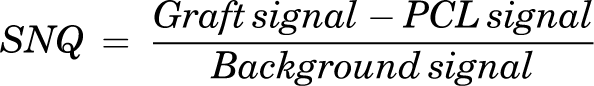Abstract
Background:
Joint infection after anterior cruciate ligament (ACL) reconstruction is a rare but serious complication.
Purpose:
To assess the effect of joint infection on the graft, cartilage, and bone tunnel using magnetic resonance imaging (MRI) after arthroscopic single-bundle ACL reconstruction with autologous hamstring tendons.
Study Design:
Cohort study; Level of evidence, 3.
Methods:
This retrospective matched cohort study included 26 patients who underwent arthroscopic single-bundle ACL reconstruction with hamstring tendon graft at the authors’ institute between January 2002 and December 2017 and developed postoperative joint infection. These patients were matched 1:3 to patients who did not sustain joint infection after ACL reconstruction (control group). MRI scans were collected at the time of follow-up. The following parameters were evaluated: graft signal-to-noise quotient (SNQ); graft signal intensity at the bone-graft interface and within the knee joint; bone tunnel enlargement at the tunnel aperture, midsection, and exit of the tibial and femoral tunnels; and cartilage integrity.
Results:
The average follow-up time was 47.8 months in the infection group and 48.5 months in the control group. Compared with the control group, the infection group had a significantly higher SNQ (20.01 ± 12.08 vs 7.61 ± 6.70; P = .014) as well as a higher signal intensity at the bone-graft interface (P = .037) and higher Howell grade (P = .031). The mean enlargement at the femoral tunnel aperture was 31.20% ± 26.76% in the infection group and 19.22% ± 20.10% in the control group (P = .037). The articular cartilage of the patellofemoral and lateral femorotibial joints showed more degenerative change in the infection group.
Conclusion:
Study findings indicated that graft ligamentization and incorporation graft maturity were inferior in patients who experienced a joint infection after ACL reconstruction compared with patients who did not.
Joint infection after anterior cruciate ligament (ACL) reconstruction is a rare complication, with reported incidence rates ranging from 0.14% to 1.8%. 7,11,14,22,26,34 It may cause cartilage damage 10 and can potentially influence graft ligamentization, bone tunnel enlargement, and development of osteoarthritis.
In the early stage of graft remodeling, avascular necrosis and hypocellularity occur at the central part of the graft, which can be seen 2 to 4 weeks after surgery in animal models. 25 Postoperative joint infection frequently occurs in the first 3 weeks after ACL reconstruction, 35 which coincides with the early phase of graft remodeling. Therefore, the effect of joint infection on graft ligamentization and healing needs to be investigated. Magnetic resonance imaging (MRI) can be used to precisely assess the homogeneity of the graft and healing at the graft-bone interface. Weiler et al 37 correlated a lower graft signal-to-noise quotient (SNQ) with better biomechanical and historical properties in sheep models.
Animal experiments have proven that joint infection can lead to rapid in situ chondrocyte death once pathogens adhere to the cartilage. 31 Researchers have noticed that injecting antibiotic-killed Staphylococcus aureus into the knee joint of a mouse can induce mild-to-moderate synovitis and bone erosion, which can last for at least 14 days. 4 In humans, the consequences of septic knee arthritis are serious; the annual risk of arthroplasty is about 6 times that of the general population. 1 Hence, a postoperative joint infection is a great threat to articular cartilage.
Bone tunnel enlargement after ACL reconstruction theoretically increases the difficulty of revision surgery. 23,28 Studies have demonstrated that tunnel expansion occurs early and primarily at the tunnel apertures, and the degree of enlargement varies with different types of graft and fixation. 36 Whether joint infection after ACL reconstruction has the potential to influence bone tunnel enlargement remains unknown.
The primary purpose of this study was to evaluate graft ligamentization and incorporation using MRI in patients with postoperative joint infection after ACL reconstruction. Our secondary aim was to assess both cartilage integrity and bone tunnel enlargement. Based on previous studies, we hypothesized that postoperative joint infection would hinder graft ligamentization, disrupt cartilage integrity, and exacerbate bone tunnel enlargement after ACL reconstruction.
Methods
Patients
Between January 2002 and December 2017, 20,549 arthroscopic ACL reconstructions were performed by senior surgeons at our institute. After surgery, 62 joint infections were diagnosed according to the American Centers for Disease Control and Prevention criteria for joint infection (see Appendix Table A1). Inclusion criteria were patients (1) aged between 16 and 50 years, (2) who underwent single-bundle ACL reconstruction with autologous hamstring tendons, and (3) who had follow-up MRI at least 1 year after surgery. Exclusion criteria were (1) a concomitant ligament injury that required surgical repair and (2) a delayed onset of postoperative joint infection (>3 months after surgery). Ultimately, 26 patients were included. The mean follow-up was 47 months (range, 12-156 months). Informed consent were provided by participants to collect their imaging files.
