Abstract
Background:
Clinically significant outcome (CSO) thresholds for the Patient-Reported Outcome Measurement Information System–Upper Extremity (PROMIS–UE) score have been previously defined after arthroscopic rotator cuff repair (RCR). However, the time required to achieve CSOs for the PROMIS–UE score is unknown.
Purpose:
To (1) determine the time required to achieve the minimal clinically important difference (MCID), substantial clinical benefit (SCB), and Patient Acceptable Symptom State (PASS) score thresholds after RCR for the PROMIS–UE questionnaire and (2) identify patient factors associated with earlier or delayed achievement of these clinical benchmarks.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
A prospectively maintained institutional database was retrospectively reviewed for consecutive patients who underwent RCR between January 2018 and January 2019. Patients were included if they completed the PROMIS–UE questionnaire both preoperatively and at standardized postoperative time intervals: 5 to 7 months (6-month time point), 11 to 13 months (1-year time point), and ≥23 months (2-year time point). Kaplan-Meier survival curves with interval censoring were used to define the cumulative percentage of patients who achieved the MCID, SCB, and PASS. Patient variables associated with earlier or delayed achievement of the MCID, SCB, and PASS were determined using Weibull parametric survival regression analysis.
Results:
Included were 105 patients undergoing RCR (age, 57.3 ± 10.3 years; body mass index, 31.5 ± 6.1 kg/m2). By 2-year follow-up, the cumulative percentage of patients achieving the MCID, SCB, and PASS was 86.7%, 76.2%, and 74.3%, respectively. The mean time required to reach the MCID, SCB, and PASS score thresholds was 9.5 ± 3.8, 10.3 ± 4.4, and 9.8 ± 4.6 months, respectively. Factors associated with delayed achievement of CSOs included greater baseline PROMIS–UE score (MCID and SCB) and workers’ compensation insurance (PASS). Greater baseline PROMIS–UE scores were associated with earlier achievement of the PASS.
Conclusion:
Most patients achieved CSOs for the PROMIS–UE within 12 months of RCR. Patient-specific factors found to be associated with earlier or delayed achievement of CSOs can be used to inform patient discussions on the expected timeline for recovery after RCR.
Keywords
Rotator cuff tears are common injuries in the United States, affecting around 40% of those who are 60 years, 25 with approximately 250,000 repairs performed annually. 5 As clinicians continue to optimize repair techniques, rotator cuff (RC) repairs (RCRs) have been shown to improve clinical outcomes in appropriately indicated patients, with low complication and revision rates. 10,33,36,38 Patient-reported outcome (PRO) measures (PROMs) have been validated and utilized to trace patient progression toward recovery after upper extremity injuries. 42 As health systems begin transitioning to value-based models of care, PROMs represent a promising objective scale and benchmark for assessing the clinical benefit of RCR. 18 However, there remains a substantial amount of heterogeneity in which outcomes are reported after RCR. 28 Current PROMs place a significant degree of question burden and respondent fatigue, leading to poor compliance rates and long-term follow-up. 40 Furthermore, these tools generate financial and administrative burdens as well as performance constraints because of the inherent subjectivity in the context of shoulder arthroscopy and RCR. 2,15,42
To address the shortcomings of PROMs, the National Institutes of Health created the Patient-Reported Outcomes Measurement Information System (PROMIS)—a patient-centered outcome assessment of 4 to 12 questions that integrates item-response theory with computer adaptive testing. In doing so, the PROMIS questionnaire comprises a single, standardized metric with favorable psychometric performance relative to legacy PROMs. 42 Specifically, the PROMIS–Upper Extremity (PROMIS–UE) questionnaire has shown unique advantages as compared with both legacy and other PROMIS instruments, including early responsiveness, faster time to completion, fewer floor and ceiling effects, decrease in test burden, and increase in compliance rates leading to a higher power in the measure, 3,27,43 particularly in the context of rotator cuff pathologies. 1,34,42
Of late, research has transitioned from reporting statistically significant improvements after surgery to determining the changes in PROM scores that correspond to clinically significant outcomes (CSOs). Various measures have been proposed to define CSOs after RCR, including the minimal clinically important difference (MCID), substantial clinical benefit (SCB), and Patient Acceptable Symptom State (PASS). 4,7 The MCID represents the minimal improvement in the PROM score that corresponds to clinically significant improvement, whereas the SCB represents the PROM score change associated with substantial clinical improvement. 17 In contrast, the PASS is defined as an absolute postoperative PROM score associated with overall satisfaction. Yet, the time required to achieve these measures after a surgical intervention has received less attention. Determining the time required for a patient to achieve CSOs provides several potential benefits, including calibrating patient and physician expectations for outcome improvement, informing physician decision making, and establishing follow-up time frames through the course of treatment. Recently, Manderle et al 29 evaluated the time necessary to achieve MCID, SCB, and PASS after RCRs for commonly utilized legacy PROM measures, such as the American Shoulder and Elbow Surgeons (ASES) score.
