Abstract
Background:
Preoperative magnetic resonance imaging (MRI) is used to estimate the quantity of tissue provided for fresh osteochondral allograft (FOCA) in the knee. Use of 3-dimensional (3D) MRI modeling software for this purpose may improve defect assessment, providing a more accurate estimate of osteochondral allograft tissue required and eliminating the possibility of acquiring an inadequate quantity of tissue for transplant surgery.
Purpose:
To evaluate the capacity of damage assessment (DA) 3D MRI modeling software to preoperatively estimate the osteochondral allograft surface area used in surgery.
Study Design:
Cohort study (diagnosis); Level of evidence, 2.
Methods:
Included were 36 patients who had undergone FOCA surgery to the distal femur. Based on the preoperative MRI scans, the DA software estimated the total surface area of the lesion as well as the surface areas of each subarea of injury: full-thickness cartilage injury (International Cartilage Repair Society [ICRS] grade 4), partial-thickness cartilage injury (ICRS grade 2-3), bone marrow edema, bone loss, and bone cyst. The probability of overestimation of graft tissue areas by the DA software was calculated using a Bayes-moderated proportion, and the relationship between the prediction discrepancy (ie, over- or underestimation) and the magnitude of the DA estimate was assessed using nonparametric local-linear regression.
Results:
The DA total surface area measurement overestimated the actual area of FOCA tissue transplanted 81.6% (95% CI, 67.2%-91.4%) of the time, corresponding to a median overestimation of 3.14 cm2, or 1.78 times the area of FOCA transplanted. The DA software overestimated the area of FOCA transplanted 100% of the time for defect areas measuring >4.52 cm2. For defects <4.21 cm2, the maximum-magnitude underestimation of tissue area was 1.45 cm2 (on a fold scale, 0.63 times the transplanted area); a plausible heuristic is that multiplying small DA-measured areas of injury by a factor of ∼1.5 would yield an overestimation of the tissue area transplanted most of the time.
Conclusion:
The DA 3D modeling software overestimated osteochondral defect size >80% of the time in 36 distal femoral FOCA cases. A policy of consistent but limited overestimation of osteochondral defect size may provide a more reliable basis for predicting the minimum safe amount of allograft tissue to acquire for transplantation.
Osteochondral defects in the knee are a common cause of pain and functional loss. 3,29 Without proper treatment, these defects progress, leading to larger areas of cartilage and/or bony injury with degenerative joint disease 10,19 and a potential indication for arthroplasty. 9 Treatment strategies include nonoperative management, debridement, marrow stimulation, or reparative/restorative techniques. The size of the lesion, patient age, and associated knee pathologies help determine the most appropriate surgical technique.
Fresh osteochondral allograft (FOCA) transplantation is a proven treatment strategy for a variety of osteochondral injuries. 3,12,22,24 Unlike other cartilage repair techniques, FOCA replaces both the cartilage and underlying subchondral bone at the site of injury and can be used to treat small focal lesions, large irregular lesions, and multicompartmental areas of disease. Prior outcome studies have reported improved mid- and long-term patient outcomes 2,28 and survivability of 86% at 5 years, 78% to 81% at 10 years, and 67% at 20 years. 11,15
Chondrocytes in osteochondral grafts have been shown to remain 80% viable up to 28 days at 98.6°F (37°C) 14,25 after osteochondral harvest. Safety supply chain protocols to ensure donor safety and prerelease screening criteria prohibit immediate tissue release, 5,23 creating a delay in supply to surgeons. The increase in FOCA procedures has led to greater demand for osteochondral allograft tissue. 5,17 While the modern logistics of acquiring and distributing osteochondral allograft tissue are more streamlined than ever, the limited supply of osteochondral tissue has temporal constraints with respect to both expiration and release.
Size matching further constrains the distribution and efficiency of tissue utilization, as routine surgical techniques include the use of intraoperative harvest of cores from sectioned articular surfaces (rather than precut plugs) for most procedures. Tissue banks routinely provide substantial excess tissue to allow surgeons adequate volume to resurface lesions. This safety margin, however, has the secondary effect of limiting supply, as often more tissue is discarded than is actually applied.
