Abstract
Background:
It is well known that glenoid osseous defects >13.5% of the glenoid width critically destabilize the shoulder, as do labral tears. Chondrolabral defects often occur with anterior dislocation of the shoulder. It is unclear whether glenoid chondrolabral defects contribute to shoulder stability and, if so, at what size they become critical.
Purpose/Hypothesis:
The purpose of this study was to determine the effect of incremental chondrolabral defect sizes on anterior shoulder stability in the setting of labral deficiency. The hypothesis was that chondrolabral defects ≥13.5% of the glenoid width will decrease anterior shoulder stability.
Study Design:
Controlled laboratory study.
Methods:
This controlled laboratory study tested 12 fresh-frozen shoulders. Specimens were attached to a custom testing device in abduction and neutral rotation with 50-N compression applied to the glenoid. The humeral head was translated 10 mm anterior, anteroinferior, and anterosuperior with the conditions of intact cartilage and labrum and anterior full-thickness chondrolabral defects of 3-, 6-, and 9-mm width. Translation force was measured continuously. Peak translation force divided by 50-N compressive force defined the stability ratio. Data were analyzed using analysis of variance.
Results:
The anterior stability ratio decreased between the intact state (36% ± 7%) and all defects ≥3 mm (≤32% ± 8%; P ≤ .023). The anteroinferior stability ratio decreased between the intact state (52% ± 7%) and all defects ≥3 mm (≤47% ± 7%; P ≤ .006). The anterosuperior stability ratio decreased between the intact state (36% ± 4%) and all defects ≥6 mm (≤33% ± 4%; P ≤ .006). A 3-mm defect equated to 10% of the glenoid width. There were moderate to strong negative correlations between chondrolabral defect size and stability ratio in the anterior, anteroinferior, and anterosuperior directions (r = –0.79, –0.63, and –0.58, respectively; P ≤ .001). There were moderate to strong negative correlations between the percentage of glenoid chondrolabral defect size to the glenoid width and the stability percentage in all directions (r = –0.81, –0.63, and –0.61; P ≤ .001).
Conclusion:
An anterior glenoid chondrolabral defect ≥3 mm (>10% of the glenoid width) significantly decreased anterior and anteroinferior stability. Chondrolabral defect size negatively correlated with stability.
Clinical Relevance:
To fully restore glenohumeral stability, in addition to labral repair, it may be necessary to reconstruct chondrolabral defects as small as 3 mm (10% of the glenoid width).
The prevalence of anterior glenoid cartilage defects (ie, damage to the articular chondral surface) in the absence of osseous defects is reported in up to 57% of anterior shoulder instability cases. 4,17,24,35 Combined osteocartilaginous defects are even more prevalent, as bone loss is reported as 70% to 97% in anterior shoulder instability and is associated with loss of overlying cartilage. 8,20,22,29 The curvature of the glenoid cartilage deepens the glenoid concavity and therefore augments shoulder stability. 31,42 Clinically, the presence of a glenoid cartilage defect is associated with an increased risk for instability recurrence. 18,19,28 Even in the absence of instability recurrence, the loss of stability may increase translation of the humeral head relative to the glenoid and thus contribute to glenohumeral osteoarthritis (OA). 26 Radiographic OA occurs in up to 60% of cases after glenohumeral instability with long-term follow-up. 2,5,7,11,23,25,27,34 Small cartilage defects may therefore increase translation and contribute to OA progression. 3,7,28 However, little is known regarding the effect of cartilage defects upon shoulder stability.
The effect of the glenoid osseous defects on stability has been thoroughly examined in biomechanical cadaveric studies. 12,36 In these studies, 25% of the glenoid width has been shown to be a critical glenoid osseous defect size. It was recently reported that glenoid bone loss of >13.5% or 17% of the glenoid width reduces postoperative clinical outcomes, and critical bone loss is considered <25% of the glenoid width. 29,30,37 These osseous defects have chondrolabral defects of at least the same size.
The purpose of this study was to determine the effect of incremental chondrolabral defect sizes on anterior shoulder stability in the setting of labral deficiency. We hypothesized that isolated chondrolabral defects ≥13.5% of the glenoid width would significantly decrease anterior shoulder stability, even in the absence of osseous defects.
