Abstract
Background:
A classification system for the graft state after superior capsule reconstruction (SCR) using magnetic resonance imaging (MRI) has not been described previously.
Purpose:
To introduce a new, MRI-based classification system for graft integrity after SCR and to evaluate the system according to postoperative outcomes.
Study Design:
Cohort study (diagnosis); Level of evidence, 3.
Method:
Included were 62 consecutive patients who underwent SCR using autologous fascia lata graft between January 2013 and April 2021. Postoperative outcomes were assessed (American Shoulder and Elbow Surgeons [ASES] score, Constant score, pain visual analog scale [pVAS], range of motion [ROM], acromiohumeral distance [AHD], Hamada grade). Graft status was classified by 2 orthopaedic surgeons on postoperative MRI in accordance with the signal intensity and the presence or extent of the tear, as follows: type 1 (hypointense signal without tear), type 2 (hyperintense signal without tear), type 3 (partial-thickness tear), type 4 (full-thickness tear with partial continuity), and type 5 (full-thickness tear with complete discontinuity). Intra- and interobserver agreement were assessed using Cohen kappa. The correlation between postoperative outcomes (ASES score, Constant score, pVAS, ROM, AHD, and Hamada grade) and the SCR graft classification system was assessed with the Pearson correlation coefficient, and the outcomes were compared according to classification type.
Results:
Patients were classified according to the new system as follows: type 1 (n = 15), type 2 (n = 20), type 3 (n = 7), type 4 (n = 8), and type 5 (n = 12). There was excellent interobserver agreement (κ = 0.819) and intraobserver agreement (κ = 0.937 and 0.919). The classification system showed a moderate to high correlation with the ASES score (r = –0.451; P = .001), pVAS (r = 0.359; P = .005), AHD (r = –0.642; P < .001), and Hamada grade (r = 0.414; P < .001). Patients classified as having types 1 and 2 showed better outcomes in terms of ASES score, pVAS, ROM, and AHD compared with type 5 patients (P ≤ .021 for all).
Conclusion:
The new classification system was highly reproducible and showed clinical utility for both radiological and clinical evaluation after SCR.
Keywords
Studies regarding superior capsule reconstruction (SCR) as an alternative treatment for massive irreparable rotator cuff tears have been increasing. 2,13,17,21,39 SCR, which utilizes the autologous fascia lata as a humeral head depressor, was introduced by Mihata et al in 2012. 27,29 SCR reinforces superior static stability and prevents superior migration of the humeral head caused by the massive rotator cuff tear. Many studies have reported favorable clinical outcomes of SCR, 2,21,25 –27,39,40 and graft healing is an important factor in this regard. 15,16,25,40
The established Sugaya classification 36 uses postoperative magnetic resonance imaging (MRI) to evaluate the quality of tendon healing using 5 grades after rotator cuff repair surgery and is useful for assessing the condition of the tendons. 24,32,41 However, healing from an SCR using autologous fascia lata is referred to as “graft healing,” which is not the same as the “tendon healing” that occurs after rotator cuff repair.
The primary aim of this study was to classify patients after SCR based on graft integrity status using an MRI-based system similar to the Sugaya classification system for rotator cuff tendon healing. The secondary aim was to evaluate clinical outcomes according to this classification. We postulated that comparisons of clinical outcomes in accordance with the new SCR graft classification system would be applicable in clinical practice.
Methods
The protocol for this study was approved by our institutional review board. Between January 2013 and April 2021, a total of 131 consecutive patients underwent SCR by a single surgeon (I.H.J.) at our hospital. All patients in this population who underwent postoperative MRI after 1 year were identified (n = 98). Among them, we excluded patients with a poor-quality MRI that was not suitable for graft evaluation (n = 10) and those who received SCR using an allograft (n = 26). Ultimately, a total of 62 patients were included in the analyses (Figure 1).

Study flow diagram. MRCT, massive rotator cuff tear; MRI, magnetic resonance imaging; SCR, superior capsule reconstruction.
SCR Surgery
After an irreparable rotator cuff tear was diagnosed via a diagnostic arthroscopy, we performed acromioplasty to reduce the friction between the subacromial undersurface and the graft. Subscapularis (SSC) repair was also performed in cases with concomitant SSC tears. The defect was measured in the medial-lateral and anterior-posterior directions.