Historically, matching donor size involved height and weight measurements of the donors and patients. 13,20 Presently, tissue banks match donor tissue with defect size via knee radiographs and/or magnetic resonance imaging (MRI) for distal femoral lesions. The donor and patient condyle size, tibial width, and defect size are compared. 18,26,28 However, there are limitations in size matching via radiographs and MRI. The femoral condyles of patients with osteochondral defects can be wider and flatter than those of patients without defects, necessitating a larger donor femoral condyle. 26 Second, prior studies have demonstrated that MRI may underestimate the true defect size. 7,16
The discrepancy in preoperative imaging and actual intraoperative defect size may be in part because of the limitations of conventional MRI scans for detecting layers of injury other than International Cartilage Repair Society (ICRS) grade 4 full-thickness areas. 16 Advanced MRI techniques such as dGEMRIC (delayed gadolinium-enhanced MRI of cartilage), T1-rho, and T2 mapping quantify the biochemical cartilage activity to further evaluate areas of partial cartilage injury. 6,21 However, these advanced techniques are limited in availability to clinicians and currently impractical.
Three-dimensional (3D) modeling software may improve the accuracy of evaluating the extent of osteochondral injury, including areas of partial-thickness cartilage injury and subchondral pathology. Prior studies have used preliminary modeling software and computed tomography imaging to match osteochondral defect depth and topography with donor tissue. 1,4,27 However, modeling software in general has not evolved beyond these studies into routine clinical practice in cartilage repair.
The purpose of this study was to evaluate the accuracy of 3D MRI modeling damage assessment (DA) software (Damage Marking Report; Episurf Medical) to predict the FOCA graft size needed for surgery. A secondary aim of this study was to evaluate which subarea of injury as identified by the DA software most consistently overestimates the area of osteochondral allograft transplanted. Clinically, a consistent overestimation of the osteochondral allograft tissue transplanted that is much smaller than the conventional safety margins would ensure sufficient quantity of graft for transplant while minimizing waste and potentially increasing the limited supply of allograft tissue. Our hypotheses were that (1) the DA software would consistently overestimate the size of the osteochondral defect compared with the area of osteochondral allograft tissue transplanted, given the ability of the software to detect partial-thickness areas of cartilage injury, and (2) the overestimation would remain within reasonable bounds.
Methods
Institutional review board approval was obtained for the study protocol. Prospectively collected patient data were retrospectively identified from the respective patient database at each of 3 clinical sites and appropriately de-identified and anonymized. Included were patients aged ≥18 years who underwent FOCA surgery to the distal femur with preoperative MRI available for review. Excluded were patients who underwent FOCA surgery for defects of the trochlea. Demographic and surgical data were collected for analysis. All patients underwent FOCA by 1 of 3 surgeons (D.C., M.P., A.V.). Patients provided informed consent for the surgery.
The de-identified preoperative MRI scans were sent to Episurf for analysis and interpretation, and a DA report for each treatable lesion was generated (Figure 1). The MRI scans were initially assessed by an Episurf specialist radiologist before being fed into the Damage Marking Report software. Standard 2-dimensional MRI scans from 1.5-T and 3-T machines were used in the present study. The DA reports included the total surface area of the lesion as well as the surface areas of each subarea of injury, which are color coded (red = full-thickness cartilage injury [ICRS grade 4]; pink = partial-thickness cartilage injury [ICRS grade 2-3]; blue = bone marrow edema; green = bone loss [ICRS grade 2-4c]; and purple = bone cyst). The total area of injury for each case corresponded to the minimal convex ovoid region covering the combined surface area of the subareas.

