Abstract
Background:
Synovial mesenchymal stem cells (MSCs) have high proliferative potential and are considered an excellent source for stem cell therapy.
Purposes:
To isolate MSCs from the synovium of ankle joints in patients with chronic lateral ankle instability (CLAI) and to compare the characteristics of MSCs derived from the synovium anterior to the talus with those from the surrounding anterior talofibular ligament (ATFL) synovium.
Study Design:
Controlled laboratory study.
Methods:
The synovium was harvested from 2 locations in the ankle, the synovium anterior to the talus and the surrounding ATFL synovium, of 14 patients who underwent arthroscopic ATFL repair for CLAI without osteochondral lesions of the talus (OLTs). Synovial fluid was also harvested. MSCs were isolated from both types of synovial tissue, as well as synovial fluid. The number of MSCs in the synovium and their viability, proliferation, colony-forming units, and potential to differentiate into adipose, bone, and cartilage tissues were determined and compared between groups. Additionally, real-time polymerase chain reaction was used to assess the differentiation capacity of adipose, bone, and cartilage tissues from both samples. The Wilcoxon signed rank test was used to compare the sample weight, number of colonies, number of nucleated cells per colony, yield obtained, and phenotypic characteristics of MSCs derived from different locations of the synovium.
Results:
No significant differences were observed in the sample weight (P = .051), number of nucleated cells per milligram (P = .272), number of colonies (P = .722), and yield obtained (P = .099) between the 2 groups. MSCs could not be isolated from synovial fluid. The frequency of oil red O–positive adipogenic colonies (P = .028) and the expression of the adipsin gene (P < .05) were significantly increased in the cells from the synovium anterior to the talus compared to those in the cells from the surrounding ATFL synovium. However, chondrogenic and osteogenic potentials were not significantly different between the 2 groups.
Conclusion:
Synovial MSCs obtained from the ankle joint had self-renewal and multilineage differentiation potential, although the adipogenesis potential of MSCs from the synovium anterior to the talus was superior to that from the surrounding ATFL synovium.
Clinical Relevance:
Both the adipose synovium and fibrous synovium in the ankle joints of patients with CLAI may be a good source of MSCs for stem cell therapy applications, whereas synovial fluid appeared unsuitable.
Keywords
Ankle arthroscopic surgery has evolved as a tool for the assessment and treatment of intra-articular abnormalities, including cartilage damage and lateral ligament injuries of the ankle joint. 5,10,33 Meanwhile, arthroscopic anterior talofibular ligament (ATFL) repair is becoming a promising and popular treatment strategy for chronic lateral ankle instability (CLAI). 1,17,31 Mesenchymal stem cells (MSCs) are a promising source for stem cell therapy, as they can be isolated from various tissues, such as bone marrow, adipose, muscle, periosteal, umbilical, and synovial tissue, and they have the potential for application in regenerative medicine. 27
MSCs are frequently used to treat cartilage damage and osteoarthritis of various joints, except ligament and tendon injuries. 6,7,11,12,16,18 The synovium has been reported as an optimal source of MSCs, as its derivatives exhibit excellent proliferative capacity and chondrogenic potential. 25 For instance, Sekiya et al 29 reported positive outcomes after the concomitant application of synovial MSCs during arthroscopic surgery for patients with cartilage injuries of the knee. Moreover, anterior cruciate ligament–derived stem cells are known for therapeutic potential in anterior cruciate ligament reconstruction. 13,20 In addition, Hatakeyama et al 9 showed that MSCs derived from the knee synovium, with a higher fatty tissue content, exhibit significantly enhanced adipogenic and osteogenic potentials compared to those of MSCs derived from the hip paralabral synovium. Kim et al 14 reported that synovial fluid and tissue isolated from the ankle joint were good sources of MSCs.
Although previous studies have investigated the characteristics of MSCs harvested from patients with osteochondral lesions of the talus (OLTs), they did not describe the differences in the characteristics of synovial MSCs based on their source location. 14 In addition, these studies did not investigate MSCs from patients with other injuries, such as CLAI. Thus, there is a lack of information on the differentiation and proliferation potentials of synovial MSCs derived from the ankle joints of patients with CLAI.
