Abstract
Background:
Platelet-rich plasma (PRP) has demonstrated beneficial effects on healing of the bone-tendon interface (BTI).
Purpose:
To determine the optimal initiation time for PRP application after rotator cuff repair in an animal model.
Study Design:
Controlled laboratory study.
Methods:
A total of 136 C57BL/6 mice were included; 40 mice were used to prepare PRP, while 96 mice underwent acute supraspinatus tendon (SST) repair. The animals were randomly divided into 4 groups: a control group and 3 groups in which PRP was injected into the injury interface immediately after surgery, on the 7th postoperative day (PRP-7d), and on the 14th postoperative day. At 4 and 8 weeks postoperatively, the animals were sacrificed, blood was collected by eyeball removal, and samples of the SST-humerus complex were collected. Histological, imaging, immunological, and biomechanical data were compared among the groups using 1-way analysis of variance with the Bonferroni post hoc test.
Results:
Histological analysis revealed that the fibrocartilage layer at the BTI was larger in the PRP-7d group compared to the other groups at both 4 and 8 weeks postoperatively. Moreover, the PRP-7d group exhibited improved proteoglycan content and distribution compared to the other groups. Enzyme-linked immunosorbent assay results demonstrated that at 4 weeks postoperatively, higher concentrations of transforming growth factor–β1 and platelet-derived growth factor–BB (PDGF-BB) were seen in the PRP-7d group versus the PRP-14d and control gruops (P < .05), and at 8 weeks postoperatively, the concentration of PDGF-BB was higher in the PRP-7d group versus the control group (P < .05). Biomechanical testing at 4 weeks postoperatively revealed that the failure load and ultimate strength of the SST-humerus complex were superior in the PRP-7d group compared to the other groups (P < .05), at 8 weeks, PRP-7d group was superior to the control group (P < .05). Additionally, at 8 weeks postoperatively, the PRP-7d group exhibited a greater trabecular number and trabecular thickness at the BTI compared to the PRP-14d and control gruops (P < .05).
Conclusion:
PRP promoted healing of the BTI after a rotator cuff injury at an early stage.
Clinical Relevance:
A PRP injection on the 7th postoperative day demonstrated superior therapeutic effects compared with injections at other time points.
A rotator cuff injury is a common cause of shoulder dysfunction and pain, affecting 7% to 30% of the general population. 11 The incidence and diagnosis of rotator cuff injuries are on the rise because of aging of the population and advancements in diagnostic techniques, respectively. 41 Self-healing is often challenging because of limited blood flow around the tendons. Initially, nonoperative treatment is preferred, but surgery becomes necessary when nonoperative measures fail. In the United States alone, there are over 4.5 million annual visits related to rotator cuff injuries, resulting in more than 300,000 rotator cuff repair procedures.12,40
Despite significant advancements in surgical techniques, the retear rate after surgery remains high. Several factors, such as tear severity and fatty infiltration, can influence retear rates. 31 Normal healing of the bone-tendon interface (BTI) involves the composition of bone, calcified fibrocartilage, uncalcified fibrocartilage, and tendon. However, repair of the insertion sites is challenging because of the unique architecture of the rotator cuff. Instead of regenerating the native insertion site, healing occurs through fibrous scar tissue formation.7,24 Presently, achieving satisfactory outcomes after repair is difficult, prompting researchers to explore interventions that modify the tissue’s biological environment. The delivery of growth factors to enhance tendon-bone healing has emerged as an appealing option, 3 and the use of platelet-rich plasma (PRP) has gained attention as a biological therapy in the field of sports medicine.29,30
PRP is a concentrated product obtained through the centrifugation of whole blood, containing a supraphysiological concentration of platelets. Upon application, platelets release various growth factors and cytokines, including platelet-derived growth factor (PDGF) and transforming growth factor (TGF), which play crucial roles in promoting injury healing by stimulating angiogenesis, cell proliferation, and differentiation.27,45 While basic studies have demonstrated the effectiveness of PRP,29,42 clinical outcomes remain controversial. Most studies to date have applied PRP immediately after repair14,28 without considering the natural tissue repair mechanisms triggered after an injury, including the induction of hematomas and early production of growth factors postoperatively. The continuous addition of high concentrations of growth factors through PRP may impact these processes and contribute to differences in clinical outcomes.8,19 Consequently, the optimal timing for PRP application after rotator cuff repair remains unclear.