Example of damage assessment (DA) of a distal medial femoral condyle osteochondral defect on Damage Marking Report software (Episurf Medical). The DA report includes the total surface area of the lesion and the surface area of each subarea of injury, which are color coded: red = full-thickness cartilage injury (International Cartilage Repair Society [ICRS] grade 4); pink = partial-thickness cartilage injury (ICRS grade 2-3); blue = bone marrow edema; and green = bone loss. Not shown: purple = bone cyst.
Surgical Technique
Arthrotomy for exposure was based on surgeon preference. Fresh osteochondral tissue was obtained from JRF Ortho, using MRI for size matching. The tissue was screened for absence of defects, aseptically harvested from donor knees, and stored at 4°C in a proprietary solution. The corresponding sized allograft to match the debrided osteochondral area of injury was prepared as described previously. 30 The matched allograft was compared with the DA estimated area of injury. Large oblong osteochondral defects were treated with the previously described snowman technique of interposing 2 dowel grafts. 26 No shell graft techniques were employed in patients in this study. Concomitant knee pathology was also addressed.
Postoperatively, patients were allowed to immediately bear weight as tolerated in a knee brace with crutch assistance, when concomitant procedures did not limit weightbearing. Full active and passive knee range of motion was prescribed for open-chain activity immediately and after brace removal with weightbearing.
Statistical Analysis
Demographic information, clinical histories, and other patient characteristics related to the surgeries were described simply by counts and associated percentages (categorical variables) or mean ± standard deviation and range of observed values (scalar variables), both aggregated across the entire cohort and stratified by the 3 contributing surgeons (labeled “Surgeon A,” “Surgeon B,” and “Surgeon C”). The probability of overestimation of graft tissue area by DA was estimated for the total area measure and each subarea measure using a Bayes-moderated proportion with a Jeffreys (ie, beta[1/2,1/2]) prior distribution, and credible intervals were calculated to cover 95% central posterior probability; note that this estimate did not represent the observed proportion in the cohort (which was just slightly larger in all cases) but was rather a best-guess prediction for the probability of overestimation that we should expect for a future case drawn from a population similar to that represented in our cohort (and thus is more useful than the observed proportion for calibrating expectations for future studies of this question).
The relationship between the prediction discrepancy (ie, over- or underestimation) and the magnitude of the DA estimate was assessed using nonparametric local-linear regression (Epanechnikov kernel with bandwidth chosen by 10-fold cross-validation); this was performed on both a difference scale (DA estimate – actual graft size) and a fold scale (DA estimate/actual graft size). Regressions were fit on the full cohort as well as separately for single-core and 2-core graft cases for the total area measure and each subarea measure calculated by the DA software. The minimum cutoff for consistent overestimation by DA was estimated by spline-based (B-spline basis with knots chosen by the Bayesian information criterion) nonlinear regression modeling of the size cutoff against the classification specificity (ie, correct classification of underprediction) at that cutoff, using the calibration function obtained from this regression to locate the smallest cutoff point where the lower bound of a 95% confidence interval for predicted specificity first exceeds 0.9 in expectation. The approximate multiplier to rescue underestimation cases (ie, reliably convert them into overestimates) was estimated via log-linear generalized linear modeling of the actual graft size against the DA estimate, with jackknife resampling of cases under a variety of aggressive (material minimization focus) and conservative (underprediction risk minimization focus) scenarios in order to arrive at a robust guess that was likely to remain valid for future cases similar to those in our cohort.
Results
Thirty-six FOCA surgeries and corresponding DA reports were analyzed, with each of the 3 surgeons providing 12 cases. The 36 patients included 15 men and 21 women with an average age of 39 years (range, 16-64 years). There were 24 osteochondral defects on the medial femoral condyle and 12 lateral femoral condyle defects. Prior surgery to the knee occurred in 22 of 36 patients (61%), including 11 of 36 (31%) with prior cartilage surgery to the area of injury. Additional preoperative data are found in Table 1.
