Abstract
Background:
Guided physiotherapy and surgical arthrolysis are effective in most patients with knee extension deficit. However, in refractory cases, posterior knee capsulotomy may be needed.
Purpose:
To assess extension restoration, pain reduction, and functional improvement after arthroscopic complete posterior knee capsulotomy in patients with extension deficit refractory to guided physiotherapy and surgical arthrolysis.
Study Design:
Case series; Level of evidence, 4.
Methods:
Included were patients with symptomatic asymmetric extension deficit >3° refractory to at least 6 months of guided physiotherapy and initial arthrolysis (15 patients with 12-month follow-up and 8 patients with 24-month follow-up). The mean duration of extension deficit was 24.6 months. An arthroscopic complete posterior knee capsulotomy was performed with transection of the posteromedial, posterolateral and central capsule, and the posterior septum. The primary outcome measure was knee extension, with hyperextension denoted as negative knee extension values. Secondary outcome measures included visual analog scale (VAS) for pain during maximum effort and exercise, International Knee Documentation Committee (IKDC) score, and Knee injury and Osteoarthritis Outcome Score (KOOS).
Results:
The mean patient age was 40.0 years (range, 26-70 years); 6 out of 15 patients had developed knee contracture after isolated anterior cruciate ligament reconstruction. The mean knee extension deficit decreased from 16.9° (range, 7° to 45°) preoperatively to -0.2° (range, -5° to 5°) at 12-month follow-up (P = .003) and to -0.3° (range, -5° to 5°) at 24-month follow-up (P = .035). The mean VAS pain score decreased from 3.5 (range, 1-6) preoperatively to 1.1 (range, 0-2) at 12-month follow-up (P = .004) and to 1.5 (range, 0-4) at 24-month follow-up (P = .005). The mean IKDC increased from 37.9 (range, 21-62) preoperatively to 63.9 (range, 46-87) at 12-month follow-up (P < .001) and to 60.9 (range, 39-80) at 24-month follow-up (P = .003). The mean KOOS increased from 45.0 (range, 30-62) preoperatively to 75.3 (range, 49-94) at 12-month follow-up (P < .001) and to 72.3 (range, 49-92) at 24-month follow-up (P = .003). There were no significant differences between 12- and 24-month follow-up in extension deficit or functional outcomes. One patient had a midcalf subcutaneous hematoma 5 weeks postoperatively, requiring evacuation.
Conclusion:
Arthroscopic complete posterior knee capsulotomy was able to restore knee extension, reduce pain, and improve function, with 12-month follow-up results sustained at 24-month follow-up.
Registration:
NCT05385393 (ClinicalTrials.gov identifier).
Extension deficit of the knee is a relatively common complication after surgical procedures of the knee as well as a component of osteoarthritis, systemic diseases, and other pathologies. In a recent meta-analysis by Scholes et al, 15 up to 1 in 3 anterior cruciate ligament reconstructions led to knee extension deficit at 1-year follow-up. Knee extension deficit is a debilitating disorder that has a significant impact on the biomechanics of the whole body.1,2,5 In cases refractory to guided physiotherapy, the treatment consists of a thorough arthrolysis of the affected knee. 4 However, if the extension deficit persists, contracture of the posterior knee capsule may develop. Therefore, both manipulation under anesthesia and arthrolysis could be insufficient. In such rare cases, the treatment consists of a posterior capsulotomy (posterior capsular release) of the affected joint.3,7,10,16 Traditionally, this procedure has usually been performed through an open approach.8,9,12,16
An arthroscopic approach is a less invasive procedure,14,18 potentially resulting in limited postoperative pain,14,18 faster recovery,7,14,18 and increased postoperative function. 7 Furthermore, arthroscopic fluid increases the space within the joint, providing the surgeon with a safer operative field and superior visualization.7,14,18
The purpose of this study was to assess the efficacy of an arthroscopic complete posterior knee capsulotomy in extension restoration, pain reduction, and functional improvement in cases of extension deficit refractory to guided physiotherapy and surgical arthrolysis.