Methods
Specimen Preparation
Thirteen fresh-frozen cadaveric shoulders were tested with a mean age of 63 years (range, 56-69 years) at time of death. These were obtained from Science Care Anatomical and the United Tissue Network. Specimens were thawed overnight at room temperature before testing. All soft tissue was removed from the scapula and humerus, except for the glenoid labrum and articular cartilage. 9 Because the labrum attaches to the articular cartilage and there is a fibrocartilaginous area between the 2, the boundary is not clearly visible macroscopically. 14,40 Therefore, the labrum was excised in the same location as the cartilage in this study. Soft tissues were removed to reduce interpatient variability and because the capsuloligamentous structures do not play a role in stability in the midranges of motion in a biomechanical study. 12 The coracoid process and acromion were resected to allow clearance for the humeral head to translate without impingement. All specimens were free of macroscopic and radiographic evidence of glenohumeral OA or prior surgery.
Mounting Specimens
Each specimen underwent a computed tomographic scan using a Siemens Sensation scanner. Image data were acquired with a 130-kV tube voltage, 512 × 512 acquisition matrix, 1.0-mm slice thickness, 0.75 pitch, and 170-mA tube current. From the images, 3-dimensional (3D) models of the humerus and scapula were created (MIMICs; Materialise). The models were used to create 3D-printed negative molds (ABS plastic [acrylonitrile butadiene styrene], DimensionElite; Stratasys) to align the bones to the testing system (Figure 1) in predefined orientations. On the humeral side, specimen-specific fixtures aligned the humerus in 60° of abduction and neutral rotation relative to the scapula. The rotation of the humeral shaft was determined by the position of the biceps groove. The humerus was attached to a cantilever arm for application of a vertical 50-N compressive force using static weights. 13,38,39 On the scapular side, a plane was fit to the mean surface of the glenoid fossa on the 3D model. Specimen-specific scapular alignment fixtures were 3D printed to hold the scapular body medial to the glenoid vault to maintain a horizontal glenoid plane orientation.

Anterior instability test apparatus. Opt, Optotrak Certus motion capture marker.
The scapular fixture was mounted on linear rails to allow anterior humeral translation, as well as a plate to provide fixed rotation in 15° increments about the scapular axis (defined vertically through the glenoid center). The linear stage was then connected via a low-stretch cable to a hydraulic testing machine (model 8800 controller, 1331 load frame; Instron) whose load cell recorded the applied translation force (Dynacell model 2527-13; Instron). Four motion capture markers with 0.1-mm accuracy and 0.01-mm resolution (Optotrak Certus; Northern Digital) were attached to the testing apparatus: (1) on the stationary base of the rails as reference, (2) on the sliding table for measures of anterior translation, (3) on the humeral alignment fixture for measures of humeral head lateral translation relative to the glenoid, and (4) on the load cell to synchronize the start of data recording between the test and measurement systems (Figure 1). Force and displacement on the Instron were sampled by WaveMatrix (Instron) at 100 Hz. The 3D coordinates of the Optotrak Certus motion capture marker were recorded at 100 Hz.
Glenoid Chondrolabral Defects
First, a retrospective review was performed of all instability operations performed at our institution over a 7-year period. Of those cases, 45 with isolated cartilage defects were identified on magnetic resonance imaging scans and segmented to create 3D models to determine the most common morphology of cartilage defects. It was determined that isolated glenoid cartilage defects were most often oriented directly anterior 15 ; thus, all simulated cartilage defects were oriented directly anterior (Figure 2). To create defects, a custom 3D printed fixture (Figure 2A) with a cutting guide contacted the anterior rim of the glenoid to act as the reference plane. The fixture had a movable slot into which a scalpel was inserted perpendicular to the plane of the glenoid at 3-mm intervals (Figure 2, B and C). Once the border of the cartilage defect was created with the scalpel, all cartilage and labrum anterior to the cut were removed with a scalpel and curette down to the subchondral bone, thereby creating a combined cartilage defect and labral deficiency.

Creating the glenoid cartilage defect. (A) A custom 3-dimensional printed fixture with a cutting guide contacted the anterior rim of the glenoid to act as the reference for defect creation. (B) The fixture contained a movable slot into which a scalpel was inserted perpendicular to the plane of the glenoid. (C) Defect cuts were made at 3-mm intervals.