All patients in the infection group were then individually matched with 3 patients who received single-bundle ACL reconstruction without postoperative infection (control group) by sex, age, tunnel drilling technique, type of graft and fixation, and similar follow-up time (Figure 1; see Appendix Table A2 for detailed matching criteria). All the matching variables were reported to be associated with graft maturity, bone tunnel enlargement, and cartilage wear. 6,30,32,36,38 All patients in the control group received MRI scans at a regular clinic follow-up visit; patients with reinjury and other complications were excluded and replaced.
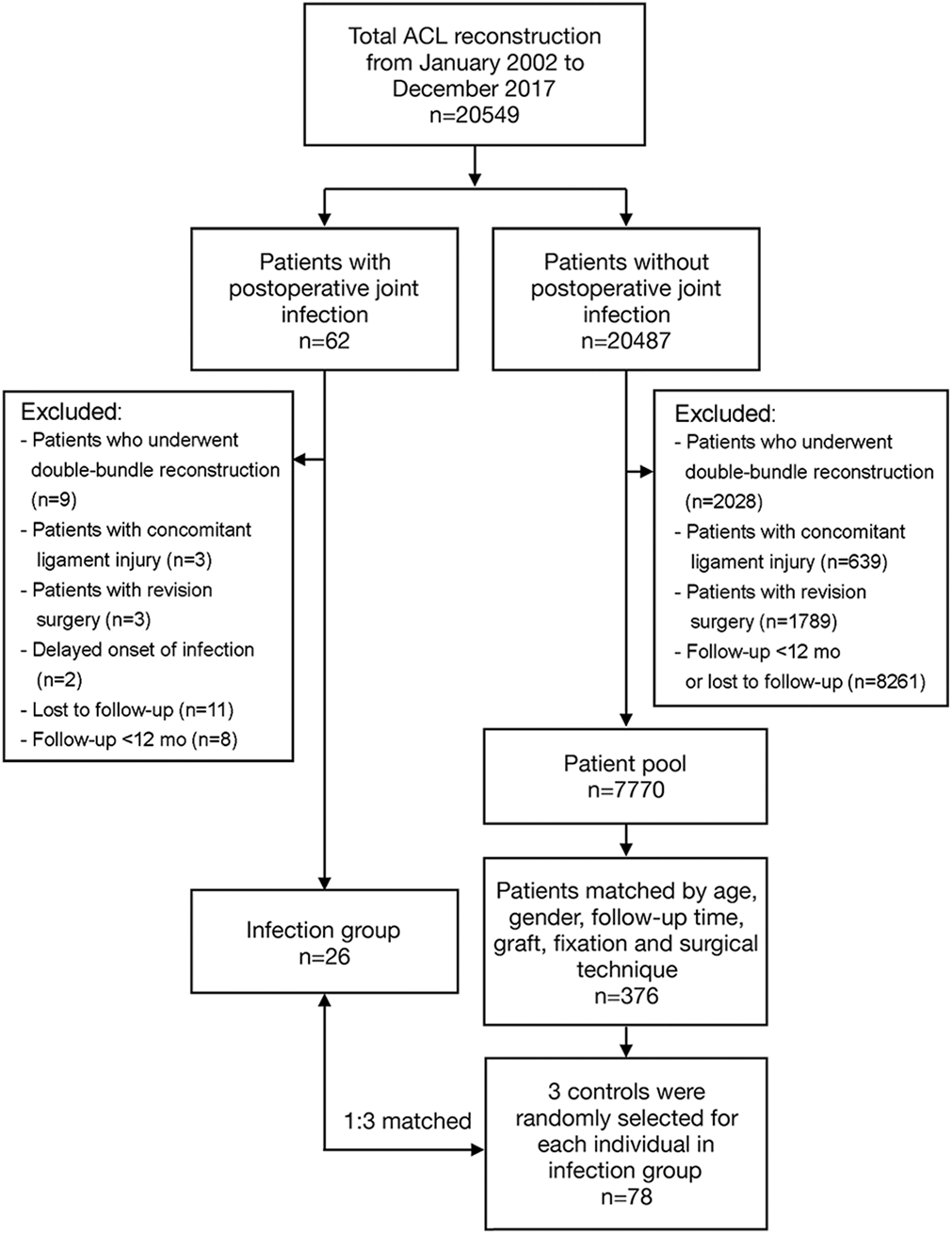
Flow diagram of patient selection. ACL, anterior cruciate ligament.