To our knowledge, no prior studies have investigated the time-dependent achievement of CSOs for the PROMIS–UE score after RCR. The purpose of the present study was to (1) determine the time required to achieve the MCID, SCB, and PASS after RCR for the PROMIS–UE questionnaire and (2) identify patient factors associated with earlier or delayed achievement of these clinical benchmarks. The authors hypothesized that the majority of patients would reach the MCID, SCB, and PASS thresholds for the PROMIS questionnaire between 6 and 12 months after surgery.
Methods
Study Design and Methods
This investigation received institutional review board approval before initiation. The study population and patient-selection process implemented in the present study are described in detail by Haunschild et al. 19 In brief, a prospectively collected database of patients from a single institution between January 2018 and January 2019 was retrospectively queried for patients who underwent primary RCR. During the study period, a total of 180 consecutive patients undergoing RCR who completed the baseline PROMIS–UE questionnaire were identified. Completed 12-month postoperative PROMIS–UE scores were used as the criteria to identify the study cohort. These data were previously collected and described in the literature. 19 In the present study, PROMIS–UE scores were subsequently administered to the same cohort at 24 months postoperatively. In addition, the electronic medical record was retrospectively reviewed for completed PROMIS–UE scores at 6 months postoperatively. Four surgeons (A.B.Y., B.F., B.J.C., and N.N.V.) in the sports medicine service at the senior author’s institution (N.N.V.)contributed patients to this registry. Trained research personnel administered baseline function and pain assessments before surgery. This information was stored in electronic data collection services (Outcome Based Electronic Research Database; Universal Research Solutions; and Patient IQ), which were utilized to collect postoperative PROMs. The PROMIS–UE score was collected at standardized, postoperative time points: 5 to 7 months (6-month time point), 11 to 13 months (1-year time point), and ≥23 months (2-year time point).
Patient Selection
Patients were included if they had undergone primary arthroscopic RCR and completed the PROMIS–UE questionnaire form both preoperatively and at a minimum of 1 postoperative time point. The exclusion criteria consisted of patients undergoing superior capsular reconstruction, rotator cuff debridement, patch augmentation repairs, revision repairs, and biologic augmentation (including platelet-rich plasma and bone marrow aspirate concentrate) to the index procedure. The medical records of identified patients were subsequently reviewed to collect pertinent patient variables, including demographic (age, sex, body mass index [BMI], smoking status, and workers’ compensation status), clinical (preoperative range of motion, duration of symptoms, and preoperative imaging), and operative details (laterality, tear size, retraction, type of repair performed, and concomitant procedures). The duration of symptoms (ie, the interval between injury and surgery) was categorized into ≤3 months and >3 months based on previous literature.
41
Tear size was classified using the methods detailed by DeOrio and Cofield
9
: small tears were
Outcome Measures
The primary outcome measure was the PROMIS–UE Version 1.2 (Appendix Table A1), 21 which was collected at baseline and each postoperative time interval. This score is measured using a T-score metric, where the mean score for the sample population is 50 ± 10 and can take any value between 20 and 80 points, with a higher score signifying a higher level of function. At 1 month before each postoperative time point (6 months, 1 year, and 2 years), patients received an email requesting the completion of the PROMIS questionnaire. A reminder email was sent every 5 days until 1 month after the 6-month and 1-year time points or until the form was submitted, allowing 2 months for completion. Patients completed the 2-year follow-up survey at a minimum of 23 months postoperatively. The PROMIS–UE score at each specific time point was then used to determine CSO achievement.
