Abstract
Background:
The docking technique is widely used to perform ulnar collateral ligament (UCL) reconstructions because of its high failure torque and reliable clinical outcomes. A double–cortical button technique was recently described, with advantages including the ability to tension the graft at the ulnar and humeral attachments and the creation of single bone tunnels.
Purpose/Hypothesis:
To compare the biomechanics between the docking and double-button UCL reconstruction techniques using cadaveric specimens. We hypothesized that there would be no difference in postoperative stiffness or maximum strength between the techniques.
Study Design:
Controlled laboratory study.
Methods:
Eight matched pairs of cadaveric elbow joints underwent controlled humeral valgus torsion cycles in a test frame. Toe region stiffness, elastic region stiffness, and maximum torque were measured during a 4-step protocol: intact, injured, reconstructed (10 and 1000 cycles), and ramp to failure. Graft strains were calculated using 3-dimensional motion capture.
Results:
After 10 cycles, intact ligaments from the docking and double-button groups exhibited mean ± SD elastic torsional stiffness of 1.60 ± 0.49 and 1.64 ± 0.35 N·m/deg (P = .827), while docking (1.10 ± 0.39 N·m/deg) and double-button (1.05 ± 0.29 N·m/deg) reconstructions were lower (P = .754). There were no significant differences in maximum torque between the docking (3.45 ± 1.35 N·m) and double-button (3.25 ± 1.31 N·m) groups (P = .777). Similarly, differences in maximum graft strains were not significant between the docking (8.1% ± 7.2%) and double-button (5.5% ± 3.1%) groups (P = .645). The groups demonstrated similar decreases in these measures after cyclic loading. Ramp-to-failure testing showed no significant differences in ultimate torque between the docking (8.93 ± 3.9 N·m) and double-button (9.56 ± 3.5 N·m) groups (P = .739).
Conclusion:
The biomechanical behavior of the double-button technique was not significantly different from that of the docking technique. Both reconstruction techniques restored joint stability, but neither fully recapitulated preinjury joint stiffness.
Clinical Relevance:
With its procedural advantages, results preliminarily support the use of the double-button reconstruction technique for UCL reconstruction as a reliable single-tunnel technique for primary or revision cases.
Keywords
Ulnar collateral ligament (UCL) injuries typically occur in overhead throwing athletes, wrestlers, and gymnasts, with increasing incidence over the past 2 decades.
12,17
Surgical reconstruction of the UCL is often required for successful return to competition, and likewise, the incidence of UCL reconstruction surgery has also increased. Leland et al
17
reported a 13% incidence of UCL reconstruction among all Major League Baseball and Minor League Baseball players, a 3% increase over 6 years
Multiple techniques for surgical reconstruction of the UCL have emerged over the past 4 decades, including several iterations of the original Jobe technique 14 and the docking technique developed by Gibson et al. 11 and Rohrbough et al. 23 Clinical observations have shown that docking reconstruction provides the highest return-to-play rates and lower complication rates when compared with the Jobe and modified Jobe techniques. 9,24,26 However, laboratory studies have demonstrated that Jobe reconstructions typically fail at the ulnar tunnel, while docking reconstructions most commonly fail at the suture-bone interface. 22,26
Several groups have described novel UCL reconstruction techniques that use 2 cortical buttons to achieve strong single-tunnel fixation for the humerus and ulna. 4,13,16,20 –22 Surgical advantages of the dual cortical button reconstruction include a smaller surgical incision and a single-tunnel bone-preserving technique that is well-suited for primary and revisions cases. In a clinical study, competitive throwers treated with the double-button technique demonstrated similar return-to-play rates to those of Major League Baseball pitchers. 11 Additionally, a biomechanical study noted that a bisuspensory reconstruction technique resulted in similar ramp-to-failure loads to the docking technique. 13 However, there is limited information regarding the biomechanical performance of double-button reconstructions during cyclic loading.
The goal of this cadaveric study was to perform double-button and docking techniques on matching pairs of cadaveric specimens and to compare the biomechanical properties of the UCL reconstructions. We hypothesized that there would be no difference in postoperative joint stiffness or reconstructed ligament strength between the reconstruction techniques.
