Abstract
Background:
Although several complications of proximal hamstring tendon ruptures have been reported in the literature, few studies have comprehensively analyzed the complication profile of proximal hamstring tendon repair.
Purpose:
To identify the overall rate of complications following proximal hamstring tendon repair and to differentiate these complications into categories.
Study Design:
Systematic review; Level of evidence, 4.
Methods:
Included in this review were studies that examined surgical repair of proximal hamstring tendon ruptures; all studies were in English and had an evidence level of 4 or higher. No restrictions were made regarding publication date or methodological quality. Data regarding complications were extracted to calculate the overall complication rate as well as the rate of major and minor complications. A quantitative data synthesis was conducted using the chi-square test to compare the proportion of patients who experienced complications with the endoscopic versus open approach.
Results:
A total of 43 articles including 2833 proximal hamstring tendon repairs were identified. The overall postoperative complication rate was 15.3% (n = 433). The rate of major complications was 4.6%, including a 1.7% rate of sciatic nerve injury, 0.8% rate of venous thromboembolism, 0.8% reoperation rate, 0.8% rerupture rate, and 0.4% rate of deep infection. Minor complications included a 2.4% rate of posterior femoral cutaneous nerve injury, 2.3% rate of persistent hamstring myopathy, 2.2% rate of persistent sitting pain, 1.8% rate of peri-incisional numbness, 1.1% rate of superficial infection, and 0.8% rate of hematoma/seroma.
Conclusion:
Proximal hamstring tendon repair is associated with an overall complication rate of 15.3%, including a 4.6% rate of major complications.
Hamstring injuries are among the most common athletic injuries in both elite and recreational athletes, accounting for 12% to 16% of all injuries in the athlete population.8,41 In a systematic review composed of 53 studies and 144 hamstring injuries in the general population, tears of the muscle or tendon accounted for 57.6% of total injuries, with the remaining 42.4% being avulsions of the hamstring tendon origin/insertion to bone. 26 Although most hamstring injuries arise at the musculotendinous junction, proximal hamstring tendon ruptures may also occur when 1 or more hamstring tendons avulse from their origin at the ischial tuberosity, typically as a result of simultaneous hip flexion and knee extension.17,38,45
Indications for surgical repair of hamstring injuries include acute semimembranosus ruptures with clear evidence of tendon retraction, acute conjoint tendon ruptures with >2 cm of retraction, failed nonoperative treatment of conjoint tendon ruptures with <2 cm of retraction, and complete ruptures of both the conjoint tendon and the semimembranosus tendon (with or without concomitant adductor avulsion).30,34 Surgical repair of proximal hamstring tendon ruptures has been shown in multiple clinical outcome studies to be superior to nonoperative management, with improved functional scores and ability to return to sport postoperatively. 20 However, operative intervention in the setting of proximal hamstring tendon rupture has been associated with several complications, including nerve injury, tendon rerupture, and persistent pain with sitting. 38
Previous studies have reported variable incidence rates of these complications, which has contributed to substantial heterogeneity in the literature pertaining to proximal hamstring tendon repair. The objective of this systematic review was to provide a comprehensive synthesis of all articles that have reported complications associated with proximal hamstring tendon repair, including quantitative estimates of the overall complication rate, as well as major and minor complication rates.
Methods
This systematic review was performed according to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. A health sciences librarian developed the search strategy utilizing a combination of keywords and specific database subject headings related to each concept, including proximal hamstring, semimembranosus, semitendinosus, biceps femoris, avulsion, tear, rupture, and outcomes (Appendix Table A1). A search was conducted within PubMed, Medline (via Ovid), Embase (via Ovid), Web of Science Core Collection, and SPORTDiscus (via EBSCOhost) between inception and May 4, 2022. No limit regarding the year of publication was imposed. Non-English and nonhuman studies were excluded from the search, and duplicates were removed using EndNote X9 (Clarivate).
