Abstract
Background:
Anterior cruciate ligament reconstruction (ACLR) using the complete tibial tunnel technique and adjustable-loop cortical suspensory fixation is known to leave a “dead space” that holds the loop device in the tibial tunnel. The consequence of the dead space and its effect on graft healing are still uncertain.
Purpose:
To investigate morphological changes in the tibial tunnel and their effect on graft healing, and to identify factors affecting bone healing in the tibial loop tunnel after ACLR with a quadrupled semitendinosus tendon autograft using adjustable suspensory fixation.
Study Design:
Case series; Level of evidence, 4.
Methods:
Included were 48 patients (34 male, 14 female; mean age, 25.2 ± 5.6 years) who underwent ACLR with a quadrupled semitendinosus tendon autograft using adjustable suspensory fixation. To evaluate tibial tunnel morphology, computed tomography was performed at 1 day and 6 months postoperatively. At 1 year postoperatively, graft healing was assessed on magnetic resonance imaging using the graft signal-to-noise quotient (SNQ). Multivariate regression and correlation analyses were performed to determine any associations between volumetric changes in bone healing and operative variables.
Results:
At 6 months after ACLR, a mean of 63.2% of the tibial loop tunnel was filled by bone. Multivariate regression analysis showed that remnant preservation was significantly associated with the loop tunnel filling rate (P < .001). At 1 year after ACLR, the tibial loop tunnel was almost completely closed (98.5%). There were no correlations between loop tunnel volume and graft integration or graft SNQ. A significant but weak correlation was found between graft tunnel volume and intratunnel graft SNQ (P = .10) as well as integration grade in the tibial tunnel (P = .30).
Conclusion:
Excellent bone filling in the tibial loop tunnel was seen at 1 year after ACLR. Remnant preservation was significantly associated with the loop tunnel filling rate. A weak correlation was found between graft tunnel volume and intratunnel graft SNQ as well as integration grade in the tibial tunnel.
There is ongoing debate about how best to reconstruct the anterior cruciate ligament (ACL) to restore knee kinematics, including which is the best fixation method. 19 Despite favorable outcomes after interference screw fixation, there are concerns related to graft biology, such as graft damage during screw insertion, a small tendon-to-bone contact area for graft integration, the presence of cyst formation around the screw, or tunnel widening. 17,24 These have led to the widespread clinical use of adjustable-loop cortical suspensory fixation in both the femur and tibia. 5,22 It allows using a single hamstring tendon harvest that produces a larger diameter graft and provides larger contact zones that favor integration. 4 However, some technical details, especially regarding tibial tunnel management, remain controversial. 8
The all-inside method is a less invasive bone-preserving technique with the potential for more rapid recovery. 2 The possible disadvantages of this technique are requiring specific surgical instruments and more extensive excision of the ACL remnant, which have the potential benefits of graft healing. 7 On the other hand, several studies have shown good clinical and radiological outcomes when the complete tibial tunnel technique with adjustable-loop cortical suspensory fixation is used. 4,22 This technique creates a “dead space” that holds the loop device in the tibial tunnel. However, the consequence of the dead space and its effect on graft healing are still uncertain. A greater awareness of bone healing in the tibial loop tunnel could reduce concerns about bone loss. Furthermore, it is possible that bone healing would result in improved graft healing via biological interactions.
The purposes of this study were to evaluate morphological changes in the tibial tunnel and to identify factors affecting bone healing in the tibial loop tunnel after ACL reconstruction (ACLR) with a 4-strand semitendinosus tendon autograft using adjustable suspensory fixation. We hypothesized that (1) acceptable bone healing would occur in the tibial loop tunnel; (2) factors such as age, smoking, and surgical parameters would affect bone healing; and (3) more complete bone healing in the tibial loop tunnel would improve graft integration and maturation.