An ankle sprain is the most common lower limb musculoskeletal injury and has the highest reinjury rate of all lower limb musculoskeletal injuries. 4 In addition, 40% of patients who sustain a lateral ankle sprain develop CLAI. 21 For these patients, ATFL repair or reconstruction is one of the treatment methods. However, the therapeutic potential of MSCs for CLAI is unclear. Previous studies have reported that OLTs are common in patients with CLAI. Sugimoto et al 30 reported that OLTs, including superficial softening and fibrillation or fissuring of cartilage, were observed in 76.8% (76/99) of patients during arthroscopic surgery for CLAI. Hadeed et al 8 reported that 53 patients (27.6%) who underwent Broström-Gould ankle ligament reconstruction had OLTs on preoperative magnetic resonance imaging scans. Thus, an arthroscopic procedure with MSC therapy may enable us to simultaneously assess and deal with concurrent lesions in CLAI cases, such as OLTs.
This study aimed to isolate MSCs from the synovium of ankle joints in patients with CLAI and to compare the characteristics of MSCs derived from the synovium anterior to the talus and those from the surrounding ATFL synovium. We hypothesized that the synovium would serve as a better source of MSCs than would the synovial fluid in patients with CLAI.
Methods
Patient Selection and Tissue Collection
The local institutional review board approved the study, and all participants provided written informed consent. Samples were originally obtained from 20 patients (13 male and 7 female; mean age, 34.7 ± 17.4 years) who underwent arthroscopic ATFL repair for CLAI between August 2015 and October 2016. CLAI was diagnosed based on at least one previous occurrence of a lateral ankle sprain and residual instability. 19,21 Inclusion criteria for CLAI were as follows: (1) mechanical instability of grade ≥2 based on physical examination and imaging results 2 and (2) ≥2 episodes of functional ankle instability. Imaging analysis included a radiographic inversion stress test and anterior drawer test, ultrasound, and magnetic resonance imaging. 