To qualify for inclusion in the systematic review, studies were required to examine the surgical repair of proximal hamstring tendon avulsions or ruptures and represent level 4 evidence or higher. Only original research studies were included. Nonoperative treatment, cadaveric studies, myositis ossificans, proximal hamstring tendinopathy, avulsion fractures of the ischial tuberosity, and proximal hamstring tendon reconstruction using allograft were excluded. All abstracts and full-text articles were stored in Rayyan QCRI, which allowed blinding of each independent reviewer to the inclusion/exclusion decisions made by the other reviewer throughout the article assessment process.
All identified articles (n = 718) were assessed for study eligibility by 2 reviewers, each of whom was blinded to the inclusion/exclusion decisions of the other reviewer. The 2 independent reviewers, a medical student (J.J.L.) and physician assistant (E.A.A.) followed an algorithm for abstract screening that was developed and piloted by the senior author (H.P.G.). After excluding all irrelevant articles during the abstract screening process (n = 661), full-text articles were obtained for all remaining abstracts (n = 57). In cases of disagreement between the 2 reviewers, blinding was removed, and the final eligibility of the article was resolved by a group consensus, with the senior author (H.P.G.) making the final decision. The 2 primary reviewers then independently performed a detailed assessment of each full-text article. A total of 43 studies met the inclusion criteria (Figure 1).

PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) diagram illustrating study inclusion and exclusion. a As the primary data search included all possible related articles, additional sources were not needed.
The included studies underwent data extraction for study design (eg, cohort, case-control, case series), level of evidence (1-4), surgical approach (open vs endoscopic), injury setting (sports, other), and mean or median follow-up time. Duplicate patient cohorts were excluded. Specific complications were also extracted from each article to calculate the overall complication rate. Complications were then further categorized into major complications (those that caused debilitating injury, required an operation, or were potentially life-threatening, such as rerupture, reoperation, sciatic nerve injury, venous thromboembolism [VTE] development, and deep infection) and minor complications (those that caused persistent symptoms without significant impairment, pain, numbness, superficial infection, and transient nerve injury, such as posterior femoral cutaneous nerve [PFCN] injury, persistent sitting pain, persistent hamstring myopathy, hematoma/seroma, peri-incisional numbness, and superficial infection) based on author consensus as well as previous literature reports.21,23
Articles that did not explicitly state the level of evidence were independently graded for level of evidence by the same 2 blinded reviewers. None of the included articles were randomized controlled trials that would qualify as level 1 evidence. Prospective cohort studies were considered as having level 2 evidence, retrospective cohort and case-control studies were considered level 3 evidence, and case series were considered level 4 evidence.
The number of overall, major, and minor complications were aggregated among all included studies and analyzed using descriptive statistics. The chi-square test was used to compare the rates of overall, major, and minor complications. Statistical analysis was performed using R statistical software package (Version 1.4.1717-3, RStudio).
Results
The characteristics of the 43 included studies are shown in Appendix Table A2. ‡ Article publication dates ranged from 1995 to 2022. Of the 43 studies included, 15 were prospective (34.9%), § and 28 were retrospective (65.1%). ‖ Level 4 case series comprised a majority of the included studies (n = 33; 76.7%) (Appendix Table A2).
Study Characteristics
The included studies comprised 2999 patients and 2833 surgeries, with some studies reporting outcomes for nonoperative patients, none of whom were included in our analysis. Some patients had bilateral injuries and received 2 surgeries, in which case both procedures were recorded. Three studies included patients who were treated with endoscopic proximal hamstring repairs,1,19,25 while 37 studies included patients who were treated with open hamstring repairs ¶ ; 3 studies11,18,32 included patients who were treated with endoscopic or open repairs. Of the studies that reported patient sex, there were 1683 male patients (62.7%) and 1003 female patients (37.3%). In studies that reported sporting level, 194 patients were professional or semi-professional athletes (6.7%), 92 were college or high school level elite athletes (3.2%), and 944 were athletes at a recreational or unspecified level (32.6%). Of the studies that distinguished between acute and chronic injuries, 706 injuries were acute (58.3%) and 506 were chronic (41.7%), with a majority of these studies defining the acute time frame as within 28 to 42 days of injury. A total of 23 studies did not report the number of patients lost to follow-up (53%). # A total of 82 patients were lost to follow-up from the studies that reported that metric (2.8%). Mean follow-up time was 3.23 years (range, 0.7-6.5 years).