Methods
After receiving ethics committee approval for the study protocol, we retrospectively evaluated the records of 186 patients who underwent single-bundle ACLR with a quadrupled semitendinosus tendon autograft in our clinic between October 2018 and June 2021. The study inclusion criteria were the following: (1) primary single-bundle ACLR using the complete tibial tunnel technique with adjustable-loop cortical suspensory fixation, (2) patients who underwent computed tomography (CT) at 1 day and 6 months postoperatively as well as magnetic resonance imaging (MRI) at 1 year postoperatively, and (3) minimum 12-month follow-up. Patients with multiligament injuries, inflammatory arthritis, cartilage defects of Outerbridge grade 2 to 4, Beighton score >4 (these patients often exhibit hyperextension that can affect graft healing because of impingement and increase tension on the ACL graft), and reruptures after surgery were excluded from the study. Adhering to these criteria, 48 patients were included.
Surgical Technique and Rehabilitation
All patients underwent anatomic single-bundle ACLR using adjustable suspensory fixation with a quadrupled semitendinosus tendon autograft. Before reconstruction, associated meniscal and cartilage injuries were treated. Remnant ACL tissue was classified in all patients according to Crain et al 6 : scarring to the posterior cruciate ligament (type 1), scarring to the roof of the intercondylar notch (type 2), scarring to the lateral femoral condyle (type 3), or no identifiable tissue remaining (type 4). In Crain type 4 knees, remnant tissue was completely resected. In other types, all residual, continuous, but biomechanically insufficient ACL tissue was preserved.
The harvested semitendinosus tendon (24-28 cm in length) was quadrupled with an adjustable-loop cortical suspensory fixation device (Lift Loop External; Orthomed) on the tibial side and a fixed-loop system (Femobutton; Orthomed) on the femoral side. The Lift Loop External consists of a 20 mm–wide titanium button and 2 loops controlled by a knotless locking mechanism. The Femobutton consists of a 10 mm–wide titanium button and a continuous loop available in 6 lengths (15-40 mm). The anatomic femoral tunnel was reamed through the anteromedial portal. First, a complete bone tunnel was created over the guide pin using a 4.5-mm drill. Next, the length of the tunnel was measured, and a socket was drilled using a router of the same size as the graft, considering a 6- to 8-mm span for “flip” movement. Then, a complete outside-in tibial tunnel was created in the central portion of the anatomic footprint. The graft was passed into the tibial tunnel, through the intra-articular space, and into the femoral tunnel. The graft was tensioned at 20° of knee flexion. The knee was flexed and extended 30 times. Then, graft tightness was examined with a probe. Finally, the entire construct was retensioned on the tibial side, and additional tying was performed over the adjustable suspensory fixation device using a nonsliding knot.
All patients followed the same postoperative rehabilitation protocol: partial weightbearing for 4 weeks with crutches, increasing knee flexion gradually, and reaching full range of motion by 6 weeks. Patients were permitted to return to sports activity after 9 months to 1 year.
Clinical Outcome Measures
The following patient and injury characteristics were recorded: age, sex, side, body mass index (BMI), time from injury to surgery, preinjury activity level, and follow-up period. Patient-reported outcomes were evaluated with the International Knee Documentation Committee (IKDC) subjective knee form, Lysholm score, and Marx activity scale at 1 year postoperatively. 1,16 Knee laxity was evaluated with the pivot-shift test and side-to-side difference on the KT-1000 arthrometer (Medmetric) at 1 year.
Radiological Evaluation
Patients underwent CT (Revolution EVO; GE Healthcare) at 1 day and 6 months after surgery. At 1 year postoperatively, all patients underwent MRI (1.5-T Magnetom Aera; Siemens Healthineers).
CT with a 1-mm slice thickness and 160-mm field of view was performed to determine the tunnel location and to examine morphological changes in the tibial tunnel. Tunnel volume measurements were performed using 3-dimensional processing software (OsiriX MD 12.0). On each slice of the axial and sagittal CT scans, the tibial graft tunnel and tibial loop tunnel were manually traced and confirmed on coronal images. The volumes of the tibial graft tunnel and tibial loop tunnel were calculated with software by measuring the traced area on each slice (Figures 1 and 2A).

The borders of the tibial graft tunnel (orange line) and tibial loop tunnel (blue line) were outlined on (A) sagittal and (B) axial computed tomography (CT) at 1 day postoperatively. (C) Three-dimensional CT reconstruction of the tibial graft tunnel (orange) and tibial loop tunnel (blue). (D-F) Similar measurements were performed at 6 months postoperatively.