Methods
Specimen Preparation and Care
For this study, we used 16 frozen arm specimens from 8 cadaveric donors (7 male, 1 female; age range, 74-87 years). Specimens were stored at –20°C and underwent 3 freeze-thaw cycles to room temperature throughout the duration of testing. The palmaris longus was harvested when present; in instances of an absent palmaris longus, the flexor carpi radialis was instead sectioned for the right and left arms (n = 2). For each specimen, a 65-mm, medially based incision was made just anterior to the medial epicondyle with a No. 10 scalpel. The flexor pronator mass was detached from the humeral origin of each specimen before all testing conditions. Once grafts were harvested, amputations were performed 20 cm proximal/distal from the elbow joint line, and soft tissue was dissected to expose 10 cm of bone on the proximal and distal ends of the humerus and forearm (Figure 1A). The exposed bone ends were then potted in polycarbonate tubes filled with polymethyl methacrylate.

(A) Schematic of the dissection of the cadaveric arm specimens with amputations of the humerus and forearm located 20 cm from the elbow joint line. Soft tissue was maintained about the joint itself, while exposed bones were potted in polymethyl methacrylate (PMMA). (B) Schematic of the arm in the testing position. The elbow was flexed to 90° with the forearm held securely in a horizontal position. A valgus torque was applied along the long axis of the humerus. (C) Photograph of the test setup. Aluminum fixtures held the bones in place, and a universal joint was used to prevent off-axis torques. Retroreflective marker clusters were rigidly affixed to the bones for 3-dimensional (3D) motion tracking.
Torsional Testing Protocol
Specimens were subjected to mechanical tests that are representative of the late cocking and early acceleration phases of a baseball pitch motion 19 by applying a valgus torsion to the humerus with the elbow flexed to 90° (Figure 1B). 3,8,15,19 To create this model, arms were rigidly affixed with a custom aluminum jig to a universal test frame (ElectroForce 3550; TA Instruments) with a 1100-N/14-N·m load/torque cell (Figure 1C). The potted end of the humerus was fixated to the load/torque cell with an in-line universal joint using blunt tip screws. The forearm was positioned in full supination such that the radius and ulna were parallel to each other. The elbow was flexed to 90°, the angle shown to undergo the highest valgus load during pitching. 3,5,8,10 To maintain this pose, the potted end of the forearm was affixed to a rigid horizontal jig. The upright humerus was rotated about its long axis by a rotational actuator at selected velocities. Time, rotation, and reaction torque were measured with the test frame at a sampling rate of 100 Hz.
Output graphs of torque versus rotation were constructed, and a custom bilinear curve-fitting algorithm (MATLAB; MathWorks) was used to identify the toe region and elastic region of each curve (Figure 2). This algorithm used the onboard least squares curve-fitting function in MATLAB to determine the best-fit slopes for the toe and elastic regions.
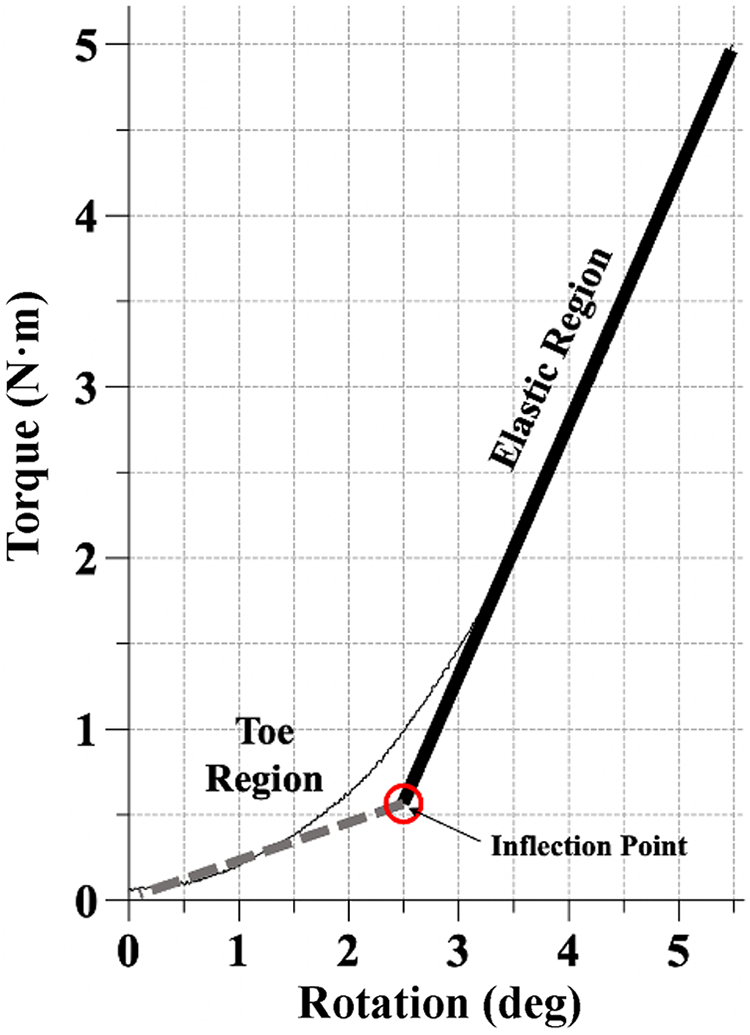
Example of a torque-rotation plot. A least squares curve-fitting algorithm was used to make best-fit linear approximations of the toe (dashed line) and elastic (solid line) regions. An inflection point represents the rotation at which the transition from the toe to elastic region occurs.
