Abstract
Background:
A knotless, tensionable primary anterior cruciate ligament (ACL) repair system preloaded with an internal brace has been released. Currently, there is no biomechanical data on the stabilization and gap formation behavior of the adjustable system when compared with fixed repairs in human ACL tissue.
Hypothesis:
That knotless adjustable suture repair with an internal brace would provide overall higher construct stability and greater load share on the ACL with less gap formation compared with fixed repair.
Study Design:
Controlled laboratory study.
Methods:
Human cadaveric knees were utilized for internal braced ACL repair constructs (each group n = 16). Two fixed groups consisting of a single-cinch loop (SCL), cortical button (SCL group), and knotless suture-anchor (anchor group) were compared with an SCL-adjustable loop device (SCL-ALD) group. Testing was performed at 4 different peak loads (50, 150, 250, 350 N) over 4000 cycles at 0.75 Hz including suture repair preconditioning (10 cycles at 0.5 Hz) for SCL-ALD. Specimens were ultimately pulled to failure with a cut internal brace. The final loading situation of the construct and ACL repair with gap formation and ultimate strength were evaluated.
Results:
Peak elongation at various peak loads showed a significantly higher (P < .001) stabilization of SCL-ALD when compared with both fixed groups. There was a significantly higher (P < .001) load share of SCL-ALD, especially at lower loads (48% of 50 N), and the gap formation remained restricted up to 250 N. With only a little load share on the fixed constructs (<6%) at lower loads (50, 150 N), gap formation in these groups started at a load of 150 N, leading to significantly higher gaps (P < .001). The ultimate failure load for SCL-ALD and anchor groups was significantly increased (P < .001) as compared with SCL. The stiffness of SCL-ALD (62.9 ± 10.6 N/mm) was significantly increased (P < .001).
Conclusion:
Internal braced knotless adjustable fixation for ACL repair with preconditioning of the suture repaired ligament increased the overall stabilization with higher load share on the ACL and restricted gap formation (<0.5 mm up to 350 N) compared with fixed suture repair. All internal braced repairs restored stability according to native ACL function.
Clinical Relevance:
Adjustable ACL repair improved the mechanical characteristics and reduced gap formation, but the overall clinical significance on healing remains unclear.
Modern-day arthroscopic approaches in the primary repair of anterior cruciate ligament (ACL) injuries sparked renewed interest, especially for the treatment of the younger athletic population. 18 Primary ACL repair, historically performed open, showed poor outcomes with longer follow-up, but the surgical approach for ACL repair has evolved to an arthroscopic procedure focusing more on proximal ACL tears with good tissue quality. ACL repair allows the preservation of normal ACL anatomy and the proprioceptive properties of the ACL fibers.21,26 In addition to the development of several minimally invasive ACL repair techniques,5,10,17 biological augmentation enhanced healing rates and implant innovations have contributed to improved clinical outcomes.15,16,22 Additional mechanical protection using an independent nonabsorbable high-strength suture augmentation along with the ACL suture repair has previously shown promising biomechanical and clinical outcomes,2,4,6,8,11,14,27 demonstrating its importance for early protection of the repair construct to allow for healing. 19
Adjustable single-cinch cortical button fixation showed optimized time-zero ACL tension, leading to significantly improved stabilization and reduced gap formation with higher ultimate strength when compared with other suture repair techniques with either suture knot tying over a button or suture anchor fixation. 1 The added strength of the internal brace further optimized the stabilization potential at loads occurring during normal daily activities by reducing gap formation and peak loads on the ACL suture repair construct. 2 However, both studies were performed in a porcine model, leaving the stabilization effect of the repair in human ligament tissue unknown. A modified ACL fixation suture was designed to allow for simplified and efficient suture passing together with an adjustable loop device (ALD) implant for incremental suture repair tensioning independent of the nonabsorbable high-strength suture augmentation. Currently, there is a lack of biomechanical data regarding the stabilization potential of adjustable ACL repair tested dynamically in the load range of early and late rehabilitation when compared with clinically described fixed internally braced ACL repair techniques using knotless anchor or suture knot tying over the femoral button.5,28
The purpose of this study was to evaluate and compare the stabilization and gap formation behavior as well as ultimate failure strength of the adjustable ACL repair technique to fixed ACL repair techniques with a knotless suture anchor and knotted cortical button fixation in a biomechanical in vitro study using a cadaveric model. It was hypothesized that internally braced ACL repair with the adjustable implant would provide for improved mechanical stabilization with reduced gap formation and higher residual loads on the ACL compared with both fixed repair techniques.