Test Conditions
During testing, the scapula was translated 10 mm 12,38,39 posterior relative to the humeral head, from a centered position on the glenoid at 2 mm/s. 13,39 With a 50-N compression force applied to the humerus, the table on which the scapula was fixed was moved in the superoinferior and anteroposterior directions. The vertical movement of the humerus was measured with the Optotrak. The center position was defined as the point where the humeral head was closest to the glenoid fossa in both directions. From each test, the peak translation force was quantified, and the stability ratio was defined as the peak translation force divided by a 50-N compression force. 9,13,36 Many similar studies have applied a 50-N compression force.36,38,39 It has been reported that the force applied to the glenoid fossa during abduction in vivo is ≥50 N. 18 Furthermore, no significant damage occurs when a displacement test is performed with a 50-N compression force. 38
All testing was performed in triplicate, and the 3 data points were averaged to generate a representative data point for each condition. The stability ratio was tested sequentially with the intact state and defects at widths of 3, 6, and 9 mm. For each condition, the stability ratio was determined in 3 directions: anterior, anterosuperior, and anteroinferior. Anterior was defined as perpendicular to the long axis of the glenoid (0°), anterosuperior as +30° superior to anterior, and anteroinferior as –45° inferior to anterior. The anterosuperior direction was included to be complete, as there may be some heterogeneity in the direction of dislocation depending on anatomic variation and variation in the force placed on the shoulder at the time of the dislocation. In our preliminary experiments, when we moved the humeral head in a +45° superior-to-anterior direction, it collided with the coracoid process. For this reason, we chose +30° as the anterosuperior direction. The order of directional testing was randomized within a given defect size by reorienting the rotational plate holding the scapular fixture between trials.
Defect Depth Measurement
The articular surface was measured to investigate the degree to which defects altered the depth of the curvature (Figure 3). The thickness of the combined chondrolabral tissue was also measured. These measurements were taken on an anteroposterior axis through the center of the glenoid fossa using a digitizer with ±0.23-mm accuracy (MicroScribe G2X; Solution Technologies). The difference in height between the anterior margin and center of the cartilage was measured as depth. The thickness of the cartilage was measured at the anterior margin.

Depth of concavity and thickness of cartilage at anterior edge.
Statistical Analysis
One-way repeated measures analysis of variance was used to compare the stability ratio of the different defect sizes in the same direction. If significant, Bonferroni tests were used to detect differences between defect sizes. Pearson correlation was used to calculate the following: the relationship between the glenoid defect size and the stability ratio; the percentage of the glenoid defect to glenoid width and the stability ratio; the relationship between the depth and the stability ratio in the anterior direction; the relationship between the thickness of the anterior articular cartilage and the stability ratio in the anterior direction; and the percentage of the depth to glenoid width and the stability ratio. P ≤ .050 was considered the threshold for statistical significance.
A power analysis was performed using G*Power 3.1 (Heinrich Heine University) to determine the number of specimens necessary to achieve 80% power. 6 Effect size was calculated from means and standard deviations of 3 cadavers calculated in pilot testing. Calculated effect sizes were 1.24 in the anterior, 0.45 in the anteroinferior, and 0.53 in the anterosuperior directions, resulting in sample sizes of 3, 11, and 15 for the anterior, anterosuperior, and anteroinferior directions, respectively. All statistical analyses were performed with SPSS Statistics 25 (SPSS Inc) and JMP 15 (SAS Institute Inc).
Results
One cadaveric specimen was excluded because an existing cartilage defect was discovered during preparation. In another specimen, there was a technical error during the test in the anterosuperior direction, so it was excluded from that analysis but included for the other directions. This resulted in anterior and anteroinferior tests in 12 cadavers and anterosuperior tests in 11 cadavers. Donors were 171 ± 13 cm tall (140-188 cm; mean ± SD [range]), weighed 65 ± 16 kg (41-99 kg), and had a body mass index of 22 ± 4 (15-32).