3-Dimensional Motion Tracking to Assess Graft Strain
We used a 3-dimensional (3D) motion capture to quantify graft strain during testing, similar to previously established methods. 25 Briefly, 6 cameras (OptiTrack Motion Track Systems; NaturalPoint) recorded the 3D motions of retroreflective marker clusters that were attached to the humerus and ulna (Figure 1C). Under static conditions, a retroreflective wand was used to determine the 3D locations of the insertion points of the UCL/graft relative to the marker clusters on the humerus and ulna. Virtual coordinate systems were established at these sites, and a gauge length for the UCL/graft was calculated by calculating the Euclidean distance between the locations. Maximum displacements during the tests were then normalized as percentage strain. Calculations of strain, rather than measures of displacement, were used to normalize differences in cadaveric specimen size. Registration errors during calibration of the capture volume were <0.2 mm. We also performed a brief experiment to confirm the accuracy of our system. Marker clusters were perturbed by 5.0 mm using the universal test frame, and 3D motions were recorded. Using data from 3 trials, the root mean square error between actuator displacement and marker displacement was 0.127 mm, which represents a 2.54% error.
Torsional Testing of Intact Specimens
A series of torsional tests was performed sequentially to test the intact, injured, and reconstructed joints. First, all specimens underwent quasistatic testing in their intact state. A valgus rotation was applied to the humerus at a controlled rate of 1 deg/s from the neutral position until 5 N·m of torque was achieved. The humerus then was returned to the neutral position. This was repeated for 10 cycles, and the maximum rotation required to reach 5 N·m was recorded. The 5-N·m threshold of torque was chosen in accordance with previous studies and represents the physiological moments exerted upon the joint in valgus rotation without damaging the construct. 3,5,8,15,19 Using the time, rotation, and torque data collected by the load/torque cell, torque versus angular displacement curves were created for each specimen.
Simulation and Confirmation of an Injury
Next, a UCL injury for the elbow specimens was simulated and confirmed with a similar test procedure. Without being removed or altered within the test frame after intact state testing, a full-thickness tear of the midsection of the anterior bundle of the UCL was created with a No. 10 scalpel. Each specimen then underwent 10 more cycles of humeral valgus rotation. Here, specimens were rotated to the angle at which 5 N·m of resistance was achieved in the intact state. On the 10th cycle, rotational stiffness was again calculated from the torque–angular displacement curve, and the maximum torque on the 10th cycle was recorded.
Surgical Reconstruction
The specimens were then organized into matched pairs of left and right elbows from the same cadaver of origin. Elbows from each pair were randomized to the docking or double-button group (n = 8; 4 left elbow specimens and 4 right elbow specimens for both groups).
Docking Technique
The docking technique, described previously, 23 was executed in the same manner for each specimen. Briefly, drill guides were used to create 3.5 mm–diameter ulnar tunnels and a 4.2-mm diameter humeral tunnel. The prepared graft was then passed through the ulnar tunnel and docked in the humeral tunnel. After cycling the joint to eliminate graft creep, a varus load was applied to the elbow before knot tying over the medial epicondylar ridge.
