting the MATS from the LATS, indicating the amount of internal tibial rotation 25 : IRTS = LATS – MATS.
On the axial view, ITRa was measured following the protocol described by Lin et al. 12 The posterior femoral condylar line (PFCL) was drawn tangent to the posterior border of the femoral condyles at the level where the trochlear groove was deepest while remaining well defined 3 (Figure 1, D and E). The posterior tibial line (PTL) was drawn tangent to the posterior border of the tibial plateau at the most distal slice where the PCL was inserted into the bone (Figure 1, D and F). The angle between the PFCL and PTL was measured as ITRa (Figure 1F). A negative value of ITRa indicated external tibial rotation.
Measurement of PTS
Measurement of PTS in lateral and medial compartments (LPTS and MPTS) was performed following the protocol described by Hudek et al. 8 The LPTS was measured on the same central lateral slice as the LATS measurement in the lateral compartment. The angle between the line perpendicular to the LTA and the line tangent to the lateral tibial plateau was measured as the LPTS (Figure 1B). For patients with an irregular shape of the lateral tibial plateau, measuring LPTS may be challenging, and the measuring protocol for this situation is shown in Appendix Figure A1.
In the medial compartment, MPTS were measured on the same central medial slice as the MATS measurement, and the same protocol was performed (Figure 1C).
Statistical Analysis
Statistical analyses were conducted using the R software (for Mac OS X GUI; Version 4.0.0; R Foundation for Statistical Computing). The variables were reported as means with standard deviations and ranges or as frequencies with percentages. The normality of continuous variables was assessed using the Shapiro-Wilk test, while the Bartlett test was utilized to test the equality of variances. P < .05 was considered statistically significant.
The chi-square test, the chi-square test with Yates continuity correction, and the Fisher exact test were utilized to identify differences in discrete variables between the ALC-injured and ALC-intact groups. Pearson correlation analysis was performed to determine the correlation between IRTS and ITRa, which was confirmed to follow a bivariate normal distribution using the Shapiro-Wilk multivariate normality test. Multivariate logistic regression models were established to identify potential associated factors for concomitant MRI-determined ALC injury using the R function “glm.” The dependent variables were categorized as 1 for ALC-injured and 0 for ALC-intact. The diagnostic performance of associated factors in predicting the presence of concomitant MRI-determined ALC injury was evaluated using receiver operating characteristic (ROC) curves and area under the curves (AUCs). The best cutoff value was calculated as the threshold that maximizes the Youden index. The Wald chi-square test P values in logistic regression models and AUC values were compared to identify which factors were most strongly associated with the outcome.
To estimate the statistical power of the study, a power analysis was performed with an acceptable alpha error of .05, utilizing the “pwr.t2n.test” function in the R package “pwr” (n1 = 57; n2 = 66; significance level = .05; alternative = “2-sided”). The effect size was calculated using the Cohen d based on the mean value of IRTS in the ALC-injured and ALC-intact groups, as well as the pooled standard deviation. The power of the study was determined to be 99.8% based on this analysis.
Results
Patient characteristics, arthroscopic findings, and MRI measurements are summarized in Table 1. Univariate analysis revealed no significant difference in descriptive data or arthroscopic findings between the ALC-injured and ALC-intact groups (Table 1). The ALC-injured group showed significantly increased LATS (P < .001), GATS (P = .002), IRTS (P < .001), and ITRa (P < .001) compared with the ALC-intact group, while no significant difference in MATS (P = .810) was identified (Table 1 and Figure 2). No significant difference was observed between the 2 groups in LPTS (P = .805) and MPTS (P = .725).
Patient Characteristics, Arthroscopic Findings, and MRI Measurements a

Data are reported as mean ± SD (range) or n (%) unless otherwise indicated. Bold P values indicate statistically significant differences between groups (P < .05). ALC, anterolateral complex; BMI, body mass index; GATS, global anterior tibial subluxation; IRTS, internal rotational tibial subluxation; ITRa, axial internal tibial rotation; LATS, lateral anterior tibial subluxation; MATS, medial anterior tibial subluxation; MRI, magnetic resonance imaging.

Comparison of the anteroposterior and rotational tibiofemoral positions between the ALC-injured and ALC-intact groups. ALC, anterolateral complex; GATS, global anterior tibial subluxation; IRTS, internal rotational tibial subluxation; ITRa, axial internal tibial rotation; LATS, lateral anterior tibial subluxation; MATS, medial anterior tibial subluxation. A statistically significant difference between groups: **P < .01; ***P < .001.