Statistical Analysis
The methodology used for statistical analysis has been described in detail in previously published literature. 11,29 Briefly, the cohort-specific MCID, SCB, and PASS threshold values were obtained from prior literature on primary RCR 19 (MCID, 4.87; SCB, 7.95; PASS, 39). The cumulative achievement of the MCID, SCB, and PASS at each postoperative period (5-7 months, 11-13 months, and ≥23 months) was determined via Kaplan-Meier survival curve analysis and interval censoring. Patients with incomplete PROMIS–UE data were included in the analysis and were designated as nonachievers of the MCID/SCB/PASS at that specific postoperative period. 11 Thus, data points for the PROMIS–UE were available for inclusion in the multivariate model for every patient at each postoperative time point.
The time required to reach the MCID, SCB, and PASS threshold values for the study population was calculated by averaging the earliest time point that each patient met the PROMIS–UE score necessary for achievement. For example, patients reaching the appropriate score thresholds at 6 or 12 months postoperatively were considered achievers of the MCID by 24 months postoperatively, regardless of whether they had 24-month PROMIS–UE scores available. 11 Weibull parametric survival regression analysis was used to determine the influence of patient factors on the time to achieve the MCID, SCB, and PASS threshold values. 11,32 Variables included in the regression analysis were chosen based on previous literature: 19 age; sex (female vs male); BMI; interval between injury and surgery (≤3 vs >3 months); current smoking status; insurance status (workers’ compensation vs other); preoperative range of motion (external rotation, forward flexion); concomitant procedures (biceps tenodesis, distal clavicle excision); preoperative PROMIS–UE score; rotator cuff tear size; and Goutallier stage. Statistical analysis was performed with RStudio software Version 4.0.4 (R Foundation for Statistical Computing).
Results
Demographic Characteristics of the Study Population
Of the 105 patients with completed preoperative and 12-month postoperative PROMIS–UE scores, 70 (66.7%) and 63 (60%) patients completed the PROMIS–UE questionnaire at 6 (range, 5-7 months) and 24 (range, 23-33 months) months after surgery, respectively. The mean age and BMI of the study population were 57.3 ± 10.3 years and 31.5 ± 6.1 kg/m2, respectively, and 30.5% were women. The most common rotator cuff tear size classification was medium (n = 46 [44.7%]). The most common degree of rotator cuff muscle fatty infiltration was Goutallier stage 1 (n = 49 [47.6%]). The demographic characteristics of the study population is fully described in Table 1.
Baseline Characteristics of the Study Population a

a BMI, body mass index; PROMIS–UE, Patient-Reported Outcome Measurement Information System–Upper Extremity; RC, rotator cuff.
b Baseline radiographs were available in 103 (98.1%) of the 105 patients in the study cohort.
Time to Achievement and PROMIS–UE Scores
The mean baseline, 6-month, 12-month, and 24-month postoperative PROMIS–UE scores for the entire study cohort were 31.6 ± 7.6, 40.9 ± 9.8, 44.4 ± 9.8, and 45.4 ± 11.6, respectively. By 2 years postoperatively, a total of 91 (86.7%) patients achieved the MCID, 80 (76.2%) achieved SCB, and 78 (74.3%) achieved PASS thresholds for the PROMIS–UE score (Table 2). However, the majority achieved these thresholds at or before 12 months postoperatively, with only a small minority achieving such thresholds between 12 and 24 months postoperatively. For those who achieved these goals, the mean time required to achieve the MCID, SCB, and PASS for the PROMIS–UE score was 9.5 ± 3.8, 10.3 ± 4.4, and 9.8 ± 4.6 months, respectively (Figure 1).
Cumulative Percentage of CSO Achievement for PROMIS–UE a

a CSO, clinically significant outcome; MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State; PROMIS–UE, Patient-Reported Outcome Measurement Information System–Upper Extremity; SCB, substantial clinical benefit.