Methods
This study presents the preliminary results of a single-arm prospective registered clinical trial (NCT05385393). The study was designed in compliance with the latest Helsinki Declaration, 19 and the study protocol was approved by a regional committee for medical research ethics. All patients provided informed consent. Inclusion criteria were age >18 years, symptomatic asymmetric extension deficit >3°, and inability to restore full knee extension after at least 6 months of guided physiotherapy and after initial knee arthrolysis. Exclusion criteria were presence of knee inflammatory signs, nonadherence to the treatment protocol, contractures due to extra-articular reasons, <90° of knee flexion, <6 months since the last surgical procedure of the affected knee, and restoration of full knee extension after the initial knee arthrolysis during the final procedure.
There were no inclusion or exclusion criteria concerning the cause of the knee extension deficit, as we wanted to assess whether posterior knee capsulotomy may be used universally in the treatment of a knee extension deficit. An a priori sample size calculation for primary outcome measure (knee extension at follow-up) was performed with alpha = .05 and beta = 0.95 for the Student matched-samples t test. Effect size was calculated based on the results from the study of LaPrade et al, 7 with a mean preoperative extension deficit of 14.7° and a mean postoperative extension deficit of 0.7°. The standard deviation was estimated from the equation (maximum - minimum)/4, resulting in standard deviations of 5.25 for preoperative value and 5.0 for postoperative value.7,17 The calculated effect size was 2.73. Analysis resulted in a necessary sample size of 5 participants. Such a small number was considered a result of a large calculated effect size of the procedure.
From 27 patients enrolled in this trial to date, we decided to analyze the results of patients with both 12-month follow-up (n = 15) and 24-month follow-up (n = 8). Figure 1 shows the flowchart of patient progression through the study.

Flowchart of patients screening, inclusion, and follow-up. *The 12-month follow-up results were not assessed in 1 patient due to leukemia relapse and intensive chemotherapy.
Surgical Technique
The arthroscopic posterior knee capsulotomy procedure was performed under general or regional anesthesia with the patient positioned supine. During the procedure, 5 arthroscopic portals were used: anterolateral, anteromedial, anteromedial parapatellar, posteromedial, and posterolateral. Initially, the diagnostic part of the arthroscopy was performed, aiming to detect adhesions or osteophytes impairing knee range of motion. Subsequently, the knee anterior and posterior arthrolysis and removal of osteophytes, if detected, was performed. The possibility of performing symmetrical hyperextension was assessed, and, if it was not possible, a complete arthroscopic posterior knee capsulotomy was performed. This procedure consisted of an isolated capsulotomy with no muscle release, performed at the level of the posterior aspect of the menisci, with transection of the posteromedial, posterolateral, and central capsule, and the posterior septum. In all patients, knees could be flexed to around 90°. Therefore, single-stage surgery was possible, because knee flexion enables posterior movement of the popliteal neurovascular bundle and makes it possible and safe to create posterior arthroscopic portals under direct visualization.
The posterior knee capsulotomy technique used in this study has been described in detail, including figures and video of each surgical step, by Malinowski et al. 10 Initially, a posteromedial portal was created under visual control with the scope inserted through the anterolateral portal and transnotch maneuver. The scissors and radiofrequency (RF) probe were introduced through a posteromedial portal and used to transect the posteromedial capsule from medial to lateral (Figure 2A). Then, the viewing portal was changed to posteromedial. The RF probe was introduced through an anteromedial parapatellar portal by means of transnotch maneuver to the posteromedial compartment and was used to extend the posteromedial capsulotomy and remove the medial part of the posterior septum. Next, the arthroscope was moved to the posterolateral compartment through the anteromedial portal over the posterior root of lateral meniscus and a posterolateral portal was created under direct visualization. The lateral part of the posterior septum was removed and posterolateral capsule was sectioned from lateral to medial (Figure 2C). When the lateral capsule was released, the arthroscope was introduced through the posterolateral portal. The RF probe, inserted through the posteromedial portal, was used to remove the remainder of the posterior septum. At the end of the procedure, scissors introduced through the posteromedial portal were used to create free space by blunt dissection between the central part of the posterior capsule and popliteal fossa connective tissue. An RF probe positioned anteriorly was introduced behind the central part of the posterior capsule to section the remainder of it (Figure 2B). Finally, careful manipulation was performed into extension until symmetrical hyperextension of the knee was attained.