Surgical Procedures
ACL reconstruction was performed by senior surgeons at our institution. A standard diagnostic arthroscopy was performed in each patient. The integrity of cartilage was assessed using the International Cartilage Repair Society (ICRS) classification. 16 Coexisting meniscal and chondral injuries were treated before ACL reconstruction.
During ACL reconstruction, autologous hamstring tendons (semitendinosus and gracilis) were harvested. The grafts were prepared on the back table. The femoral and tibial tunnels were drilled according to the size of the graft. The tibial tunnel was drilled using a tibial guider, which was placed at the center of the ACL footprint to achieve anatomic reconstruction. In the infection group, we used the transtibial technique in 13 patients and the anteromedial portal in the rest of the patients to drill the femoral tunnel. On the femoral side, grafts were fixed with a cortical device (Endobutton with continuous-loop suture; Smith & Nephew). A staple, Intrafix screw (Depuy Mitek), or bioabsorbable interference screw (Biosure; Smith & Nephew) was used on the tibial side.
Joint Infection
The average time to the presentation of infection was 17 days (range, 5-62 days) after surgery. Of the 26 patients included, 19 patients had an acute onset of symptom (<3 weeks) and 7 patients had a subacute onset of symptoms (3 weeks-3 months). The symptoms at onset included fever, knee swelling, severe knee pain, local erythema, local warmth, and decreased range of motion.
Once joint infection was suspected, patients were emergently admitted. Synovial fluid aspiration and culture were done in all patients suspected to have an infection. Among the patients included in our study, 14 cultures showed coagulase-negative Staphylococcus, 3 showed Staphylococcus aureus, and 9 had a negative result (the diagnosis was made according to symptoms and synovial fluid test). Immediate intravenous antibiotics were initiated after aspiration. Twenty-two patients underwent urgent arthroscopic irrigation and debridement and synovectomy with graft retention. Four patients continued nonoperative treatment because of improved symptoms and received aspirational irrigation and drainage in addition to intravenous antibiotics. All the patients were eventually cured of the infection and were discharged after their symptoms resolved and laboratory results returned normal. After discharge, oral antibiotics were administrated for another 2 weeks.
MRI Evaluation
A 3.0-T MRI unit (General Electric Healthcare) with an 8-channel coil was used for measurement. A proton density–weighted fat suppressed turbo-spin echo sequence was taken to calculate the SNQ of the graft as described by Cavaignac et al 6 (Figure 2). Graft signal intensity was measured at the proximal, middle, and distal ends of the graft. The SNQs of the 3 segments (proximal, middle, distal) and the average SNQ were determined using the following equation:

Measurement of SNQ. Five regions of interest (ROIs; circles) were placed on the graft (proximal, middle, distal), posterior cruciate ligament, and background (ROI = 0.05 cm2). SNQ, signal-to-noise quotient.
To determine graft maturity based on the graft signal intensity within the knee joint, we used the Howell classification 13 :
Grade 1: Homogeneous, low-intensity signal indistinguishable from the posterior cruciate ligament and patellar tendon.
Grade 2: Normal ligament signal over at least 50% of its volume, intermingled with portions that have increased signal intensity.
Grade 3: Increased signal intensity over at least 50% of its volume, intermingled with portions that have a normal ligament signal.
Grade 4: Diffuse increase in signal intensity without strands with a normal ligament appearance.
The protocol described by Ge et al 8 was used to measure graft healing at the bone-graft interface in the tibial tunnel:
Grade 1: Low intensity, no fibrosis at the bone-graft interface, full attachment.
Grade 2: High intensity over a portion of the interface.
Grade 3: High intensity over the entire bone-graft interface, poor attachment.
The anteroposterior and mediolateral diameters at the tunnel aperture, midsection, and exit of tibial and femoral tunnels were measured on T1-weighted images (in millimeters) using a refined method based on that described by Weber et al 36 (Figure 3). Enlargement of the tibial and femoral tunnels was calculated with the following formula:

(A and C) Measurement of the anteroposterior diameter of the femoral and tibial tunnels. The tunnel cross section on the sagittal view was an ellipse, so measurement was performed along the minor axis to reflect the true diameter. (B and D) Measurement of the mediolateral diameter of the femoral and tibial tunnels. FTa, femoral tunnel aperture; FTe, femoral tunnel exit; FTm, femoral tunnel midsection; TTa, tibial tunnel aperture; TTe, tibial tunnel exit; TTm, tibial tunnel midsection.
The cartilage integrity at the primary ACL reconstruction was assessed using ICRS grading 16 (grade 0: normal cartilage; grade 1: superficial lesions, fissures and cracks, soft indention; grade 2: lesions extending down <50% of cartilage depth; grade 3: cartilage defects extending down >50% of cartilage depth; grade 4: complete loss of cartilage thickness) and recorded in the surgical notes. The sum of the ICRS grades in each of the compartments was calculated; for example, the sum of the ICRS grade for patellar and trochlear cartilage represents the cartilage integrity of the patellofemoral joint (PFJ). At follow-up, cartilage integrity was assessed according to the Whole-Organ Magnetic Resonance Imaging Score (WORMS) 27 of the knee on sagittal and axial T2-weighted Fast Spin Echo images (Figure 4).