The fascia lata was harvested from the ipsilateral thigh and prepared as a double-folded 2-layer graft. A running suture with Ethibond (Ethicon) 2-0 in the graft margin was used. A graft of at least 6 mm in thickness was obtained in the final preparation, which was recorded in the operation record. After debridement of the superior margin of the glenoid, 2 or 3 suture anchors (1.7-mm Suture fix Anchor; Smith & Nephew) were inserted from the 10-o’clock to 2-o’clock position according to the defect size. A sliding locking knot suture was used on each anchor. A double-row suture bridge method was used for humeral side fixation. Two threaded anchors (4.5-mm Healicoil; Smith & Nephew) were inserted anteriorly and posteriorly to the medial row of the footprint, respectively, and the graft was fixed with a mattress suture. Remnant rotator cuff tissue or subacromial bursa were sutured using remaining strings from the medial row anchors and fixed with 2 knotless anchors (4.5-mm; Footprint Anchor; Smith & Nephew) inserted into the lateral row of the footprint. All graft fixations were performed in a 30° shoulder abduction and neutral rotation position. (Figure 2). In 38 of 62 patients (61.29%), mesh was implemented between the grafts to enhance stiffness. 16

Graft preparation and superior capsule reconstruction procedure. (A) Polypropylene mesh augmentation. (B) Marginal running suturing. (C) The mean thickness of the graft was at least 6 mm. (D) Suture anchors were inserted in the 10-o’clock to 2-o’clock direction of the superior surface of the glenoid. (E) After fixation of the graft at each side of the glenoid and the humerus. (F) Remnant tissues including rotator cuff tendon and bursa tissues were repaired on the graft (over the top technique) and fixed.
Postoperative Rehabilitation
Immobilization for 6 to 8 weeks with an abduction brace was performed in all patients. After immobilization, a passive range of motion (ROM) exercise program was started. After a full ROM was achieved, strengthening exercise programs including elastic band exercises and periscapular muscle strengthening exercises were commenced. Patients were recommended to return to daily activities within a tolerable range at 3 months after surgery, while leisure sports activities were allowed 1 year after surgery.
Patient Data, Radiological Assessment, and Shoulder MRI
Patient information (age, sex, underlying disease, and history of previous surgeries) was collected through a review of electronic medical records. Clinical outcomes (ROM, pain visual analog scale [pVAS], American Shoulder and Elbow Surgeons [ASES] score, and Constant score) measured preoperatively and at 1 year postoperatively were retrieved from the medical records.
The Hamada classification system was used to evaluate the stage of cuff tear arthropathy on plain radiographs taken preoperatively and 1 year postoperatively. The acromiohumeral distance (AHD) was measured using preoperative and 1-year postoperative plain radiographs obtained with the shoulder in a neutral position.
Pre- and postoperative shoulder MRIs were performed using a 3-T scanner (Ingenia; Philips). The following parameters were used on the scanner: axial and coronal T2 fat saturation (repetition time [TR] = 4700 ms; echo time [TE] = 65 ms), coronal T1 (TR = 640 ms; TE = 21 ms), and coronal and sagittal T2 (TR = 2880 ms; TE = 80 ms). Weighted images were acquired. The slice thickness was 2 mm with an interslice gap of 0.5 mm (field of view, 150 mm; image matrix, 512 × 512). The imaging data were jointly reviewed and evaluated by 2 orthopaedic surgeons (J.-B.L. and J.W.Y.). To evaluate preoperative characteristics of the shoulder pathology, fatty infiltration (FI) of the rotator cuff muscle and tendon retraction were assessed using preoperative MRI. The preoperative FI of the rotator cuff muscle was assessed using the Goutallier classification: grade 0 for normal muscle, grade 1 for muscle with fatty streaks, grade 2 for muscle with greater FI, grade 3 for muscle with equal FI, and grade 4 for muscle with lesser FI. 7,10,35 The degree of tendon retraction was evaluated using the Patte classification on coronal and axial views of the shoulder 16,33 : grade 1, in which the tear stump of the tendon is retracted and located before the lateral articular margin; grade 2, in which the stump is at the level of the humeral head; grade 3, in which the stump is at the glenoid level; and grade 4, in which the stump is located medially to the glenoid level. 33
MRI Assessment of Graft Integrity
Postoperative MRI was performed at 1 year postoperatively to evaluate the graft integrity. We identified the graft tissue between the inserted anchors to reduce the possibility of misreading torn graft when the MRI cut direction was not parallel to the graft direction. To determine the integrity of the graft, several consecutive cuts of the images were checked in the coronal and sagittal views (Figure 3).