In this study, we aimed to investigate the effects of PRP injections at different initiation times on healing of the BTI after rotator cuff repair in mice. The healing process was assessed through comprehensive histological, immunological, imaging, and biomechanical evaluations. We hypothesized that PRP injections on the 7th day postoperatively would be more favorable for injury recovery.
Methods
The experimental protocol of this study was approved by the local animal ethics committee. A total of 136 male C57BL/6 mice (age, 8 weeks; weight, 20-22 g; free from specific pathogens) were used. Previous studies have found that rodents are the most suitable and commonly used species for investigating rotator cuff injuries. 43 Overall, 40 mice were used to prepare PRP, and 96 mice underwent supraspinatus tendon (SST) repair of the right forelimb according to previous methods.20,30
Briefly, all mice were subjected to routine disinfection. Anesthesia was induced via an intraperitoneal injection of 0.3% pentobarbital sodium, and the mice were positioned in the left lateral decubitus position. A lateral incision was made in the right shoulder region, and subcutaneous tissue was bluntly separated. The position of the deltoid muscle was identified and minimally detached to expose the SST. The SST was secured using 6-0 PDS suture (Ethicon) in a row of 8 stitches. Subsequently, the SST was separated from the humeral head junction, and any remaining fibro-soft bone was removed. Bone tunnels were drilled from the posteromedial to anterior aspects of the humeral head, and PDS suture was passed through the tunnels to connect the SST to its original insertion site before attaching the tendon to the humeral head, stretching the tendon to confirm its proper tension, and covering it with saline gauze to keep moist. PDS suture was tightened, the deltoid muscle was repositioned, and the skin was closed using nonabsorbable suture (Ethicon). All animals received intramuscular buprenorphine (0.05 mg/kg/d; TIPR Pharmaceutical) for 3 consecutive days postoperatively, and animals were inspected for wound infections or other complications. Only mice with well-healed incisions were included, and mice with an infection at the incision site and contaminated samples were excluded. No animal had a surgical incision infection. Postoperatively, the mice were observed to awaken naturally, placed in cages for normal activities, and fed routinely, and the limbs were not braced.
The mice were randomly divided into 4 groups (n = 24 per group): control group, PRP injected immediately after surgery (PRP-0d), PRP injected on the 7th postoperative day (PRP-7d), and PRP injected on the 14th postoperative day (PRP-14d). The control group received a postoperative injection of 0.1 mL saline only at the site of the injury. The other 3 groups received an injection of preprepared PRP, consisting of a mixture of 0.1 mL PRP, 20 μL 10% calcium chloride, and 10 U bovine thrombin, into the injury interface at different time points after surgery. All injections were administered into the injury interface by a physician (C.Z.) knowledgeable in mouse anatomy.
The mice were euthanized at 4 and 8 weeks postoperatively, and both blood samples and the SST-humerus complex (SSTH) were collected for histomorphometric, immunological, and biomechanical testing at these postoperative time points. Additionally, micro–computed tomography (micro-CT) was performed at 8 weeks postoperatively to assess the morphological structure of the subchondral bone in the rotator cuff. A flowchart illustrating the study design is shown in Figure 1.

Flowchart depicting the study design. micro-CT, micro–computed tomography; PRP, platelet-rich plasma.
PRP Preparation and Analysis
PRP was prepared through allogeneic blood collection in this study, mice (age, 8 weeks; weight, 20-22 g) underwent eyeball removal, and approximately 5 mL of blood was collected from 5 mice. Blood was placed in a preprepared 0.5-mL centrifuge tube (Labnet) containing 3.2% trisodium citrate as an anticoagulant. PRP was prepared using the second centrifugation method of Landesberg et al. 17 Initially, the samples were centrifuged at 200g for 10 minutes. The entire supernatant was pipetted to a depth of 3 mm below the crossing interface. This was followed by a second centrifugation, again at 200g for 10 minutes. The upper three-quarters of the supernatant was discarded, and the remaining portion constituted PRP. In addition to being used for injections, portions of PRP were used for blood analysis. Specifically, 0.2 mL of whole blood and PRP were added to separate test tubes, and the platelet concentration was determined using an automated hematology analyzer.