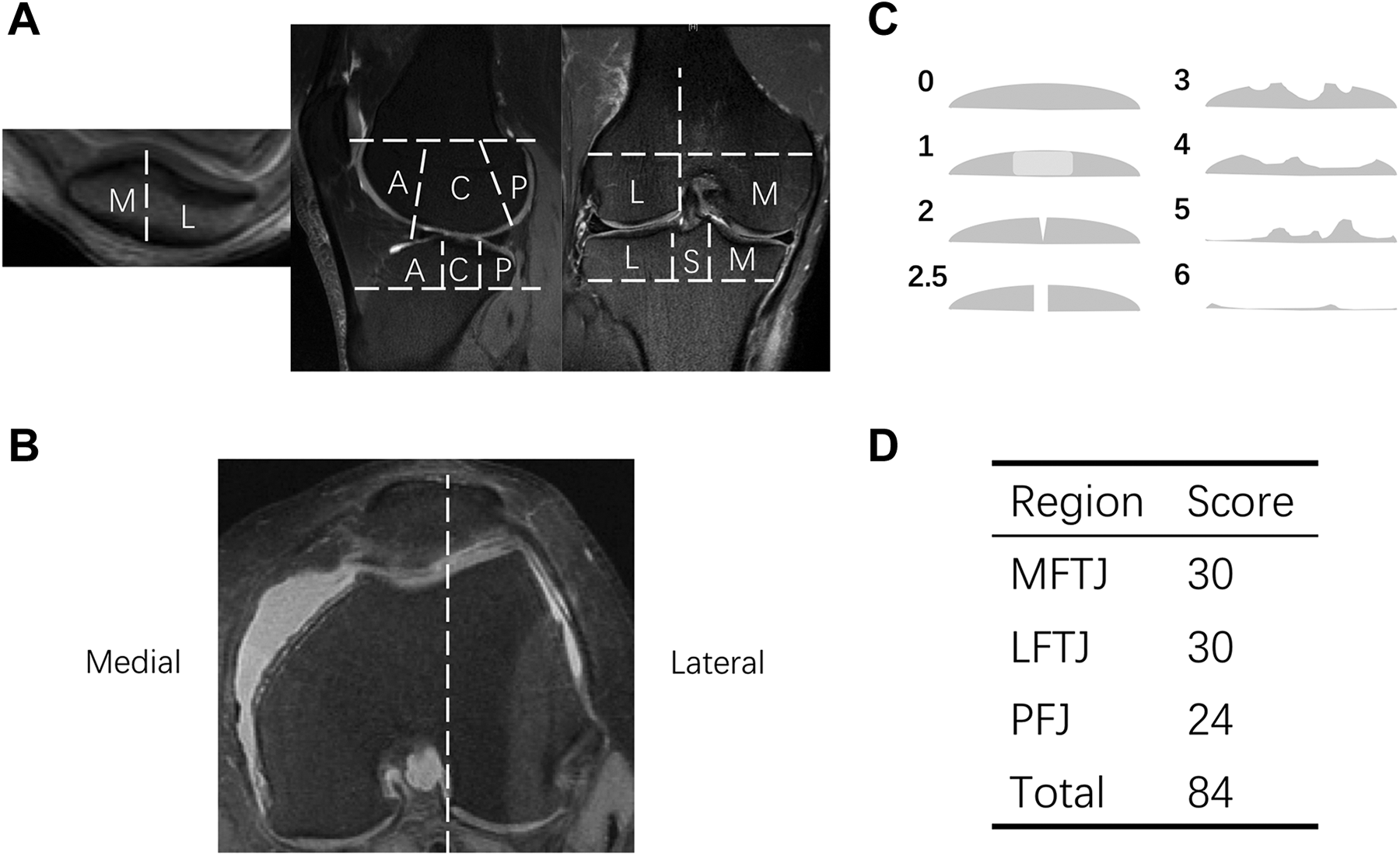
Whole-Organ Magnetic Resonance Imaging Score (WORMS). (A and B) Regional subdivision of articular surfaces. (C) Eight-point scale for scoring articular cartilage signal and morphology. (D) Maximum score of articular cartilage in different joint compartments. A, anterior; C, central; L, lateral; LFTJ, lateral femorotibial joint; M, medial; MFTJ, medial femorotibial joint; P, posterior; PFJ, patellofemoral joint; S, subspinous.