MRI was performed to evaluate the tibial tunnel condition and to determine graft maturation and integration. All participants underwent our standard knee MRI protocol including 2-dimensional (2D) proton density–weighted turbo spin echo (TSE) with fat suppression in 2 planes (sagittal and axial), 2D T1-weighted TSE in the coronal plane, and 2D T2-weighted TSE with fat suppression in the coronal plane. To evaluate graft healing, a T2-weighted TSE without fat suppression sequence was used with parallel imaging using generalized autocalibrating partially parallel acquisition (GRAPPA) with the following parameters: voxel size, 0.8 × 0.8 × 0.8 mm; field of view, 160 × 160 mm; repetition time, 1050 milliseconds; echo time, 70 milliseconds; bandwidth, 413 Hz/pixel; slice thickness, 0.8 mm; base resolution, 192; phase resolution, 100; and acceleration factor, 2. Signal intensity was determined by taking measurements at 6 circular regions of interest, each with a 0.21-cm2 area: (1) quadriceps tendon at 2 cm proximal to the patella, (2) proximal portion of the intra-articular graft, (3) midsubstance, (4) distal portion of the intra-articular graft, (5) intratunnel graft within the tibial tunnel, and (6) background at 2 cm anterior to the patellar tendon (Figure 2B). The signal-to-noise quotient (SNQ) was calculated for each graft region with the following formula: SNQACL = (SIGNALACL – SIGNALQuadriceps)/SIGNALBackground.
Graft integration was classified using the 3-grade method according to Ge et al 9 : grade 1 with low signal intensity at the bone-graft interface with full attachment, grade 2 with medium signal intensity, and grade 3 with high signal intensity with poor attachment. Graft integration in the tibial tunnel was evaluated on axial and sagittal oblique MRI with proton density–weighted TSE sequences (Figure 2C).

(A) Volumetric measurements were performed on magnetic resonance imaging (MRI) at 1 year postoperatively. (B) Placement of the 6 regions of interest used to calculate the signal-to-noise quotient: (1) quadriceps tendon (2 cm proximal to the patella), (2) proximal site, (3) midsubstance, (4) distal site, (5) intratunnel site, and (6) background (2 cm anterior to the patellar tendon). (C) Graft integration was evaluated with an axial proton density–weighted MRI sequence.
To assess intraobserver and interobserver reliability, the measurements were repeated by the first author (A.E.O.) after at least 2 weeks. These were matched to the findings of an experienced radiologist (E.G.).
Statistical Analysis
All statistical analyses were performed using SPSS (version 22.0; IBM). Data were expressed as the mean ± standard deviation. Univariate and multivariate linear regression analyses using the enter method were performed to assess factors associated with loop tunnel filling at 6 months. After considering multicollinearity, we selected age, sex, BMI, graft size, remnant preservation, and smoking as independent variables. Correlations between graft and loop tunnel volume at 1 year, clinical outcomes, and graft SNQ at 1 year were analyzed using the Pearson correlation test. P < .05 was considered the threshold for significance. An intraclass correlation coefficient of >0.8 was considered excellent, and a value between 0.5 and 0.8 was considered good. Power analysis showed that our sample size of 48 was sufficient to detect significant factors with an effect size (Cohen d) of 0.6, type I error at 5%, and power of 80%.
Results
Clinical Outcomes
Overall, 48 patients (34 male, 14 female) with a mean age of 25.2 ± 5.6 years who underwent ACLR using the complete tibial tunnel technique with adjustable suspensory fixation were included in the study. A remnant-preserving procedure was performed in 33 patients. The mean follow-up was 25.7 ± 6.8 months. At 1 year postoperatively, the mean IKDC score was 82.4 ± 8.5 (range, 54-96), the mean Marx activity score was 6.8 ± 3.7 (range, 3-10), and the mean side-to-side difference in anterior tibial translation was 1.5 ± 1.1 mm. Detailed patient demographics and clinical outcomes are shown in Table 1. Intraobserver and interobserver reliability values for radiological measurements are shown in Table 2.