Comparisons between (A) arthroscopic view and (B-G) postoperative MRI. (A) Arthroscopic view from a standard lateral portal. The autologous fascia lata graft was placed between the glenoid and humerus and fixed with anchors at each side. The yellow arrows indicate the locations of the glenoid anchors, the large white arrows indicate the locations of the humerus anchor, the horizontal white line represents the width of the graft, the red line represents the virtual line of the coronal section MRI, and the red asterisk represents a pseudotear shown in (G). (B-D) T2-weighted sagittal view postoperative MRI scans. (B) Anchors were inserted into the glenoid (white arrows). (C) Midpoint of the graft. The horizontal white line represents the graft width. (D) Anchors were inserted into the humerus (white arrows), and the graft was placed between the anchors. (E-G) T2-weighted fat-suppressed coronal view postoperative MRI scans. The graft was placed between the anchors (white arrows). (G) A graft tear–like finding (red asterisk), but a pseudolesion due to the direction of MRI acquisition (red line), which was not parallel to the graft. MRI, magnetic resonance imaging.
The thickness of the graft was checked in both the sagittal and coronal views and was categorized as normal, partial tear, or a complete tear according to the depth of the tear in the graft. The intrasubstance hypersignal intensity of the graft was determined when there was only a signal change without graft tear. The proposed graft classifications after SCR were as follows (Figure 4): type 1, graft with no tear and with homogeneously low intensity on each image; type 2, graft with no tear with intrasubstance hypersignal intensity; type 3, graft with a partial-thickness tear; type 4, graft with a full-thickness tear but with partial integrity; and type 5, graft with a full-thickness tear and complete discontinuity.

Schematic images and postoperative MRIs of SCR grafts classified into 5 categories. (A) Type 1, graft with no tear and with homogeneously low intensity on each image. (B) Type 2, graft with no tear with intrasubstance hypersignal intensity. (C) Type 3, graft with a partial-thickness tear. (D) Type 4, graft with a full-thickness tear but with partial integrity. (E) Type 5, graft with a full-thickness tear and complete discontinuity. MRI, magnetic resonance images; SCR, superior capsule reconstruction.
Intra- and Interobserver Reliability of SCR Graft Classifications
Two orthopaedic shoulder specialists (J.-B.L. and J.W.Y.) participated in the reproducibility assessment of the new classification system for graft healing after SCR. Each observer independently classified the graft status twice in accordance with this new system, with an interval of at least 4 weeks between assessments.
Clinical and Radiological Outcomes
An independent examiner (J.W.Y) who was not involved in any of the surgeries conducted the clinical assessments of the study patients. The preoperative and 1-year postoperative clinical outcomes (ASES score, Constant score, pVAS, and ROM) and radiological (AHD and Hamada classification 12 ) outcomes were compared according to SCR graft type under the new classification. Due to the small number of included study patients, types 1 and 2 were combined into group A (without tear), types 3 and 4 were combined into group B (tear but with continuity), and type 5 was regarded as group C (without continuity) for statistical analysis.
Statistical Analysis
Quantitative data are described as mean ± standard deviation and qualitative data as number and percentage. Data sets for measured parameters were compared using the Mann-Whitney U test for continuous data and the Fisher exact test for categorical data. For intergroup comparisons (groups A versus group B versus group C), analysis of variance with Tukey post hoc test was used. The intra- and interobserver reliability of the MRI assessments were calculated using the Cohen kappa coefficient (κ), 4 with κ values interpreted as described by Landis and Koch 18 : <0 (no agreement), 0 to 0.20 (slight agreement), 0.21 to 0.40 (fair agreement), 0.41 to 0.60 (moderate agreement), 0.610 to 0.80 (substantial agreement), and 0.81 to 1.00 (almost perfect agreement). The Pearson correlation coefficient (r) was used to evaluate the correlations between the clinical outcomes and the new SCR classification system, in which r values <0.3 were considered low, 0.3 to 0.6 moderate, and >0.6 high. The significance level for all comparisons was set at P < .05. All descriptive and analytic analyses were conducted using SPSS version 21.0 (IBM Corp).