Statistical Analysis
A power analysis was performed. A minimal sample size of 9 in each group was required to detect a difference of 1 in the SNQ between the groups. For the power analysis, the α value was set at .05 and the β value at 0.8. All statistical analysis was performed with SPSS (version 20.0; IBM Corp). Numeric variables are denoted as mean ± standard deviation (SD), and ordinal variables are denoted as median (range). The chi-square test was used to compare categorical variables. The Student t test was applied to analyze the difference of SNQ and bone tunnel enlargement in the infection and control groups. The Wilcoxon signed rank test was used for cartilage integrity. P < .05 was considered statistically significant.
Results
Patient Characteristics
The groups were equivalent in terms of age, sex, follow-up time, surgical technique, type of graft, and fixation type (Table 1).
Patient and Surgery Characteristics in the Infection and Control Groups a
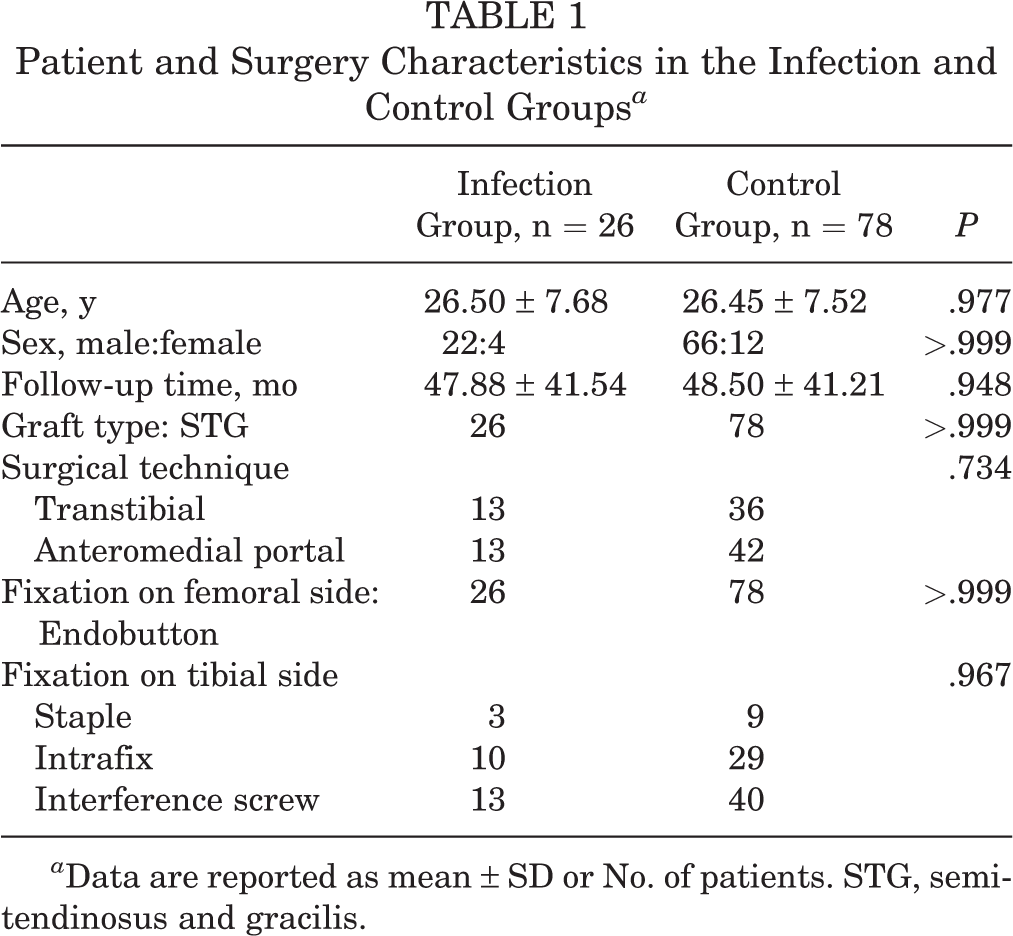
a Data are reported as mean ± SD or No. of patients. STG, semitendinosus and gracilis.
Graft Ligamentization and Healing
The mean SNQ was 20.01 ± 12.08 in the infection group and 7.61 ± 6.70 in the control group (P = .014) (Figure 5). The homogeneity of the graft signal was significantly inferior in the infection group, both within the knee joint (P = .031) and at the bone-graft interface (P = .037) (Table 2).

Signal-to-noise quotient (SNQ) at the proximal, middle, and distal sites of the graft and the average graft SNQ. Error bars indicate ±2 SE. *Statistically significant difference between groups (P < .05).