Patient Characteristics and Clinical Outcomes (n = 48) a

a Data are reported as mean ± SD unless otherwise indicated. BMI, body mass index; IKDC, International Knee Documentation Committee.
Intraobserver and Interobserver Reliability of Radiological Measurements a
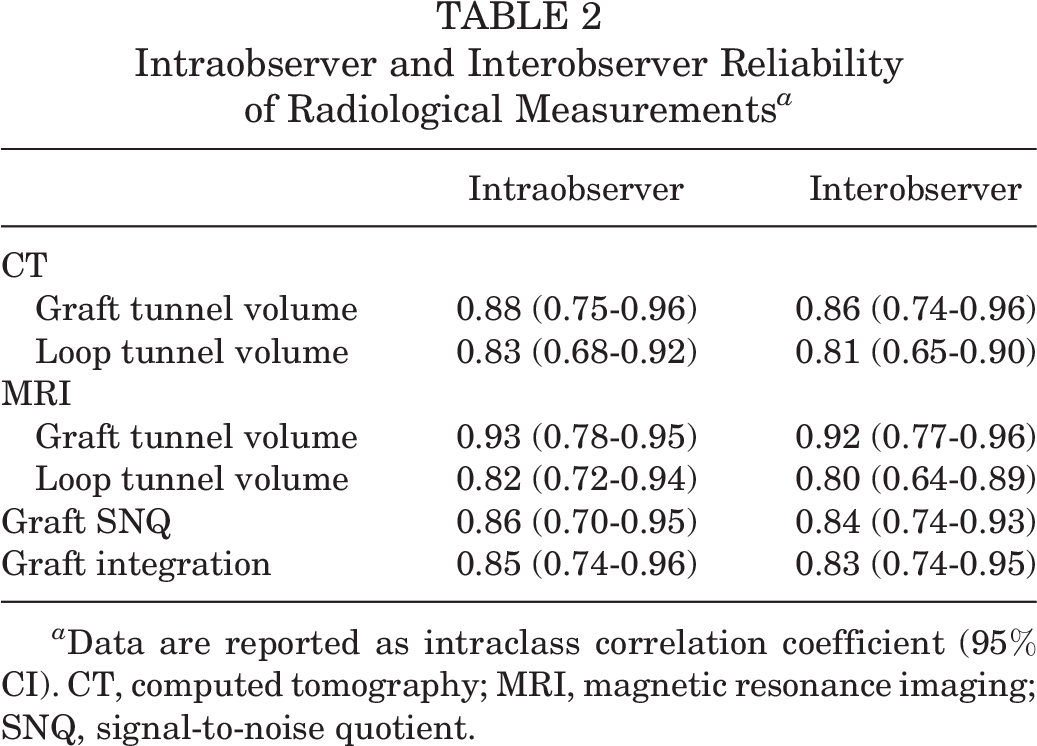
a Data are reported as intraclass correlation coefficient (95% CI). CT, computed tomography; MRI, magnetic resonance imaging; SNQ, signal-to-noise quotient.
CT Outcomes
Based on multiplanar reconstruction images on CT, both the graft and loop tunnel volume were measured (Table 3). At 1 day postoperatively, the mean graft tunnel volume was 1.16 ± 0.4 cm3 (range, 0.91-2.65 cm3), and the mean loop tunnel volume was 2.37 ± 0.5 cm3 (range, 0.75-3.53 cm3). At 6 months after surgery, the mean graft tunnel volume was 1.38 ± 0.6 cm3 (range, 1.11-2.93 cm3), and the mean loop tunnel volume was 0.92 ± 0.3 cm3 (range, 0.09-2.18 cm3). At 6 months, a mean of 63.2% of the tibial loop tunnel was filled by bone. Univariate analysis showed that graft size, remnant preservation, and smoking were significantly associated with the loop tunnel filling rate (Table 4). However, on multivariate analysis, no significant association was found between bone healing in the tibial loop tunnel and age, sex, BMI, graft size, or smoking.
Tibial Tunnel Volume at Different Postoperative Time Points a

a Data are reported as mean ± SD (range) unless otherwise indicated.