Methods
Testing Groups
Fixed internally braced ACL repair techniques based on knotless suture anchor (Anchor) and single-cinch loop (SCL) cortical button fixation as described in previous research were tested biomechanically in a human model and compared with a new clinically available knotless SCL-ALD fixation.1,2 Cyclic testing was performed at 4 different load levels (each n = 4), resulting in 16 test samples for each group (for a total of 48).
Specimen Preparation
Overall, 24 paired cadaveric tibias with preserved ACL and femurs (59.7 ± 5.5 years and 82.4 ± 17.8 kg) provided by the Science Care donor bank were inspected visually after soft tissue resection to ensure the structural integrity of the native ACL without tears. The human specimens were randomized equally in a balanced incomplete block design into 3 groups without showing any statistical differences. The use of a balanced incomplete block diagram provides an optimal efficiency design statistical model to benefit from paired specimen similarity while analyzing 3 treatment groups. The ACL was released from the femoral footprint using a scalpel with the tibia potted in a 2-component fast-cast-resin polyurethane embedding (Huntsman Advanced Materials) in line with the anatomic ACL long axis. Embedding was carried out until 2 cm distal to the predefined tibial tunnel exit point to leave sufficient space for internal brace knot tying.
The ACL was lengthened to 30 mm along the longitudinal axis from the center of the tibial footprint measured with a digital caliper. According to previously published testing using porcine tissue, 2 the same bone tunnels were prepared using ACL aiming devices. The tibia was equipped with a 3.5-mm tunnel through the midportion of the ACL footprint, while a 2.4-mm pin was passed through the lateral wall of the notch in the center of the native femoral ACL footprint with a proximal lateral cortex exit point at a distance of 15 mm proximal and 5 mm anterior to the lateral epicondyle. 9 A bone block of 32 mm in diameter and 35 mm in length was extracted along the guide pin by using a cylinder drill and sawing off the medial bone portion. A continuous 3.5 mm–diameter tunnel for cortical button specimens and an anchor-specific drill hole of 3.7 mm in diameter were prepared (Figure 1). The embedded tibia and femoral-sided bone block docked in a custom-made steel fixture were secured to the base plate and actuator, respectively, of a dynamic testing machine (ElectroPuls E10000; Instron) using clamps. The initial distance from the femoral cortex to the ACL footprint was set to 65 mm to allow reattachment of the ACL stump to the femoral block. Prepared bones were stored at -20°C and thawed at room temperature overnight before biomechanical testing. All tests were performed at room temperature, and soft tissue was kept moist with physiological saline solution during preparation and testing.