In the anterior direction, there was a decrease in stability ratio between the intact state (36% ± 7%) and the 3-, 6-, and 9-mm defects (32% ± 8%, 24% ± 6%, 15% ± 4%, respectively; P ≤ .023) (Figure 4). Differences were detected for all combinations of defect size (P ≤ .001). In the anteroinferior direction, there was again a decrease in stability ratio between the intact state (52% ± 7%) and the 3-, 6-, and 9-mm defects (47% ± 7%, 42% ± 6%, 38% ± 8%; P ≤ .006). Differences were again detected for all combinations of defect size (P ≤ .017). In the anterosuperior direction, there was a decrease in stability ratio between the intact (36% ± 4%) and 6- and 9-mm defects (33% ± 4% and 28% ± 4%; P ≤ .006). There were also differences detected with the 3- and 6-mm defects as compared with the 9-mm defects (P ≤ .001). The mean glenoid width and length were 31.2 ± 2.5 and 42.4 ± 3.2 mm. Thus, a 3-mm defect was equivalent to approximately 10% of the glenoid width.

The relationship between glenoid cartilage defect size and the stability ratio. Data are shown as mean ± SD. *Statistically significant difference between defect sizes.
There were moderate to strong negative correlations between defect size and stability ratio in the anterior, anteroinferior, and anterosuperior directions (r = –0.79, –0.63, and –0.58, respectively; P ≤ .001) (Figure 5A). There were moderate to strong negative correlations between the percentage of the defect size to the glenoid width and the stability ratio in the anterior, anteroinferior, and anterosuperior directions (r = –0.81, –0.63, and –0.61; P ≤ .001) (Figure 5B).
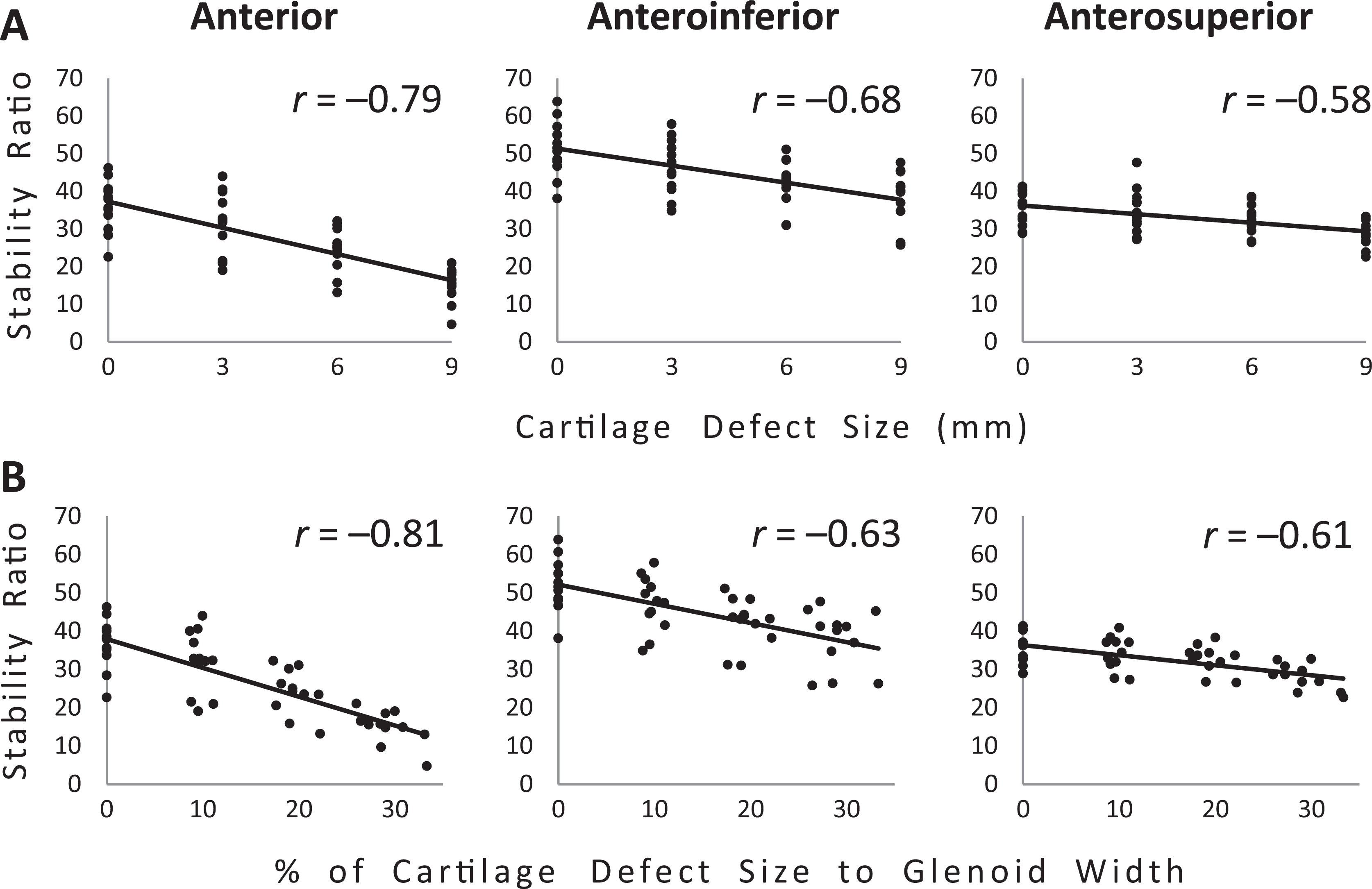
Correlations between cartilage defect size (A) in millimeters and (B) as a percentage of the glenoid width and the stability ratio in the anterior, anteroinferior, and anterosuperior directions.