Micro-CT
Micro-CT was used to evaluate the microstructure of newly formed bone at the BTI, following established protocols. Specimens were fixed in paraformaldehyde for 24 hours and rinsed with distilled water. Subsequently, the specimens were placed in a micro-CT scanner (Scanco Medical). 44 Image acquisition and data analysis were performed using CTAn, DataViewer, and CTvol software (SkyScan). The scanning parameters were as follows: resolution, 10.4 μm; energy, 55 kVp; exposure time, 0.4 seconds; rotation step, 0.36°; and rotation, 180°. The region of interest was selected to include the subchondral bone area at the interface of the supraspinatus muscle and tendon. Morphometric parameters of the bone structure were analyzed, including bone volume fraction (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), and trabecular separation (Tb.Sp).
Histology
Specimens were fixed in 10% neutral buffered formalin for 24 hours at room temperature and then decalcified in EDTA (ethylenediaminetetraacetic acid) decalcifying solution. After decalcification, the specimens were dehydrated using graded ethanol solutions and embedded in paraffin. The paraffin blocks were sectioned at a thickness of 5 μm along the coronal plane using a microtome. Hematoxylin and eosin (H&E) staining or safranin O/Fast Green staining was performed on the sections. H&E staining was used to observe the histological structure of the BTI, and safranin O/Fast Green staining was used to observe the proteoglycan (PG) content and distribution in the fibrocartilage layer at the BTI. The slices were digitally scanned and imaged (Pannoramic DESK, P-MIDI, P250; 3DHISTECH), and image browsing software (CaseViewer C.V.2.3; 3DHISTECH) was used for assessing SSTH healing.
Sections stained with safranin O/Fast Green were used for semiquantitative analysis, and the fibrocartilage area was measured using CaseViewer software. Image-Pro Plus software (Media Cybernetics) was used to evaluate the integrated optical density (IOD), which represents the PG content. There were 3 rectangular regions randomly selected from the fibrocartilage layer (100 × 100 μm 2 ), and the IOD values were averaged to determine the PG content of the regenerated fibrocartilage layer. Semiquantitative analysis of histological images was performed in a blinded fashion.
Immunology
Serum samples were collected at 4 and 8 weeks postoperatively to assess relevant growth factors using an enzyme-linked immunosorbent assay kit. 16 Blood was obtained by removing the eyeballs of the mice. Following the manufacturer’s instructions (Thermo Fisher Scientific), serum samples were subjected to antibody coating, with PDGF-BB or TGF-β1 antibody alone, while the standard solution was placed in separate wells. The plate was then incubated at 37°C for 60 minutes, followed by 5 washes with the washing solution. After drying, a color development reagent was added, and the plate was incubated at 37°C for 10 minutes to remove any excess light. Finally, a stop solution was added to terminate the color development reaction. Data were acquired and recorded using a microplate reader (Thermo Fisher Scientific).
Biomechanical Testing
The cross-sectional area (CSA), failure load, and ultimate strength of the specimens were assessed using a materials testing system. Before testing, the SSTH was fixed in a clamp with sandpaper. The humerus and SST were clamped separately, and the SSTH was pretreated under a 0.05-N tensile force for 1 minute. The width and thickness of the healed area were measured using a caliper, and the CSA was calculated (CSA [mm2] = width [mm] × thickness [mm]). Subsequently, the specimens were placed in a materials testing machine (2710-004; Instron). A 60° of abduction angle was employed for uniaxial pulling experiments to closely approximate the anatomic position of the SST. The SSTH was pulled at a rate of 1 mm/min until failure. 36
Statistical Analysis
Statistical analysis of the experimental data was conducted using SPSS software (Version 26.0; IBM). All quantitative data were expressed as means with standard deviations, and differences among the groups were analyzed using 1-way analysis of variance with the Bonferroni post hoc test. Statistical significance was set as P < .05, while high statistical significance was set as P < .01.
A priori power analysis was performed using G*Power software (Version 3.1, Germany) to determine the sample size required to achieve a statistical power of 80% at an alpha level of .05. Based on the calculations, a sample size of 21 mice in each group was deemed sufficient to detect the anticipated differences. Therefore, 96 mice (24 per group) were enrolled in this study.
Results
Analysis of Blood Components
Analysis of the composition of blood revealed that the platelet concentration in PRP was 1422.33 ± 139.68× 109/L, which was approximately 4.4 times higher than the concentration in whole blood (323.43 ± 30.74×109/L).
