Abstract
Background:
Quadriceps muscle atrophy remains a limiting factor in returning to activity after anterior cruciate ligament reconstruction (ACLR). Blood flow restriction (BFR) therapy may accelerate quadriceps strengthening in the perioperative period.
Purpose:
To evaluate postoperative isometric quadriceps strength in patients who underwent ACLR with a perioperative BFR program.
Study Design:
Randomized controlled trial; Level of evidence, 1.
Methods:
Patients indicated for ACLR were randomized into 2 groups, BFR and control, at their initial clinic visit. All patients underwent 2 weeks of prehabilitation preoperatively, with the BFR group performing exercises with a pneumatic cuff set to 80% limb occlusion pressure placed over the proximal thigh. All patients also underwent a standardized postoperative 12-week physical therapy protocol, with the BFR group using pneumatic cuffs during exercise. Quadriceps strength was measured as peak and mean torque during seated leg extension and presented as quadriceps index (percentage vs healthy limb). Patient-reported outcomes (PROs), knee range of motion, and quadriceps circumference were also gathered at 6 weeks, 3 months, and 6 months postoperatively, and adverse effects were recorded.
Results:
Included were 46 patients, 22 in the BFR group (mean age, 25.4 ± 10.6 years) and 24 in the control group (mean age, 27.5 ± 12.0 years). At 6 weeks postoperatively, the BFR group demonstrated significantly greater strength compared with the controls (quadriceps index: 57% ± 24% vs 40% ± 18%; P = .029), and the BFR group had significantly better Patient-Reported Outcomes Measurement Information System–Physical Function (42.69 ± 5.64 vs 39.20 ± 5.51; P = .001) and International Knee Documentation Committee (58.22 ± 7.64 vs 47.05 ± 13.50; P = .011) scores. At 6 weeks postoperatively, controls demonstrated a significant drop in the peak torque generation of the operative versus nonoperative leg. There were no significant differences in strength or PROs at 3 or 6 months postoperatively. Three patients elected to drop out of the BFR group secondary to cuff intolerance during exercise; otherwise, no other severe adverse events were reported.
Conclusion:
Integrating BFR into perioperative physical therapy protocols led to improved strength and increased PROs at 6 weeks after ACLR. No differences in strength or PROs were found at 3 and 6 months between the 2 groups.
Registration:
NCT04374968 (ClinicalTrials.gov identifier).
Anterior cruciate ligament (ACL) injury is a common occurrence in elite and recreational athletes, leading to a 9- to 12-month rehabilitation process after surgery.15,23,32 Several parameters must be met during the gradual advancement of this process before patients and athletes are cleared to return to sport (RTS). 19 These include achieving goals in range of motion, strength, balance, and proprioception and advancing through sport-specific drills such as cutting and pivoting.1,10 After ACL reconstruction (ACLR), quadriceps atrophy and weakness of the surgical leg are known to cause side-to-side strength asymmetry,20,31 which may last 9 months postoperatively. 28 An athlete may not RTS before attaining adequate strength, as altered lower extremity mechanics due to weakness is a significant risk factor for reinjury or contralateral injury.13,29 However, ACLR rehabilitation does not allow for aggressive early strengthening in order to protect the maturing graft.
Blood flow restriction (BFR) therapy during rehabilitation protocols is a potential modality to accelerate quadriceps strength gains in the early postoperative period. BFR uses a pneumatic tourniquet system during exercise that maintains arterial flow while occluding venous return. 37 The tourniquet effect in BFR takes advantage of the effects of hypoxia, inducing muscle tension and metabolic stress, simulating high-intensity exercise without exceeding postoperative weight restrictions.26,30,33-35 With a low-risk profile and relatively low cost, BFR use in the clinical setting has preceded high-level studies examining its benefits. 36 The early literature suggests enhanced muscular strength and hypertrophy while using BFR with low-resistance loads.3,6,37 Low-resistance exercise allows patients to rehabilitate earlier from injury or surgery within the parameters of their postoperative restrictions, while BFR potentially reduces atrophy and weakness.3,8,12,17 Therefore, the reported benefits of BFR are important to study in patients after ACLR, whose path to RTS is contingent on regaining quadriceps muscle strength.
Although perioperative BFR therapy has gained popularity and merit, available studies have significant limitations, such as short follow-up and nonstandardized protocols and outcome measures, which have led to a lack of consensus regarding recommendations for use. 17 Therefore, the purpose of this investigation was to evaluate a perioperative BFR program in patients undergoing ACLR, with the primary outcome of isometric quadriceps strength at 3 months after surgery. We hypothesized that utilization of BFR during physical rehabilitation both before and after surgery would lead to improved quadriceps strength at the final 3-month follow-up.