Graft Signal Intensity Within the Knee Joint and at the Bone-Graft Interface a
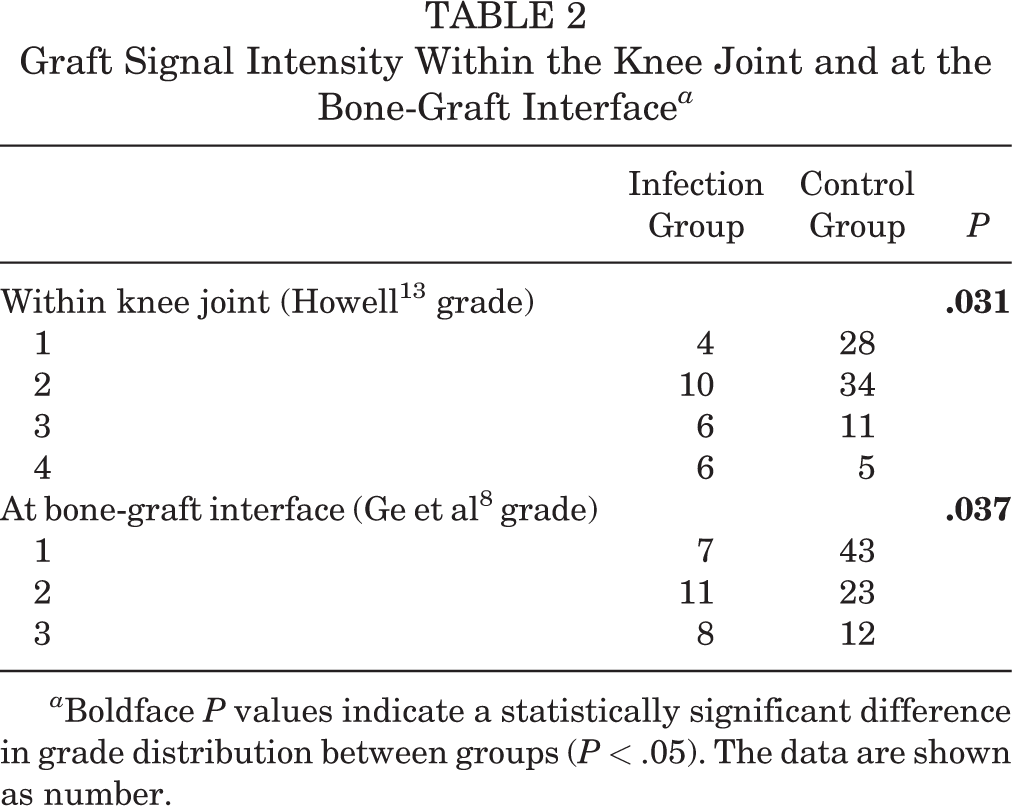
a Boldface P values indicate a statistically significant difference in grade distribution between groups (P < .05). The data are shown as number.
Cartilage Integrity
There was no significant between-group difference in the grade of cartilage lesion in each compartment at the time of surgery. At follow-up, the cartilage of the PFJ and lateral femorotibial joint (LFTJ) in the infection group was observed to have higher-grade lesions.
Further investigation found that the cartilage lesion in the medial femorotibial joint (MFTJ) was worse than that in the LFTJ at the primary reconstruction in both groups (P = .002); however, the grade of the cartilage lesion in the LFTJ became significantly worse than that in the MFTJ at follow-up (P < .001) (Table 3).
Cartilage Morphology Under Arthroscopy at Surgery and on MRI scans at Follow-up a

a Boldface P values indicate a statistically significant difference between groups (P < .05). The data are shown as median (range). Dashes indicate not applicable. ICRS, International Cartilage Repair Society; LFTJ, lateral femorotibial joint; MFTJ, medial femorotibial joint; PFJ, patellofemoral joint; WORMS, Whole-Organ Magnetic Resonance Imaging Score.
Bone Tunnel Enlargement
Bone tunnel enlargement was significantly larger at the femoral tunnel aperture in the infection group; however, there was no difference in the degree of bone tunnel enlargement elsewhere (Table 4).
