Abstract
Background:
Ghrelin, an amino acid hormone secreted primarily from the stomach, can regulate bone metabolism, regulate inflammation via suppressing proinflammatory cytokines, and suppress expression of matrix metalloproteinases (MMPs).
Purpose:
To measure synovial fluid levels of ghrelin in young patients with anterior cruciate ligament (ACL) tear to assess the role of ghrelin as a potential biomarker for cartilage injury.
Study Design:
Controlled laboratory study.
Methods:
This study included 120 patients who underwent ACL reconstructionbetween January 1, 2016, and May 31, 2021. We categorized 60 patients with acute cartilage injury (International Cartilage Regeneration & Joint Preservation Society grade 2 or 3) as the acute group and 60 patients with no acute cartilage injury as the nonacute group, with the healthy contralateral knee of each patient acting as the control group (n = 120). Synovial fluid samples were collected from the knees in the operating room before ACL reconstruction. We assessed the inflammatory biomarkers interleukin (IL)-6, MMP-1, MMP-9, and MMP-13, as well as serum ghrelin level and Mankin score, and results were compared between the 3 study groups with the Mann-Whitney U test.
Results:
Lower serum ghrelin levels in the synovial fluid were found in the acute group compared with the nonacute group and healthy controls (232.4 vs 434.4 vs 421.5 pg/mL, respectively; P < .001). Ghrelin level in the synovial fluid was significantly and positively correlated with IL-6 (r = 0.4223; P < .0001), MMP-13 (r = 0.3402; P < .0001), and Mankin score (r = 0.1453; P = .0244).
Conclusion:
In patients with ACL injury, ghrelin synovial fluid was significantly differently expressed in patients with cartilage injury and no cartilage injury.
Clinical Relevance:
Ghrelin synovial fluid has the potential to be a biomarker to predict acute cartilage injury in patients with ACL injury.
The anterior cruciate ligament (ACL) is crucial to stabilizing the knee joint, connecting the femur to the tibia. Around 40% of sports injury is caused by ACL injury, 22 which leads to knee instability 30 and results in poor knee-related quality of life. 25,39 Treatment for ACL injuries in athletes often involves reconstructing the ACL, including removing the torn ligament and replacing it with a tendon graft that is usually taken from another part of the knee. 7,12,25
ACL rupture leads to tibial anterior displacement. 37 A considerable proportion of patients with collision injury have bone marrow edema, 10,18 during which the articular cartilage secretes proinflammatory cytokines into the joint space. 2,28,41 These inflammatory biomarkers at the time of ACL injury, as well as the chondral impaction, contribute to the development of osteoarthritis (OA). 1 Articular cartilage is hyaline cartilage, which lacks blood supply and nerve tissue nutrition, resulting in a weak ability to repair itself, making it difficult to heal after injury. Diagnosing acute cartilage injury has been a challenging problem. In most cases, the diagnosis is according to the imaging (such as magnetic resonance imaging [MRI]) as well as knee pain symptoms, instability of the knee, and functional restrictions. 3,5 Early-stage cartilage injury is particularly difficult to diagnose, resulting in a longer time to treatment. 40 Thus, it is of interest to find a reliable parameter to detect early acute cartilage injury in patients.
Inflammatory biomarkers in the synovial fluid are specific to the joint and are also common in people who have some forms of inflammatory arthritis, such as rheumatoid arthritis and osteoarthritis. 21 Inflammatory markers of the knee joint are important in diagnosing early cartilage metabolism after ACL injury. 29 Thus, a synovial fluid biomarker profile characteristic is a possible approach to identify highly clinically relevant cartilage injury.
Ghrelin is an amino acid hormone that is secreted primarily in the stomach. 44 Widely known as the “hunger hormone,” ghrelin is the endogenous ligand of receptors for the growth hormone secretagogue. 20 In addition to the growth hormone–releasing function, ghrelin has a broad variety of other functions, 13 such as regulation of dietary intake, stimulation of insulin secretion, and regulation of glucose balance and immune system. 33 Ghrelin can regulate bone metabolism by an autocrine/paracrine mode 31 and via the direct effect on the bone cells, 6 promoting the proliferation and inhibiting apoptosis of osteoblasts. 4,8,19 Furthermore, ghrelin increases the level of chondroitin sulfate type 4, and it enhances proteoglycan synthesis by promoting hypertrophy and participating in chondrocyte metabolism. 4 It has been confirmed previously that ghrelin regulates inflammation via suppressing proinflammatory cytokines, such as interleukin (IL)-6 and IL-1β, 11 and that ghrelin suppresses expression of matrix metalloproteinases (MMPs), including MMP-1, MMP-9, and MMP-13, in human chondrocytes. 24 Thus, ghrelin may be an important diagnostic marker for cartilage injury.