Demographic Data of the Study Patients Overall and by Surgeon a
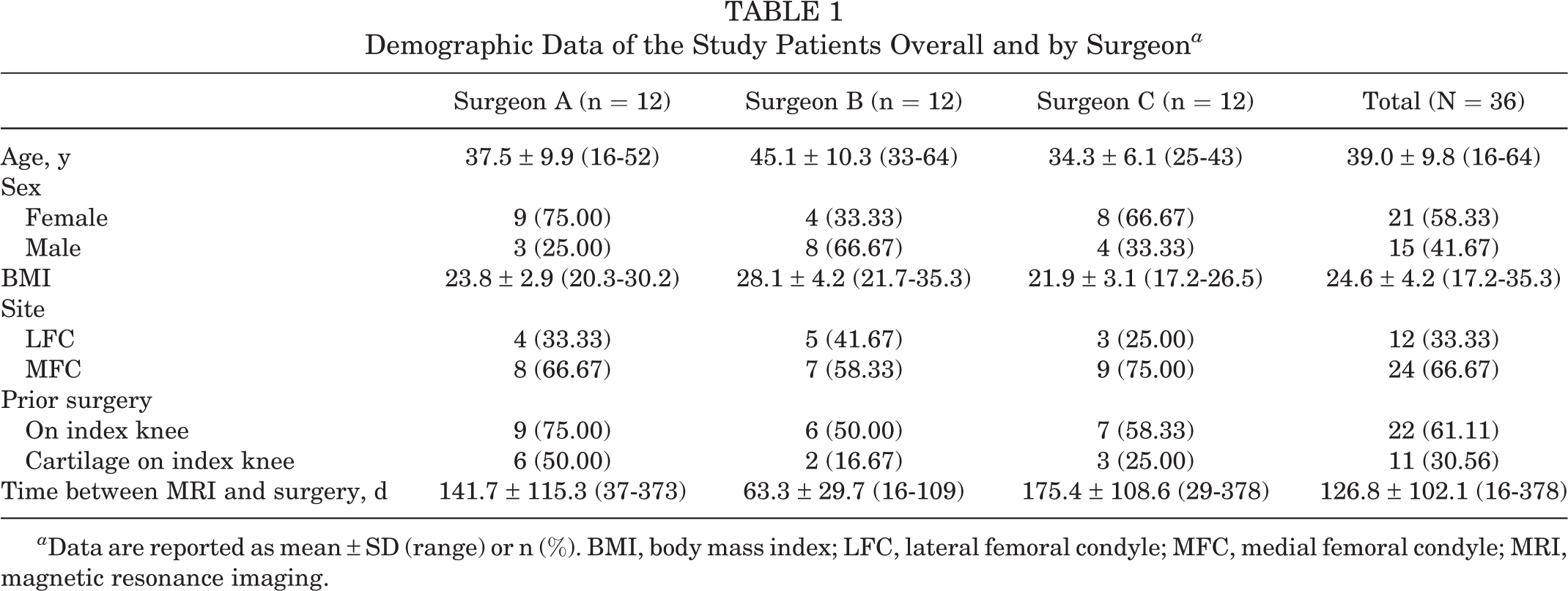
a Data are reported as mean ± SD (range) or n (%). BMI, body mass index; LFC, lateral femoral condyle; MFC, medial femoral condyle; MRI, magnetic resonance imaging.
The 36 FOCAs included 30 single-core plugs and 6 overlapping 2-core plugs. The average graft area was 3.40 cm2 (range, 1.13-7.07 cm2). Concomitant procedures were performed in 32 of 36 patients (89%) (see Table 2 for details). Patient factors including time from MRI to surgery and history of prior cartilage surgery did not appear to meaningfully affect the accuracy of the DA estimation of osteochondral defect size (Table 3); however, we did observe 1 case of extreme overestimation (DA estimate >12 times the actual graft applied) and 2 cases of injury area estimates at roughly the full size of an adult hemicondyle (>20 cm2), and possibly by coincidence, all 3 of these cases involved the medial femoral condyle site with no history of prior surgery at the site (and 2 of the 3 patients were men).