Enlargement at Tunnel Aperture, Midsection, and Exit of Tibial and Femoral Tunnels a
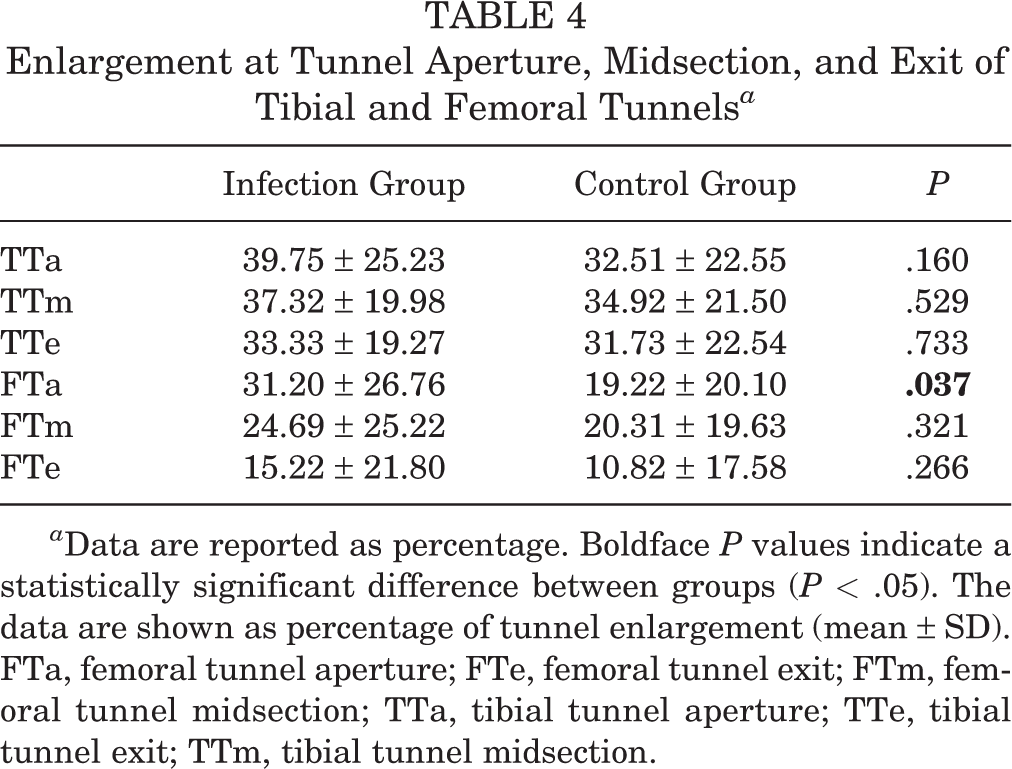
a Data are reported as percentage. Boldface P values indicate a statistically significant difference between groups (P < .05). The data are shown as percentage of tunnel enlargement (mean ± SD). FTa, femoral tunnel aperture; FTe, femoral tunnel exit; FTm, femoral tunnel midsection; TTa, tibial tunnel aperture; TTe, tibial tunnel exit; TTm, tibial tunnel midsection.
Discussion
The main finding of the present study was that graft maturity was inferior in patients who experienced joint infection after ACL reconstruction as the graft SNQ, Howell scale, and signal at the bone-graft interface were significantly higher in the infection group. In addition, joint infection was found to be associated with worse articular cartilage degeneration and enlargement of the femoral tunnel at the aperture.
In this study, in order to obtain more convincing results, we matched the factors that may play a role in graft maturity and bone tunnel enlargement in both groups, including age, follow-up time, type of graft and fixation, and bone tunnel placement technique. 6,30,32,36,38
Acute infectious arthritis can cause a great deal of damage to the joint within a short span of time. Researchers have found that bacteria can directly invade joint tissue, causing cell death and extracellular matrix degradation. 24,31 Meanwhile, the rapid bacterial proliferation consumes nutrients and oxygen in the synovial fluid, hindering cell repair and growth. Besides direct damage, acute inflammation invoked by bacterial infection promotes the production of inflammatory cytokines and the infiltration of inflammatory cells, such as neutrophils and macrophages. These cells and molecules can cause cell damage and extracellular matrix degradation, leading to further destruction of the joint. 20 Autoimmune-mediated tissue injuries induced by molecular mimicry between the host and pathogen may have latent disturbance on the joint after elimination of the pathogen. 3,19
To achieve final ligamentization, tendon graft will go through 3 characteristic phases: (1) early phase with central graft avascular necrosis and hypocellularity, (2) proliferation phase with revascularization, and (3) ligamentization phase with collagen fiber rearrangement and cell redifferentiation that resembles normal ACL. 15,18,25 Postoperative joint infection frequently occurred in the early stage after surgery, which coincides with the avascular necrosis phase of graft remodeling. The mean SNQ value of the graft in the infection group was 20.01 ± 12.08, which was significantly higher than the mean value in the control group (7.61 ± 6.70; P = .014). The SNQ values of single-bundle hamstring tendon graft reported in other literature range from 5.2 ± 4.5 at 1 year to 2.98 ± 5.48 at 2 years after surgery, 6,17 which are comparable to our result in the control group. The findings were of great clinical importance. Calculation of the SNQ value is a good method to assess graft maturity and has been found to be associated with type of fixation and surgical technique. 2,17 In the present study, these factors were matched. The higher SNQ in the infection group indicates diminished graft revascularization and biomechanical properties. 5,9,29,37
The cartilage integrity observed under arthroscopy at the primary reconstruction was similar between the infection and control groups. At follow-up, the cartilage lesion of the PFJ and LFTJ in the infection group was worse than that of the controls, implying that postoperative joint infection can damage articular cartilage. We noticed that the grade of cartilage lesions in the weightbearing region of the LFTJ and medial trochlea was worse in the infection group; however, no significant difference was found in the medial compartment between groups. At the time of reconstruction, the baseline cartilage integrity in the medial compartment was worse than that in the lateral compartment in both groups, while at follow-up, the cartilage lesion in the lateral compartment became worse than that in the medial compartment in both groups. Other research found similar results. 21 An in vivo biomechanical study also supports these findings. 12 Thus, we deduce that the main reason for the spatial characteristics of cartilage lesions before and after ACL reconstruction is the change in knee biomechanics, while joint infection only contributes as a co-factor that synergizes the progression of cartilage degeneration in the PFJ and LFTJ. In the medial compartment, the contact pressure abates after ACL reconstruction, making joint infection alone inadequate to cause obvious differences between the groups.