2 Patients with multiligament injuries, such as ATFL and deltoid ligament injuries, were excluded from this study. Patients with a history of surgery for CLAI and concomitant fractures were also excluded. All patients were refractory to nonoperative treatment options, such as rest, bracing, anti-inflammatory agents, proprioceptive training, and ankle strengthening for at least 3 months. 15,19 At the time of surgery, the conditions of the talus and tibial cartilage were assessed and classified according to the International Cartilage Repair Society classification system. Overall, 6 patients with CLAI and concomitant OLTs were ultimately excluded, resulting in 14 patients (9 male and 5 female; mean age, 33.3 ± 19.4 years) being included in the study (Figure 1).
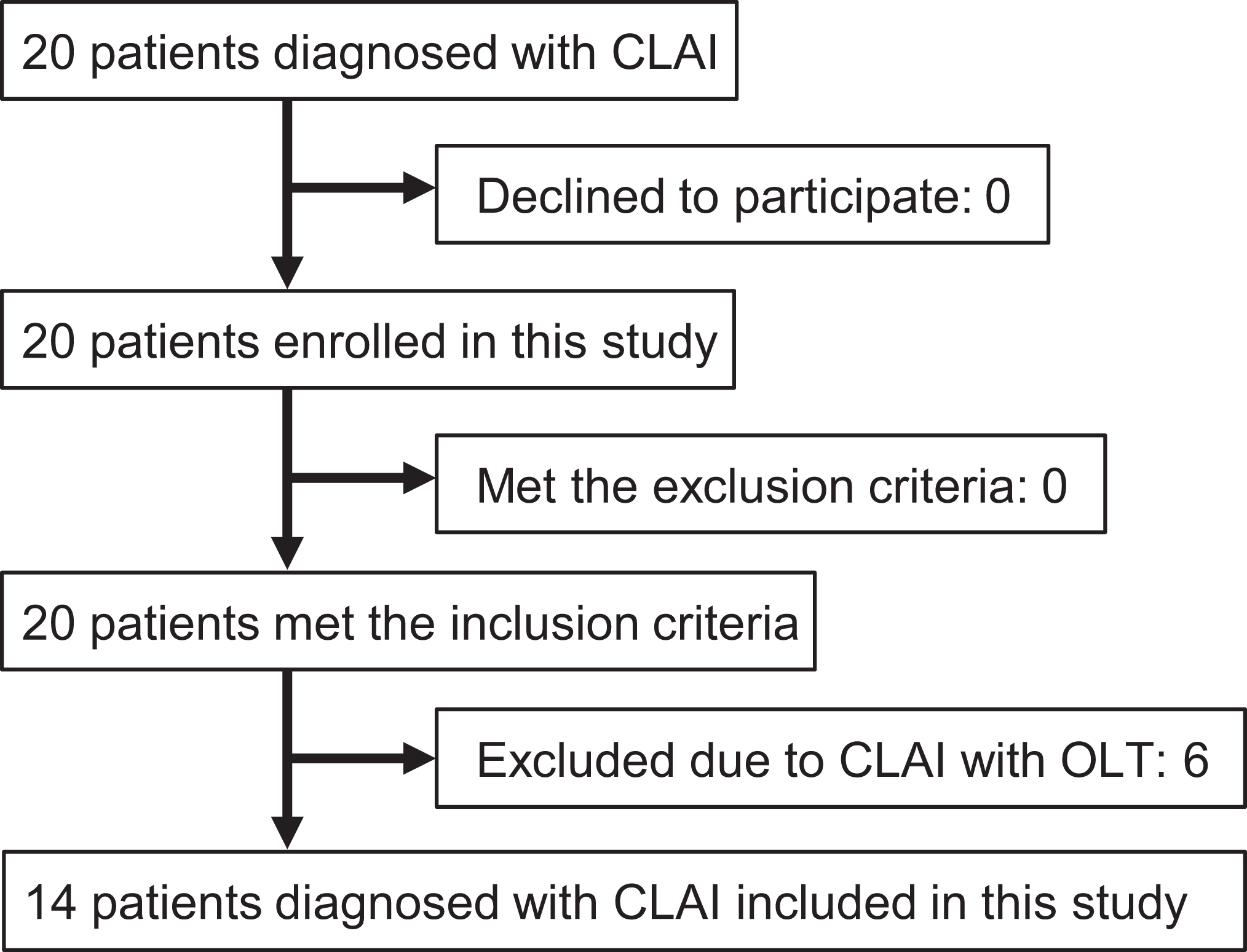
Flowchart illustrating the inclusion and exclusion criteria. CLAI, chronic lateral ankle instability; OLT, osteochondral lesion of the talus.
Synovial fluid and the synovium of the ankle were obtained during arthroscopic surgery with the patient under general anesthesia. Synovial fluid was obtained from the ankle joints after an injection of saline solution (5 mL). To minimize variations in sampling, we harvested specimens from the synovium anterior to the talus and the surrounding ATFL synovium during surgery. Arthroscopic ATFL repair was performed as described in a previous report. 31 For harvesting the surrounding ATFL synovium, the medial midline portal and accessory anterolateral portal were used as viewing and working portals, respectively. We refer to the synovium anterior to the talus as the “anterior fat pad” throughout this article in accordance with a previous report. 3
Isolation of Cells
MSCs were isolated as described previously. 9,23,24,32 Synovial fluid was diluted with phosphate-buffered saline (PBS), filtered using a 70-µm nylon filter (Becton Dickinson), and placed in culture dishes (area, 60 cm2; Nalge Nunc International).