Details of FOCA Surgery Overall and by Surgeon a

a Data are reported as mean ± SD (range) or n (%). ACL, anterior cruciate ligament; FOCA, fresh osteochondral allograft; LCL, lateral collateral ligament; MCL, medial collateral ligament.
Sensitivity of Error in DA Prediction to Patient Factors a

a BMI, body mass index; DA, damage assessment; LCL, lateral collateral ligament; MCL, medial collateral ligament; MRI, magnetic resonance imaging.
b Substantially attenuated when the largest 2 (ie, >95th percentile of) overestimates are omitted:
Any prior surgery on index knee: –0.01 (95% CI, –0.05 to 0.03)
Prior cartilage surgery on index knee: –0.12 (95% CI, –0.33 to 0.08)
MFC site (left): 0.40 (95% CI, –0.21 to 1.03)
MFC site (right): 0.40 (95% CI, –0.21 to 1.02)
Male sex: 0.07 (95% CI, –3.02 to 3.15)
The DA software overestimated the area of FOCA tissue transplanted in 30 of 36 cases, which included 25 single-core plugs and 5 overlapping 2-core plugs. The DA underestimated the area of FOCA tissue transplanted in 6 of 36 cases, including 5 single-core plugs and 1 overlapping 2-core plug. The proportions of single-core versus 2-core plug transplants among both over- and underestimates are consistent with the overall proportion of 2-core plugs in the cohort (6/36).
The total surface area as calculated by the DA software overestimated the actual area of FOCA tissue transplanted an estimated 81.6% (95% CI, 67.2%-91.4%) of the time (Figure 2A), which corresponded to a median overestimation of 3.14 cm2, or 1.78 times the area of FOCA transplanted. The calculated partial-thickness cartilage injury area (pink; Figure 2B) overestimated the area of FOCA tissue transplanted 71.1% (95% CI, 55.5%-83.5%) of the time, which corresponded to a median overestimation of 2.33 cm2, or 1.65 times the area of FOCA transplanted. However, the calculated full-thickness cartilage injury area (red; Figure 2C) overestimated the area of FOCA tissue transplanted only 16% (95% CI, 5.7%-33.7%) of the time, which corresponded to a median underestimation of 1.84 cm2, or 0.32 times the area of FOCA transplanted. The calculated bony edema area (blue; Figure 2D) overestimated the area of FOCA tissue transplanted only 36.7% (95% CI, 21.3%-54.5%) of the time, which corresponded to a median underestimation of 1.22 cm2, or 0.54 times the area of FOCA transplanted. The calculated area of subchondral bone loss (green; Figure 2E) overestimated the area of FOCA tissue transplanted only 36.4% (95% CI, 13.7%-65.2%) of the time, which corresponded to a median underestimation of 1.45 cm2, or 0.71 times the area of FOCA transplanted. The calculated area of bone cyst (purple, Figure 2F) overestimated the area of FOCA tissue transplanted only 10% (95% CI, 1.1%-38.1%) of the time, which corresponded to a median underestimation of 3.09 cm2, or 0.37 times the area of the FOCA transplanted. Notably, the green and purple subareas were very infrequently observed in our DA reports, and occasionally the red or blue subareas (usually both if either one) were also not observed; thus, for use in general (ie, applying to all eligible patients) perhaps only the pink subarea is available in all or a majority of cases.

Difference between estimated osteochondral lesion size according to damage assessment software and actual fresh osteochondral allograft tissue transplanted for (A) total surface area, (B) partial-thickness cartilage injury area, (C) full-thickness cartilage injury area, (D) bone marrow edema area, (E) bone loss area, and (F) bone cyst area. Pr, probability.