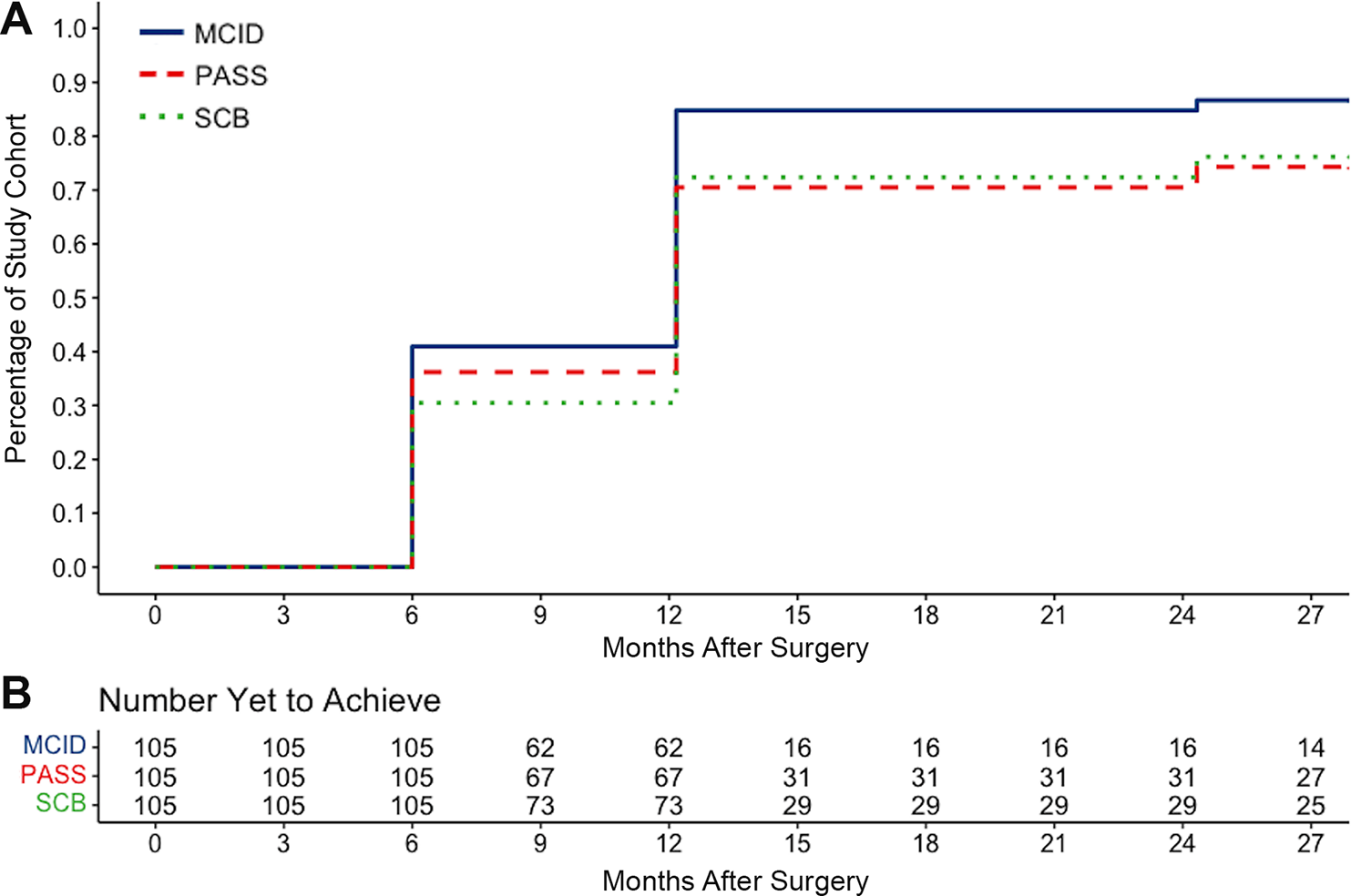
Proportion of study patients who achieved the MCID, SCB, and PASS thresholds for the PROMIS–UE score by postoperative time point. The chart displays the number of patients yet to achieve clinically significant outcomes at each time point. MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State; PROMIS–UE, Patient-Reported Outcome Measurement Information System–Upper Extremity; SCB, substantial clinical benefit.