Methods
The Consolidated Standards of Reporting Trials (CONSORT) statement was employed to conduct this prospective randomized controlled trial (RCT). This RCT was registered with ClinicalTrials.gov, and institutional review board approval was obtained. The hypothesis was formulated before study initiation. Patients gave consent to participate in the study and were randomized using 1:1 allocation utilizing simple randomization computer software (MD Anderson Cancer Center) into a BFR or non-BFR rehabilitation physical therapy protocol. Inclusion criteria included patients aged ≥14 years scheduled to undergo a primary ACLR and evaluated at our clinic <3 months from the date of injury. Patients were excluded if they had a personal or family history of bleeding disorders, deep venous thrombosis, same joint surgery in the past year, peripheral artery disease, uncontrolled hypertension, active anticoagulation use, tobacco use, a body mass index >40 kg/m2, and an inability to tolerate the BFR cuff. All patient data were kept in a secure digital database during their involvement in the study, and all patient data were subsequently deleted. Because collected outcomes were a combination of objective measures and self-reported data by the patients, this study was not blinded, and patients were made aware of their treatment group after consenting to participate in the study.
A power analysis performed before beginning data collection demonstrated that with 16 patients per group, we would be able to detect an effect size (the detectable difference in standard deviation units) of 0.83 with 80% power with a significance level set to .05. A sample of 45 patients was thus targeted to allow for complete data collection.
A total of 130 patients were evaluated for potential inclusion in this study, of whom 61 met exclusion criteria and 23 declined to participate. A total of 46 patients who had been evaluated by 3 fellowship-trained sports surgeons (K.O., V.M.) between June 2020 and February 2021 met the inclusion criteria, agreed to participate in the study, and were enrolled. Of the 46 patients, 22 were randomized into the BFR group and 24 into the control group. Figure 1 shows the CONSORT flow diagram of patient inclusion.

Consolidated Standards of Reporting Trials (CONSORT) flow diagram of patient inclusion in the study. ACL, anterior cruciate ligament; BFR, blood flow restriction.
Intervention
Patients who were diagnosed with an ACL tear and were indicated for reconstruction were identified and gave consent at their initial clinic visit (ICV). Before surgery, all patients indeterminant of treatment group were instructed on a home exercise program by a licensed physical therapist. The home exercise program consisted of exercises performed 5 times a week for the 2 weeks before surgery as a form of “prehabilitation.” Exercises included quadriceps contractions in end-range extension, straight-leg raises, long-arc quadriceps sets, and quarter squats. All exercises were performed for 75 repetitions with a repetition scheme of 30-15-15-15, allowing for 30 seconds of rest between sets. Patients were educated on how to properly perform the required exercises and were asked to sign a log that would be returned during the preoperative visit to demonstrate compliance. Patients randomized to the BFR cohort received personalized limb occlusion pressure (LOP) measured using a Doppler ultrasound placed on the dorsalis pedis pulse. Patients in the BFR group were provided a single-chamber pneumatic torniquet (Smart Tool Plus) and instructed to set the pressure to 80% LOP when performing prehabilitation exercises at home. Patients were instructed to leave the cuff inflated for the duration of each exercise, ensuring a rest period of at least 2 minutes between exercises with the cuff deflated. All patients were shown how to inflate and deflate the cuff properly and tested on their compliance in the clinic before taking home the cuff. Graft choice was determined through shared decision-making between the clinician and the patient based on activity level and patient-specific factors.
After ACLR, all patients indeterminant of treatment group underwent the same postoperative rehabilitation protocol. Patients who did not have a meniscal repair were made full weightbearing immediately postoperatively in a brace. Patients who underwent meniscal repair were kept in a brace that limited flexion to 90° for 6 weeks and also did not perform exercises with >90° of knee flexion during this time. Rehabilitation sessions were all performed at our institution's physical therapy facilities under the guidance of licensed physical therapists who were certified in BFR therapy. The postoperative rehabilitation sessions for all patients began by postoperative day 3. Patients in the BFR group began incorporating BFR into their rehabilitation as early as their first session as long as there was not excessive pain, effusion, or leg edema. Patients in the BFR group began with ischemic preconditioning at 100% occlusion pressure at day 3 postoperatively and then progressed to BFR with exercises over the first 2 sessions. Patients performed the exercises using a 30-15-15-15 repetition scheme for a total of 75 repetitions with the BFR cuff set to 80% LOP. Individuals in the BFR group performed all repetitions with the cuff inflated, followed by 1 minute of cuff deflation between sets. Both groups underwent the same protocol with exercises performed 2 to 3 times per week, and all patients were encouraged to continue the exercises at home in between physical therapy sessions (all without BFR). Patients started with supine quadriceps sets, side-lying hip abduction, calf raises, supine straight-leg raises, long-arc quadriceps sets from 90° to 45° of knee flexion, and quarter squats, both with and without BFR. Once patients were able to complete those exercises, they were progressed to full squats, forward and side lunges, step-ups, and full-range long-arc quadriceps sets. At the 3-month mark, patients were progressed to usual heavy-resistance training and BFR was discontinued.