After ACL reconstruction, the bone tunnel undergoes a remodeling process before attaining complete graft-to-bone healing. Researchers have found that inflammatory mediators in synovial fluid can enter the space between the graft and bone after ACL reconstruction and lead to osteolysis. 33 Thus, we hypothesized that postoperative septic arthritis can exacerbate bone tunnel enlargement. In the infection group, the mean enlargement was 31.20% ± 26.76% at the femoral tunnel aperture, while in the control group, the mean enlargement was 19.22% ± 20.10% (P = .037). However, although the degree of bone tunnel enlargement elsewhere was larger in the infection group, no significant difference was found. One possible reason for this may be that joint infection contributes to the early stage of bone tunnel enlargement, but the exact effect is not able to be detected years after the remodeling process. Weber et al 36 found that both tibial and femoral tunnels are dynamic, with tunnel expansion for the first 12 to 24 weeks, followed by tunnel closure until 2 years after surgery. The early tunnel enlargement was significant during the first 6 postoperative weeks, which might be further enhanced by acute infection. Years after tunnel closure, the early effect was compensated and became hard to detect. All patients involved in this study were followed up for at least 12 months; hence, there was scarce radiological evidence of early bone tunnel enlargement. Nevertheless, systematic errors from MRI measurement should not be ignored. It was hard to distinguish 3 segments on the sagittal view in the case of more vertical and shorter femoral tunnels, which could cause inaccuracy when measuring the femoral tunnel aperture.
Limitations
There are several limitations to this study. First, the follow-up rate was low. Of the 62 patients who experienced a joint infection after single-bundle ACL reconstruction, only 26 patients had valid MRI scanning. Hence, the true pattern of bone tunnel and graft remodeling might have been misinterpreted. However, the present cohort is the largest cohort reported to date; further effort will be made to collect follow-up statistics and investigate clinical outcomes after joint infection. Second, because we could not obtain an oblique sagittal view of the bone tunnel, we had to estimate the correct anteroposterior diameter on MRI scans. Moreover, interobserver and intraobserver agreement of the MRI measurements were not calculated. Third, we did not collect information on concomitant injuries of the patients. Nevertheless, there was no reported relationship between concomitant injury and bone tunnel widening or graft remodeling after surgery.
Currently, there are no relative studies investigating the mechanism behind inferior graft maturity and cartilage injury caused by joint infection. Because we cannot obtain a specimen from patients who have an infection, further animal experiments are needed to continue to observe the actual effect of bacterial infection on ACL graft and cartilage.
Conclusion
Graft ligamentization and incorporation graft maturity were inferior in patients who experienced joint infection after ACL reconstruction, as the graft SNQ, Howell scale, and signal at the bone-graft interface were significantly higher in the infection group.
Footnotes
APPENDIX
Patient Matching Criteria a

| Variable | Criteria |
|---|---|
| Sex (male or female) | Strictly matched |
| Age | Allow ±1-y difference |
| Follow-up time | Allow ±3-mo difference |
| Graft type (hamstring tendon autograft or BPTB autograft) | BPTB graft was excluded |
| Fixation on femoral side (Endobutton or interference screw) | Interference screw was excluded |
| Fixation on tibial side (staple or Intrafix screw or interference screw) | Patients with staple were strictly matched; perfect match was desirable in patients with intratunnel fixation, while similar fixation was allowed when perfect match was not found in the pool |
| Surgical technique (transtibial technique or anteromedial portal) | Perfect match was desirable, while other technique was allowed when perfect match was not found in the pool because the transtibial technique was outdated |
a BPTB, bone–patellar tendon–bone.
Final revision submitted June 28, 2022; accepted July 12, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the National Research and Development Program of China (grant No. 2018YFF0301100). AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from Peking University Third Hospital.