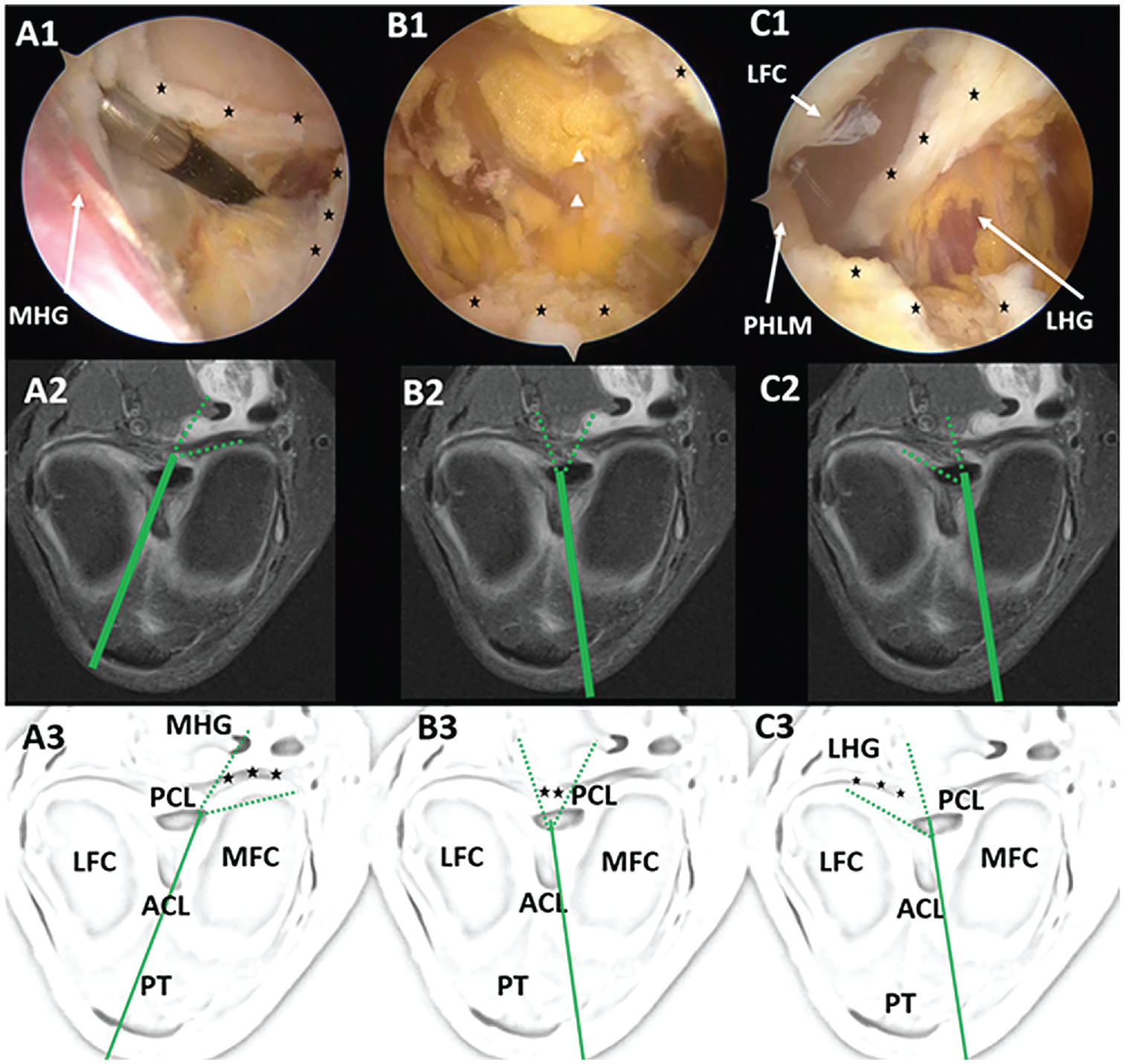
Arthroscopic view and presentation of visualization method of performed posterior knee capsulotomy. A patient with fluid in the popliteal space is shown for a better view of the posterior contour of the capsule. Top row: arthroscopic view; middle row, axial MRI scan; bottom row, labeled schematics. Asterisks indicate the capsule. (A1-A3) Posteromedial compartment, with arthroscope inserted through the anterolateral portal. (B1-B3) Central view, after septum resection, with arthroscope inserted through the anteromedial portal. Arrowheads in B1 represent connective tissue in front of popliteal neurovascular bundle. (C1-C3) Posterolateral compartment, with arthroscope inserted through the anteromedial portal. (A1-C1) After transection; (A3-C3) intact. Solid green line indicates arthroscope; dashed green lines indicate area of visualization. ACL, anterior cruciate ligament; LFC, lateral femoral condyle; LHG, lateral head of gastrocnemius; MFC, medial femoral condyle; MHG, medial head of gastrocnemius; MRI, magnetic resonance imaging; PCL, posterior cruciate ligament; PHLM, posterior horn of lateral meniscus; PT, patellar tendon.