The correlation analysis between IRTS and ITRa showed a strong positive correlation between the 2 variables (rP = 0.809; P < .001), indicating that there would be collinearity between these 2 variables if they were simultaneously included in a multivariate model (Figure 3). To account for this collinearity, only 1 of these variables was included in the multivariate model to describe the rotational tibiofemoral position. When IRTS was included in the model, multivariate logistic regression analyses revealed that IRTS (odds ratio [OR], 1.247; P < .001) and GATS (OR, 1.270; P = .016) were associated factors for the presence of concomitant MRI-determined ALC injury (Table 2). When ITRa was included in the model, similar results were obtained (OR, 1.145 [P = .002] for ITRa; OR, 1.335 [P = .004] for GATS) and are summarized in Appendix Table A1.

A scatter plot showing a strong positive correlation (rP = 0.809) between IRTS (measured on sagittal MRI) and ITRa (measured on axial MRI). ALC, anterolateral complex; IRTS, internal rotational tibial subluxation; ITRa, axial internal tibial rotation; MRI, magnetic resonance imaging.
Results of Multivariate Logistic Regression Showing Associated Factors for the Presence of Concomitant MRI-Determined ALC Injury a

Dependent variable: 1 = ALC-injured, 0 = ALC-intact (n = 123). Dash indicates value is not relevant to the results. Bold P values indicate statistical significance (P < .05). ALC, anterolateral complex; BMI, body mass index; GATS, global anterior tibial subluxation; IRTS, internal rotational tibial subluxation; LPTS, lateral posterior tibial slope; MPTS, medial posterior tibial slope; MRI, magnetic resonance imaging; OR, odds ratio.
The ROC analyses evaluating the diagnostic performance of predicting the presence of concomitant MRI-determined ALC injury are summarized in Table 3 and Figure 4. The best cutoff values for IRTS and GATS were 3.1 mm (specificity, 84.2%; sensitivity, 59.1%) with an AUC of 0.734 and 4.7 mm (specificity, 63.2%; sensitivity, 63.6%) with an AUC of 0.658, respectively (Table 3 and Figure 4). The P values in the logistic regression models (Table 2) and the AUC values (Table 3 and Figure 4) were compared among these associated factors, indicating that IRTS (P < .001; AUC = 0.734) showed a stronger association with the outcome than GATS (P = .016; AUC = 0.658), suggesting that concomitant MRI-determined ALC injury can result in a more significant internal rotational subluxation than anterior subluxation of the tibia. When ITRa was included in the analyses, similar results were obtained (cutoff value of 6° for ITRa; specificity, 71.9%; sensitivity, 63.6%; AUC = 0.712) and are summarized in Table 3 and Appendix Figure A2.
ROC Analyses of IRTS, ITRa, and GATS for Predicting the Presence of Concomitant MRI-Determined ALC Injury a

ALC, anterolateral complex; AUC, area under the receiver operating characteristic curve; GATS, global anterior tibial subluxation; IRTS, internal rotational tibial subluxation; ITRa, axial internal tibial rotation; MRI, magnetic resonance imaging; ROC, receiver operating characteristic.

ROC curves evaluating the diagnostic performance of IRTS and GATS in predicting the presence of concomitant MRI-determined ALC injury. The best cutoff values were calculated as the thresholds that maximize the Youden index and are presented as “threshold (specificity, sensitivity)” in the figure. ALC, anterolateral complex; AUC, area under the receiver operating characteristic curve; GATS, global anterior tibial subluxation; IRTS, internal rotational tibial subluxation; MRI, magnetic resonance imaging; ROC, receiver operating characteristic.
Discussion
The most important finding of the present study was that increased IRTS and ITRa were observed in ACL-injured patients with concomitant MRI-determined ALC injury compared with the ALC-intact group, indicating that the quantitative MRI measurements of the altered rotational tibiofemoral position were significantly associated with the MRI-determined diagnosis of concomitant ALC injury. Furthermore, we found that an IRTS threshold of 3.1 mm demonstrated a specificity of 84.2% and a sensitivity of 59.1% in indicating the presence of concomitant MRI-determined ALC injury. For ITRa, the threshold was 6° with a specificity of 71.9% and a sensitivity of 63.6%.
The preoperative diagnosis of concomitant ALC injury remains a clinical challenge because of the lack of reliable examinations for assessing rotational knee instability. Although the pivot-shift test under anesthesia is commonly used to evaluate internal rotational knee instability,13,19 the grading results rely on the examiner's perception 11 and cannot be quantitatively analyzed. Moreover, the characterization of ALC abnormalities on MRI has raised debate, as only 1 or 2 images can be detected in the coronal view, given the length and width of the ALL. 18 Miyaji et al 15 also reported that MRI-determined anterolateral capsule injury was not associated with the pivot-shift test in ACL-injured knees. In addition, poor interobserver reliability in diagnosing ALL injuries in the setting of an ACL tear has been reported. 22 Therefore, in this study, to ensure accurate classification of the ALC-injured and ALC-intact groups, 3 experienced surgeons independently evaluated the MRI scans while being blinded to each other's assessments. We excluded 187 patients with indistinct ALC structures and those with inconsistent diagnoses from 3 examiners. It should be acknowledged that this exclusion process may introduce enrollment bias. However, our analysis of descriptive data indicated no significant differences between the 2 groups, and the multivariate analysis further demonstrated that the potential influence was minimal.