Outcomes and Measures
Participants completed this study across a 6-month postoperative period with data collected at 6 time points: (1) their ICV, (2) the day of surgery, (3) the first postoperative visit (within 2 weeks from the day of surgery), (4) the 6-week follow-up visit, (5) the 3-month follow-up visit, and (6) the 6-month follow-up visit. At the ICV, the patients’ anthropometric and demographic information was collected. This included age, body mass index, sex, quadriceps circumference (measured 15 cm proximal to the superior pole of the patella), and leg length for torque calculation (measured from the lateral knee joint line to 5 cm proximal to the distal aspect of the lateral malleolus).
Objective biomechanical measures were collected using a handheld dynamometer (Lafayette Instruments) in a standardized manner previously described in the literature. 9 This methodology consisted of placing the patients in a seated position with their leg over the end of the clinic bed and the knee at 90° of flexion. A belt was then placed across the patients’ thighs to minimize movement during strength testing and to hold the hips in place. A handheld dynamometer was then positioned behind the leg of the clinic bed using a flat attachment, and a belt was placed around the dynamometer and 5 cm proximal to the distal aspect of the lateral malleolus (Figure 2). Patients were then instructed to extend their knee and encouraged verbally to use maximal effort. This protocol has been previously evaluated in comparison with traditional isokinetic quadriceps strength testing and found to correlate with findings on isokinetic testing as well as producing high-reliability measurements.2,21,24
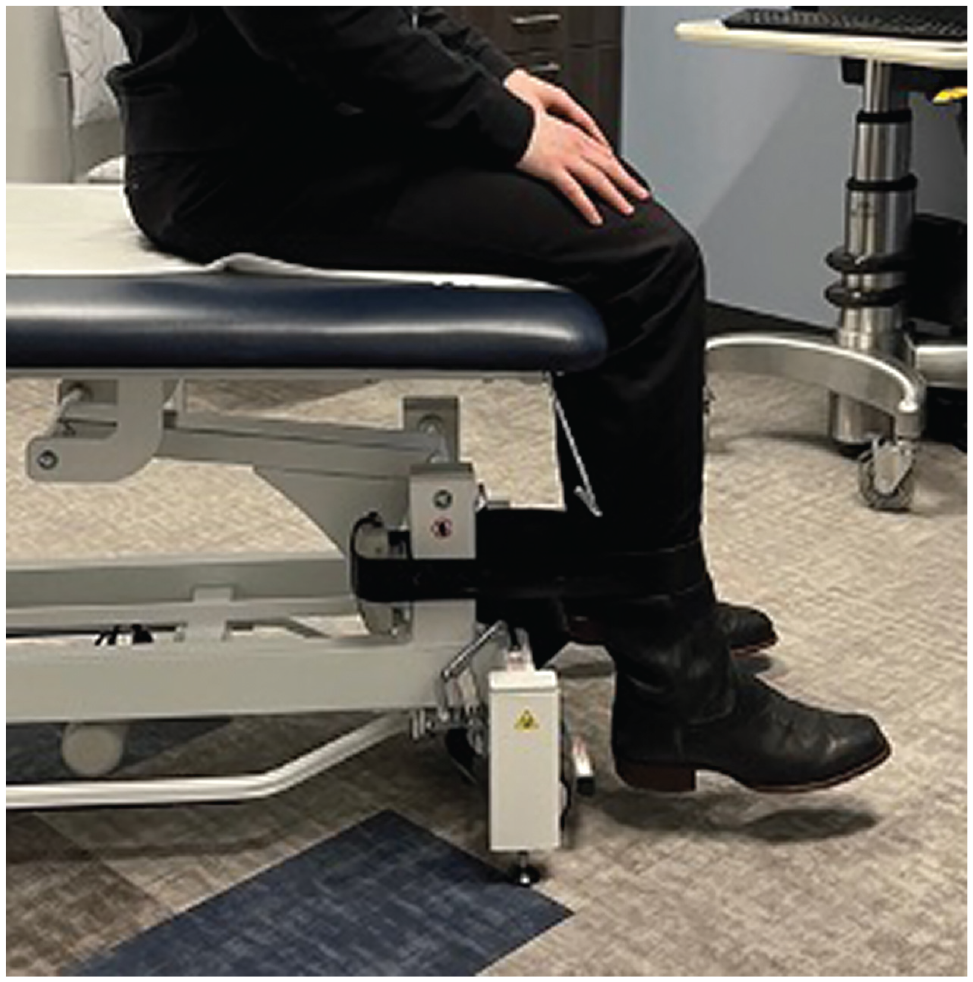
Demonstration of belt-stabilized handheld dynamometer placement used to measure quadriceps strength. An additional gait belt was used to stabilize the hips at 90° of flexion.