Double-Button Technique
For the remaining 8 specimens, the double–cortical button suspension technique was performed as previously described 4 (see clinical radiographs in Figure 3). The autograft was first prepared by being folded equally into half, and FiberWire (Arthrex) suturing was used to secure the closed loop end in a luggage-type fashion and to secure the free ends using a Krakow running locked stitch. Cortical buttons (Arthrex, Inc) were placed in a sliding-type fashion around the FiberWire at the proximal and distal ends. A guide pin was then placed at the sublime tubercle and directed to exit the radial cortex of the ulna distal to the proximal radial ulnar articulation.

Radiographs of the single-tunnel dual suspensory button technique from a clinical case. 4 (A) A coronal plane radiograph with arrows oriented perpendicularly to the flat surface of the 2 buttons. The humeral tunnel remains distal to the olecranon fossa, and the ulnar tunnel exits distal to the proximal radioulnar joint. (B) An oblique image with the same buttons (arrows). (C) A sagittal plane radiograph with the same buttons (arrows).
The elbow was guided through a flexion-extension arc to identify the isometric point, and a suture loop was fixed to the ulnar guide pin and used to identify the humeral origin of the UCL at this point. The humeral guide pin was drilled bicortically to the lateral cortex of the humerus, with care to keep the guide pin distal to the olecranon fossa, and confirmed using fluoroscopy. A reamer ranging in size from 5 to 5.5 mm was used to ream the medial cortex of the ulna and humerus to accommodate the graft to a minimum depth of 15 mm. The graft-and-button construct was then placed through the ulna and secured by flipping the button. The graft was tensioned until the graft advanced 15 to 20 mm into the osseous socket of the sublime tubercle, and the humeral cortical button was then introduced through the humeral tunnel and flipped.
After confirmation of the button placement using fluoroscopy, the graft was tensioned until 20 mm was positioned within the humeral tunnel. The proximal and distal ends were then pulled sequentially for final tensioning, and the elbow range of motion was cycled to confirm graft isometry and to reduce graft creep. If needed, graft tension was adjusted and this process was repeated. The graft was secured by suturing the excess end back to itself at the tunnel apertures using a free taper needle and tying the sutures down with a triple overhand knot.
Functional Assessment
Additional mechanical tests were performed to assess postoperative joint function. Reconstructed specimens underwent the quasistatic test used for the injured state, followed by a cyclic test (1000 cycles at 1 Hz 19 ) and finally a ramp-to-failure test (1 deg/s). 8,15 Failure was defined as the point immediately before a sudden decrease in torque. If clear, instantaneous failure did not occur, the maximum torque measured throughout 25° of humeral valgus rotation was recorded. This represents a reasonable value for the maximum range of motion of humeral valgus rotation. 7 A summary of the overall testing protocol is provided in Figure 4.

(A) The rotational velocity, cutoff threshold, and number of cycles performed for each phase of the torsional tests. (B) Representative torque-rotation plot of results from the elbow joint after 10 cycles of loading: (a) intact (black), (b)injured (dotted red), and (c) reconstructed (dashed blue).Dash indicates not applicable.
Results
The full-thickness UCL tear led to significant changes in joint function versus the intact state, and there were no significant differences in injury severity between the docking and double-button groups. Comparison between the study groups showed no significant differences in toe region stiffness, elastic region stiffness, and maximum torque. Although the mean graft strain values for the docking group were consistently higher than those for the double-button group, there were no significant differences between reconstruction groups (Table 1).
Comparison Between Groups of Mechanical Values According to Testing Stage a

a Data are presented as mean ± SD.
Specimens within the docking and double-button groups saw a statistically significant decrease in stiffness and maximum torque between the intact and injured conditions (Table 2, Figure 5). When compared with the intact state, the injured specimens in the docking group demonstrated a 66% decrease in toe region stiffness, a 65% decrease in elastic region stiffness, and a 66% decrease in maximum torque (P < .001 for all), and the injured specimens in the double-button group had a 74% decrease in stiffness in the toe region, a 73% decrease in the elastic region, and a 71% decrease in maximum torque (P < .001 for all).
P Values for Within-Group Comparison of the Mechanical Values According to Nondestructive Testing Stages a

a Bold P values indicate statistically significant difference between testing stages (P < .05).