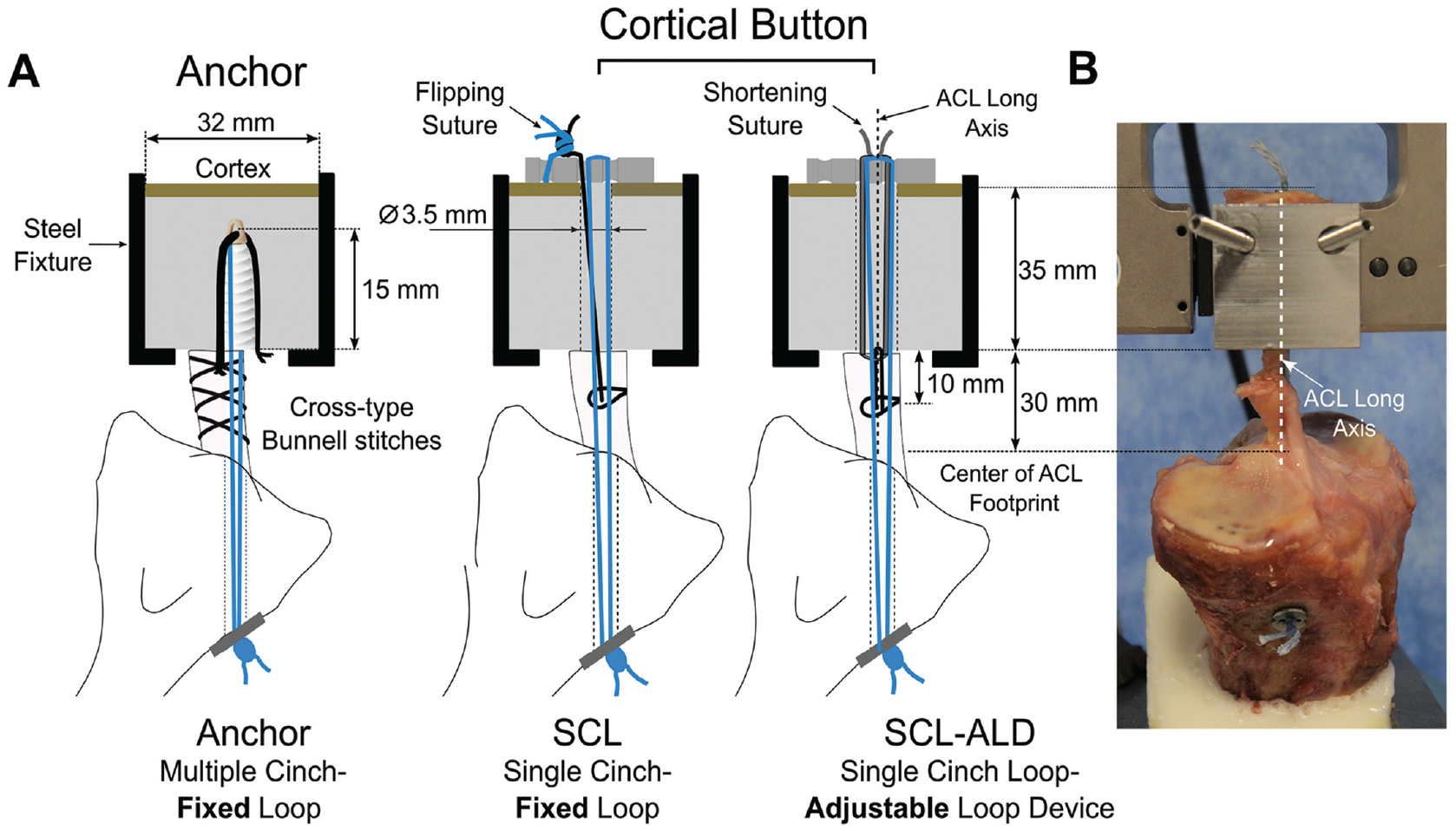
(A) Schematic illustration of the bone tunnel and fixation-related definitions of the internal brace ACL repair groups. (B) Final experimental setup. ACL, anterior cruciate ligament; SCL, single cinch-fixed loop; SCL-ALD, single-cinch loop-adjustable loop device.
Repair Techniques with Internal Brace
Two previously reported fixed repair techniques and the new adjustable ACL repair technique were utilized in addition to a high-strength suture tape (FiberTape 2 mm, Arthrex) to reattach the ACL to the femoral bone.1,2 The fixed suture repair techniques were based on SCL cortical buttons and multiple cross-stitch anchor fixation. The SCL fixation was created by passing a suture (FiberSnare, Arthrex) at a distance of 10 mm from the native ligament torn site through and around the ACL for closing the cinch by transferring the suture through the looped end.
The surgical technique steps in Figure 2 demonstrate the method of securing the SCL-ALD repair fixation using a continuous suture loop (FiberRing, Arthrex) with a preassembled shuttle loop and passing suture without reflecting the actual testing setup. For SCL fixation, mainly the anteromedial ACL bundle was reattached. The cortical button was shuttled through the femoral tunnel and flipped on the cortex with the internal brace looped over the central holes of the button. For SCL specimens, the No. 2 flipping suture remained in position for later femoral suture knot tying; otherwise, it was removed.