Biomechanical outcomes were recorded during all visits except the first preoperative visit because of concern for pain and healing in the early postoperative period. Collected metrics included peak quadriceps torque (N·m), mean torque (N·m), and knee range of motion (degrees) in both the operative and nonoperative legs. The quadriceps index (QI) was calculated as the mean torque generation of the operative leg divided by that of the nonoperative leg to determine the percentage of strength compared with the healthy limb. Return to baseline was calculated by comparing QI at follow-up to that at the ICV, with values at or exceeding the ICV considered a return to baseline. Patient-reported outcomes (PROs) collected at each time point consisted of visual analog scale (VAS) for pain, the International Knee Documentation Committee (IKDC) score, and the Patient-Reported Outcomes Measurement Information System (PROMIS) Physical Function (PF), Pain Interference (PI), and Depression (D) domains.
Statistical Analysis
The primary outcome of this study was mean quadriceps torque generation in the operative leg at 3 months postoperatively, as this correlated with the completion of BFR. Patients were tested again at 6 months postoperatively to determine if any differences between groups were maintained with longer follow-up. Torque values (N·m) were calculated from the data collection by using measurement in centimeters from the lateral knee joint line to 5 cm proximal to the distal aspect of the lateral malleolus and multiplying this value by the force measurements (N) recorded by the dynamometer. Since 3 force measurements were taken at each time interval, the mean of the 3 measurements was used during all statistical analyses.
Continuous variables are summarized as means and standard deviations, and categorical variables are presented as frequencies and percentages. Outlier analysis was performed and individual strength testing observations >2 or <2 standard deviations were removed before analysis. Because of the nonnormality and skewness of the data set, nonparametric equivalents were used in the analysis instead of conventional parametric tests. Statistical testing has been performed using the Wilcoxon rank-sum test for the 2-group comparisons and the Wilcoxon signed-rank test for the evaluations of change within each separate group. Effect size was evaluated using Cohen d, with thresholds of >0.80 for large, 0.80 to 0.50 for moderate, and <0.05 for small. The threshold for statistical significance was set at P < .05. In addition, P values between .05 and .10 represent statistically nonsignificant borderline results. Statistical analysis was performed using SAS Version 9.4 (SAS Institute Inc).