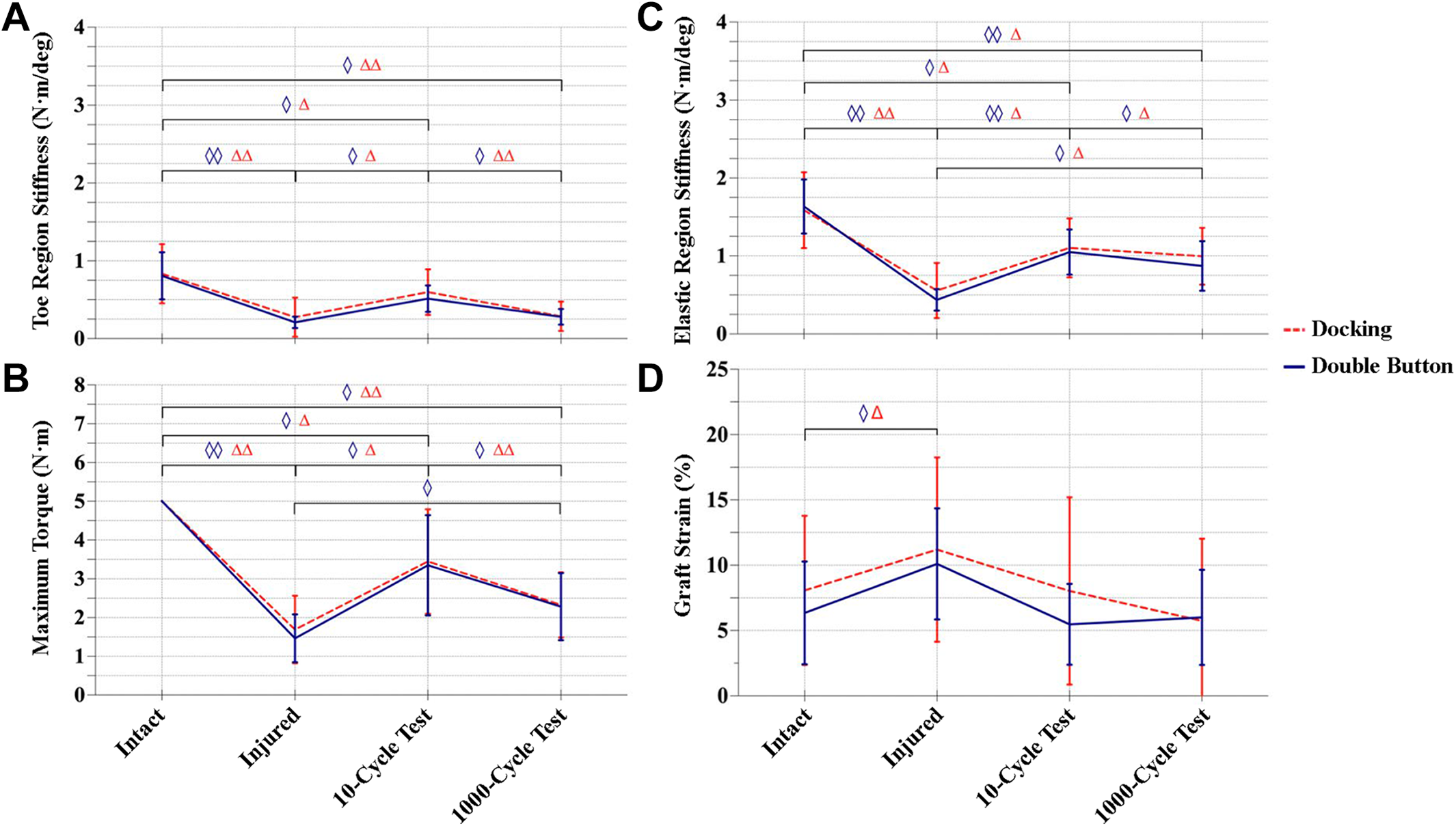
Changes in mechanical properties during cyclic testing of specimens in the docking and double-button groups: (A) toe region stiffness, (B) elastic stiffness, (C) maximum torque, and (D) ligament/graft strain. Data are presented as mean ± SD. Statistically significant differences: (◊) P < .05 between specimens in the double-button group; (Δ) P < .05 between specimens in the docking group; (◊◊) P < .001 between specimens in the double-button group; and (ΔΔ) P < .001 between specimens in the docking group.