Results
Patients
Among the 62 study patients (mean age, 65.2 ± 8.5 years), 21 (33.9%) were men. A concomitant SSC repair was performed in 9 (14.5%) patients. In most of the patients, the tear margin was retracted to glenoid level or more retracted medially (Patte grade 3 in 48 [77.4%] and grade 4 in 8 [12.9%]). Preoperative mean AHD was 5.06 ± 2.11 mm. The mean follow-up duration was 28.5 ± 17.7 months. Table 1 lists the patient demographics and preoperative clinical and radiological findings.
Demographic, Clinical, and Radiological Characteristics of the Study Patients (N = 62) a
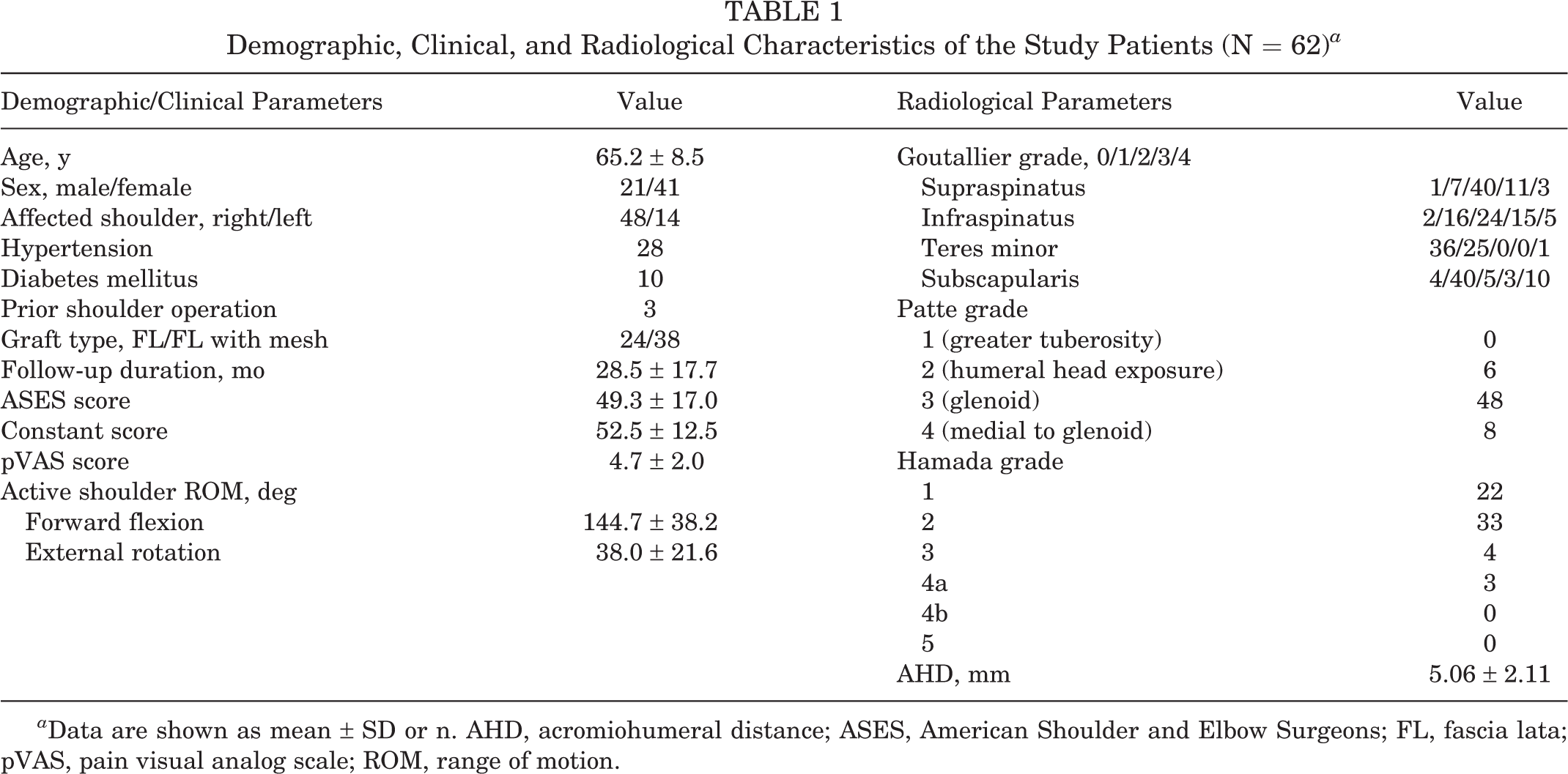
a Data are shown as mean ± SD or n. AHD, acromiohumeral distance; ASES, American Shoulder and Elbow Surgeons; FL, fascia lata; pVAS, pain visual analog scale; ROM, range of motion.