Results
The characteristics of the 46 study patients are shown in Table 1. There were no significant differences in patient characteristics between the BFR and control groups.
Characteristics of Patients in the Study (N = 46) a
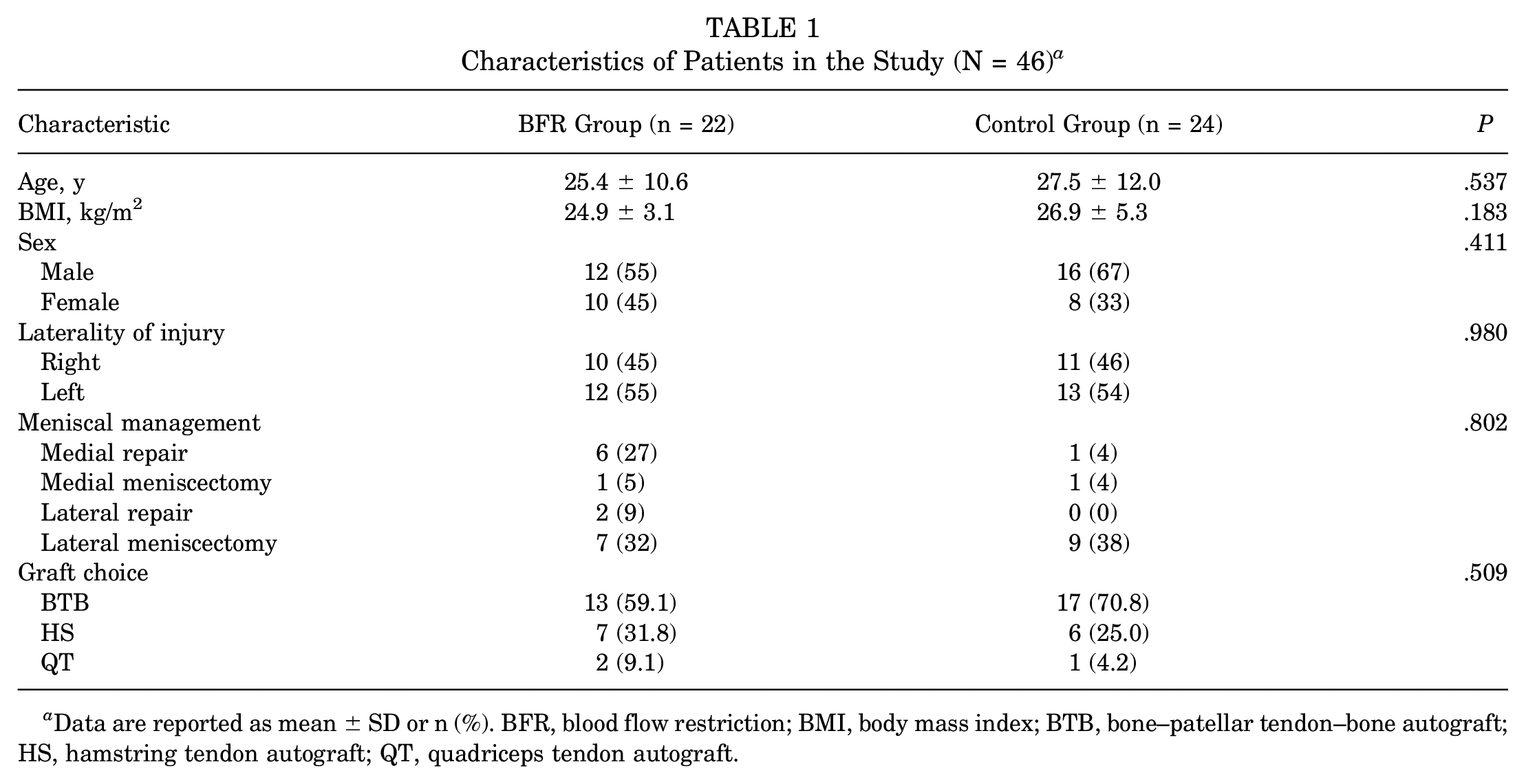
Data are reported as mean ± SD or n (%). BFR, blood flow restriction; BMI, body mass index; BTB, bone–patellar tendon–bone autograft; HS, hamstring tendon autograft; QT, quadriceps tendon autograft.
During the ICV, both the operative and nonoperative extremity baseline measures were evaluated for quadriceps circumference, peak quadriceps torque, mean quadriceps torque, time to peak torque, knee range of motion, VAS pain, IKDC score, and PROMIS scores (Table 2). The only significant difference between the groups at ICV was the IKDC score, which was lower for the BFR cohort (P = .017).
Initial Clinic Visit Measurements a

Data are reported as mean ± SD. Boldface P value indicates a statistically significant difference between groups (P < .05). BFR, blood flow restriction; D, Depression; IKDC, International Knee Documentation Committee; PF, Physical Function; PI, Pain Interference; PROMIS, Patient-Reported Outcomes Measurement Information System; VAS, visual analog scale.
From the ICV to the preoperative visit, both groups demonstrated improvements in strength over the course of the prehabilitation program, with no difference in QI between the BFR and control groups. All patients, regardless of treatment group, experienced a decline in mean and peak quadriceps torque generation at 6 weeks postoperatively in the operative leg (Table 3). At 6 weeks postsurgery, patients in the control group demonstrated a significantly greater drop in the peak torque generation of the operative leg compared with the nonoperative leg, whereas patients in the BFR group did not. At 3 months postoperatively, both cohorts had not returned to their baseline peak or mean quadriceps strength. At 6 months postoperatively, both cohorts experienced improved peak torque generation compared with the ICV in the operative leg; however, no statistically significant difference was found between cohorts.