Currently, there is no published literature explaining the relationship between ghrelin concentrations in synovial fluid in patients after ACL injury. The purpose of this study was to measure synovial fluid levels of ghrelin in young patients with ACL injury to assess its potential role as a diagnostic biomarker for cartilage injury. We hypothesized that ghrelin levels would be lower in patients with cartilage injury than in those with healthy knees.
Methods
Study Patients
The protocol for this study was approved by a medical ethics review committee, and all study participants provided written informed consent. A total of 120 patients scheduled for ACL reconstruction between January 1, 2016, and May 31, 2021, were recruited prospectively in this study. All patient diagnoses of ACL injury were confirmed according to clinical and MRI examinations. Our inclusion criteria were patients scheduled to undergo isolated arthroscopic ACL reconstruction. Excluded were patients <18 years, those with primary OA of any joint or severe systemic or chronic disease, those who had previous knee surgery or an intra-articular injection 3 months before surgery, and those with an insufficient amount of synovial fluid after aspiration. In this study, patients were not allowed to receive steroids or hyaluronic acid injection during the analysis.
The patients were divided into those with acute cartilage injury (acute group; n = 60) and those without acute cartilage injury (nonacute group; n = 60), with the healthy contralateral knee of each patient acting as the control group (n = 120). Acute cartilage injury was defined as International Cartilage Regeneration & Joint Preservation Society (ICRS) grade 2 or 3 at 1 month before study enrollment.
Intraoperative Procedures
Synovial Fluid Collection
Synovial fluid samples (5 mL) were collected with the help of the superolateral portal location using a local anesthetic injection or a numbing spray before knee debridement and ACL reconstruction. We collected the maximum amount of synovial fluid that egressed for evaluation. All samples were stored on ice during transport. After synovial fluid samples were collected, the samples were centrifuged immediately to remove cells and joint debris and then stored at -80°C until analysis.
Degree of Cartilage Injury
Cartilage injury was assessed intraoperatively by 2 reviewers (D.J. and R.L.) using the ICRS score.
Analysis of Cartilage Injury Markers
Blood samples were taken from all patients and the serum was frozen at a temperature of -80°C before surgery. Radioimmunoassay kit was used to evaluate the serum and synovial fluid ghrelin levels. We chose inflammatory biomarkers that are commonly found in ACL injury and cartilage degeneration (IL-6, MMP-1, MMP-9, MMP-13) and measured their levels using enzyme-linked immunosorbent assay. Serum markers, including albumin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT), were also analyzed. The precision of the intra-assay was ≤5% and interassay is ≤4%. All parameters were measured twice by 2 reviewers (D.J. and R.L.). The coefficient of variation between the reviewers ranged from 2.8% to 5.4%.
We assessed the histopathological gradation of the severity using the grading system proposed by Mankin et al. 27 This is a reliable test of histological scoring of cartilage changes with evaluations of structure, matrix staining, cellular abnormalities, as well as tidemark integrity. The total score ranges from 0 to 14, with 0 meaning normal and 14 meaning most severe injury. 43
Statistical Analysis
Statistical analyses were performed with SPSS 23 (IBM) and GraphPad Prism (Version 9.0.1). Baseline characteristics were compared for all continuous variables with mean values and categorical variables with numbers. We compared ghrelin levels between the acute group, the nonacute group, and healthy controls using Mann-Whitney U test with P < .05 considered statistically significant. The association of ghrelin levels in synovial fluid and serum with inflammatory biomarkers and Mankin score were assessed with the Spearman correlation coefficient (r).
Results
Baseline Characteristics of the Study Groups
The 120 study patients were between 21 and 38 years of age, and the interval between injury and surgery was 4 to 6 weeks. Baseline patient characteristics; albumin, AST, and ALT levels; and Mankin score according to the study groups are summarized in Table 1.
Baseline Characteristics of the Study Patients a

a Data are reported as mean ± SD unless otherwise indicated. ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; IU, international units.
Ghrelin Levels and Inflammatory Biomarkers
Ghrelin levels and inflammatory biomarkers in the acute, nonacute, and control groups are summarized in Table 2. There was no statistical difference in the serum ghrelin level between the acute and nonacute groups (263.9 vs 328.8). Patients in the acute group had higher levels of IL-6 (12.8 ± 3.4 vs 5.0 ± 3.4 pg/mL; P = .012) and MMP-13 (48.5 vs 13.6 pg/mL; P = .015) than patients in the nonacute group. Patients in the acute group also had lower synovial fluid ghrelin level compared with patients in the nonacute group as well as with their own healthy knees (232.4 vs 434.4 vs 421.5 pg/mL; P < .001) (Figure 1). Synovial fluid ghrelin levels in the knees with and without acute cartilage injury compared with healthy control knees. Synovial fluid ghrelin level was significantly and positively correlated with IL-6 (r = 0.4223; P < .0001), MMP-13 (r = 0.3402; P < .0001), and Mankin score (r = 0.1453; P = .0244) (Figure 2).