Micro-CT
According to micro-CT, the PRP-7d group exhibited superior healing compared to the other groups at 8 weeks after surgery. Analysis results of the morphological and structural parameters of subchondral bone are presented in Figure 2. There was no significant difference in the BV/TV among the groups (control: 45% ± 5%; PRP-0d: 48% ± 6%; PRP-7d: 49% ± 6%; PRP-14d: 47% ± 5%). However, the PRP-7d group showed a higher Tb.N (3.48 ± 0.34/mm) compared to the PRP-14d group (2.84 ± 0.28/mm; P < .01), and control group (2.57 ± 0.27/mm; P < .01), there was no significant difference between PRP-7d and PRP-0d group (3.07 ± 0.33/mm). Additionally, the PRP-0d group exhibited a superior Tb.Th (38.82 ± 4.66 μm) compared to the control group (31.08 ± 4.97 μm; P < .05), there was no significant difference between PRP-0d and PRP-14d group (34.58 ± 4.61 μm), while the PRP-7d group showed the highest Tb.Th (42.48 ± 4.71 μm). No significant differences were observed in the Tb.Sp among the 4 groups (control: 78.67 ± 8.24 μm; PRP-0d: 73.29 ± 7.65 μm; PRP-7d: 70.42 ± 8.28 μm; PRP-14d: 76.70 ± 7.82 μm).

(A) Micro–computed tomography of the supraspinatus tendon–humerus complex at 8 weeks postoperatively. The dashed box indicates the region of interest (ROI). Scale bar = 500 μm. (B) Quantitative analysis of the ROI. Error bars represent standard deviations. *P < .05. **P < .01. BV/TV, bone volume fraction; PRP, platelet-rich plasma; PRP-0d, PRP injected immediately after surgery; PRP-7d, PRP injected on the 7th postoperative day; PRP-14d, PRP injected on the 14th postoperative day; Tb.N, trabecular number; Tb.Sp, trabecular separation; Tb.Th, trabecular thickness.
Histology
Figures 3 and 4 display H&E staining and safranin O/Fast Green staining, respectively, at 4 and 8 weeks postoperatively. The H&E staining results revealed that during the early healing stage after repair of a rotator cuff injury, the PRP-7d group exhibited significantly superior healing of the fibrocartilage layer at the BTI compared to the PRP-0d, PRP-14d, and control groups. Moreover, healing observed in the PRP-0d group was better than that in the control and PRP-14d groups.
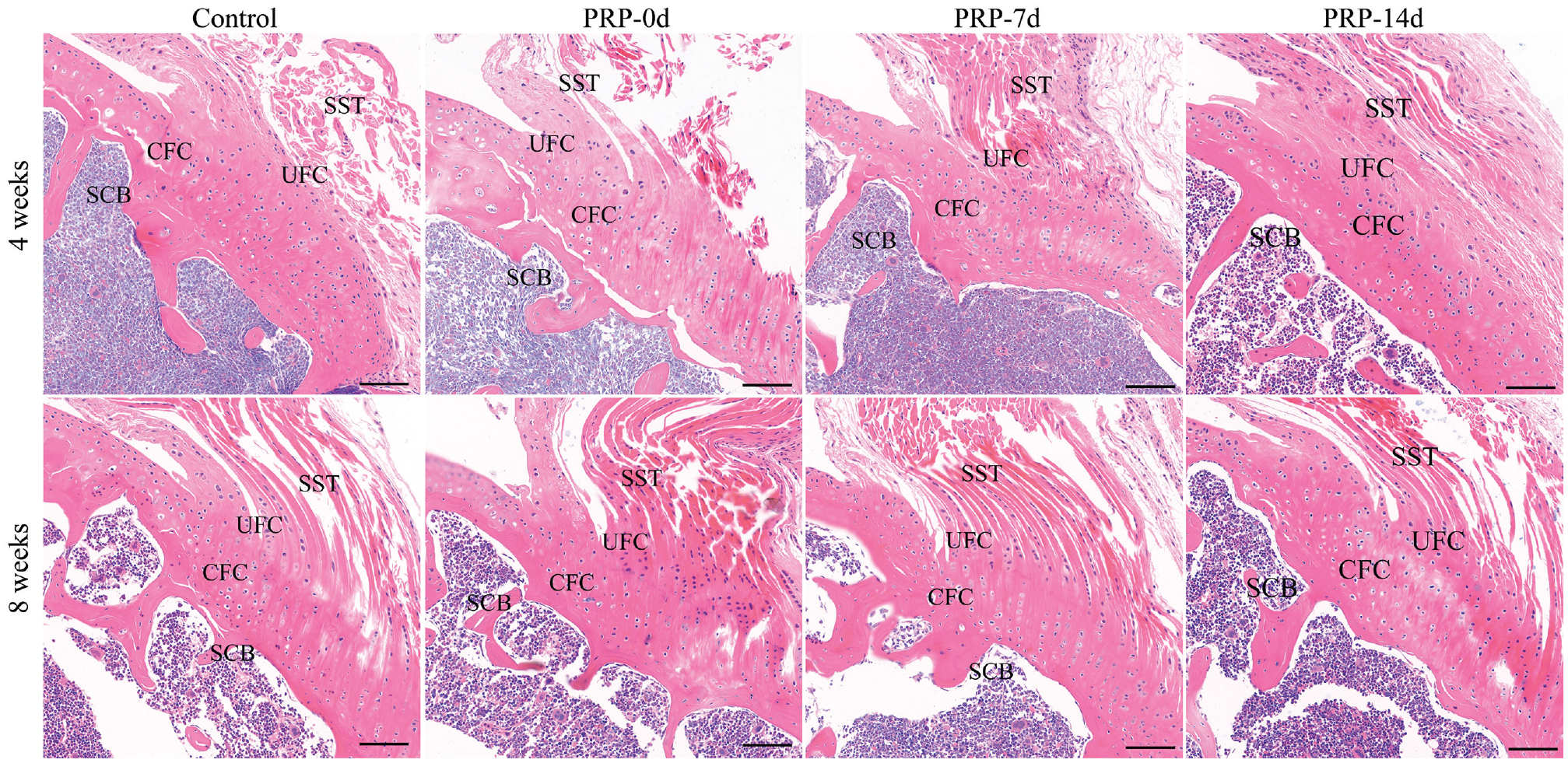
Hematoxylin and eosin staining of the supraspinatus tendon (SST)–humerus complex at 4 and 8 weeks postoperatively. Scale bar = 50 μm. CFC, calcified fibrocartilage; PRP, platelet-rich plasma; PRP-0d, PRP injected immediately after surgery; PRP-7d, PRP injected on the 7th postoperative day; PRP-14d, PRP injected on the 14th postoperative day; SCB, subchondral bone; UFC, uncalcified fibrocartilage.