Surgical technique steps for SCL ACL repair fixation with adjustable loop device loading. (A,B) The passing suture was stitched through the proximal ACL and (C) transferred through the continuous suture loop for closing and tightening the cinch by advancing the shuttling loop. For ALD (TightRope II; Arthrex) assembly with the cinch loop in place, (D) the guiding strand attached to the closed adjustable loop was passed through the shuttle loop eyelet and (E) transferred through the cinch by pulling on the opposing shuttle loop suture. (F) The ALD assembly was finalized by passing the open strand through the closed loop and (G) pulling it through the predefined path using the threading help with the assembly card. (H) The guiding suture was removed thereafter. ACL, anterior cruciate ligament; ALD, adjustable loop device; SCL, single-cinch loop.
Three cross-type Bunnell stitches for each No. 2 suture limb connecting both ACL bundles with the final suture locking passes through the ACL below the most proximal Bunnell stitches were utilized for knotless ACL repair suture anchor fixation. 1 The 2 free tibial ends of the internal brace were shuttled through the tibial tunnel and left loose. The internal brace for knotless anchor fixation was looped together with the repair suture over the anchor eyelet with the 2 free ends on the tibial side. 2
Construct Fixation
A manual 50-N pull over 5 seconds was performed using a spring-loaded tensiometer for preconditioning of the repair sutures to ensure homogeneous engagement and reduced settling effects. 2 Suture repair fixation with the test machine actuator locked in position allowed for tracking of the construct tension.
Knotless suture anchor fixation in the femoral bone and SCL repair suture knot tying to the externally positioned button flipping suture with 4 half hitches using an arthroscopic knot pusher was performed for femoral fixation. The SCL-ALD specimen was manually tensioned to a defined ACL time-zero preload (50 N) by alternating pulling on the loop shortening strands and kept knotless. The tibial sided free ends of the internal brace were knotted over a button (TightRope ABS; Arthrex) with 4 half-hitch knots using tightening handles to adjust the tension level to approximately 50 N measured by the test machine (ElectroPuls E10000; Instron). The time-zero position for ACL repair and internal brace fixation served both as a reference for later dynamic elongation analysis and simulated a knee in full extension (joint space of 30 mm).
Biomechanical Testing
The load was applied in line with the ACL and tunnel axis to simulate a worst-case loading scenario at a frequency of 0.75 Hz over 4000 cycles. 2 Additional precycling of adjustable repair samples for a total of 10 cycles at 0.5 Hz between the time-zero position and -3 mm of slackening simulated intraoperative knee flexion activity between full extension and 90° of flexion.2,13 Thereafter, ACL repair retensioning (50 N) in the time-zero position was performed with the ALD left knotless. Overall, cyclic testing at 4 peak load levels representative of in vivo ACL loads during early and late rehabilitation (50, 150, 250, and 350 N) and a constant valley load of 10 N was performed with each construct loaded at the same peak load level over 4000 cycles.20,23 Finally, for all specimens, the internal brace was cut before pull to failure testing (50 mm/min), starting from the time-zero position, to provide information on the isolated ACL suture repair in relation to the overall construct stability. Load-displacement data during cycling and pull to failure were recorded using Wavematrix software (ElectroPuls E10000; Instron) with a sampling rate of 500 Hz.
Outcome Data
Final peak elongation (sp) at various load levels (Figure 3) as an indicator for the stabilization potential of a group was used for comparative analysis. Gap formation (sGap) and residual load (FR) at final peak elongation were determined during pull to failure for various ACL repairs with cut internal brace. Gap formation represents plastic deformation (laxity) with no load (<1 N) on the repair construct. The residual load on the repair (FR) at final peak elongation quantifies the amount of load transferred over the suture repair after the application of 4000 load cycles. Ultimate load and stiffness were determined for all constructs in the linear portion of the load-elongation curve.

Schematic illustration of the initial (first) and final (4000th) load cycle hysteresis curves with pull-to-failure progression of the isolated ACL suture repair after cutting the IB. Metrics for comparison included sP, sGap, and FR as well as ultimate load and stiffness during pull to failure (not shown). ACL, anterior cruciate ligament; FR, residual load on the ACL repair; IB, internal brace; sGap, gap formation; sP, final peak elongation; w/o, without.