Factors Associated With Bone Healing at 6 Months a
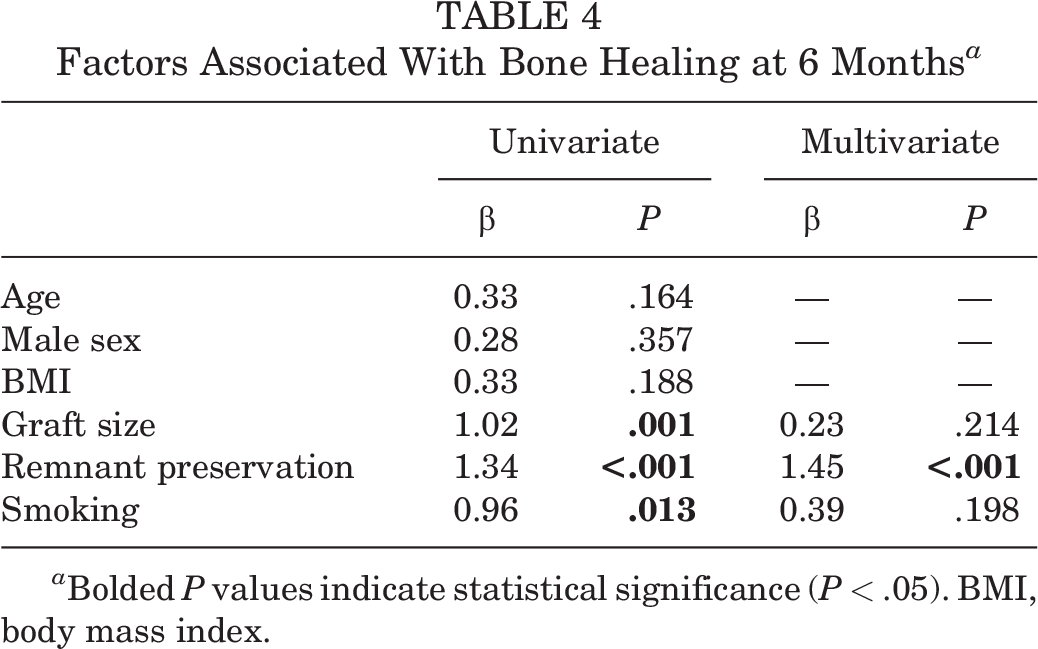
a Bolded P values indicate statistical significance (P < .05). BMI, body mass index.
MRI Outcomes
Similar volumetric measurements were performed on MRI at 1 year. The mean graft tunnel volume was 1.21 ± 0.6 cm3 (range, 1.05-2.91 cm3), and the mean loop tunnel volume was 0.13 ± 0.1 cm3 (range, 0.08-0.21 cm3). The tibial loop tunnel was almost completely closed (98.5%) by bone at 1 year postoperatively (Figure 3).
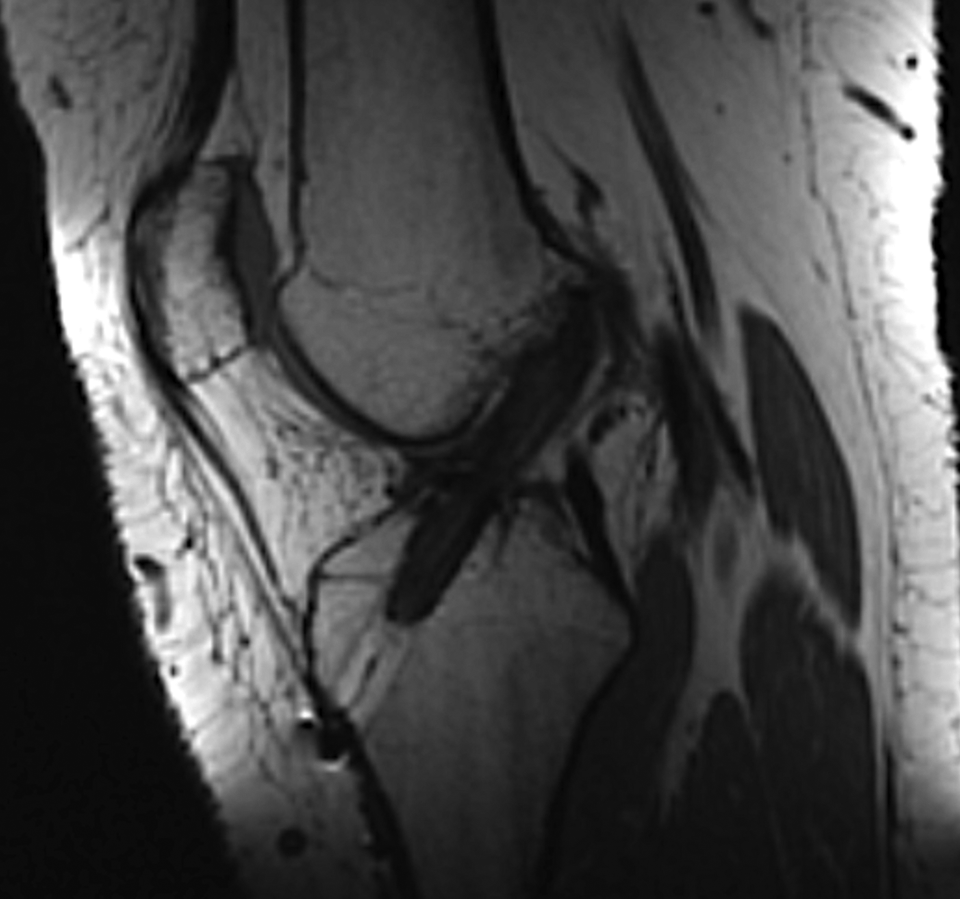
Magnetic resonance imaging at 1 year postoperatively demonstrating the intra-articular graft with a lower signal and a completely closed tibial loop tunnel.
The mean SNQ of the intra-articular graft (4.8 ± 2.5) was significantly higher than that of the intratunnel graft (3.6 ± 1.8) at 1 year postoperatively. Graft integration in the tibial tunnel was assessed as grade 1 in 27 patients, grade 2 in 16 patients, and grade 3 in 5 patients. Correlations between tunnel volume and clinical and radiological outcomes at 1 year are shown in Table 5. There were no correlations between loop tunnel volume and graft integration or graft SNQ. A significant but weak correlation was found between graft tunnel volume and intratunnel graft SNQ as well as integration grade in the tibial tunnel.
Correlation Between Tibial Tunnel Volume, Graft Healing, and Clinical Outcomes at 1 Year a

a Data are reported as r values. Bolded P values indicate a statistical significance (P < .05). IKDC, International Knee Documentation Committee; SNQ, signal-to-noise quotient.
Discussion
The principal findings of this study were that all patients obtained excellent bone filling in the tibial loop tunnel at 1 year and that remnant preservation was significantly associated with the loop tunnel filling rate. Furthermore, graft SNQ and graft integration were not affected by morphological changes in the tibial loop tunnel.
The most important aspect of the all-inside ACLR technique is the drilling of closed sockets instead of full tibial tunnels. 7 However, several reports have raised the concern of an association of this technique with insufficient graft tensioning due to socket-graft length mismatch. 10,14 Furthermore, it requires retrograde drilling using a FlipCutter with the potential for increased damage to remnant ACL tissue compared to outside-in drilling. 7 In contrast to the all-inside technique, successful reconstruction without specific surgical equipment could be achieved via complete outside-in tibial tunnel drilling using adjustable suspensory fixation. 4,5 Modification of the all-inside technique would result in more stable and biological fixation even if the tibial bone stock is sacrificed. The present study showed that tibial bone loss was not a concern. All patients achieved excellent bone healing in the tibial loop tunnel by 1 year postoperatively. Thus, our first hypothesis was confirmed.