Postoperatively, the patient performed gravitational hyperextension by keeping the heel of the operated leg on a support for 5 minutes each hour starting at the first postoperative day. At 2 weeks after surgery, patients started supervised physiotherapy 1 to 2 times per week for the next 3 months. Patients ambulated using crutches for 6 weeks with weightbearing depending on the postoperative knee condition and the patient’s tolerance. For this period of time, volume of walking was decreased to the minimum to avoid an excessive knee postoperative inflammatory reaction.
Outcome Measures
The primary outcome measure was knee extension at 12- and 24-month follow-up. Knee passive extension was measured with a goniometer with the patient lying supine.7,11,18 Two authors (K.M. and M.M.) performed this measurement independently twice with a 5-minute interval between measurements to minimize recall bias, and the worse outcome (ie, larger extension deficit or less hyperextension) was recorded in each case and used for analysis. Knee extension was assessed before surgery under anesthesia, immediately after surgery under anesthesia, and at 6-week and 3-, 6-, 12-, and 24-month follow-up. Knee hyperextension was denoted as the negative value of knee extension. Dynamic knee hyperextension, defined as an ability of the patient to actively elevate the heel off the examination table with the thigh pressed toward the table (Figure 3), was considered as the best possible outcome for the restoration of knee extension and was assessed at 12- and 24-month follow-up.