Baseline Ghrelin Level and Synovial Fluid Inflammatory Biomarkers According to Study Groups a

a Values presented as mean and standard deviation if not otherwise stated. Boldface P values indicate statistically significant differences among the 3 groups (P < .05). IL, interleukin; MMP, matrix metalloproteinase.
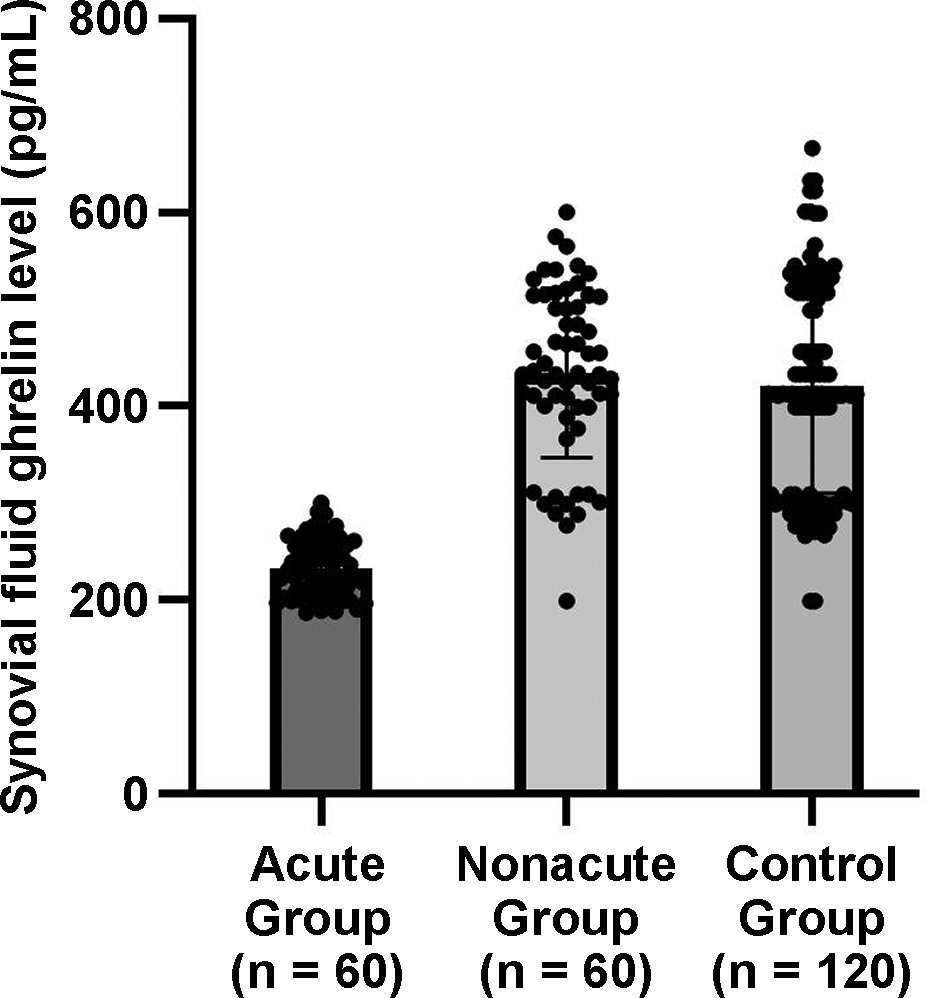
Synovial fluid ghrelin levels in patients ACL reconstruction with or without acute cartilage injury compared with the levels in their own healthy knees (N = 240).
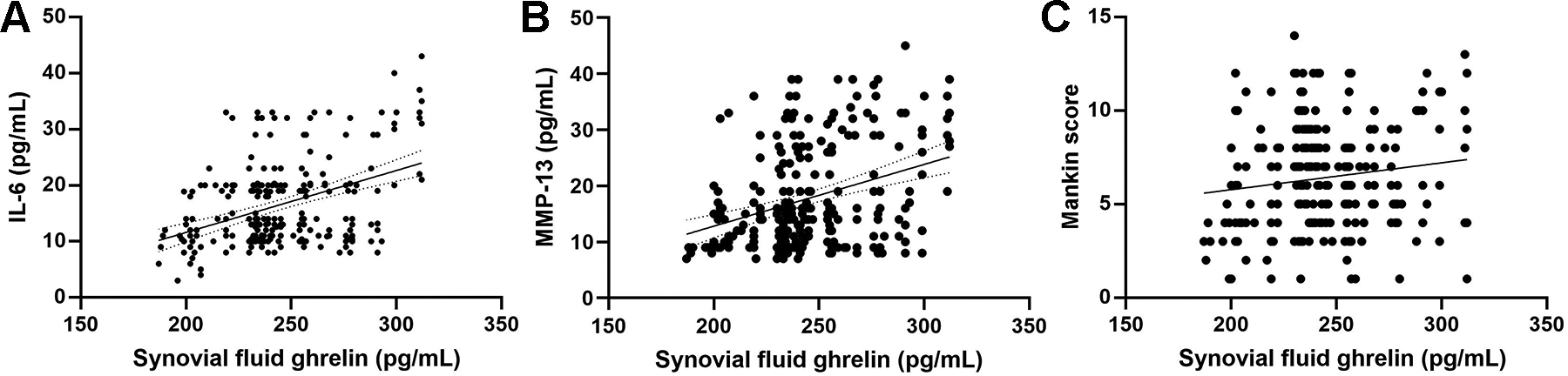
Correlation of synovial fluid ghrelin with (A) IL-6, (B) MMP-13, and (C) Mankin score. IL, interleukin; MMP, matrix metalloproteinase.