Both UCL reconstruction techniques led to significant improvements in stiffness and maximum torque after 10 cycles as compared with the injured state; however, neither technique achieved preinjury function (Figure 5). Specimens reconstructed using the docking method showed a 114% increase in stiffness in the toe region as compared with the injured condition (P = .008), but stiffness was still 28% lower than the intact condition (P = .049). In the elastic region, stiffness increased by 96% versus the injured condition (P = .002) but was still 31% less stiff than the intact condition (P = .006). Specimens reconstructed with the docking method only achieved 69% of maximum torque compared to intact specimens (5 N·m) after 10 cycles (P = .014) (Table 2).
Specimens reconstructed using the double-button method showed a 143% increase in toe region stiffness as compared with the injured condition (P = .002), but stiffness was still 37% lower than the intact specimens (P = .044). Similarly, for elastic stiffness, specimens showed a 139% increase in stiffness versus the injured condition (P < .001), but this was still 36% lower than intact specimens (P = .002). Double-button reconstruction led to 65% maximum torque as compared with the intact state (P = .007) (Figure 5, Table 2).
After 1000 cycles, there were significant degradative changes in stiffness and maximum resistive torque of the reconstructions, and these changes were similar between groups. When compared with immediate postreconstruction analysis (10 cycles), specimens reconstructed with the docking technique demonstrated a 52% decrease in toe region stiffness (P < .001) and a 10% decrease in elastic region stiffness (P = .002). Maximum torque for these specimens decreased by 33% during the same interval (P < .001). From 10 to 1000 cycles, specimens in the double-button reconstruction group showed a 45% decrease in toe region stiffness (P = .002), a 17% decrease in elastic region stiffness (P = .031), and a 30% decrease in maximum torque (P = .005) (Figure 5, Table 2).
Strains of the intact ligaments were 8.1% ± 5.7% and 6.4 ± 3.9% (mean ± SD) for the docking and double-button groups, respectively; these values increased significantly to 11.2% ± 7.1% and 10.1% ± 4.3% in the injured condition (P = .004 and .016, respectively). Surgical reconstruction restored strain values in the docking (8.1% ± 7.2%) and double-button (5.5% ± 3.1%) groups, which were similar to strains observed in the intact state. These graft strains did not change significantly after 1000 cyclic loads (Figure 5D, Table 2).
Of 8 double-button specimens, 6 failed instantaneously, as defined by a sharp, instantaneous drop in torque. This was the case for only 2 of 8 docking specimens. It was difficult to accurately determine the exact failure mode for each specimen, so this information was not included in the data set.
Discussion
Results from this experiment suggest that the docking and double-button reconstruction techniques result in similar joint stiffness and strength of reconstruction. Specifically, there were no major differences between the groups as regards joint stiffness, reaction torque, or graft strain at initial fixation (10 cycles) or after cyclic loading (1000 cycles). The 2 reconstruction methods withstood similar maximum torque before failure and demonstrated similar stiffness during ramp-to-failure tests.
In the clinical setting, our group previously published on the outcomes of 23 patients with a minimum 2-year follow-up, reporting a 82.6% return-to-play rate and a statistically significant improvement in visual analog scale scores. 22 Mirzayan et al 21 described a different dual–cortical button suspension technique for UCL reconstruction, and their short-term clinical outcomes were equally as encouraging, with 83% of patients demonstrating good to excellent Conway scores and a mean return-to-play time of 9 months. While long-term outcome data are still lacking, results from the current study suggest that the biomechanics of the double-button cortical fixation are similar to other well-established techniques.
The results from this experiment support the findings of a similar biomechanical study by Jackson et al 13 that used a slightly different surgical technique and was limited to ramp-to-failure testing. Our technique creates a humeral tunnel that runs from the humeral isometric insertion of the anterior bundle of the UCL across the elbow to the lateral column, with the tunnel remaining within the humeral trochlea. This technique consistently produces tunnel lengths of 40 to 60 mm. In contrast, the technique described by Jackson et al employs a tunnel that begins at the UCL “insertion” and exits the medial column at the intermuscular septum, consistently creating a tunnel length of only 20 mm. The technique described by Jackson et al employs an Arthrex TightRope system. In contrast, our technique employs cortical distal biceps buttons, which are smaller than the ones used in their study. In terms of experimental design, Jackson et al measured valgus laxity at discrete angles of 30°, 60°, 90°, and 120° of flexion, finding no differences in laxity in reconstructed specimens as compared with the native state. In the current study, we chose to perform cyclic valgus rotation of the specimens at 90° of flexion to mimic the motion of a baseball pitch, and we found a significant decrease in stiffnesses and maximum torques after 1000 cycles versus the initial 10 cycles. Notably, this decrease was observed in the double-button and docking group specimens. Jackson et al performed ramp-to-failure testing at 70° of flexion while our group did so at 90°. While maximum torques differed between these studies, both support the bisuspensory fixation technique in minimizing the most common method of failure of existing techniques—bone tunnel fracture and graft-suture pullout.