Factors Associated With Time to Achieve MCID, SCB, and PASS
Regarding the MCID, greater preoperative PROMIS–UE scores (hazard ratio [HR], 0.938 [95% CI, 0.909-0.968]; P < .001) were significantly associated with delayed achievement. No factors were significantly associated with earlier achievement of the MCID (Table 3). Regarding the SCB, greater preoperative PROMIS–UE scores (HR, 0.909 [95% CI, 0.875-0.944]; P < .001) were significantly associated with delayed achievement. No factors were significantly associated with earlier achievement of the SCB (Table 4). Regarding the PASS, workers’ compensation insurance status (HR, 0.405 [95% CI, 0.186-0.881]; P = .022) was significantly associated with delayed achievement. Earlier achievement of the PASS was significantly associated with greater preoperative PROMIS–UE scores (HR, 1.048 [95% CI, 1.016 -1.082]; P = .003) (Table 5).
Patient Factors Associated With Longer or Shorter Time to Achieve MCID for PROMIS–UE a

a The bold P value indicates statistical significance (P < .05). BMI, body mass index; HR, hazard ratio; MCID, minimal clinically important difference; PROMIS–UE, Patient-Reported Outcomes Measurement Information System–Upper Extremity.
b The reference category for comparison was small rotator cuff tear size.
c The reference category for comparison was Goutallier stage 0/1. There were no patients with Goutallier stage 4.
Patient Factors Associated With Longer or Shorter Time to Achieve SCB for PROMIS–UE a

a The bold P value indicates statistical significance (P < .05). BMI, body mass index; HR, hazard ratio; PROMIS–UE, Patient-Reported Outcomes Measurement Information System–Upper Extremity; SCB, substantial clinical benefit.
b The reference category for comparison was small rotator cuff tear size.
c The reference category for comparison was Goutallier stage 0/1. No patients were Goutallier stage 4.
Patient Factors Associated With Longer or Shorter Time to Achieve PASS for PROMIS–UE a

a Bold P values indicate statistical significance (P < .05). BMI, body mass index; HR, hazard ratio; PASS, Patient Acceptable Symptom State; PROMIS–UE, Patient-Reported Outcomes Measurement Information System–Upper Extremity.
b The reference category for comparison was small rotator cuff tear size.
c The reference category for comparison was Goutallier stage 0/1. No patients were Goutallier stage 4.
Discussion
The present study defined the timeline of the MCID, SCB, and PASS achievement for the PROMIS–UE score after RCR and identified several important patient factors associated with delayed and earlier achievement of these CSO thresholds. The majority of the study cohort achieved the MCID, SCB, and PASS thresholds for the PROMIS–UE score within 12 months after RCR. Patients who did not achieve these thresholds by 12 months postoperatively were unlikely to do so at the 24-month follow-up. The mean time required to achieve the MCID was 9.5 ± 3.8 months, 10.3 ± 4.4 months for the SCB and 9.8 ± 4.6 months for the PASS. Patient factors significantly associated with earlier achievement of CSOs included greater preoperative PROMIS–UE scores (for PASS). Patient factors significantly associated with delayed achievement of CSOs included greater preoperative PROMIS–UE scores (for MCID and SCB) and workers’ compensation insurance status (for PASS). More severe RC disease on preoperative imaging (eg, RC tear size, Goutallier stage) was not significantly associated with delayed achievement of CSOs. The findings of the present study may be beneficial when educating and counseling patients on the expected timeline of recovery after arthroscopic RCR.
The PROMIS–UE form has been extensively evaluated for its psychometric properties and efficacy against legacy PROs. 39,42 Backed by the National Institutes of Health, it is currently used in numerous point-of-care practices across the United States, given its dynamically efficient short form and computer adaptive testing format. 27 Nonetheless, as patients become increasingly interested in the results of the surveys completed during their care, future iterations of the PROMIS–UE should consider providing results in plain language for patients to easily interpret, including graphical representations and contextualized statistics. In addition, it is of importance to more accurately characterize the expected timeline for improvement after RCR using the PROMIS–UE tool, as this may influence patient expectations and the allocation of limited resources postoperatively.