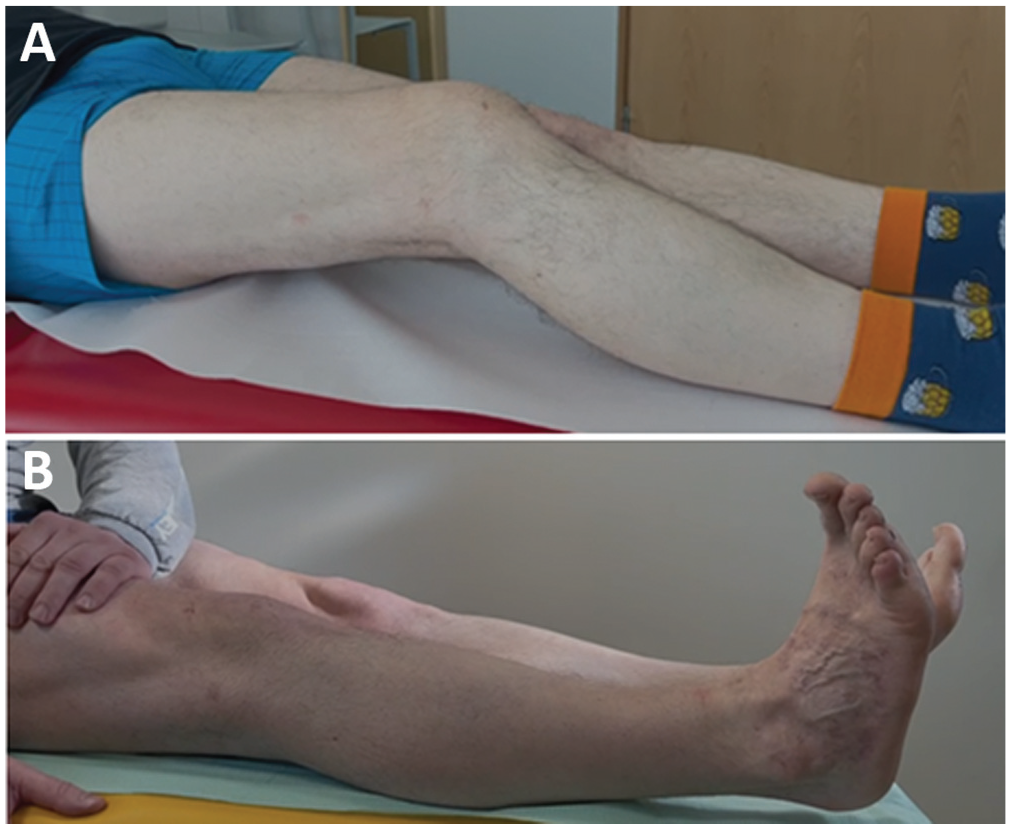
Examples from patients of (A) preoperative knee extension deficit of 30° and (B) dynamic hyperextension possible at 12-month follow-up.
Secondary outcome measures included visual analog scale (VAS) score of pain during maximum effort and exercise (range, 0-10 [worst pain]) and the International Knee Documentation Committee Questionnaire (IKDC) and the Knee injury and Osteoarthritis Outcome Score (KOOS) at 12- and 24-month follow-up6,13 All secondary outcome measures were assessed before surgery and at 6-week and 3-, 6-, 12-, and 24-month follow-up.
Data Management and Statistical Analysis
Collected data were analyzed statistically using Statistica Version 13.3 software (StatSoft Poland). Results of both primary and secondary outcome measures were compared from the preoperative time point to the 12-month and the 24-month follow-up, as well as between the 12- and 24-month follow-up times. First, the Shapiro-Wilk test was performed to assess the normality of the distribution. In case of normal distribution, the Student t test for paired samples was used. In case of nonnormal distribution, Wilcoxon signed-rank test was used. Because multiple paired-sample tests were conducted for each subgroup, the Bonferroni correction was used to multiply achieved P values by 3 before comparing with the significance threshold. The threshold for significance was set at the standard P < .05.
Results
Initial knee arthrolysis did not restore symmetrical hyperextension of the knee in any of the evaluated patients, and therefore complete posterior knee capsulotomy was performed in each patient. Consecutive patients with a knee extension deficit were included in the trial. The mean patient age was 40.0 years (range, 26-70 years), and 3 patients were female. The mean duration of knee contracture was 24.6 months (range, 6-60 months). Of the 15 patients in this study, 6 developed a knee contracture after an isolated anterior cruciate ligament reconstruction (ACLR); in 3 other cases the reason was isolated osteoarthritis; and in 1 case each, osteoarthritis and autoimmune disorder, systemic disease, articular fracture, bone infarctions and infection and systematic disease, ACLR with confirmed infection, and ACLR with an autograft osteochondral transfer. In none of the patients who had undergone ACLR was the extension lost immediately after ACLR. Irrespective of the contracture etiology, all patients had at least 0° of contralateral knee extension. Some of the patients underwent previous knee arthrolysis with partial meniscectomy and loose cartilage debridement. However, none of the patients underwent osteophyte removal and none had posterior portals created; therefore, it can be safely assumed that only an anterior arthrolysis was performed in the previous procedures. Study population characteristics are summarized in Table 1.