The biomechanical testing protocol used in this study closely mimics other experiments, 3,5,6,8,18 and comparable measures made in this study strengthen our confidence in the results of the current experiment. Our findings suggest that, regardless of the technique used, UCL reconstruction did not restore elbow joint stiffness to its native state. Several biomechanical studies that have used the docking method as a control group have also reported lower torsional stiffness and ultimate torque as compared with the native UCL. 3,18 Armstrong et al 2 performed a biomechanical analysis of 4 reconstruction methods of the medial collateral ligament in the elbow, including docking and button methods, and found that no technique recapitulated native stiffness. Dugas et al 8 used a modified Jobe technique and reported elastic stiffness values of 1.28 ± 0.49 N·m/deg, which is similar to the stiffness value of 1.10 ± 0.39 N·m/deg in this study. Ciccotti et al 6 cited a mean failure torque of 8.2 ± 4.5 N·m when testing the docking reconstruction technique, which is comparable with the failure torque of the docking method in this study (8.93 ± 3.9 N·m).
The stiffnesses and failure torques in this study were substantially lower than in several previously published experiments, and differences are likely due to differences in donor age (80.5 years in the current study) and/or test protocol. Bernholt et al 3 reported a rotational elastic stiffness of 3.0 ± 0.4 N·m/deg in specimens that underwent the docking reconstruction, higher than what was found in the current study (1.10 ± 0.39 N·m/deg). Indeed, a younger cadaveric specimen sample group was used in their study (52.4 years). Two other studies cited mean failure torques ranging from 20.9 to 30.6 N·m in reconstructed elbows, 1,8 while the mean values in this experiment were lower. The mean donor ages for these studies were 43.0 and 63.0 years. Discrepancies in ultimate torque may be caused by differences in test protocols. The aforementioned studies continued to apply torsion to the humerus until failure, whereas we limited our maximum valgus rotation to 25°, which is a reasonable threshold for the varus/valgus range of motion in the flexed elbow joint.
The 3D motion capture technique provided us with the capability to measure strains throughout testing. This represents an improvement over previous studies that measured graft strain using Vernier calipers in static poses. 2,3,8 However, our method fails to characterize local changes in graft strain along the entire length of the graft, which includes lengths of tissue inside bone tunnels and at the button-graft interface. Additionally, there were small errors, in measuring displacement that is inherent to motion capture technology.
Limitations
This study had several notable limitations. First, the cadaveric nature of the study has inherent limitations, most notably a lack of healing. The absence of graft ingrowth into the bone likely results in joint stiffness and ultimate failure loads that are lower than what would occur with a healed reconstruction. The lack of healing also precludes our ability to gauge the longitudinal efficacy of the reconstruction. The soft tissues and dynamic stabilizers, which provide compressive stability to the elbow, were detached in the present cadaveric model. This resulted in isolated loading of the graft that may be larger in magnitude than what is experienced clinically. This study also did not include in vivo analyses, such as risk of button placement in the humerus or ulna, risk of failed deployment of the button, and postoperative symptoms related to hardware. Finally, the results of this study were underpowered (statistical power = 0.10), and it is estimated that a sample size of 135 would be required to achieve a power of 0.8. Because of the limited power of the study, it is impossible to confirm or reject our hypothesis.
Conclusion
A novel double-button technique for UCL reconstruction provided similar biomechanics to the widely used docking method. Both reconstruction techniques restored joint stability, but neither fully recapitulated preinjury joint stiffness. Further research is required to better understand the differences between these methods of UCL reconstruction.
Footnotes
Final revision submitted May 11, 2022; accepted July 5, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: Funding was provided by the Murray Blackman SE Fellowship Fund, an internal grant from the University of Pennsylvania. Surgical supplies used in this study were provided by Arthrex. G.R.H. has received consulting fees from FX Shoulder USA, Lima USA, Limacorporate, and Medical Device Business Services. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto. This study was supported by NIH/NIAMS P30AR069619.
Ethical approval was not sought for the present study.