While previous studies have established CSO thresholds at discrete time points after RCR, 7,16,19,23 few have investigated the time-dependent achievement of CSOs in the postoperative period. In a cohort of 203 primary RCR patients, Manderle et al 29 demonstrated that the mean time required for patients to achieve the MCID for the ASES, Single Assessment Numeric Evaluation (SANE), and Constant score was 5 to 7 months after surgery, whereas the SCB and PASS were achieved between 7 and 9 months postoperatively. In contrast, the present study found a slightly longer mean time of 9 to 11 months for the achievement of the MCID, SCB, and PASS thresholds for the PROMIS–UE score. Although strong correlations have been found between the PROMIS–UE and ASES scores in patients undergoing RCR, 1,13,24 recent literature demonstrated that the PROMIS–UE underperforms psychometrically relative to the ASES. 24 Lavoie-Gagne et al 24 performed psychometric validation of the PROMIS–UE computer adaptive test in 107 patients undergoing arthroscopic RCR. The authors found that the PROMIS–UE had a strong correlation with the ASES and minimal floor and ceiling effects; however, most of the individual PROMIS–UE question item responses demonstrated poor reliability corresponding to shoulder function. In the present study, the longer mean time-to-achievement and lower overall achievement rates of the MCID and SCB compared with the findings of Manderle et al support this finding and may indicate that the PROMIS–UE is less sensitive relative to the ASES for measuring improvement after RCR. Caution must be exercised when choosing PROs for reporting research results, as intrinsic differences in these metrics may create the appearance of more rapid or delayed recovery in the same patient cohort. As such, advocating for the continued standardization of outcome reporting after RCR is crucial.
The variable impact of preoperative outcome scores on the achievement of different CSOs has been well-established in the literature. Haunschild et al, 19 in a cohort of 105 primary RCR patients observed for a minimum of 12 months, demonstrated that lower baseline PROMIS–UE scores were associated with the achievement of the MCID and SCB. In contrast, higher baseline PROMIS–UE scores were associated with the achievement of the PASS. Analogous trends have been demonstrated in several studies assessing the time required to achieve CSOs after shoulder surgery. 11,29 In 203 primary RCR patients, Manderle et al 29 found that greater preoperative ASES, SANE, and Constant-Murley scores were associated with delayed achievement of the MCID. This is consistent with the findings of the present study, where greater preoperative PROMIS–UE scores were associated with delayed achievement of the MCID and SCB, but earlier achievement of the PASS. The opposite impact that baseline scores have on achievement of the MCID/SCB compared with the PASS is due to differences in how these metrics are calculated. 19,37 The MCID and SCB quantify the clinically meaningful improvement in functional status from baseline, whereas the PASS represents an absolute PRO score threshold corresponding to overall satisfaction. 31 In general, patients with already high levels of functioning at baseline are less likely to experience the functional improvement required to surpass the MCID and SCB, and vice versa. On the contrary, for patients with higher baseline scores, less improvement is necessary to achieve a satisfactory outcome (ie, the PASS score threshold) after surgery. These findings may help inform patient selection for RCR. For example, if a patient’s baseline PROMIS score is relatively high, then a period of conservative management and watchful waiting may be considered until a lower baseline PROMIS score is achieved such that clinically significant improvement is perceived by the patient more rapidly in the postoperative period.
Workers’ compensation insurance status was found to be an independent predictor of delayed achievement of the PASS. The negative association between workers’ compensation status and outcomes after shoulder surgery has been established in previous literature. 8,14,20,30 When controlling for covariates such as age, education level, work demands, and preoperative expectations, workers’ compensation status remains an independent predictor of poor outcomes after RCR. 20 Several potential explanations for this finding include higher rates of noncompliance with postoperative rehabilitation compared with nonworkers’ compensation patients 6 and decreased satisfaction from the inability to return to full-duty heavy work secondary to functional limitations or physician recommendation. In addition, the present study demonstrated that more severe RC disease documented on preoperative imaging (eg, RC tear size, Goutallier stage) was not significantly associated with delayed achievement of CSOs for the PROMIS–UE questionnaire. Previous literature has reported that preoperative RC tear size and fatty infiltration are associated with tendon integrity and risk of retear after RCR. 12,22,26 Although somewhat counterintuitive, research has demonstrated a disconnect between imaging findings of RC disease and patients’ subjective experience of shoulder pain and function, 35 which may explain the present findings. Alternatively, it is possible that the PROMIS–UE questionnaire is not sensitive enough for the assessment of shoulder-specific recovery after RCR. 24 Nonetheless, our findings suggest that patients undergoing RCR may expect comparable timelines for achievement of clinical improvement regardless of the preoperative RC disease state.