Statistical Analysis
In this study, repair techniques were the independent variables, and all metrics for comparison parameters were the dependent variables; sP, sGap, and FR as well as ultimate load and stiffness were defined as the primary outcome variables. Statistical analysis was performed using Sigma Plot Statistics for Windows (Version 13.0; Systat Software).
The statistical analysis included a one-way analysis of variance (ANOVA) with Tukey post hoc test performed for pairwise analysis of primary outcome variables. Significance was defined as P ≤ .05 and the desired power level was set at 0.8. The Shapiro-Wilks test was used to confirm each dataset followed a normal distribution. A nonparametric Kruskal-Wallis test was used for datasets that failed this test. For Kruskal-Wallis tests that found significance, a Tukey post hoc test was conducted to further analyze the differences. The observed post hoc average power values of all 1-way ANOVA tests were much higher than the desired power level of 0.8, leading us to conclude that our sample size was sufficient.
A 1-way analysis of covariance (ANCOVA) was used for regression analysis of the overall concluding regressive load-displacement dependencies over the total load spectrum. The Shapiro-Wilks test was used to confirm that each dataset followed a normal distribution. For ANCOVA tests that were considered significant, a Holm-Sidak post hoc test was performed for pairwise analysis. Significance was defined as P ≤ .05 and the desired power level was set at 0.8. Data analysis was performed with MATLAB (Version R2019a; MathWorks).
Results
The results of peak elongation and gap formation as well as regression analysis for each mean outcome data as a function of applied peak loads with R2 values are shown in Table 1. Linear regression curves of the peak loads in dependence of peak elongation provided accuracy in the order of at least R2 = 0.92 for all groups.
s P and sGap by Group Across all Load Levels With Corresponding Regression Curves and Goodness-of-fit a

Data are shown as mean ± SD unless otherwise indicated. ACL, anterior cruciate ligament; SCL, single-cinch fixed loop; SCL-ALD, single-cinch adjustable loop device; sGap, gap formation; sP peak elongation.
Values in parentheses indicate the percentile displacement with ACL repair under loads in relation to peak elongation ((sP - sGap)/sP).
Functional zones of the mean and standard deviation peak elongation results at various loads of internally braced ACL repair techniques (Figure 4) were established to indicate the overall stabilization potential in relation to native ACL functional data. 3 While a combined functional zone was established for the Anchor and SCL groups because of similar peak elongation behavior (P = .87), the ANCOVA test showed a statistically higher (P < .001) stabilization potential of the adjustable ACL repair technique (SCL-ALD) when compared with both fixed groups.
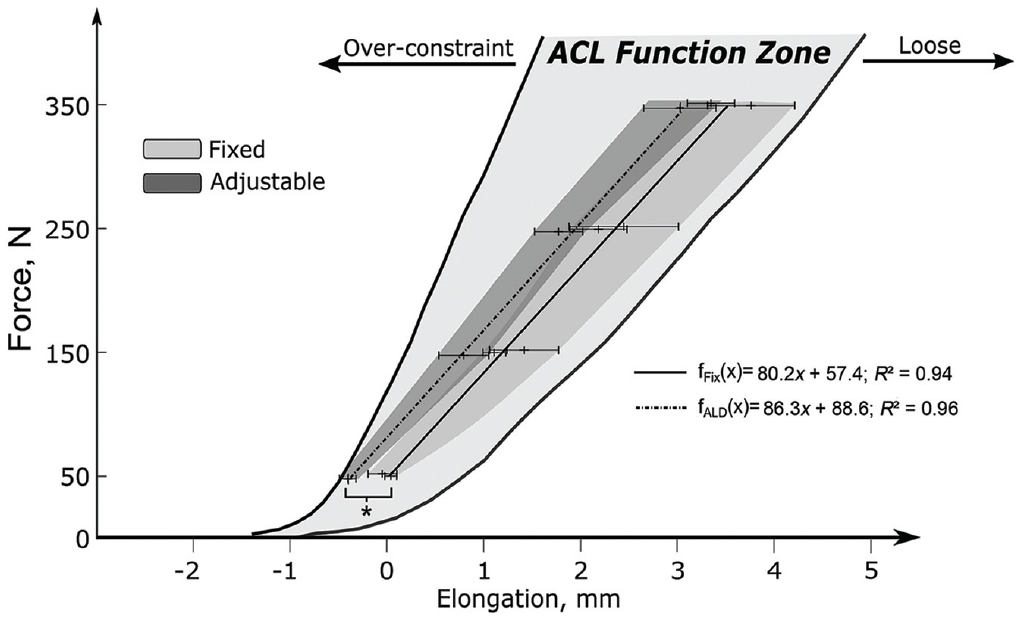
Mean peak elongation with standard deviation for applied load levels. The functional zones indicate the stabilization potential of ACL repair with internal brace augmentation with reference to the native ACL function zone. 3 *Statistically significant difference (P < .001, ANCOVA). ACL, anterior cruciate ligament; ALD, adjustable loop device; ANCOVA, analysis of covariance.