Complications
The overall complication rate for a total of 2833 proximal hamstring repairs was 15.3% (n = 433). The rate of major complications was 4.6% (n = 129), including a 1.7% rate of sciatic nerve injury (n = 48), 0.8% rate of VTE (n = 25), 0.8% reoperation rate (n = 23), 0.8% rerupture rate (n = 22), and 0.4% rate of deep infection (n = 11) (Table 1). The rate of minor complications was 10.5% (n = 297). Minor complications included PFCN injury (2.4%; n = 69), persistent hamstring myopathy (2.3%; n = 64), persistent sitting pain (2.2%; n = 61), peri-incisional numbness (1.8%; n = 50), superficial infection (1.1%; n = 31), and hematoma/seroma (0.8%; n = 22) (Table 1).
Incidence of Major and Minor Complications a
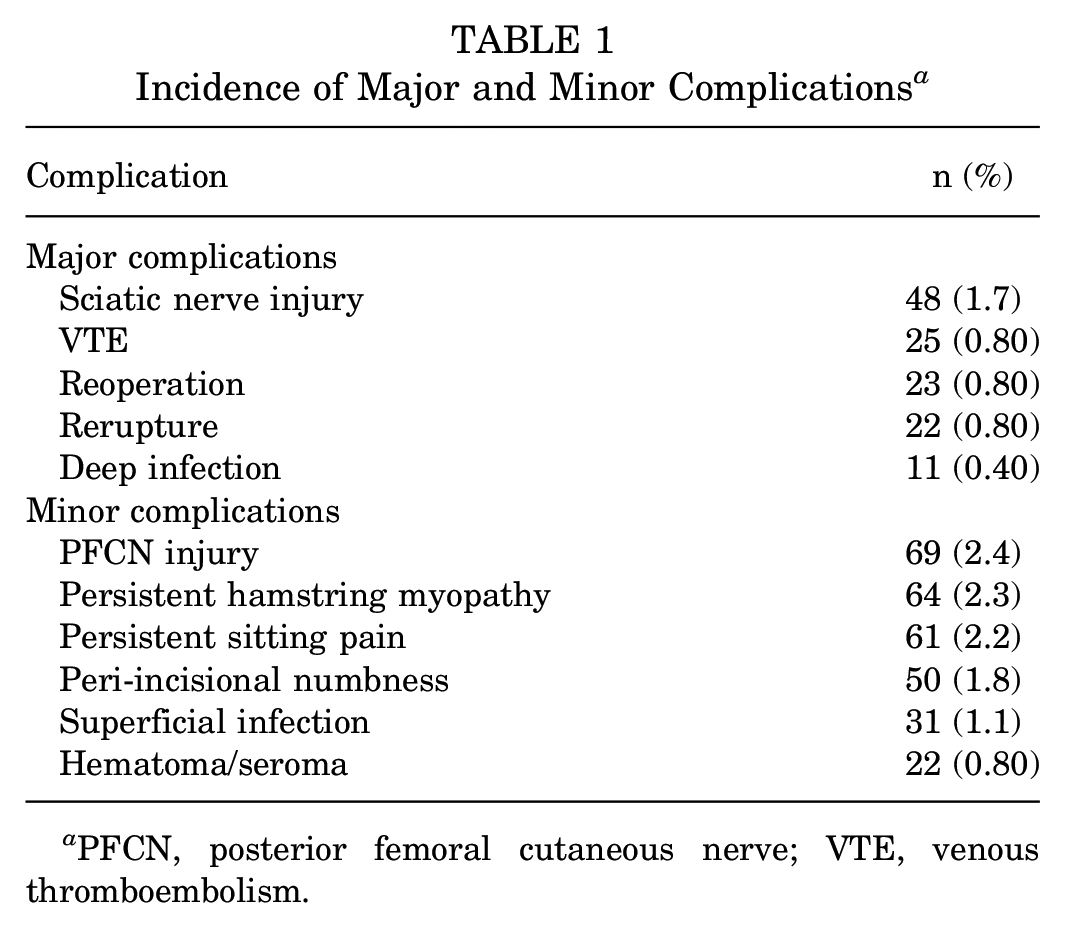
PFCN, posterior femoral cutaneous nerve; VTE, venous thromboembolism.