In this study, we performed quantitative measurements to describe the altered rotational tibiofemoral position, and we hypothesized that an increased internal tibial rotation would be observed, given that the ALC is considered a secondary stabilizer to the ACL and contributes to internal rotational knee stability.4,9,21 Our results revealed that increased IRTS and ITRa were both significantly associated with the presence of concomitant MRI-determined ALC injury, as confirmed by multivariate logistic regression models. These findings support our initial hypothesis. In clinical practice, the measurement of this altered rotational tibiofemoral position can serve as a potential indicator for patients with a concomitant ALC injury, especially considering that routine 3 mm-thick MRI may not provide enough information for clinical decision-making.
Furthermore, we observed that an increase in LATS and GATS was associated with concomitant MRI-determined ALC injury, while MATS was not significantly associated. An increase in internal rotation of the tibia leads to an increase in LATS and a relative decrease in MATS, resulting in a more pronounced increase in IRTS, while the variation in GATS is subtle. Therefore, our findings indicate that a concomitant MRI-determined ALC injury can result in both internal rotational subluxation and anterior subluxation of the tibia compared with an isolated ACL injury without MRI-determined ALC injury. To determine which direction of subluxation is more strongly associated with the outcome, we performed logistic regression analyses and found that IRTS and ITRa had higher AUC values and lower P values than GATS (Tables 2 and 3, Figure 4), suggesting that a concomitant MRI-determined ALC injury can result in a more significant variation in rotational rather than anteroposterior tibiofemoral position. These findings are consistent with a recent biomechanical study 21 and address a gap in the clinical and radiological assessment of the ALC function.
Another noteworthy finding was the identification of an IRTS threshold of 3.1 mm and an ITRa threshold of 6° as the best cutoff values indicating the MRI-determined ALC injury. However, the absolute values of the AUCs and sensitivities of IRTS and ITRa suggest that they may not be considered sufficiently “strong” indicators to predict the diagnosis of concomitant ALC injury independently. This observation is reasonable given the complex nature of ALC injuries, as relying solely on a single variable may not sufficiently capture the diverse characteristics of ALC involvement. Nonetheless, these findings provide promising insights and lay the foundation for future studies to develop a clinical prediction model for ALC injury.
Limitations
There are several limitations to this study that should be considered. One limitation is the slice thickness of 3 mm, which may impact the accuracy of the measurements. In addition, the height of the tibia available on the images and the knee flexion angle at the time of MRI could also influence the results. To address the potential bias from the variation in knee flexion angle, we suggested estimating this angle before inclusion and controlling for this factor (Appendix Figure A1). However, it may still be challenging to ensure that all patients have the same knee flexion angle, especially in a retrospective analysis. While acknowledging this limitation, it is important to consider that it may improve the generalizability of the findings. Furthermore, we provided detailed measuring protocols to improve the reliability of the results.
Several confounding factors—such as concomitant meniscal injuries, a long injury-to-MRI time,14,25,26 and steep PTS 24 —could also have affected the results. To control for these variables, we only included nonchronic ACL-injured patients with an injury-to-MRI time of <3 months. Univariate analyses showed no significant differences in descriptive data and the PTS between the 2 groups, indicating that these variables were controlled. This study did not include the pivot-shift test; thus, no direct evidence was provided to support the association between these quantitative MRI measurements and internal rotational knee instability. The idea of future research focusing on the association between the cutoff value of IRTS or ITRa and high-grade pivot shift is intriguing.
Conclusion
The presence of concomitant MRI-determined ALC injury in ACL-injured patients was associated with a significant increase in IRTS and ITRa compared with those with intact ALC, indicating that these MRI measurements of the altered rotational tibiofemoral position could serve as potential quantifiable indicators for identifying concomitant ALC injury in clinical practice.