The mean cartilage depth was 4.0, 3.1, 1.9, and 1.0 mm for the intact and 3-, 6-, and 9-mm defects, respectively. There was a moderate positive correlation between cartilage depth and stability ratio in the anterior direction (r = 0.51; P = .001). There was a moderate positive correlation between the percentage of cartilage depth to glenoid width and the stability ratio (r = 0.55; P ≤ .001) (Figure 6).

Correlations between the percentage of cartilage depth to glenoid width and the stability ratio in the anterior direction.
The mean thickness of the anterior articular cartilage was 3.5, 2.2, 2.2, and 2.0 mm for the intact and 3-, 6-, and 9-mm defects, respectively. There was a weak negative correlation between the maximal thickness of the anterior articular cartilage and stability ratio in anterior direction (r = –0.34; P = .016). There was a weak positive correlation between the percentage of cartilage thickness to glenoid width and the stability ratio (r = 0.30; P = .021) (Figure 7).
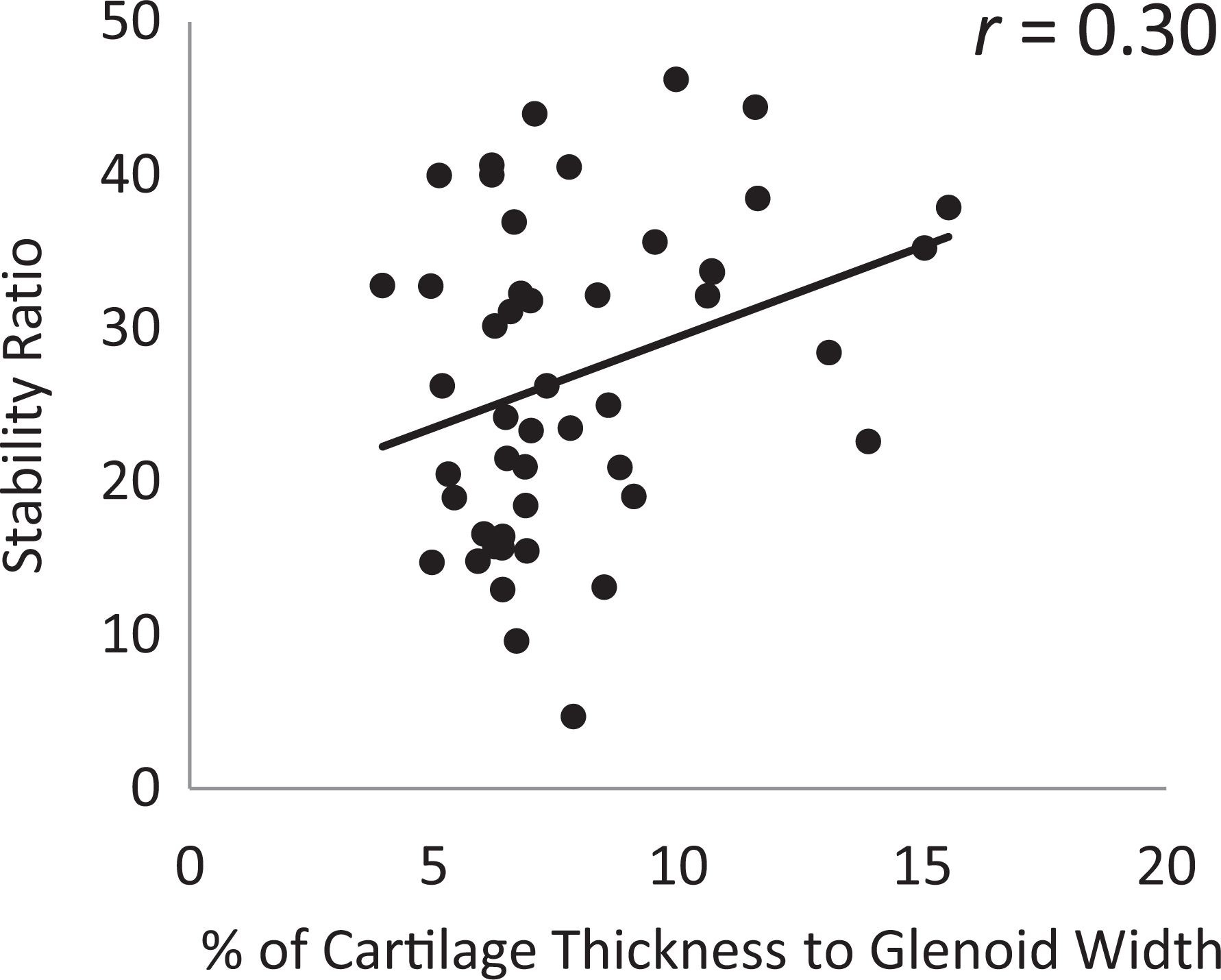
Correlations between the cartilage thickness (percentage) to the glenoid width and the stability ratio in the anterior direction.
Discussion
The present study showed that even a 3-mm glenoid chondrolabral defect, which equates to 10% of the glenoid width, significantly reduced glenohumeral stability in the anterior and anteroinferior directions in the setting of labral deficiency. This defect size was smaller than that in our hypothesis. Defect size and defect depth inversely correlated with the stability ratio, suggesting that larger defects result in larger decreases in stability and that the curvature of the glenoid cartilage contributes to glenohumeral stability.