Regarding major complications, 12 studies reported postoperative sciatic nerve injuries with wide variability, ** ranging from 1.7% to 17.4%; 12 studies reported VTE complications, †† occurring at a rate of 0.5% to 5.3%; and 12 studies reported rerupture ‡‡ and 10 studies reported reoperation. §§ Rerupture and reoperation were reported with wide variability, ranging from 1.5% to 12.2% and 1.1% to 16.7%, respectively. Four studies reported rerupture in patients where reoperation was not indicated.22,29,44,45
Regarding minor complications, 15 studies reported PFCN injury, with rates ranging between 0.8% to 45.5% ‖‖ ; 10 studies reported postoperative persistent hamstring myopathy, with rates ranging from 2.9% to 63.6% ¶¶ ; and 15 studies reported superficial infection, with rates ranging from 0.8% to 6.5%. ## Regarding infection, 10 studies reported superficial infections that resolved with oral antibiotics, a 1 study reported a superficial infection requiring an additional operative washout, 10 1 study reported a superficial infection that resolved with local debridement and dressing changes, 12 and 4 studies did not report treatment regimen for the superficial infection.11,37,43,46
Discussion
The findings of this systematic review suggest that, despite successful outcomes associated with proximal hamstring tendon repair, there is a substantial risk of sustaining an intraoperative or postoperative complication. For the 2833 repairs included, the overall complication rate was 15.3%, including a 4.6% rate of major complications and 10.5% rate of minor complications.
We defined major complications as those that caused debilitating injury, required an operation, or were potentially life-threatening. Of the studies to report sciatic nerve injury, Subbu et al 44 assessed the largest number of patients, with a rate of 12 sciatic nerve injuries in 112 patients (10.7%). Birmingham et al 9 reported the highest incidence of injury, with 4 sciatic nerve injuries in 23 patients (17.4%). Injury to the sciatic nerve, although seemingly rare, significantly impacts a patient's quality of life by producing pain, paresthesias, and foot drop. Deep venous thrombosis (DVT) is a concerning complication after lower extremity orthopaedic surgery, given the potential for pulmonary embolism. 17 Engler et al 17 specifically assessed the incidence of DVT associated with proximal hamstring rupture, reporting 5 DVTs in 132 operatively treated patients (3.8%). This is higher than our observed frequency of 0.8%.
We defined minor complications as those that caused persistent symptoms without significant impairment, such as pain, numbness, superficial infection, and transient nerve injury. Although less catastrophic than sciatic nerve injury, injury to the PFCN presents a quality-of-life issue if a patient cannot sit comfortably for the rest of their life. The study by Klingele et al 23 observed the highest frequency of PFCN injury, which occurred in 5 of their 11 patients (45.5%). We used hamstring myopathy as a general term to represent hamstring pain, muscle weakness, muscle cramps, muscle tightness, and tendinitis due to the heterogeneity of similar symptoms reported in the reviewed literature and found wide variability of this complication (2.9% to 63.6%) in the current review.