Limitations
Several limitations warrant consideration when interpreting the results of the present study. First, the absence of complete PROMIS–UE data at each postoperative time point may have resulted in a nonnegligible degree of measurement bias. It is possible that the responses at 6 and 24 months postoperatively were biased toward those patients choosing to respond. However, the compliance rates achieved (6 months, 66.7%; 24 months, 60%) were in accordance with previous literature 19 and decreased the impact of this potential limitation. Second, the PROMIS–UE data were collected at fixed, 6-month intervals after surgery. As such, the calculated mean time to achieve the MCID, SCB, and PASS thresholds in this study reflects the postoperative time points that data were collected and may be limited in this regard. It is possible that obtaining PRO data at earlier time points (eg, at 6 and 12 weeks postoperatively) would provide a more accurate description of the time-dependent nature of outcome achievement. Collecting more granular data in the first 6 months of surgery can be challenging and increases the risk of questionnaire fatigue and poor PRO compliance. Third, outcome data were contributed by 4 surgeons at the authors’ institution, each of which may have slight variations with respect to surgical technique and postoperative rehabilitation protocols. Last, several concomitant procedures (ie, distal clavicle excision, biceps tenodesis) commonly performed at the time of primary RCR at our institution were included, which may have influenced outcome achievement. However, these additional procedures were controlled for when performing the multivariable regression analysis. It is possible that the results of the present study do not generalize to patients undergoing isolated RCR or different concomitant procedures.
Conclusion
Most patients achieved CSOs for the PROMIS–UE within 12 months of RCR. Patient-specific factors found to be associated with earlier or delayed achievement of CSOs can be used to inform patient discussions on the expected timeline for recovery after RCR.
Footnotes
Final revision submitted November 28, 2022; accepted January 1, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: M.C.F. has received grant support from Acumed and Arthrex; education payments from Medwest, Smith & Nephew, and Zimmer; consulting fees from Encore Medical; and hospitality payments from Stryker. G.E.G. has received consulting fees from AzurMeds, Bioventus, DePuy, Encore Medical, Stryker, and Wright Medical; nonconsulting fees from Wright Medical; royalties from Encore Medical, Stryker, and Wright Medical; honoraria from Wright Medical; and hospitality payments from Arthrex and Smith & Nephew. A.B.Y. has received education payments from Medwest, consulting fees from JRF Ortho and Olympus America; nonconsulting fees from Arthrex; honoraria from JRF Ortho; and hospitality payments from Stryker. B.F. has received education payments from Medwest and consulting fees from Stryker. B.J.C. has received education payments from Endo Pharmaceuticals; consulting fees from Acumed, Aesculap Biologics, Anika Therapeutics, Bioventus, Endo Pharmaceuticals, Flexion Therapeutics, Geistlich Pharma North America, Ossic, Ossio, Pacira Pharmaceuticals, Smith & Nephew, and Vericel; nonconsulting fees from Aesculap Biologics, Arthrex, LifeNet Health, Pacira Pharmaceuticals, and Terumo BCT; royalties from Arthrex; honoraria from Vericel; and hospitality payments from GE Healthcare. N.N.V. has received education payments from Medwest; consulting fees from Arthrex, Medacta, Smith & Nephew, and Stryker; nonconsulting fees from Arthrex and Smith & Nephew; royalties from Arthrex, Smith & Nephew, and Graymont Professional Products; and hospitality payments from Relievant Medsystems. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Rush University Medical Center (ref No. 18032803).