We found that a glenoid chondrolabral defect of 10% of the glenoid width statistically reduced the stability ratio. Yamamoto et al 36 performed a similar biomechanical study of anterior stability by creating a stepwise anterior bone defect. They reported that a bone defect >20% of the glenoid width statistically decreased the stability ratio. Because osseous defects result in the loss of the subchondral bone and its overlying cartilage and labrum, we hypothesized that an isolated chondrolabral defect and an osteochondral defect would demonstrate similar trends with regard to glenohumeral joint stability. The statistically significant effect upon stability started at a smaller defect size than what we expected. Interestingly, a recent finite element analysis showed that the critical size of the osteochondral defect was 16% of the width of the glenoid, 16 demonstrating that effects may be seen at smaller defect sizes than previously thought. All in all, 16% of the width of the glenoid is the same as a 5.0-mm defect in our study. In addition, clinical studies revealed that osteochondral defects of 13.5% to 20% of the glenoid width or less significantly reduced clinical results, which is again closely reflective of our findings. 29,30 A total of 13.5% to 20% of the glenoid width is the same as a 4.2- to 6.2-mm defect in our study. Our results provide further evidence that smaller defects may have a larger effect on stability than previously expected and may therefore require reconstruction to provide stability. These similar findings from computer modeling studies, cadaveric biomechanical studies, and clinical studies all provide support for lowering the critical threshold at which osseous or osteochondral grafting is performed: <20%. However, additional clinical studies are needed to determine whether a chondrolabral defect width of 3 mm is clinically critical.