Limitations
This study is limited in that we could not perform a formal quantitative systematic review comparing open versus endoscopic approach for the repair of proximal hamstring tendon avulsions. There is a paucity in the existing literature on endoscopic repair for these types of injuries; thus, the data cannot be compared objectively with data from the more common open approach. There was significant heterogeneity in temporally defining an acute versus chronic hamstring tendon injury, which made it difficult to assess outcomes in that regard. One would expect an increase in complications associated with repair of chronic injury, as chronic injury has less preserved native anatomy, higher incidents of tendon retraction, and more scar tissue. All these factors contribute to a more complicated repair, but we were unable to assess how they affected our data. As stated earlier, the lack of clear classification of a major versus minor complication may explain the high rates of complications and variation in the studies we assessed. Over 50% of the studies did not report the number of patients lost to follow-up. This, combined with the 82 patients that were reported as lost to follow-up, suggests that more complications may have occurred than were reported in the literature. There is a level of ambiguity regarding what is recognized as a complication. For example, we included peri-incisional numbness as a minor complication because many of the studies in the existing literature had done so, but really this is probably an expected sequelae of having an operation of any kind. The effect of this may be an artificially inflated complication rate for our review as well as those that chose to report it as a minor complication. Lastly, the granularity of detail reporting whether or not patients experienced more than 1 complication along with the timepoint at which complications occurred was unclear, making any further analyses down those avenues difficult.
Conclusion
Proximal hamstring tendon repair is associated with an overall complication rate of 15.3%, including a 4.6% rate of major complications.
Footnotes
Appendix
Characteristics of Included Studies a

| Lead Author (Year) | Study Design | LOE | Patients/ Surgeries (Sex), n | Age, y b | Follow-up, y b | Surgery Technique | Complications |
|---|---|---|---|---|---|---|---|
| Aguilera-Bohórquez (2021) 1 | Case series | 4 | 3/3 (NR) |
48.3 ± 16.2 | Median, 3.6 | Endoscopic | Deep infection |
| Aldridge (2012) 2 | Case series | 4 | 23/23 (10 M/13 F) |
42 (25-58) | 3.1 | Open | PFCN injury, persistent hamstring myopathy, persistent sitting pain |
| Arner (2019) 3 | Case series | 4 | 64/70 (27 M/37 F) |
47.3 (16-65) | 6.5 | Open | VTE, PFCN injury, superficial infection, peri-incisional numbness, persistent hamstring myopathy, rerupture, reoperation, sciatic nerve injury |
| Asokan (2021) 4 | Prospective cohort | 2 | 980/980 (766 M/214 F) |
Cohort 1: 29 ± 6.7
c
Cohort 2: 27 ± 5.2 d |
NR | Open | VTE |
| Ayuob (2020) 5 | Case series | 4 | 20/20 (9 M/11 F) |
M: 27.4 ± 3.8 F: 30.6 ± 5.4 |
2.3 | Open | Superficial infection, hematoma/seroma |
| Ayuob (2020) 6 | Case series | 4 | 64/64 (42 M/22 F) |