(A) Safranin O/Fast Green staining of the supraspinatus tendon (SST)–humerus complex at 4 and 8 weeks postoperatively. Scale bar = 50 μm. Comparison between groups of the (B) fibrocartilage area and (C) integrated optical density (IOD), representing the proteoglycan content of the regenerated fibrocartilage layer. Error bars represent standard deviations. *P < .05. **P < .01. CFC, calcified fibrocartilage; PRP, platelet-rich plasma; PRP-0d, PRP injected immediately after surgery; PRP-7d, PRP injected on the 7th postoperative day; PRP-14d, PRP injected on the 14th postoperative day; SCB, subchondral bone; UFC, uncalcified fibrocartilage.
The safranin O/Fast Green staining results indicated that at 4 weeks after surgery, all 3 PRP groups exhibited a noticeable fibrocartilage layer at the injury interface (Figure 4B). The regeneration area in the PRP-7d group was superior to that in the other 2 PRP groups as well as the control group, while the PRP-0d group showed better regeneration than the PRP-14d and control groups. At 8 weeks after surgery, the PRP-7d group demonstrated improved PG content and distribution in the regenerated fibrocartilage layer at the injury interface compared to the other groups. Similarly, the PRP-0d group exhibited significantly better PG content than the control group. On semiquantitative analysis, the IOD of the regenerated fibrocartilage area and PG content in the repair area of the PRP-7d group were significantly higher compared to those of the PRP-14d and control groups at 4 and 8 weeks after surgery (P < .05) (Figure 4C).
Immunology
TGF-β1
At 4 weeks after surgery, the concentration of TGF-β1 in the PRP-7d group (1.23 ± 0.21 ng/mL) was significantly higher compared to that in the PRP-14d (0.83 ± 0.17 ng/mL; P < .01), and control (0.68 ± 0.14 ng/mL; P < .01) groups, there was no significant difference between PRP-7d and PRP-0d group (0.97 ± 0.19 ng/mL). Furthermore, the concentration of TGF-β1 in the PRP-0d group was higher than in the control group (P < .05). At 8 weeks after surgery, there were no significant differences in the TGF-β1 concentration among the 4 groups (control: 0.54 ± 0.10 ng/mL; PRP-0d: 0.61 ± 0.07 ng/mL; PRP-7d: 0.69 ± 0.11 ng/mL; PRP-14d: 0.57 ± 0.10 ng/mL) (Figure 5A).