The magnitude of load transferred over the ACL repair at the end of testing was quantified and found to be significantly higher at all applied load levels (P < .001) for the adjustable ACL repair technique when compared with the fixed groups (Figure 5). The Anchor group demonstrated a significantly higher amount of load transferred over the repair when compared with the SCL group at higher load levels (250 N and 350 N).

Mean and median loads with percentage load share of various ACL repair techniques for different applied peak loads with statistical analysis. Statistically significant difference: *P < .05; **P < .001; mean power = 0.87. ACL, anterior cruciate ligament; SCL, single-cinch fixed loop; SCL-ALD, single-cinch adjustable loop device.
Concomitant with the higher loadbearing capability, the adjustable ACL repair fixation also resulted in significantly smaller gap formation than the fixed ACL repair groups. While both fixed ACL repair groups showed increasing gap formation from the peak load level of 150 N and higher, the adjustable technique showed no gap formation up to 250 N and a significantly smaller gap of 0.05 ± 0.05 mm at the final peak load of 350 N (Figure 6).
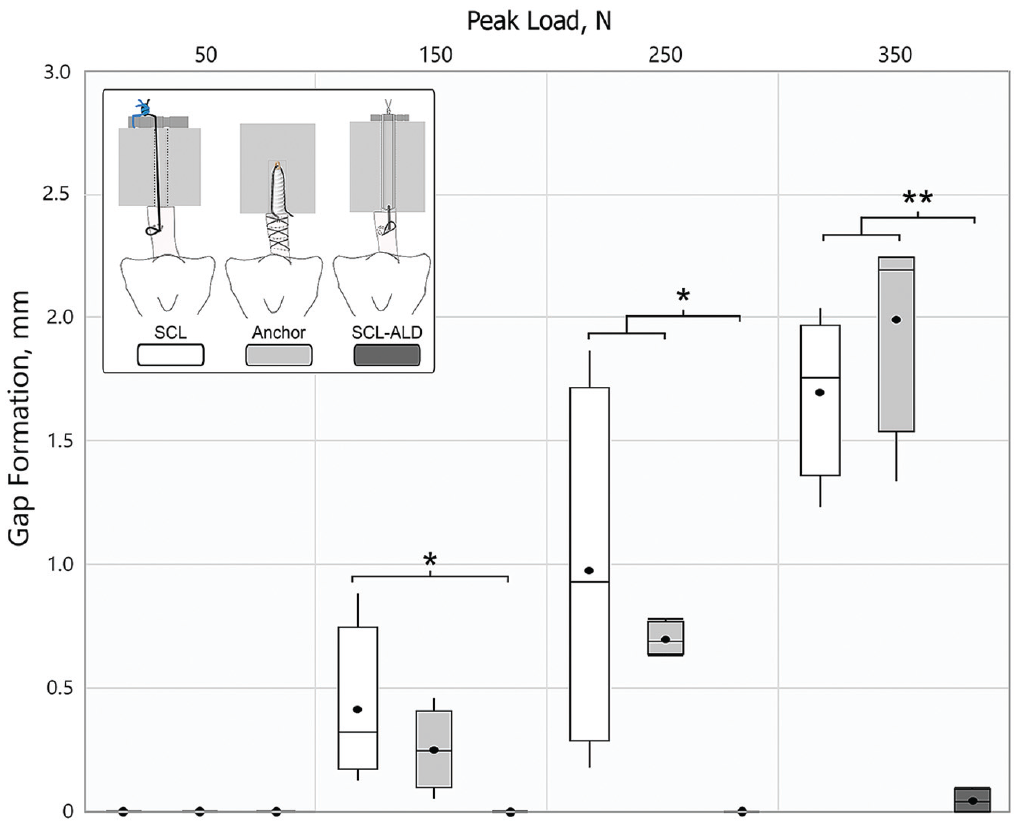
Mean and median gap formation data for different peak loads with statistical analysis. Statistically significant difference: *P < .05, **P < .001; mean power = 0.87. SCL, single-cinch fixed loop; SCL-ALD, single-cinch adjustable loop device.