The following procedures were performed according to previously described protocols. 9,23,24 Synovial samples from both locations were digested in 3 mg/mL of collagenase type V (Sigma-Aldrich) in α-modified Eagle’s minimal essential medium (α-MEM; Invitrogen) at 37°C. After 3 hours, the digested nucleated cells were seeded in α-MEM with 10% fetal bovine serum (FBS) at 103, 104, and 105 cells per culture dish (area, 60 cm2), with 6 dishes for each cell concentration. A high concentration of FBS was used to induce the largest number of colony-forming units in cell cultures derived from synovial fluid. The cells were incubated at 37°C with 5% CO2 and 95% humidity. After 3 to 4 days, the medium was replaced to remove nonadherent cells. Adherent cells were then cultured for 14 days at passage 0 without refeeding. After 14 days, 3 dishes for each concentration were stained with 0.5% crystal violet. The optimal initial cell density was determined using the following criteria: (1) the resultant colony size was unaffected by contact inhibition and (2) the maximum number of colonies was achieved. The cells from the remaining 3 dishes at passage 0 were then harvested using trypsin-EDTA (Invitrogen), counted, replated at 50 cells/cm2 in a dish (area, 150 cm2), and cultured for 21 days. The cultured cells were cryopreserved at passage 1 until further use. To expand the cells, we thawed a frozen vial and cultured the cells in culture dishes (area, 60 cm2) for 4 days. These cells (passage 2) were used for further analyses as previously described. 9,23,24
Estimation of Yield Obtained
To investigate the proliferation potential of synovial MSCs from ankle joints, we estimated the number of live cells at passage 0 obtained from the entire volume of each tissue and compared it to the final count of live cells from our cultures.
Histological Analysis
Synovial tissue obtained from the ankle joints was fixed in 4% paraformaldehyde (PFA) solution with 0.1 mol/L of phosphate buffer (pH 7.2) and embedded in paraffin, and 5-µm sections were prepared using a microtome. The tissue sections were stained with hematoxylin and eosin following a standard protocol. 9,23,24,32
In Vitro Expandability
To examine the in vitro expandability of the cells from each mesenchymal tissue, we replated passage 1 cells at 50 cells/cm2 in culture dishes (area, 150 cm2) every 14 days until their growth potential was exhausted. 26 To determine cell viability in each passage, we quantitatively evaluated the cells obtained from the 2 locations using the LIVE/DEAD Viability/Cytotoxicity Kit for mammalian cells (Invitrogen). In addition, a colorimetric assay was conducted by plating passage 4 and passage 7 cells at 5000 cells per well in a 24-well plate (Nalge Nunc International) in 500 µL of complete medium for 7 days. After incubation, 50-µL Cell Counting Kit-8 solution (highly water-soluble tetrazolium salt [WST-8]; Dojindo) was added to each well and incubated for 1 hour. The cells were counted using a spectrophotometer multiplate reader at 450 nm (SH1000; Corona Electric). 9 Cell viability was evaluated and expressed as a percentage of the viable cell counts from the surrounding ATFL synovium (100%).
Differentiation Assays
Adipogenesis Based on Colony-Forming Assays
A total of 100 passage 2 cells were plated in dishes (area, 60 cm2) and cultured in a complete medium for 14 days. Then, the medium was replaced with adipogenic α-MEM supplemented with 10% FBS, 10−7 M of dexamethasone (Sigma-Aldrich), 0.5 mM of 3-isobutyl-1-methylxanthine (Sigma-Aldrich), and 50 μM of indomethacin (WAKO) for an additional 21 days. These cultured cells were fixed in 4% PFA and stained with fresh oil red O (Sigma-Aldrich). The number of oil red O–positive colonies was counted. The same adipogenic cell cultures were subsequently stained with crystal violet, and total cell colonies were counted. 9,23,24,32
Osteogenesis Based on Colony-Forming Assays
A total of 100 passage 2 cells were plated in dishes (area, 60 cm2) and cultured for 14 days. Then, the medium was replaced with osteogenic differentiation medium consisting of α-MEM supplemented with 10% FBS, 10−9 M of dexamethasone, 20 mM of β-glycerophosphate (WAKO), and 50 μg/mL of ascorbic acid-2-phosphate for an additional 21 days. After 4% PFA fixation and positive colonies, mineralized bone nodule formation was assessed via double labeling for minerals (von Kossa stain) and alkaline phosphatase (ALP). The same osteogenic cultures were subsequently stained with crystal violet, and total cell colonies were counted. 9,23,24 Yellow colonies <2 mm in diameter were discounted.