M: 25.7 ± 3.8 F: 28.4 ± 3.4 |
2.4 | Open | PFCN injury, hematoma/seroma, rerupture, reoperation |
| Barnett (2015) 7 | Case series | 4 | 132/132 (77 M/55 F) |
42.5 ± 12.2 | 4.5 | Open | VTE, PFCN injury, superficial infection, hematoma/seroma, persistent hamstring myopathy, rerupture, reoperation, sciatic nerve injury, persistent sitting pain |
| Birmingham (2011) 9 | Case series | 4 | 23/23 (15 M/8 F) |
46 (19-65) | 3.6 | Open | Peri-incisional numbness, sciatic nerve injury |
| Blakeney (2017) 10 | Prospective cohort | 2 | 94/96 (45 M/49 F) |
Median: 50 (IQR: 16-74) | Median, 2.8 | Open | Superficial infection, deep infection |
| Bowman (2019) 11 | Case series | 4 | 58/58 (25 M/33 F) |
51.1 ± 12.0 | 2.4 | Endoscopic and open | Superficial infection, peri-incisional numbness, persistent hamstring myopathy, persistent sitting pain |
| Bowman (2013) 12 | Case series | 4 | 17/17 (3 M/14 F) |
43 (19-64) | 2.7 | Open | PFCN injury, superficial infection, deep infection, peri-incisional numbness, persistent hamstring myopathy |
| Brucker (2005) 13 | Case series | 4 | 8/8 (6 M/2 F) |
40 (23-60) | 2.8 | Open | Peri-incisional numbness, reoperation |
| Cain (2018) 14 | Case series | 4 | 3/3 (2 M/1 F) |
45 (20-67) | 1 | Open | No complications reported |
| Chahal (2012) 15 | Case series | 4 | 13/13 (8 M/5 F) |
44.6 (27-63) | 3.1 | Open | Persistent hamstring myopathy, sciatic nerve injury, persistent sitting pain |
| Cohen (2012) 16 | Case series | 4 | 52/52 (26 M/26 F) |
47.7 (17-66) | 2.8 | Open | VTE, PFCN injury, peri-incisional numbness, sciatic nerve injury |
| Engler (2019) 17 | Case series | 4 | 132/132 (NR) |
NR | NR | Open | VTE |
| Factor (2021) 18 | Case series | 4 | 6/6 (5 M/1 F) |
48 (20-61) | 2.3 | Endoscopic and open | Reoperation |
| Fletcher (2021) 19 | Case series | 4 | 30/31 (6 M/24 F) |
52.0 ± 14.2 | 3.7 | Endoscopic | Rerupture, reoperation |
| Kanakamedala (2023) 21 | Case series | 4 | 38/38 (16 M/22 F) |
51.4 ± 9.9 | 4.9 | Open | VTE, superficial infection, deep infection, rerupture, reoperation, sciatic nerve injury |
| Kayani (2020) 22 | Case series | 4 | 41/41 (31 M/10 F) |
M: 39.4 ± 8.3 F: 38.5 ± 6.9 |
2.4 | Open | Superficial infection, rerupture, sciatic nerve injury |
| Klingele (2002) 23 | Retrospective cohort | 3 | 11/11 (7 M/4 F) |
41.5 (21-51) | 2.8 | Open | PFCN injury, persistent hamstring myopathy, persistent sitting pain |
| Konan (2010) 24 | Case series | 4 | 10/10 (8 M/2 F) |
29.2 ± 4.29 | NR | Open | PFCN injury |
| Kurowicki (2020) 25 | Case series | 4 | 19/19 (6 M/14 F) |
46.2 (18-63) | 1.9 | Endoscopic | Persistent hamstring myopathy, reoperation, persistent sitting pain |
| Lefevre (2013) 27 | Case series | 4 | 34/34 (25 M/9 F) |
39.3 ± 11.4 | 2.3 | Open | Persistent sitting pain |
| Lefevre (2013) 28 | Case-control | 3 | 34/34 (25 M/9 F) |
39.3 ± 11.4 | 2.3 | Open | Hematoma/seroma, persistent hamstring myopathy, persistent sitting pain |
| Léger-St-Jean (2019) 29 | Case series | 4 | 34/38 (21 M/13 F) |
Cohort 1: 47.1 ± 13.4
e
Cohort 2: 46.2 ± 19.9 f |
4.1 | Open | VTE, deep infection, rerupture, sciatic nerve injury |
| Lempainen (2006) 31 | Case series | 4 | 47/48 (32 M/15 F) |