There was a general trend (not observed in the green or purple subareas) of an increased amount of overestimation by the DA software with increasing estimated defect area (Figure 2). For the total area measure, a larger area of mean overestimation occurred in the 2-core plug grafts (8.20 cm2) compared with the single-plug grafts (4.60 cm2), although on the fold scale (ie, relative to the estimated defect area), the mean percentage overestimation was about the same for single-core and 2-core plug grafts (2.09 times vs 2.10 times). The total area measure overestimated the area of FOCA transplanted 100% of the time for defect area estimates >4.52 cm2 (ie, this was the smallest observed value in the cohort such that all values at or above it were overestimates), but the gap between this and the largest underestimated value (3.79 cm2) was wide.
Using a statistical classification model, we determined that the cutoff point corresponding to the lowest expected risk of misclassification (overestimate vs not) in future cohorts similar to ours was 4.21 cm2; at this cutoff, there was 95% confidence that at least 90% of future cases at or just above the cutoff would be overestimated by the DA software (and the probability of overestimation is naturally expected to increase rapidly for predictions substantially above the cutoff). For defects <4.21 cm2, the maximum-magnitude tissue area of underestimation was 1.45 cm2, or on the fold scale, 0.63 times the transplanted area; this is approximately two-thirds of what is required, so a plausible heuristic is that multiplying a small (ie, <4.21 cm2) DA-measured area of injury by a factor of ∼1.5 would yield an overestimation of the tissue area transplanted most of the time. (The minimum sufficient factor was 1.58 in our cohort, and we expect that a more conservative choice such as 2.0 would likely cover nearly all cases.)
We also analyzed the 3 largest overestimations where the DA software overestimation was 10 times the tissue area transplanted, or 20 cm2 more area estimated compared with the amount of tissue transplanted. In 2 of the 3 cases, the area of bone edema (blue) was 3 times the area of cartilage injury estimated by the DA software. In the third case, there were 2 adjacent areas of cartilage injury (pink) estimated by the DA software, while only 1 area underwent FOCA transplantation.
Discussion
The most important finding of the current study is that the DA software routinely overestimated the amount of FOCA tissue transplanted in surgery. The total area of injury measured by the DA software most consistently overestimated (>80% of the time) the amount of osteochondral allograft tissue transplanted. The area of partial cartilage injury (pink) was the second most consistent overestimation (∼70% overestimated). The estimated amounts of osteochondral allograft tissue transplanted in focal areas of full-thickness cartilage loss (red) and bony injury (blue, green, and purple) were much more inconsistent and were often not even observed for individual cases (ie, where these classes of injuries did not exist). The overestimation by the DA software of the area of allograft tissue transplanted is likely because of the software’s overestimation of peripheral areas of partial-thickness cartilage injury that are not detectable surgically.
Currently, the osteochondral area of injury and size matching of the osteochondral allograft required are determined by preoperative assessment of radiographs and MRI. Donor allograft hemicondyles are size-matched to patients by measuring the coronal tibial width using these modalities. Additionally, MRI is used to evaluate the osteochondral defect size. However, preoperative MRI often underestimates defect size. Gomoll et al 16 reported that in 37 patients undergoing autologous chondrocyte implantation surgery, 85% had larger areas of cartilage debridement than the area of injury indicated on MRI. Campbell et al 7 reported that in 92 osteochondral defects, MRI underestimated the defect area on average by 70% compared with arthroscopy. We found that the DA software underestimated the osteochondral area transplanted only 16.7% of the time. The underestimation occurred in smaller osteochondral defects (<4.21 cm2), with a maximum underestimation of 1.45 cm2. The underestimation in smaller osteochondral defects could be rescued by multiplying the area of injury by ∼1.5. For example, a 4.2 cm2 defect as measured by the DA software could be viewed as requiring at least 4.2 × 1.5 = 6.3 cm2 of allograft tissue (or conservatively, up to perhaps 4.2 × 2 = 8.4 cm2 of allograft tissue).
The primary purpose of this study was to evaluate DA software’s ability to estimate the osteochondral defect size in comparison to the FOCA tissue required for surgical care. A more accurate estimation of the amount of osteochondral allograft tissue required could allow precut osteochondral plugs to be more routinely utilized, rather than hemicondyles or whole distal femora. This could lead to increases in overall tissue supply and potentially decreasing the cost to providers and patients. Additional research with a larger cohort of patients could allow us to ascertain specific instances (eg, pathology type) where the DA software could most improve clinical practice.