In Vitro Chondrogenesis Using Pellet Culture Assays
A total of 250,000 passage 3 cells were placed in a 15-mL polypropylene tube (Becton Dickinson) and centrifuged at 450g for 10 minutes. The pellets were cultured in chondrogenesis medium (high-glucose Dulbecco’s modified Eagle’s medium [Invitrogen] supplemented with 500 ng/mL of BMP-2, 10 ng/mL of transforming growth factor–β3 [TGF-β3; R&D Systems], 10−7 M of dexamethasone [Sigma-Aldrich], 50 μg/mL of ascorbic acid-2-phosphate, 40 μg/mL of proline, 100 μg/mL of pyruvate, and 50 mg/mL of ITS+ Premix [Becton Dickinson]). 9,23,24 For the microscopic assessment, the pellets were embedded in paraffin, cut into 4-µm sections, and stained with toluidine blue. 28
Immunohistochemistry
Tissue specimen sections 4 µm thick were deparaffinized and incubated with primary anti-human collagen type II antibody (1 mg/mL; ab34712 [Abcam]) for 30 minutes at 4°C. The sections were rinsed with PBS repeatedly and incubated with immune-peroxidase polymer secondary antibody (Histofine; Nichirei Biosciences) for 30 minutes. Antibodies were visualized after treatment with (ChemMate DAB+ Chromogen; DakoCytomation) for 5 minutes. 9,23,24,32
Epitope Profiling
A total of 1 million passage 3 cells were resuspended in 200 µL of PBS containing fluorescein isothiocyanate– or phycoerythrin-coupled antibodies against CD34, CD44, CD45, CD90, CD147, CD271 (BD Biosciences), CD117 (eBioscience), CD105, CD166 (Ancell), and vascular endothelial growth factor receptor–2 (VEGFR-2; R&D Systems) and incubated for 30 minutes at 4°C. The cells were then washed with PBS and resuspended in 350 µL of PBS for analysis. Fluorescein isothiocyanate– or phycoerythrin-coupled nonspecific mouse Immunoglobulin G (BD Biosciences) was used as an isotype control. Cell fluorescence was evaluated via flow cytometry using an EC800 instrument (SONY), and data were also analyzed using the EC800 instrument. 9,23,24,32
Quantitative Real-Time Reverse Transcription Polymerase Chain Reaction