Characteristics of Study Population a

ACLR, anterior cruciate ligament reconstruction; OATS, osteochondral autograft transfer system.
Mean knee extension deficit decreased from 16.9° (range, 7°-45°) to -0.2° (range, -5° to 5°) at 12-month (P = .003) and to -0.3° (range, -5° to 5°) at 24-month (P = .035) follow-up (Table 2 and Figure 4). There was no significant difference between 12- and 24-month follow-up (P = .99). In all patients, symmetrical hyperextension was achieved intraoperatively after the manipulation. In none of the patients was it necessary to remove the anterior cruciate ligament graft to achieve symmetrical extension. Dynamic hyperextension was achieved in 7 of 15 patients at 12-month follow-up and in 4 of 8 patients at 24-month follow-up (Table 2).
Comparison of Extension Deficit and Functional Outcomes Between Preoperative and Follow-up Times a

IKDC, International Knee Documentation Committee score; KOOS, Knee injury and Osteoarthritis Outcome Score; NA, not available; Preop, preoperatively; VAS, visual analog scale. Boldface P values indicate statistically significant difference compared with preoperative value (P < .05). Knee hyperextension was denoted as the negative value of knee extension deficit.
The 12-month follow-up results were not assessed in patient 7 due to leukemia relapse and intensive chemotherapy.

Change in mean knee extension deficit in degrees at different time intervals from the surgery. Error bars represent ranges.
The mean VAS pain score decreased from 3.5 (range, 1-6) to 1.1 (range, 0-2) at 12-month (P = .004) and to 1.5 (range, 0-4) at 24-month (P = .005) follow-up (Figure 5.). There was no significant difference between 12- and 24-month follow-up (P = .99).

Mean VAS score for pain during maximum effort and exercise at different time intervals before and after surgery. Error bars represent ranges. VAS, visual analog scale.
The mean IKDC increased from 37.9 (range, 21-62) to 63.9 (range, 46-87) at 12-month follow-up (P < .001) and to 60.9 (range, 39-80) at 24-month follow-up (P = .003) (Figure 6). The mean KOOS increased from 45.0 (range, 30-62) to 75.3 (range, 49-94) at 12-month follow-up (P < .001) and to 72.3 (range, 49-92) at 24-month follow-up (P = .003) (Figure 6). There was no significant difference between 12- and 24-month follow-up for either IKDC score (P = .11) or KOOS (P = .99).

Mean IKDC and KOOS at different time intervals before and after surgery. Error bars represent ranges. IKDC, International Knee Documentation Committee score; KOOS, Knee injury and Osteoarthritis Outcome Score.
Regarding safety of the procedure, there were no permanent complications, such as neurovascular damage or instability of the knee. One patient presented with a subcutaneous hematoma of the midcalf 5 weeks after the procedure, requiring evacuation (Figure 7). None of the other patients have been reoperated to date.