Currently, distal femoral osteochondral allograft tissue is offered in condyles, hemicondyles, and small premade plugs for distal femoral lesions. For smaller lesions with an area <2 cm2 (1.6 cm in diameter), small premade plugs can be used. For lesions >2 cm2, hemicondyles are often provided to ensure adequate tissue is available, which can leave substantial excess allograft tissue waste (Figure 3). Ideally, preoperative imaging would consistently overpredict the defect size by a minimal amount to accurately predict the size and area of allograft tissue required. This could lead to a more precise amount of allograft tissue provided to the surgeon, leading to less waste of tissue and a potentially associated decreased surgical cost. Our results suggest that the DA software may provide a less wasteful preoperative estimation of allograft tissue required for surgery than current standard-of-practice methods and may serve as a useful tool in determining a patient’s ultimate allograft tissue size requirements.

Photograph of excess allograft femoral hemicondyle tissue waste after cored osteochondral allograft plug removal using current preoperative patient-donor matching.
Limitations
The primary limitation of our study is the sample size; however, the 36 cases were a fair representation of the spectrum of injury and core samples seen in the clinical practice of the contributing surgeons, and we believe they provide for a very adequate initial analysis or “pilot” type data. Additionally, only defects of the distal femoral condyles were included in the study. Further investigation of the DA software’s ability to evaluate the unique morphology of patellar and trochlear osteochondral defects is warranted. Another potential limitation relates to the combination of 1.5-T and 3-T MRI scans used in this study. The accuracy of 1.5-T and 3-T MRI scans was not compared. In theory, using images with a greater field strength could result in even greater DA precision.
The majority of patients had surgery <6 months after the preoperative MRI. A longer period between MRI and surgery increases the risk of the osteochondral injury increasing in size. 8 Additionally, many patients will have had prior cartilage debridement procedures. Accounting for prior cartilage surgery and time from MRI to surgery did not meaningfully affect the accuracy of the DA estimation, nor was there any compelling relationship between magnitude of overestimation and either of these factors (Table 3). Our ability to assess the value of either of these variables for explaining prediction error was limited by the relatively small sample size (36 cases) and the presence of a few outliers with much larger than typical prediction errors.
Conclusion
Current use of MRI may not accurately assess the area of osteochondral injury to be treated. We report that the DA, a 3D modeling software, overestimated osteochondral defect size in the majority of 36 distal femoral FOCA cases. Consistent overestimation of osteochondral defect size would provide a circumstance where tissue provided for osteochondral allograft surgery would be adequate for surgical reconstruction and might allow conservation of valuable tissue concurrently, if the overestimation amount does not exceed current levels of waste. As FOCA surgery increases in clinical practice and tissue availability becomes more limited, a more efficient and accurate application of allograft tissue resources could further increase access for patients and potentially decrease cost.
Footnotes
Final revision submitted October 14, 2022; accepted November 9, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This research project was funded by a grant from JRF Ortho. M.P. and D.C. are executive board members of the MOCA (Metrics of Osteochondral Allograft) Study Group, a public service organization sponsored by JRF Ortho. S.G.M. has received education payments from Steelhead Surgical. M.P. has received consulting fees from Arthrex, JRF Ortho, and Zimmer Biomet; speaking fees from Arthrex; honoraria from Flexion Therapeutics and JRF Ortho; and royalties from Anika Therapeutics, Arthrosurface, and Arthrex. A.V. has received consulting fees from Arthrex and Stryker, nonconsulting fees from Arthrex and Smith & Nephew, and honoraria from Vericel. D.C. has received education payments from Steelhead Surgical, consulting fees from JRF Ortho and Vericel, and honoraria from JRF Ortho. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Oregon Health & Science University (reference No. 22415).