SCR Graft Classifications and Intra- and Interobserver Agreement
According to the new SCR classification of the postoperative graft status, 15 patients were classified as type 1 (24.2%), 20 as type 2 (32.3%), 7 as type 3 (11.3%), 8 as type 4 (12.9%), and 12 as type 5 (19.4%). The intraobserver agreement among the assessments was almost perfect, with a mean κ coefficient of 0.937 and 0.919 in each observer. The interobserver agreement was almost perfect, with a mean coefficient of 0.819.
Clinical and Radiological Outcomes According to SCR Graft Type
After surgery, patients in groups A, B, and C showed significant increases in their ASES (P < .001, P < .001, and P = .003, respectively) and pVAS (P < .001, P < .001, and P = .014, respectively) values compared with the preoperative levels. The Constant score was significantly elevated only in groups A and B (P < .001 for both). Improvement in forward flexion (FF) after SCR was only noted in group A (P = .016). External rotation showed no significant difference among the 3 groups. AHD was increased significantly in groups A and B (P < .001 and P = .004, respectively).
Postoperative ASES was significantly higher in group A (82.5 ± 7.4) than in group C (69.9 ± 10.8; P < .001). Postoperative pVAS was significantly lower in group A (1.19 ± 0.78) than in group C (2.08 ± 0.67; P = .021). Postoperative AHD was the highest in group A (P < .001). In terms of postoperative Hamada classification, group A showed a higher degree of improvement than group B (P = .030). Group C showed no improvement after SCR and there was 1 patient who showed progression in cuff tear arthropathy (Table 2).
Clinical and Radiological Outcomes According to SCR Graft Type a

a Data are shown as mean ± SD or n. Boldface P values indicate statistically significant difference between groups compared as indicated (P < .05). AHD, acromiohumeral distance; ANOVA, analysis of variance; ASES, American Shoulder and Elbow Surgeons; ER, external rotation; FF, forward flexion; NA, not applicable; Preop, preoperative; Postop, postoperative; pVAS, pain visual analog scale; ROM, range of motion; SCR, superior capsule reconstruction. Group A = types 1 and 2 (without tear); Group B = types 3 and 4 (tear but with continuity); group C = type 5 (tear without continuity).
b Comparison among groups A, B, and C
c Comparison between preoperative and postoperative data.
d Chi-square analysis between groups A and B.
Correlation of SCR Classification System With Clinical/Radiological Outcomes
The new SCR graft classification system showed a moderate to high degree of correlation with ASES (r = -0.451; P = .001), pVAS (r = 0.359; P = .005), ROM FF (r = -0.496; P < .001), AHD (r = -0.642; P < .001), and Hamada classification (r = 0.414; P < .001) (Table 3).
Correlation between SCR Classification System and Clinical/Radiological Outcomes a

a Boldface P values indicate statistical significance (P < .05). AHD, acromiohumeral distance; ASES, American Shoulder and Elbow Surgeons; ER, external rotation; FF, forward flexion; IR, internal rotation; pVAS, pain visual analog scale; ROM, range of motion; SCR, superior capsule reconstruction.
Discussion
Our SCR graft classification method showed almost perfect inter- and intraobserver reliability. Furthermore, this classification system showed moderate to high correlations with clinical (ASES and pVAS) and radiological (AHD and Hamada classification) outcomes. Type 5 classification in this system, which denotes a complete discontinuity of the graft, was significantly associated with poor clinical and radiological outcomes that were indicative of a failed SCR.
In the histological evaluation of SCR grafts, second-look arthroscopy and biopsy graft specimens are considered to be the gold standard; however, they are invasive and thus not ideal for clinical follow-up. 38 Graft healing in the orthopaedic field is therefore mostly evaluated using MRI. 9,11,22,34,37,42,43 A detailed description of the graft status using MRI is important to distinguish whether the graft is healing or if a pathologic condition (eg, partial tear, total rupture) has emerged. Our new classification for the graft status after SCR uses 5 different grading scores. This system will therefore help orthopaedic surgeons or radiologists to describe the graft state in clinical practice. It will also have utility in describing graft changes in future studies.