In the midrange of motion, the joint capsule is lax; thus, the joint capsule does not stabilize the joint. 10,18,21 In these ranges, the negative intra-articular pressure and concavity-compression effect are thought to provide stability to the glenohumeral joint. 12 Our results demonstrate that the glenoid cartilage curvature deepens the concavity of the glenoid and thereby contributes to stability. 31,42
Specifically, our results suggest that there is a dose-response relationship between the size of the chondrolabral defect width and the stability ratio, illustrated by inverse correlations ranging from –0.58 to –0.81 depending on the direction of translation. Therefore, chondrolabral defects <10% of the width of the glenoid fossa can contribute to anterior instability and may need to be taken into consideration. This loss of joint stability could result in an increase in translation, which could contribute to recurrence and further joint damage and OA. Future research will be necessary to understand whether it may be necessary to reconstruct cartilage defects with osteochondral allografting to fully restore glenohumeral stability when performing labral repair in the cartilage defect. In addition, a biomechanical model should evaluate the prior suggestion that removing 2 to 5 mm of cartilage at the anterior glenoid margin promotes biological labral healing during recurrent shoulder instability surgery. 1,32,33
Our study demonstrated that as the width of the chondrolabral defect increased, the depth of the chondrolabral surface decreased and the stability ratio decreased. Thus, chondrolabral defects decrease stability when the depth of the glenoid fossa decreases. Therefore, chondrolabral defects result in loss of the concavity-compression mechanism, even without glenoid bone loss. There was a weak correlation between the maximal thickness of the anterior articular chondrolabral and stability ratio in the anterior direction (r = –0.34). It has been reported that the cartilage surface of the glenoid has a smaller radius of curvature than the osseous surface. 41 As such, the chondrolabral surface on the glenoid provides more concavity for the concavity-compression mechanism than the glenoid bone alone. From our results, the effect is considered to be weak in the central part of the glenoid fossa. This effect is also directionally dependent: in the anterior and anteroinferior directions, the stability ratio was reduced at ≥3 mm, but in the anterosuperior direction, the stability ratio was reduced at ≥6 mm. It may be important to repair anterior and anteroinferior chondrolabral defects.
Limitations
This study has several limitations. First, the cartilage defects created may differ from in vivo cartilage defects, as those herein were created parallel to the long axis of the glenoid using a scalpel. Significant efforts were undertaken to study the morphology of real cartilage defects, which showed that in vivo cartilage defects often have more complex and irregular borders with rounded edges. These complex and irregular boundaries may predispose recurrent instability in a preferential direction or have no influence at all. More research is needed on this topic, as the simplified defect geometry used here was chosen to focus our understanding on the incremental nature of defect size using a repeatable template for its creation. Second, in this study, the head of the humerus was translated linearly by 10 mm, and this range of displacement did not cause dislocation. In our testing, complete dislocations were avoided to limit additional damage attributed to the humerus impacting the anterior rim of the glenoid and to preserving the ability to perform repeated testing. Here, 10-mm translation was sufficient to find the repeatable peak translation force, and further translation was unlikely to have altered our results. Specifically,10 mm means different to small and large glenoid. It would have been better to normalize the translation by the size of the humeral head or the glenoid. Third, the rate of translation in the present study was 2.0 mm/s. However, a dislocation typically occurs at much faster displacement rates in high-energy injuries. We expect the incremental nature of the stability ratio to hold regardless of speed, given its consistent trends in relation to defect size. Fourth, we did not compare the stability ratio with and without labral repair for each cartilage defect.. So the effect of labral repair in the setting of a cartilage defect on stability is not known. Fifth, since the boundary between cartilage and labrum is difficult to distinguish with the naked eye, the labrum was not considered in isolation in this study. As such, the differential effects of labrum and cartilage alone are not known.
Conclusion
An anterior glenoid chondrolabral defect >3 mm (>10% of the glenoid width) statistically decreased anterior and anteroinferior stability. Chondrolabral defect size negatively correlated with stability.
Footnotes
Final revision submitted July 14, 2022; accepted July 27, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: R.Z.T. has received consulting fees from Cayenne Medical and Mitek and royalties from Shoulder Innovations, Wright Medical, and Zimmer; he also has stock/stock options in Conextions, Intrafuse, and Kator. P.N.C. has received consulting fees from DJO and DePuy, speaking fees from DePuy, and royalties from DePuy and Responsive Arthroscopy. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by the University of Utah.