Cells that underwent adipogenesis or osteogenesis were trypsinized, and total RNA was isolated using the RNeasy Plus Mini Kit (Qiagen) according to the manufacturer’s protocol. The chondrogenic pellet was homogenized using the BioMasher (Nippi), and total RNA was prepared from a single pellet using the RNeasy Plus Micro Kit (Qiagen). First-strand cDNA was reverse transcribed from total RNA using the QuantiTect Reverse Transcription Kit (Qiagen).
cDNA was assayed 3 times using TaqMan probes for adipogenesis, osteogenesis, and chondrogenesis. For adipogenesis, adipsin (complement factor D [CFD]; Hs00157263 m1), peroxisome proliferator-activated receptor γ (PPARG; Hs01115513 m1), and lipoprotein lipase (LPL; Hs00173425 m1) probes were used. For osteogenesis, collagen type I alpha 1 (COL1A1; Hs00164004 m1), ALP placental type (ALPP; Hs03046558 s1), Runt-related transcription factor 2 (RUNX2; Hs00231692 m1), and bone γ-carboxyglutamate protein (BGLAP; Hs01587814 g1) probes were used. For chondrogenesis, collagen type II alpha 1 (COL2A1; Hs00264051 m1), collagen type X alpha 1 (COL10A1; Hs00166657 m1), and sex-determining region Y-box 9 (SOX9; Hs00165814 m1) probes (all from Applied Biosystems) were used. For an endogenous control gene, gene expression assays were performed using the TaqMan Array Human Endogenous Control (Applied Biosystems) with s001658 (ACTB; Hs99999903 m1) to normalize gene expression (data not shown). The StepOnePlus real-time Polymerase Chain Reaction (PCR) system (Applied Biosystems) was used for quantification and analysis with StepOnePlus software (version 2.0; Applied Biosystems). 9,23,24,32
Statistical Analysis
The Wilcoxon signed rank test was performed to assess the differences in isolation status and characteristics of the cells derived from the synovium. The sample weight, number of colonies, number of nucleated cells per colony, yield obtained, and cell surface markers were compared between groups based on the location of the synovium. Statistical analyses were performed using the SPSS software package (Version 24; IBM Corp). The level of significance was set at a P value of <.05. Prior power analysis was performed using G*Power (Version 3.1; Universitat Dusseldorf). The primary outcome in this study was MSCs, based on the number of colonies, obtained from the synovium. Previous similar studies have performed power analysis using the number of colonies. 23 Based on a previous report, 11 cases were required for the following conditions: effect size, 1.08; α error, .05; and power, 0.95. 23