Mean Differences in Strength Between Groups and Between ICV and Postoperative Time Points a
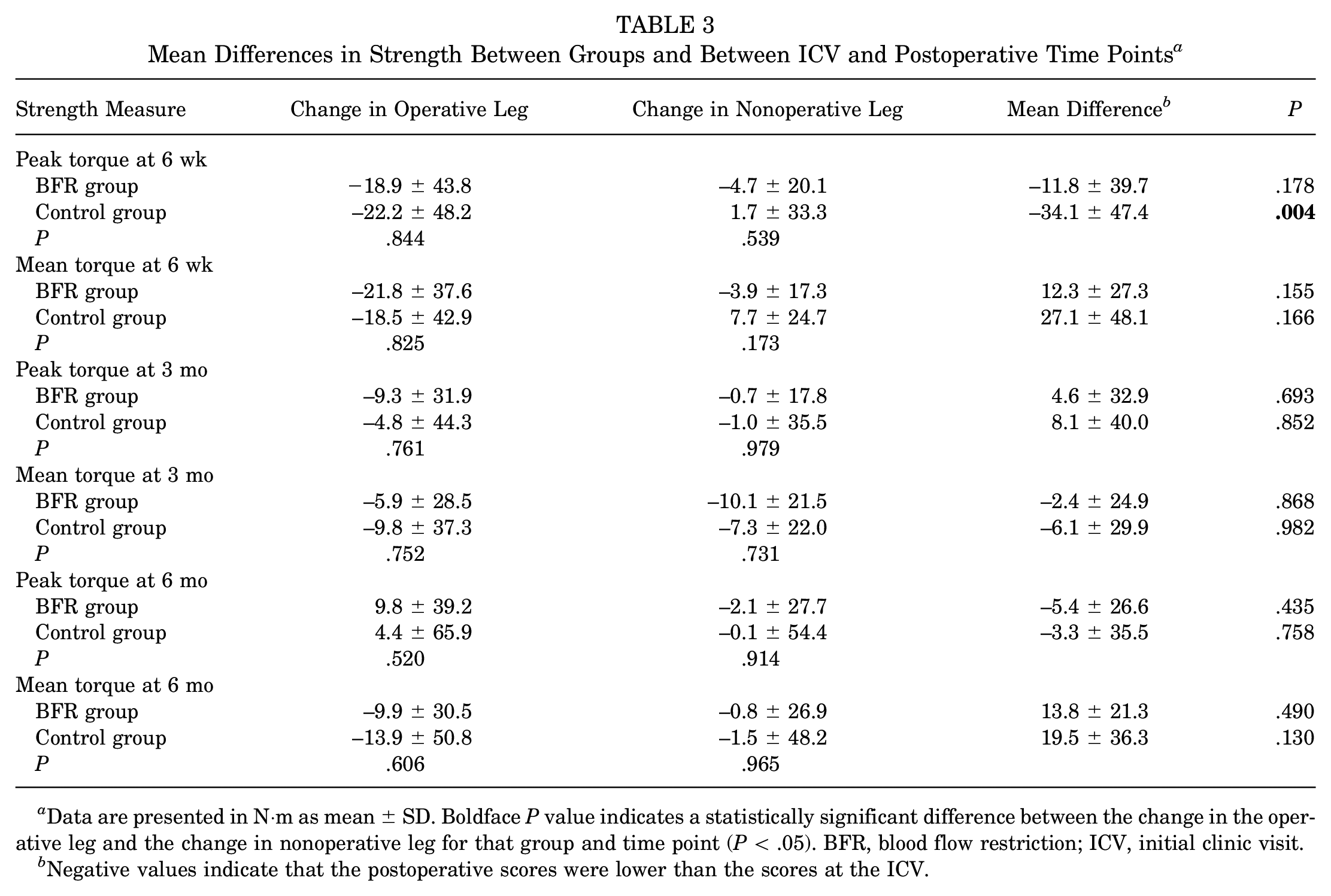
Data are presented in N·m as mean ± SD. Boldface P value indicates a statistically significant difference between the change in the operative leg and the change in nonoperative leg for that group and time point (P < .05). BFR, blood flow restriction; ICV, initial clinic visit.
Negative values indicate that the postoperative scores were lower than the scores at the ICV.
When evaluating the QI between cohorts, we found a statistically significant difference between cohorts at 6 weeks postoperatively, with the BFR group demonstrating a significantly greater mean QI than the control cohort (57% ± 24% vs 40% ± 18%; P = .029; effect size, 0.80). There were no other statistically significant differences in force generation at all other time points (Table 4 and Figure 3). At 3 months, a greater proportion of patients returned to their baseline mean QI in the BFR group (43.8%) compared with the control group (36.4%); however, this was not statistically significant (P = .322). There were no reported severe adverse events such as paresthesia, pressure injury, or blood clots. Three patients in the BFR cohort reported pain intolerance and discomfort with the torniquet set to 80% LOP and elected to discontinue BFR between the 2- and 6-week follow-up appointments. One patient in the BFR group experienced an ACL graft retear between their 3- and 6-month visits and discontinued follow-up.
Quadriceps Index Between Cohorts at Each Time Point a

Data are reported as mean ± SD. Boldface P value indicates a statistically significant difference between groups (P < .05). BFR, blood flow restriction; ICV, initial clinic visit.

Quadriceps index (QI) for mean torque across each clinic visit. *Statistically significant difference between blood flow restriction (BFR) and control (P < .05). ICV, initial clinic visit; Preop, preoperative.