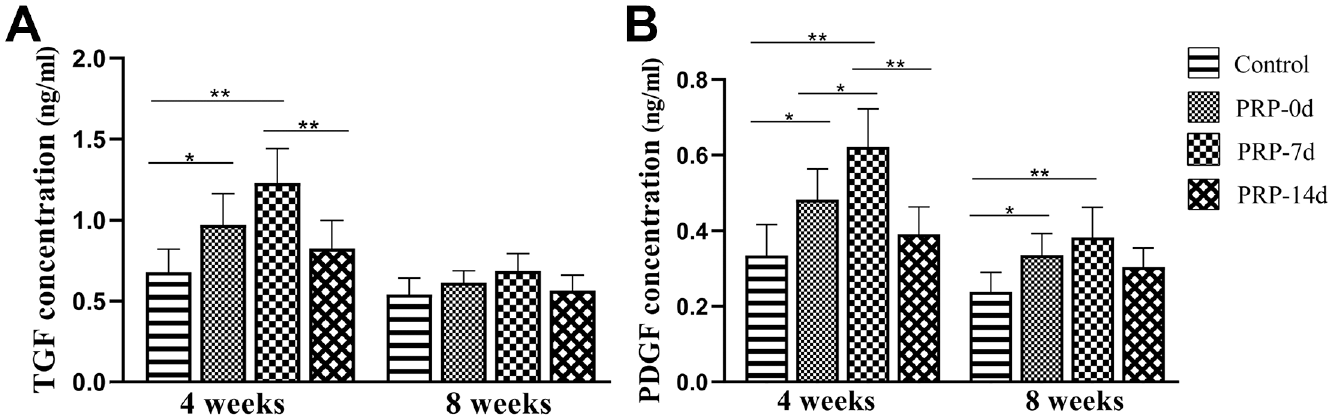
Concentration of (A) TGF-β1 and (B) PDGF-BB analyzed by enzyme-linked immunosorbent assay at 4 and 8 weeks after surgery. Error bars represent standard deviations. *P < .05. **P < .01. PDGF, platelet-derived growth factor; PRP, platelet-rich plasma; PRP-0d, PRP injected immediately after surgery; PRP-7d, PRP injected on the 7th postoperative day; PRP-14d, PRP injected on the 14th postoperative day; TGF, transforming growth factor.
PDGF-BB
At 4 weeks after surgery, the concentration of PDGF-BB in the PRP-7d group (0.62 ± 0.10 ng/mL) was significantly higher than that in the PRP-0d (0.48 ± 0.08 ng/mL; P < .05), PRP-14d (0.39 ± 0.07 ng/mL; P < .01), and control (0.34 ± 0.08 ng/mL; P < .01) groups. Moreover, the PRP-0d group exhibited a higher PDGF-BB concentration compared to the control group. At 8 weeks after surgery, the PDGF-BB concentration decreased in all groups compared to 4 weeks, and all PRP groups (PRP-0d: 0.34 ± 0.06 ng/mL; PRP-7d: 0.38 ± 0.08 ng/mL; PRP-14d: 0.31 ± 0.05 ng/mL) showed a higher concentration compared to the control group (0.24 ± 0.05 ng/mL). However, no significant differences were observed among the 3 PRP groups (Figure 5B).
Biomechanical Testing
The CSA was not significantly different between the groups at 4 weeks postoperatively (control: 1.35 ± 0.26 mm2; PRP-0d: 1.22 ± 0.26 mm2; PRP-7d: 1.12 ± 0.23 mm2; PRP-14d: 1.28 ± 0.22 mm2) or 8 weeks postoperatively (control: 1.11 ± 0.19 mm2; PRP-0d: 1.07 ± 0.17 mm2; PRP-7d: 1.03 ± 0.18 mm2; PRP-14d: 1.08 ± 0.19 mm2) (Figure 6A).

Biomechanical testing results at 4 and 8 weeks postoperatively for (A) cross-sectional area, (B) failure load, and (C) ultimate strength. Error bars represent standard deviations. *P < .05. **P < .01. PRP, platelet-rich plasma; PRP-0d, PRP injected immediately after surgery; PRP-7d, PRP injected on the 7th postoperative day; PRP-14d, PRP injected on the 14th postoperative day.