Discussion
Patients in the acute cartilage injury group had higher levels of IL-6 (12.8 ± 3.4 vs 5.0 ± 3.4 pg/mL; P = .012), and MMP-13 (48.5 vs 13.6 pg/mL; P = .015) than patients in no acute cartilage injury group. Patients with acute cartilage injury had lower synovial fluid ghrelin level compared with patients with no acute cartilage injury and with their own healthy knees (232.4 pg/mL versus 434.4 pg/mL versus 421.5 pg/mL; P < .001). Ghrelin level in the synovial fluid was correlated with IL-6 (r = 0.4223; P < .0001), MMP-13 (r = 0.3402; P < .0001), and Mankin score (r = 0.1453; P = .0244).
Ghrelin is brain-gut peptide secreted largely in the stomach 9 and functions in growth plate chondrocyte regulation. 42 A study by Wu et al 45 indicated an increase of ghrelin in OA patients and suggested its association with knee symptoms. This study was in accordance with a previous study suggesting that synovial fluid ghrelin is expressed differently between knees with cartilage injury or with no cartilage injury and contralateral controls. 42 Thus, synovial fluid, which is accessible during ACL injury, was found to be a reliable intra-articular parameter. 36 The results of the current study indicated that synovial fluid ghrelin was significantly differently expressed in patients with cartilage injury and no cartilage injury. The most meaningful thing is that no differences were found from serum samples, which illustrates that synovial fluid ghrelin is a better biomarker in predicting cartilage injury, with higher sensitivity. The reason for its sensitivity might be that synovial fluid ghrelin is derived directly from the knee.
Ghrelin is important in the regulation of cartilage-specific genes on chondrocytes. 23,34 This analysis showed that ghrelin level in the synovial fluid was correlated with IL-6 as well as MMP-13. Furthermore, IL-6 has been proved to play a vital role in the pathogenesis of bone diseases. 14,16 High levels of IL-6 were associated with greater prevalence of osteophytes in the whole tibiofemoral compartment. 38 Serum levels of IL-6 are associated with knee cartilage loss in the elderly population, indicating that low level inflammation plays a role in the pathogenesis of knee OA. 38 In accordance with previous publications, 16,17 the level of IL-6 was much higher in patients with cartilage injury than in patients with no cartilage injury.
MMP-13 is an important enzyme that targets cartilage for degradation. 15 It has the function of degrading proteoglycan, type 4 collagen, and type 9 collagen. MMP-13 can regulate the activation of other MMPs. Synovial fibroblasts are among a variety of cells that produce MMP-13. 32,35 The level of MMP-13 in the synovial fluid of patients with OA was correlated with disease severity, suggesting that MMP-13 is indicating cartilage destruction. 26 This study indicated that MMP-13 was higher in the ACL reconstruction with cartilage injury group than in the no cartilage injury group. Synovial fluid ghrelin was correlated with MMP-13. This study also suggested that ghrelin level in the synovial fluid was correlated with Mankin score, proving its prognostic function.
Limitations
As this was an observational study, the sample size was relatively small. Thus, further large-scale clinical studies in the field are needed. A biomarker has the potential to be used widely only when it can be reversed using interventions; for ghrelin synovial fluid, this seems to be the situation. When patients are diagnosed with cartilage injury at an early stage, more effective interventions are needed to help patients with the recovery process.
Conclusion
Based on study findings in patients with ACL injury, ghrelin synovial fluid has the potential to be a biomarker to predict acute cartilage injury and is correlated with levels of IL-6, MMP-13, and Mankin score. More clinical trials are needed to further confirm this.
Footnotes
Acknowledgement
The authors thank all patients and all research team members for their contribution to this study.
Final revision submitted January 28, 2023; accepted February 22, 2023.
The authors declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University (reference No. C2016150).