Footnotes
Appendix
Results of Multivariate Logistic Regression Showing Associated Factors for the Presence of Concomitant MRI-Determined ALC Injury (Including ITRa) a
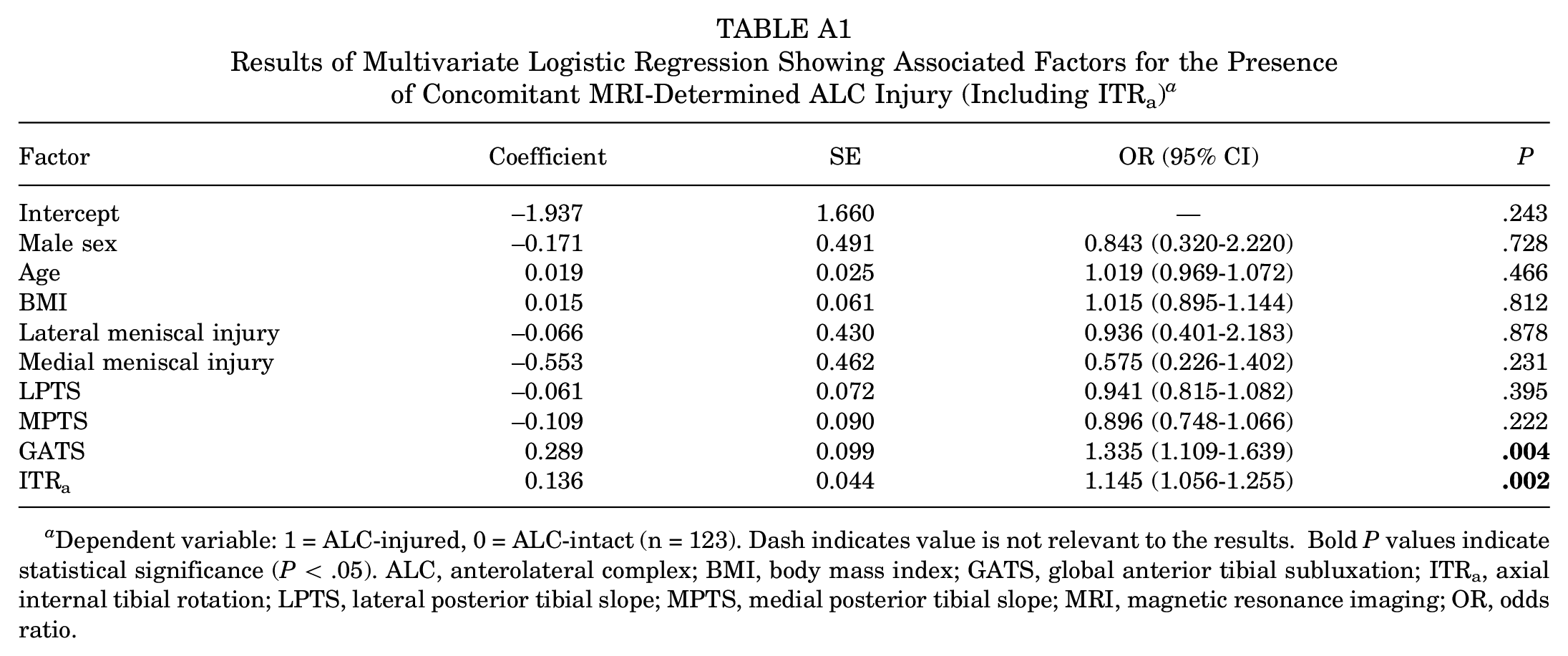
| Factor | Coefficient | SE | OR (95% CI) | P |
|---|---|---|---|---|
| Intercept | −1.937 | 1.660 | — | .243 |
| Male sex | −0.171 | 0.491 | 0.843 (0.320-2.220) | .728 |
| Age | 0.019 | 0.025 | 1.019 (0.969-1.072) | .466 |
| BMI | 0.015 | 0.061 | 1.015 (0.895-1.144) | .812 |
| Lateral meniscal injury | −0.066 | 0.430 | 0.936 (0.401-2.183) | .878 |
| Medial meniscal injury | −0.553 | 0.462 | 0.575 (0.226-1.402) | .231 |
| LPTS | −0.061 | 0.072 | 0.941 (0.815-1.082) | .395 |
| MPTS | −0.109 | 0.090 | 0.896 (0.748-1.066) | .222 |
| GATS | 0.289 | 0.099 | 1.335 (1.109-1.639) |
|
| ITRa | 0.136 | 0.044 | 1.145 (1.056-1.255) |
|
Dependent variable: 1 = ALC-injured, 0 = ALC-intact (n = 123). Dash indicates value is not relevant to the results. Bold P values indicate statistical significance (P < .05). ALC, anterolateral complex; BMI, body mass index; GATS, global anterior tibial subluxation; ITRa, axial internal tibial rotation; LPTS, lateral posterior tibial slope; MPTS, medial posterior tibial slope; MRI, magnetic resonance imaging; OR, odds ratio.
Final revision submitted August 9, 2023; accepted August 21, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research support was received from the National Natural Science Foundation of China grant (82072403) and the Clinical Cohort Project Program of Peking University Third Hospital (BYSYDL2021014). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Peking University Third Hospital (ref No. M2020433).