Results
Macroscopic and Histological Features of Synovium
Specimens harvested intraoperatively from the anterior fat pad and the surrounding ATFL synovium are shown in Figure 2A. Based on macroscopic analysis, the anterior fat pad was yellowish and floated partially in PBS. In contrast, the surrounding ATFL synovium appeared white and sank to the bottom of PBS (Figure 2B). Sections from both synovial samples were stained with hematoxylin and eosin, which revealed that the anterior fat pad consisted of both fibrous and fatty tissues, whereas the surrounding ATFL synovium appeared to have more fibrous tissue than the anterior fat pad had (Figure 2C).
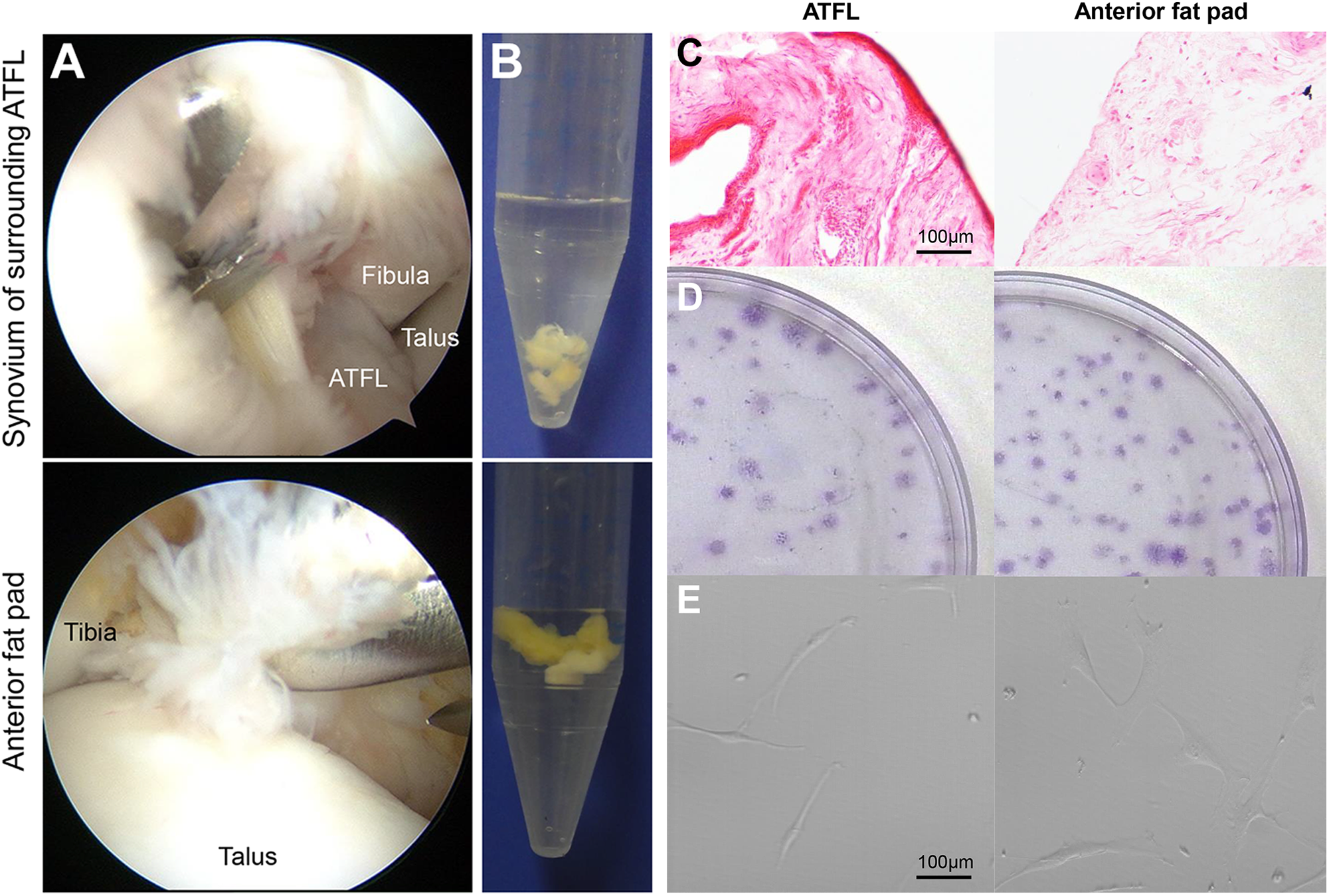
Comparison of cells derived from the surrounding anterior talofibular ligament (ATFL) synovium and from the anterior fat pad. (A) Arthroscopic observation and sample harvesting during surgery from 2 locations of synovial tissues: surrounding ATFL synovium (upper) and anterior fat pad (lower). (B) Macroscopic features of the respective tissue samples. (C) Histological analysis based on hematoxylin and eosin staining. Scale = 100 μm. (D) Representative cell colonies derived from the synovium of a donor, which were stained with crystal violet, showing large colonies with nucleated cells after plating at 104 cells per dish. (E) Representative morphological features of the cells magnified at 14 days (passage 0), showing fibroblastic spindle cell morphology. Scale = 100 μm.
Cell Isolation From Tissue
Tissue samples from the 2 synovial locations, the surrounding ATFL synovium and the anterior fat pad, were compared for various attributes, as described herein (Table 1). There were no significant differences in the sample weight, number of nucleated cells per milligram, number of colonies, number of nucleated cells per colony, or yield obtained between samples obtained from the 2 locations (Figure 2D). However, the number of colonies and yield obtained in patients with CLAI and OLTs were significantly higher than in patients with only CLAI (Table 2). The cells from both synovial tissue locations appeared to be of similar fibroblastic cell morphology (Figure 2E).