At the 6-week follow-up, the BFR group demonstrated significantly better PROMIS-PF scores (42.69 ± 5.64 vs 39.20 ± 5.51; P = .001) and IKDC scores (58.22 ± 7.64 vs 47.05 ± 13.50; P = .011) than the control group. At the 3-month follow-up, the BFR and control cohorts demonstrated no statistically significant difference between operative leg quadriceps circumference as a percentage of the nonoperative leg (98.56% ± 4.87% vs 98.66% ± 4.19%; P = .944), knee range of motion as a percentage of the nonoperative leg (96.17% ± 6.51% vs 93.18% ± 12.06%; P = .986), VAS score (0.79 ± 0.80 vs 0.50 ± 0.89; P = .191), IKDC score (61.54 ± 10.51 vs 61.09 ± 9.95; P = .855), PROMIS-PF score (46.76 ± 4.83 vs 46.88 ± 3.44; P = .726), PROMIS-PI score (50.95 ± 7.77 vs 52.14 ± 5.46; P = .254), and PROMIS-D score (39.30 ± 6.94 vs 40.53 ± 8.72; P = .872). Patients were evaluated out to 6 months, and there remained no difference in PROs.
Discussion
In this prospective RCT, we evaluated patients undergoing a 2-week preoperative and 12-week postoperative BFR rehabilitation program after ACLR compared with a control group with 6 months of follow-up and found that at 6 weeks post-ACLR the patients in the BFR group demonstrated a higher mean QI and reduced mean difference in peak torque generation between legs when compared to the control group. Additionally, at the 6-week follow-up, the BFR group demonstrated improved PROMIS-PF and IKDC scores compared with the control group. There were no other statistically significant differences between groups at any follow-up points. There were 3 patients who were unable to complete the BFR therapy because of intolerance to the intervention.
BFR training has gained popularity as patients and providers seek an expeditious return to sporting activity. Currently, studies are mixed as to the efficacy of BFR in the perioperative period surrounding ACLR, and there is no consensus on optimal uses or protocols in this patient population.4,22 Several studies have been conducted evaluating the use of BFR after ACL surgery. Hughes et al 12 conducted an RCT of 24 patients who performed postoperative rehabilitation with either low-intensity BFR resistance training or heavy-load resistance training starting 2 weeks after ACLR, for a total of 8 weeks. As in the present investigation, an 80% LOP was used for these patients; however, BFR was only used during the leg-press exercise, which was conducted twice per week during the study period for a total of 16 BFR sessions. The authors noted that while patients did not show increases in maximal isotonic quadriceps strength, the BFR group had significant improvements in multiple PROs compared with the control group. Hughes et al 12 concluded that there were potential advantages to incorporating BFR into the early post-ACLR period when patients may not be able to tolerate heavier resistance exercises. Additionally, as in our investigation, patients in the BFR group had consistently higher PROs at 6 weeks, highlighting the potential benefit that BFR may provide to early postoperative function.
Lambert et al 18 conducted a similar study to our investigation, in which patients were immediately started on BFR therapy after ACLR in conjunction with their usual physical therapy protocol, integrating BFR into select exercises. The authors found a protective effect of BFR on lean muscle mass and bone mineral density measured on dual-energy x-ray absorptiometry scans at 6 and 12 weeks after ACLR, representing another potential benefit of BFR therapy. 18 In an RCT investigating 44 patients undergoing ACLR, Ohta et al 27 found that performing low-load exercise with BFR for the first 16 weeks after ACLR led to greater isometric and isokinetic knee flexion and extension torque generation, as measured by a dynamometer, compared with a group performing the same physical therapy protocol without BFR. The ratio of injured to uninjured leg isometric force generation dropped significantly less in the BFR group (92% to 84%) compared with the control group (94% to 63%) at the final 16-week follow -up. To our knowledge, the current study is the first study to evaluate the combination of pre- and postoperative rehabilitation using BFR therapy. While there were no statistically significant differences at 12 or 24 weeks in the present cohort, the BFR group did demonstrate improved mean torque generation at 6 weeks after surgery compared with the control group. These findings demonstrate the potential benefits of BFR training in the early perioperative period for patients with ACL, including quadriceps muscle strength preservation and improved PROs. Interestingly, we found that BFR therapy may lead to quicker recovery of baseline quadriceps strength. However, this finding was only statistically significant at the 6-week follow-up; therefore, it is difficult to say if this would lead to clinically significant improvements. It is possible that this may assist in earlier return to work and more normal activities of daily living, however, as quadriceps strength is often the limiting factor to RTS. Further along in the rehabilitation process, future longer-term studies should evaluate if BFR therapy leads to faster RTS.