There are few studies exploring changes in the tibial loop tunnel over time. In their prospective cohort study, Colombet et al 4 assessed graft integration with different fixation techniques and showed that the tibial loop tunnel was completely filled in 33% of patients at 6 months postoperatively. However, it remains unknown how biomechanical factors interact with tibial loop tunnel changes. The present study was focused on differences in bone filling in the tibial loop tunnel. Volumetric analysis showed that the mean loop tunnel filling rate was 63.2% at 6 months compared to that at time zero. Furthermore, bone healing was not affected by age, sex, BMI, graft size, or smoking, but it was supported by remnant preservation. The most possible reason is related to the mechanical effect of remnant tissue that limits the flow of synovial fluid into the tibial tunnel by covering the tunnel aperture. 20,21,26 Several studies have reported that synovial fluid contains high levels of inflammatory cytokines that inhibit bone formation, and it tends to propagate into the tibial tunnel under the effects of gravity. 11,31 Also, Sun et al 25 showed that the preservation of remnant tissue decreased fluid leakage into the tibial tunnel. Therefore, our second hypothesis was partially confirmed.
ACL graft healing depends on several factors, including anatomic placement, graft choice, proper technique, and biological environment. 19 Graft healing is basically a biological process that occurs in 2 distinct areas: intra-articular maturation and intratunnel integration. 28 It has been categorized into 3 different phases: namely, inflammation, proliferation, and maturation. The early healing phase is characterized by graft necrosis during the first 3 to 6 months, and some studies found that this process has an adverse effect on biomechanical properties of the graft. 12 On the other hand, bone healing occurs via a similar biological process in the tibial loop tunnel. 30 Therefore, it was hypothesized that the process of bone healing in the tibial loop tunnel may have an effect on graft healing by biological interactions in the current study. However, no correlations were found between bone filling and graft SNQ or graft integration at 1 year. A possible explanation for the absent relation is the differences in time points. Bone healing in the tibial loop tunnel largely occurred within 6 months after surgery. However, MRI studies have demonstrated that the graft healing and integration processes continue until 2 years. 3 While it remains uncertain how graft healing affects tunnel morphology, the present study showed that the loop tunnel filling rate did not influence graft healing at 1 year. Our third hypothesis was therefore rejected.
The interaction between tunnel morphology and graft integration remains debatable. 29 Naraoka et al 20 demonstrated that there was no association between tunnel enlargement and graft integration grade. In contrast, Putnis et al 23 showed that tunnel contraction was positively correlated with graft signal intensity, and they reported that this could be a useful indicator of graft integration. In the present study, a similar correlation was found between graft tunnel volume and intratunnel graft SNQ as well as integration grade in the tibial tunnel. Histological studies revealed that the graft integration process is a continuum of biological changes, which included the fibrovascular interface at the early stage and Sharpey-like fibers at the later stage. 15 Bone ingrowth with tightly interfaced fibers within the tunnel was also demonstrated. 13 At 1 year, the graft tunnel volume was reduced in comparison to the volume at 6 months but still larger than that at baseline in the current study. This suggests that bone ingrowth into the tunnel began after 6 months and enhanced graft integration. However, the underlying biological interactions between graft integration and tunnel enlargement or contraction are far from understood.
Limitations
This study has several limitations, including its retrospective nature. First, bone healing in the tibial loop tunnel is basically a biological process, and it is possible that it is influenced by more than the aforementioned factors. Second, although the clinical utility of assessing graft healing on MRI is becoming increasingly recognized, the SNQ is highly variable depending on the scanner characteristics. 27 Third, the clinical follow-up was short, and the evaluation of graft integration was performed at 1 year instead of a longer time period. Fourth, tunnel volume measurements might be underestimated on MRI compared with those on CT. 18 Further, healing in the femoral tunnel, which may be an important area for healing of the entire graft, was not evaluated. Future studies with a longer follow-up are required to add further evidence of graft-tunnel interactions over time. Beyond these limitations, we believe that the present study provides important new information about morphological changes in the tibial tunnel after ACLR.
Conclusion
In the present study, we demonstrated that all patients obtained excellent bone filling in the tibial loop tunnel at 1 year and that remnant preservation was significantly associated with the loop tunnel filling rate. Furthermore, a weak correlation was found between graft tunnel volume and intratunnel graft SNQ as well as integration grade in the tibial tunnel.
Footnotes
Final revision submitted November 30, 2022; accepted January 8, 2023.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Ondokuz Mayıs University (protocol No. 2022/241).