Pull to Failure
All specimens reached the regular test end and were pulled to failure. The ultimate failure load for the SCL-ALD and Anchor groups was found to be significantly increased (P < .001) when compared with the SCL group (Figure 7). Ultimate stiffness was significantly increased (P < .001) for the SCL-ALD (62.9 ± 10.6 N/mm) when compared with both fixed groups and between the Anchor (33.7 ± 5.8 N/mm) and SCL (14.6 ± 4.1 N/mm) groups. In the SCL configuration failure occurred as knot slippage, whereas suture slippage on the anchor fixation site and of the tissue was observed for the Anchor and SCL-ALD groups, respectively.

Ultimate failure data of isolated tested ACL repair with statistical analysis. *Statistically significant difference, P < .001; mean power = 1. ACL, anterior cruciate ligament; SCL, single-cinch fixed loop; SCL-ALD, single-cinch adjustable loop device.).
Discussion
The most important finding of this study was that the newly released knotless adjustable ACL repair implant provided improved stabilization with significantly less gap formation and higher load-bearing capability than the fixed repair techniques in load-sharing configuration with the internal brace augmentation in a human cadaveric model. While SCL and Anchor groups showed growing gap formation beginning at a peak load level of 150 N, reaching a final gap of around 2 mm, the adjustable technique showed no gapping up to 250 N and hardly any gap formation (<0.5 mm) at 350 N. Overall, all internally braced ACL repair techniques provided sufficient mechanical stabilization to completely restore stability in the native ACL functional zone.
The advantages of ACL repair include preserving the native neurovascular anatomy and proprioception while eliminating graft site morbidity with the ease of performing primary ACL reconstruction as a revision for failed repair. 26 Moreover, patients undergoing arthroscopic primary ACL repair have been shown to have less daily awareness of their knee as compared with patients undergoing reconstruction. 24 In addition to optimizing the biological healing environment and a more careful patient selection focusing on acute proximal ACL tears with good remnant tissue quality,15,16,21 innovative technology such as knotless adjustable suture repair may further improve clinical outcomes. Suturing of the femoral ACL stump and fixing the sutures through bone tunnels by knot tying over a button or suture anchor next to the insertion site has a limited stabilization potential in relation to the in vivo requirements of the native ACL function. 1 Approaches described more recently, including biological or mechanical augmentation in addition to suture repair, revealed promising biomechanical and clinical results.2,4,6,8,11,14,27 The internal brace augmentation strengthens the repair construct and restores knee stability during the critical time of early mobilization and healing. To the best of our knowledge, this study is the first to evaluate and compare the mechanical performance of the new clinically available knotless adjustable ACL repair system with conventional cortical button or suture anchor fixation in load-sharing configuration with the internal brace in a human cadaveric model.