In our study patients, image analysis was performed using an MRI reading protocol that clearly defined the location and graft status with high reproducibility. The distinction between the graft and the surrounding tissue is an important factor in accurately determining the graft state on an MRI scan. 16,20 In some cases, distinguishing the graft from the surrounding tissue is difficult due to graft remodeling. In addition, if the direction of the graft and that of the MRI are not parallel, the possibility of misdiagnosing a graft tear should also be considered. Importantly, our imaging analysis with an MRI reading protocol showed almost perfect intra- and interobserver agreements.
The SCR graft classifications from our new system showed a moderate correlation with the clinical outcomes. Previous studies have reported that graft healing is the key to achieving favorable outcomes after SCR regardless of the graft materials used. 5,25,26 Pain from subacromial impingement, muscle weakness, and restricted active shoulder ROM are common symptoms of irreparable rotator cuff tear. 6,8,23,31 Defects in the superior capsule and posterosuperior rotator cuff tendons cause the loss of superior stability. 1,14,28,29 More severe symptoms may be caused by loss of stability of the glenohumeral joint. 3 In our present study, the graft healing group showed better clinical outcomes in terms of the ASES and pVAS values.
Our SCR classification system showed a moderate to high correlation with the AHD and Hamada classifications. Mihata and colleagues have reported that a healed graft provides superior stability and leads to significant increases in AHD. 27 In cases of failed graft healing, another study reported that a loss of superior stability leads to humeral head superior migration and subsequent progression of cuff tear arthropathy. 25 In our present study, the SCR graft classification showed significant correlations with AHD (r= -0.642; P < .001) and with the Hamada classification (r = 0.414; P < .001), suggesting that graft healing improves AHD and Hamada classification. However, Denard et al reported that preoperatively severely decreased AHD and advanced Hamada grade could be correlated with postoperative graft failure. 5 The causal relationship between AHD and postoperative graft healing is thus still unclear, and further research is needed to investigate this.
In this present study, group A (without tear) was associated with the best clinical outcomes, followed by group B (tear with graft continuity) with intermediate clinical outcomes. Group C (tear with graft discontinuity) patients had relatively poor clinical outcomes. In previous MRI-based studies, comparative analyses were conducted in populations with graft tears and those with graft healing. 19,20,25,27,40 It is notable that, in relation to the postoperative graft status of the autologous fascia lata, the spectrum varies from no tear with hypointense signal to a full-thickness tear with complete discontinuity. To the best of the authors’ knowledge, there has been no consensus or detailed descriptions regarding the definition of graft failure. In our current investigation of the association between clinical results and different graft types, the clinical outcomes (ASES, pVAS) and radiological outcomes (AHD and Hamada grade) were the best among patients with a type 1 graft status and the worst among patients with a type 5 graft status, whereas types 2, 3, and 4 did not show significant differences with type 1. Considering these results, type 5 (complete discontinuity) could be considered to indicate a failure of the SCR, both clinically and radiologically.
A previous study on graft classifications was conducted on patients in whom SCR was performed using alloderm. 30 In that report, the authors also used 5 categories to stratify the graft condition according to the presence and location of the tear as follows: intact, tear from the glenoid, midsubstance tear, tear from the tuberosity, and absent graft. However, there was no further description of the graft state in that study, which limited its capacity to explain and understand the changes that occurred due to autograft remodeling. Therefore, the classification system for SCR using alloderm may lead to some limitations in studies on SCR using autografts.
Limitations
One limitation of our study was the small number of cases with impaired integrity of the graft. The numbers of patients with each designated graft state, especially types 3, 4, and 5, were too small for detailed analyses. Future studies with a larger sample size are necessary to perform analysis with the 5 subgroups of the classification. Another limitation of our study was the relatively short follow-up duration, which hindered us from analyzing the association of the proposed classification with long-term clinical outcomes. Future studies with a larger sample size and longer follow-up durations are needed to confirm the usefulness and suitability of our proposed grading system. Lastly, as all cases were treated with SCR using autologous fascia lata, there is a lack of evidence for the generalizability of this classification system to other graft types.
Conclusion
The new SCR graft classification system introduced in this study was highly reproducible and showed clinical utility for both radiological and clinical evaluation following SCR. This system may support future studies regarding SCR with a consistent report of MRI-based outcomes.
Footnotes
Final revision submitted April 7, 2023; accepted May 3, 2023.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Asan Medical Center (No. 2022-0996).