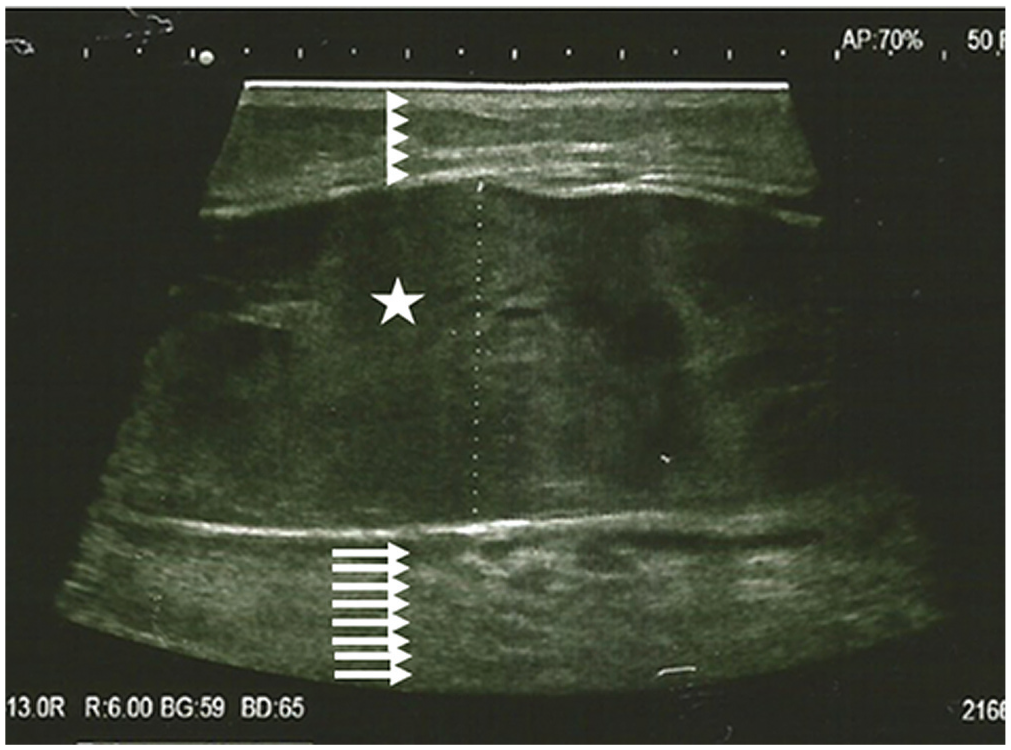
Ultrasound image of subcutaneous hematoma of the midcalf 5 weeks after arthroscopic complete posterior capsulotomy. Arrowheads represent skin and subcutaneous tissue; star represents hematoma; and arrows represent muscle belly.
Discussion
The most important finding of this study was that arthroscopic complete posterior capsulotomy was an efficacious treatment for knee extension deficit refractory to guided physiotherapy and surgical adhesion debridement, with 12-month follow-up results sustained at 24-month follow-up. This is the first trial assessing the efficacy of an arthroscopic complete posterior capsulotomy in knee extension restoration, pain reduction, and functional improvement with a prospective design and registration.
To our knowledge, 4 studies to date have assessed the outcomes of arthroscopic posterior knee capsulotomy in knee extension deficit treatment.7,11,14,18 Table 3 compares the study population, surgical technique, and design of these studies with those of the current study.
Study Population, Surgical Technique and Design: Comparison With the Literature a

ACLR, anterior cruciate ligament reconstruction; CAM, capsular attachment of the menisci; Gastroc, gastrocnemius; MM, medial meniscus; MPFL, medial patellofemoral ligament; N, no; NP, no registered protocol; NR, not reported; OA, osteoarthritis; OATS, osteochondral autograft transfer system; OCD, osteochondritis dissecans; P, registered protocol; PHMM, posterior horn of medial meniscus; PL, posterolateral; PM, posteromedial; Pro, prospective; PVNS, pigmented villonodular synovitis; Ret, retrospective; SDs, systemic disease; Y, yes.