The advantages of the knotless, adjustable implant for ACL primary repair fixation include efficient arthroscopic suture passage as well as the ability to (re)tension the construct. In contrast to fixed suture repair techniques with limited tensioning ability, 1 tensioning of the adjustable repair implant can be performed in any degree of knee flexion, including intraoperative retensioning after cycling the knee through a full range of motion. Adjustable tensioning in this study was performed at simulated full knee extension; thus, tensioning in higher knee flexion angles may “overtension” the ACL repair, leading to higher stress at the ACL fixation site and suture-induced initial gap formation. Similarly, the internal brace was always secured with the knee joint in full extension. Intraoperative preconditioning of the repair construct can further reduce the ACL tissue to the femoral wall. Final fixation of the independent internal brace augmentation after the suture repair preconditioning process protects the weaker suture repair from excessive loads and gap formation.1,2 Despite the unequal mechanical stabilization potential of the repaired ACL, load sharing in weaker suture repairs is a critical aspect of the augmentation concept as the biological healing process of the reapproximated ACL depends on a load stimulus.7,25 Loading of the fixed suture construct repaired ligament was, in general, limited over all applied peak loads, but especially low at smaller loads (<5% of 50 N), with the internal brace as the primary stabilizer. Suture repair preconditioning with an adjustable implant improved primary fixation at the time of ACL reapproximation by reducing initial settling effects and gap formation. Less gapping, equivalent to ACL repair tension loss (<1 N) due to knot slippage or suture-induced settling effects could be associated with higher fixation strength and load-bearing capability during simulated early and late rehabilitation loading. Thus, load sharing occurred with a higher amount of load transferred over the adjustable suture-repaired ligament. Although the clinical implications of the initial tension and the optimal amount of load for ACL healing remain unknown, immediate postoperative loading with gradually increasing loads transferred over the suture-repaired ligament according to early and late rehabilitation activities without losing contact with the femoral wall should provide an appropriate mechanical stimulus for the healing tissue.7,25 Overstretching with greater gap formation of the suture repaired ACL from the femoral wall or overprotection of the ACL repair with insufficient load stimulus for ligament healing may induce a disordered fibrosis process impairing the quality of scar tissue formation. 29
Direct comparison of current results to studies using shear loading according to an anterior drawer test for anteroposterior laxity testing is difficult due to the different test protocols and test setups.8,12 Based on previous biomechanical testing on the treatment of acute proximal ACL tears with suture repair and internal brace augmentation in porcine tissue, current test results are in line with these findings. 2 In general, adequate mechanical stabilization according to native ACL function was found for various internally braced ACL repair techniques with limited gap formation. Slight differences in the load-bearing and gap formation behavior between related ACL repair groups in both studies may have occurred because of (1) differences in the primary fixation stability of implant devices and (2) differences in the structural properties between human and porcine tissue. Further biomechanical investigation using 2 loops for fixation of the anterior and posterior ligament bundles may help to evaluate further potential improvements of the primary stabilization with adjustable ACL repair fixation.
Limitations
We acknowledge some limitations to the current study. In contrast to variable in vivo loading conditions in the human knee during daily activities, the load application vector was in line with the ACL and internal brace long axis in this study by in vitro worst-case loading conditions for biomechanical testing. The mean age of human donors was older than patients undergoing an ACL repair and therefore may not be representative of a clinical setting. Inconsistent structural properties on the human tissue due to age- and sex-related effects between groups may had an influence on the biomechanical outcome. Thus, the current test methodology is an in vitro simulation of the in vivo loading environment, and the obtained biomechanical performance of various ACL repairs could differ from clinical practice. Variable knot tying or tensioning of the repair depending on the surgeon’s experience may also influence the clinical outcome in practice. Although often multiple suturing for ACL repair fixation is performed in the operating theater, this biomechanical work focused on the performance of a single-cinch repair fixation. Furthermore, this study does not take into consideration the effects of biological healing for ACL repair and patients’ clinical outcomes.
Conclusion
Internal braced knotless adjustable fixation for ACL repair with preconditioning of the suture repaired ligament increased the overall stabilization with higher load share on the ACL and restricted gap formation (<0.5 mm up to 350 N) compared with fixed suture repair. All internal braced repairs restored stability according to native ACL function.
Footnotes
Final revision submitted April 28, 2023; accepted May 19, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: Arthrex provided research support for this study. S.B., D.R., O.H., and C.A.W. are employees of Arthrex. P.A.S. and S.H. are consultants for Arthrex. P.A.W. has received education payments from Elite Orthopedics and United Orthopedics; consulting fees from Arthrex; nonconsulting fees from Arthrex, Kairos Surgical, and Medical Device Business Services; and royalties from Arthrex. S.H. has received education payments from Liberty Surgical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