Regarding the failure load, at 4 weeks postoperatively, the PRP-0d group (3.39 ± 0.43 N) and PRP-7d group (4.06 ± 0.36 N) exhibited higher values compared to the PRP-14d group (3.04 ± 0.37 N) and control group (2.72 ± 0.44 N). The PRP-7d group showed superiority over the PRP-0d (P < .05), PRP-14d (P < .01), and control (P < .01) groups, while the PRP-0d group was superior to the control group (P < .05). At 8 weeks after surgery, the failure load was higher in the PRP-7d group (5.64 ± 0.40 N) compared to the PRP-14d (4.71 ± 0.41 N; P < .05) and control (4.38 ± 0.51 N; P < .01) groups (Figure 6B).
Regarding ultimate strength, at 4 weeks after surgery, the PRP-0d group (2.84 ± 0.47 MPa) and PRP-7d group (3.70 ± 0.53 MPa) exhibited higher values compared to the PRP-14d group (2.42 ± 0.46 MPa) and control group (2.06 ± 0.49 MPa). The PRP-7d group was superior to the PRP-0d group (P < .05), PRP-14d group (P < .01), and control group (P < .01). At 8 weeks postoperatively, the PRP-7d group (5.55 ± 0.62 MPa) exhibited significantly higher ultimate strength compared to the control group (4.06 ± 1.02 MPa; P < .05) (Figure 6C).
Discussion
The findings of this study demonstrate that PRP administration in the early postoperative period enhanced the healing process, with a PRP injection on the 7th day yielding superior therapeutic effects compared to the other groups.
It is important to consider the timing of biological therapies during tissue healing to achieve optimal outcomes. Existing research supports the notion that inflammation is a crucial stage of the healing process. Early stages of tissue healing involve cell proliferation and migration in which the inflammatory environment plays a pivotal role in promoting regenerative processes through various cytokines. However, excessive inflammation can lead to increased pain, fibrosis, and hypertrophic scarring. 15 Studies have shown that during the early phases of an injury, the body releases cytokines, and platelet aggregation attracts different cell growth factors to the injury site, resulting in increased growth factor expression and noticeable hematoma formation, which triggers inflammation. 8 Supplementing exogenous growth factors too early may result in excessive cytokine concentrations, exacerbating inflammation and impeding tissue healing, 33 and the inflammatory response may override any anabolic agents added at this time. It is reported that platelet concentrations at 3 to 5 times the baseline value provide optimal therapeutic effects, while high platelet concentrations may inhibit collagen synthesis. Administering PRP on the 14th postoperative day may miss the early healing phase, with limited impact on tissue recovery. Additionally, platelets derived from megakaryocytes have an in vivo half-life of approximately 7 days, and growth factors remain intragranular alphas until platelet activation.10,19 Therefore, we believe that when a hematoma begins to resolve on the 7th postoperative day and the cytokine concentration decreases, the introduction of a significant amount of growth factors through PRP supplementation can better facilitate the healing process at the injury site and sustain an optimal growth environment for tissue recovery. 1 These findings align with the results obtained in our study, further supporting the therapeutic benefits of PRP administration during the early postoperative period.
Tendon tissue healing typically progresses through 3 phases: the inflammatory phase (occurs within days of the injury), the proliferative phase (days to 2 weeks), and the remodeling phase (2 weeks to years).13,21 During the inflammatory phase, macrophages release growth factors that shift the healing process from inflammation to the proliferation of various cytokines that promote angiogenesis, which typically spans a 2-week duration before transitioning to the remodeling phase. 34 The remodeling phase, being the third phase of tissue healing, entails the reorganization of newly deposited collagen in the damaged area, with the recovery time adjusted based on the extent of the injury. 25 The selection of different initiation times in our study was aligned with these 3 healing phases.
PRP acts as a concentrated source of platelets and contains a variety of growth factors and cytokines required for healing, effectively promoting bone and soft tissue healing by accelerating effects such as cell proliferation and angiogenesis.5,10 PRP can also promote BTI healing. 37 Although platelets release a variety of growth factors, TGF, PDGF, and vascular endothelial growth factor (VEGF) are most relevant in the injury healing cascade, 39 and the concentration of growth factors was used to judge tissue healing in this study.