There also have been multiple investigations that did not find advantages to using BFR therapy after ACLR. An RCT conducted by Curran and colleagues 7 found no significant differences in maximal isokinetic or isometric knee extension torque, rectus femoris muscle volume on ultrasound, or IKDC scores between the BFR and control groups when tested 18 weeks after surgery after 8 weeks of BFR starting at week 10 postsurgery. Iversen et al 14 also did not appreciate any differences in muscle cross-sectional area on magnetic resonance imaging between a BFR group and a control group after 2 weeks of low-intensity exercises with and without BFR after ACLR. In their investigation, BFR was performed with a standard inflation pressure, rather than a personalized LOP, which may have limited its effect. In comparison with the previous studies, the present investigation used preoperative BFR and postoperative BFR starting as soon as postoperative day 3, and BFR was discontinued when the patients were able to tolerate high-intensity exercise. Additionally, incorporating BFR into a preoperative therapy program can allow patients to become familiar with exercise in the cuff, as well as condition the muscles to prevent delayed-onset muscle soreness that can occur when using BFR. 5 Overall, the significant heterogeneity of currently published BFR protocols must be noted, with the above studies using different combinations of exercise intensity, duration, and timing of BFR use, as well as LOPs. Thus, future research is needed not only to determine if BFR is a viable modality for improving outcomes after ACLR, but also to establish optimal standardized protocols to maximize patient outcomes.
In order to investigate the safety and patient tolerance of BFR therapy, Hughes et al 11 conducted an additional study specifically investigating pain and comfort with BFR in the post-ACLR rehabilitation period. The mean reported knee pain was better in the BFR group than in the control group, likely secondary to the lighter loads used during exercise. However, reported muscle pain was higher with the use of BFR in their cohort. Minniti et al 25 conducted a systematic review on the safety of BFR therapy and found that 4 of 322 patients analyzed dropped out of their respective studies because of cuff intolerance. Their review found 1 instance of upper extremity deep vein thrombosis and 1 instance of rhabdomyolysis, classified as rare adverse reactions. It must be noted that 3 patients elected to drop out of the present study secondary to BFR intolerance, which may be secondary to this reported increase in muscle pain, which represented a sizable proportion of the cohort. Aside from the cuff intolerance, there were no reported serious complications of BFR therapy in our cohort. Overall, complications of BFR remain low in both the current literature and the present investigation, and BFR remains a safe modality for the vast majority of patients.
Limitations
There are several limitations to this investigation. One is that multiple patients were lost to follow-up, as well as a large number of excluded patients. Each patient was called once per week for 3 weeks surrounding their proposed appointment time, and despite this, there was still difficulty in obtaining follow-up measurements, which may have also been influenced by the ongoing COVID-19 pandemic throughout the study period. While prior investigations have validated the use of handheld dynamometers in measuring isometric knee extension torque, the utility of these measurements as a surrogate for overall quadriceps function likely does not fully represent the patients’ complete quadriceps function.16,24 We also recruited all patients evaluated at our clinics with an ACL tear with plans to undergo reconstruction; thus, there was a variability in the age, sex, preoperative activity level, and athletic ability of our patient population, which may have also contributed to the rather large variation in measured strength via the dynamometer. While there was no statistically significant difference, there was also variability in meniscal work as well as graft choice, which may have contributed to the difference between groups. This is important to note as hamstring and quadriceps tendon autografts have been associated with less of a negative impact on quadriceps strength postoperatively. Additionally, while we disseminated our physical therapy protocol to each physical therapist, there could be variability in therapy based on provider and location. While the 6-month follow-up represents a relatively short follow-up period, BFR was only used for the first 3 months, and longer-term studies will be useful to determine if there are lasting effects of BFR training. While there were some statistically significant differences between groups, these may not translate to clinically significant differences. Patients and providers were also not blinded to their treatment group, which has the potential to introduce bias.
Conclusion
Integrating BFR into perioperative physical therapy protocols led to improved strength and increased PROs at 6 weeks after ACLR. No differences in strength or PROs were found at 3 and 6 months between the 2 groups. These findings suggest that perioperative BFR therapy in ACLR may lead to increased strength and quicker quadriceps recovery in the early postoperative period after ACLR.
Footnotes
Final revision submitted May 19, 2023; accepted May 30, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: K.R.O. has received grant support from Arthrex; education payments from Arthrex, Endo Pharmaceuticals, and Smith & Nephew; nonconsulting fees from Arthrex and Smith & Nephew; consulting fees from Endo Pharmaceuticals and Smith & Nephew; honoraria from Wright Medical; and hospitality payments from Medical Device Business Services, Stryker, and Zimmer Biomet. J.S.T. has received education payments from Pinnacle. L.S.K. has received education payments from Pinnacle and Medwest Associates. T.R.J. has received consulting fees from Biogen; hospitality payments from Exactech and Zimmer Biomet; and education payments from DePuy Synthes, Pinnacle, and Smith & Nephew. V.M. has received consulting fees from Pacira Pharmaceuticals and education payments from Pinnacle. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Henry Ford Hospital (No. 13080).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