Creation of transseptal portal was described; however, resection of the remaining part of posterior septum was not. 18
Transection of PL capsule was performed in some cases, whereas in other cases isolated PM capsule transection was performed. 14
As to the efficacy of knee extension restoration, results of the current study were comparable with the literature,7,11,14,18 confirming that arthroscopic posterior knee capsulotomy is effective for this pathology (Table 4). However, there were some differences in methodology between mentioned studies. As to assessment of knee extension, in the studies by LaPrade et al 7 and Reinholz et al, 14 hyperextension was denoted with negative values, similar to the current study. On the other hand, Wierer et al 18 compared extension deficit with the contralateral limb, and therefore no negative values were used. In a study by Mariani, 11 there were also no negative values; however, comparison of deficit angle with the contralateral limb was not described. We are aware that including negative values results in achieving slightly better measurement outcomes than in the studies of Mariani 11 and Wierer et al 18 ; however, this advantage may very well be purely semantic. Therefore the achieved values in our study can be compared directly only with results of LaPrade et al 7 and Reinholz et al. 14 Slightly superior results of the current study than in the studies of LaPrade et al 7 and Reinholz et al 14 may, however, be due to differences in surgical technique (Table 3).
Knee Extension Restoration: Comparison With the Literature a

IQR, interquartile range; NR, not reported.
Since last surgery.
Regarding pain reduction, Wierer et al, 18 Reinholz et al, 14 and the current study assessed VAS pain. While Wierer et al 18 did not elaborate as to whether the VAS pain score was assessed at rest or with use, Reinholz et al 14 assessed the VAS at rest and with use separately. In the current study, VAS was assessed as the worst pain during maximum effort and exercise. Despite differences in reporting (eg, medians vs means), comparable pain reduction was achieved in those 3 studies (Table 5). As to functional outcomes, IKDC score was assessed by Reinholz et al 14 and the current study, although preoperative IKDC values were not available for Reinholz et al 14 due to the retrospective design of their study. Results at follow-up were comparable with those of this study; however, once again reporting differences (medians vs means) occurred (Table 5). Tegner scores achieved by Wierer et al 18 were higher than those achieved by Reinholz et al 14 (Table 5). Other outcome measures such as the Lysholm (used by Wierer et al 18 ) or KOOS (used by the current study) could not be compared directly. However, Wierer et al 18 and Reinholz et al 14 reported significant improvement in functional outcomes, regardless of the functional score used for assessment.
Pain and Functional Outcomes: Comparison With the Literature a

IKDC, International Knee Documentation Committee score; IQR, interquartile range; KOOS, Knee injury and Osteoarthritis Outcome Score; NR, not reported; VAS, visual analog scale for pain.
Limitations
We recognize this study has some limitations. This study lacked a control group and therefore blinding and randomization was not possible. However, we are not aware of a different treatment modality other than posterior knee capsulotomy that is effective in the treatment of knee extension deficit refractory to guided physiotherapy and thorough joint arthrolysis.3,10 It was therefore considered unethical to leave a control group with no treatment due to the highly debilitating nature of the disease.1,2,5 Further, the study group was relatively heterogeneous, especially in comparison with previous works. The patients’ history and concurrent pathologies were numerous, including osteoarthritis, status post-ACLR, reconstructive procedures, and fracture care. In this instance, gross physiological, biomechanical, and perhaps biochemical differences as to the causative factor for the extension deficit may exist, thereby affecting the expected outcomes of patients in this cohort. On the other hand, this heterogeneity of the group allows for assessment of whether posterior knee capsulotomy may be used universally in treatment of extension knee deficit. Subgroup analysis (ie, men vs women, different etiological types of knee extension deficit) is not yet viable due to an insufficient number of patients; however, patient are being screened continuously for inclusion in the trial.
Conclusion
According to the preliminary results of this trial, arthroscopic complete posterior knee capsulotomy was efficient in knee extension restoration, pain reduction, and functional improvement in cases refractory to guided physiotherapy and surgical arthrolysis, with 12-month results sustained at 24-month follow-up.
Footnotes
Final revision submitted August 8, 2023; accepted August 21, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: N.I.K. has received hospitality payments from Encore Medical and Zimmer Biomet. R.F.L. has received royalties from Arthrex and Smith & Nephew; consulting fees from Smith & Nephew; and nonconsulting fees from Linvatec and Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from District Medical Chamber in Lodz (Reference No. K.B.-11/18).