TGF-β1 plays a crucial role in all phases of tissue healing by acting as a stimulator and healing agent at the injury interface. 23 It stimulates cell migration, modulates protease activity, and promotes fibrin binding, thereby facilitating the healing process. 46 Shortly after a tissue injury, there is an increase in TGF-β1 mRNA expression, particularly during the initial inflammatory response. Studies on rotator cuff repair have shown a significant increase in the concentration of TGF-β1, which effectively promotes cell proliferation and reduces the expression of proinflammatory factors.2,22 PDGF-BB, on the other hand, acts as an angiogenic factor and is associated with the late inflammatory phase and the early proliferative phase. 35 Tokunaga et al 35 found that rats treated with PDGF-BB for a rotator cuff injury exhibited better collagen fiber alignment and biomechanical properties compared to the control group, particularly during the early stage. Chan et al 6 injected growth factors into a BTI injury in rats and showed that supplementation on the 7th postoperative day resulted in a higher loading peak and pyridine content, a collagen cross-link associated with tendon healing maturation and biomechanical strength. Denapoli et al 9 conducted a comparative study on different times of PRP treatment after a tendon injury and concluded that the timing of the PRP injection was crucial. They found that injecting PRP on the 7th day led to improved exercise capacity, reduced fibrotic areas, and higher concentrations of growth factors such as PDGF compared to other time points.
In a study conducted by Snow et al 33 comparing PRP injected approximately 2 weeks postoperatively with saline, it was observed that delaying the application of PRP did not improve function. This lack of improvement could be attributed to the fact that the injection was administered when tissue had already entered the remodeling phase and collagen deposition had begun. During the early stages of recovery, which are crucial for healing, 32 the activation of a larger number of growth factors plays a more important role. The limited benefit of PRP supplementation at that stage was also reflected in the immunological results.
The regeneration of cartilage is a critical factor in the tissue recovery of bone-tendon injuries, particularly in relation to the biomechanical properties of the joint. Our study found that a PRP injection on the 7th postoperative day was most effective for promoting cartilage interface healing; this may be attributed to the impact of PRP on mesenchymal stem cells. In vivo, under the influence of growth factors such as VEGF and PDGF, mesenchymal stem cells migrate to the site of the tendon-bone injury around 5 days after the injury and differentiate into fibroblasts, chondrocytes, and osteoblasts. This process gradually establishes the connection between the tendon and bone.18,26,38 An injection of PRP on the 7th postoperative day may have maintained the optimal concentration of cytokines for a longer duration, effectively promoting rotator cuff healing.
The typical fibrocartilage layer is characterized by the uniform embedding of chondrocytes in an extracellular matrix composed of PGs and collagen. Beck et al 4 found linear and orderly arranged collagen fibers toward the natural tendon insertion and hypertrophic chondrocytes with a clear basophilic matrix in PRP-treated rats at 3 weeks after surgery, whereas the collagen fibrils in the control group were smaller in caliber and qualitatively less developed. This illustrates that PRP improved collagen orientation and density and increased the glycosaminoglycan content in tissue. Previous studies have shown that the application of PDGF at 7 days after an injury has a more pronounced effect on the maturation and biomechanical strength of tenocytes than early application. 6 Jiang et al 15 injected leukocyte-rich or leukocyte-poor PRP a week after tendinous bone lesions in rabbits and observed the biomechanical results at 6 weeks after surgery. They found increased overall biomechanical parameters in the PRP group compared to the control group. These findings suggest that PRP application enhances the biomechanical properties of newly formed structures at the BTI of the rotator cuff in mice. Moreover, over longer healing periods, tissue undergoes additional mechanical and biological changes, further supporting the positive effects of PRP. 26
Limitations
There were several limitations to this study. First, the specific cytokine parameters of PRP were not comprehensively analyzed. It is known that different cell counts, including leukocytes, can influence the therapeutic effects of PRP. 10 Second, the experimental period was relatively short, and future studies should include extended follow-up periods to better understand long-term outcomes. Third, to standardize PRP, allogeneic PRP was used. However, it is important to note that this approach may potentially intensify the inflammatory response. Lastly, it should be acknowledged that the model used in this study represents an acute repair scenario, whereas most rotator cuff tears in the clinic are chronic in nature.
Conclusion
PRP promoted healing of the BTI after a rotator cuff injury at an early stage in a murine animal model. A PRP injection on the 7th postoperative day demonstrated superior therapeutic effects compared with injections at other time points.
Footnotes
Final revision submitted May 22, 2023; accepted July 31, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Research support was received from the Hubei Provincial Natural Science Foundation (2019CFB397), Research Fund for Young Teachers of Wuhan Sports University and Donghu Scholar Program of Wuhan Sports University (2019), and Young and Middle-Aged Scientific Research Team Project of Wuhan Sports University (21KT14). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Wuhan Sports University (No. 2019020).