33 (16-61) | 3 | Open | Superficial infection, peri-incisional numbness, rerupture, reoperation |
| Maldonado (2021) 32 | Case series | 4 | 50/50 (16 M/34 F) |
46.1 ± 13.0 | 4.8 | Endoscopic and open | PFCN injury, hematoma/seroma |
| Mansour (2013) 33 | Case series | 4 | 10/10 (10 M/0 F) |
27.2 (23-30) | NR | Open | No complications reported |
| Orava (1995) 35 | Case series | 4 | 8/8 (6 M/2 F) |
40 (22-53) | 5.7 | Open | Sciatic nerve injury |
| Pihl (2021) 36 | Retrospective cohort | 3 | 50/37 (26 M/24 F) |
50.9 ± 9.8 | 5.5 | Open | PFCN injury |
| Pihl (2019) 37 | Retrospective cohort | 3 | 47/33 (16 M/17 F) |
51 (34-68) | 3.9 | Open | VTE, superficial infection |
| Rust (2014) 38 | Retrospective cohort | 3 | 72/58 (32 M/19 F) |
Cohort 1: 49.8 (25-74)
g
Cohort 2: 40.7 (14-62) h |
3.8 | Open | PFCN injury, superficial infection, deep infection, rerupture, reoperation |
| Sallay (2008) 39 | Case series | 4 | 25/25 (13 M/12 F) |
43.5 (20-69) | 0.7 | Open | VTE, peri-incisional numbness |
| Sarimo (2008) 40 | Case series | 4 | 41/41 (21 M/20 F) |
46 (18-71) | 3 | Open | VTE, PFCN injury, superficial infection, deep infection, hematoma/seroma, rerupture, reoperation |
| Shambaugh (2017) 42 | Retrospective cohort | 3 | 25/14 (5 M/9 F) |
46.98 ± 9.73 | 3.56 | Open | Hematoma/seroma, peri-incisional numbness |
| Skaara (2013) 43 | Case series | 4 | 31/31 (16 M/15 F) |
51 (27-73) | 2.5 | Open | PFCN injury, superficial infection |
| Subbu (2015) 44 | Case series | 4 | 112/112 (76 M/36 F) |
29 ± 8.5 | 2 | Open | Superficial infection, rerupture, sciatic nerve injury |
| van der Made (2022) 45 | Case-control | 3 | 59/26 (15 M/11 F) |
Median: 51 (IQR: 45-56) | 1 | Open | Rerupture |
| Willinger (2020) 46 | Case series | 4 | 94/94 (53 M/41 F) |
53.8 ± 12.3 | 4.68 | Open | VTE, superficial infection, hematoma/seroma, reoperation, sitting pain, peri-incisional numbness |
| Wilson (2019) 47 | Case series | 4 | 67/67 (30 M/37 F) |
48 ± 12 | NR | Open | Sciatic nerve injury |
| Wilson (2017) 48 | Retrospective cohort | 3 | 162/67 (30 M/37 F) |
48.3 ± 11.7 | 0.25 | Open | PFCN injury |
| Wood (2020) 49 | Case series | 4 | 156/156 (74 M/82 F) |
49.2 (21.5-78.9) | 3 | Open | VTE, PFCN injury, deep infection, persistent hamstring myopathy, sciatic nerve injury |
F, female; IQR, interquartile range; LOE, level of evidence; M, male; NR, not reported; PFCN, posterior femoral cutaneous nerve; VTE, venous thromboembolism.
Age data are reported as mean ± SD or mean (range) unless otherwise indicated. Follow-up data are reported as mean unless otherwise indicated.
Mechanical deep vein thrombosis prophylaxis.
Mechanical deep vein thrombosis prophylaxis plus chemical prophylaxis.
Acute repair (<4 weeks from injury to surgery).
Chronic repair (>4 weeks from injury to surgery).
Acute repair (<6 weeks from injury to surgery).
Chronic repair (>6 weeks from injury to surgery).
Final revision submitted May 2, 2023; accepted May 19, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: J.C.D. has received education payments from Supreme Orthopedic Systems, consulting fees and royalties from Zimmer Biomet, and nonconsulting fees from Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