Data of Cell Samples Obtained from the Synovium (n = 14) a

a Values are presented as mean ± SD. Wilcoxon signed rank test. ATFL, anterior talofibular ligament.
Data of Cell Samples Obtained From Patients With CLAI and Those With CLAI and OLTs a
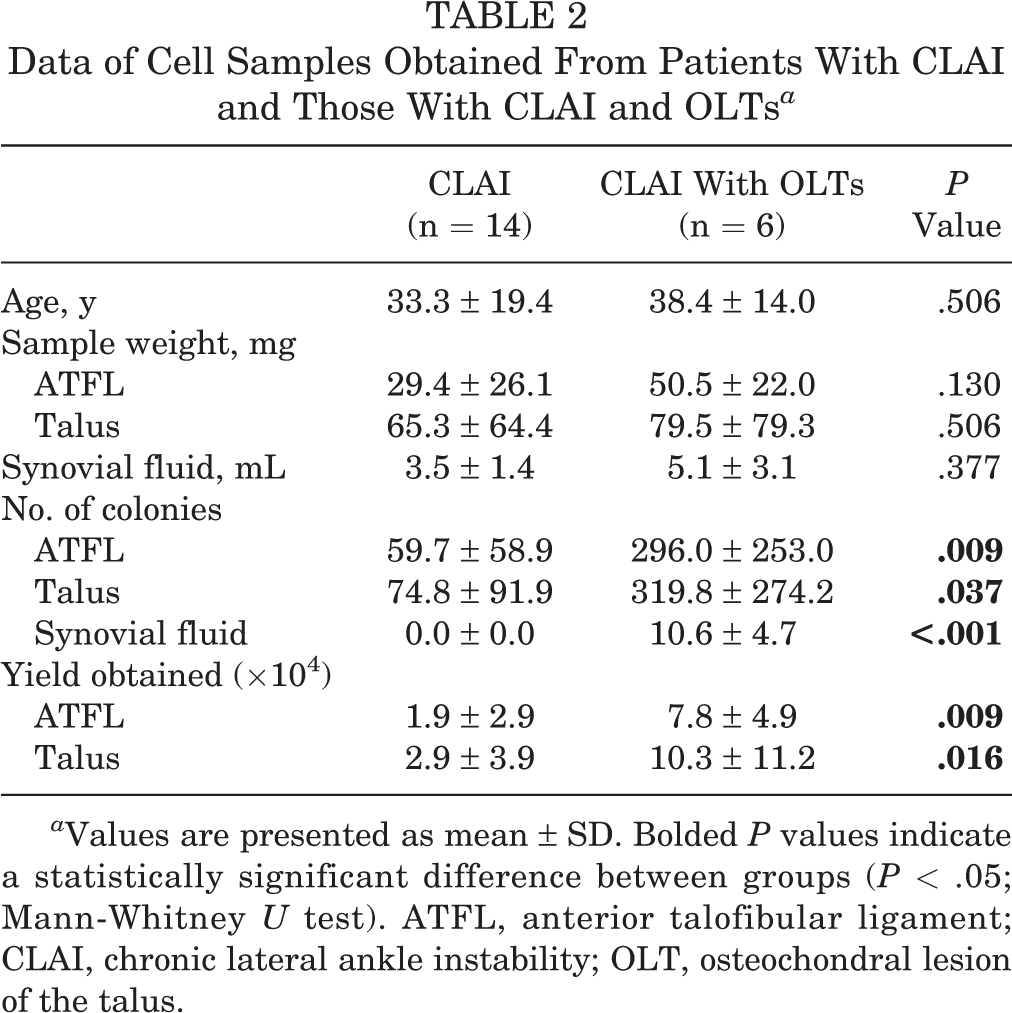
a Values are presented as mean ± SD. Bolded P values indicate a statistically significant difference between groups (P < .05; Mann-Whitney U test). ATFL, anterior talofibular ligament; CLAI, chronic lateral ankle instability; OLT, osteochondral lesion of the talus.
Cell Isolation From Synovial Fluid
Colonies were not observed in any samples obtained from the synovial fluid of patients with CLAI. On the other hand, colonies were observed from the synovial fluid of patients with CLAI and OLTs (Table 2).
In Vitro Expandability
Expandability was lost after 8 passages for both types of synovium-derived cells (Figure 3A). Using the LIVE/DEAD Viability/Cytotoxicity Kit, we determined both types of cells at passage 4 to be alive (Figure 3B). At passage 7, half of both cell types were dead. The Cell Counting Kit-8 assay analysis also showed that the viability of the cells derived from both synovial sources at passage 4 was high (ATFL vs talus: 100% vs 94%, respectively), while half of both cell types were dead at passage 7 (ATFL vs talus: 49% vs 46%, respectively) (Figure 3C).

Proliferation potential and cell viability of mesenchymal stem cells (MSCs). (A) Cells derived from 2 locations of synovial tissues, the surrounding anterior talofibular ligament (ATFL) synovium and the anterior fat pad, were analyzed for their expansion potential. Passage 1 cells from both samples were plated at 50 cells/cm2 on dishes (150 cm2). Thereafter, the cells were replated at 50 cells/cm2 every 14 days until their expansion potential was lost. Expandability was lost at passage 8 for both groups. n = 4 in each group. (B) Representative images of the results obtained from the LIVE/DEAD Viability/Cytotoxicity Kit. At passage 4, green fluorescence was observed in all cells derived from both groups, confirming that these cells were alive. At passage 7, half of the cells in both groups were dead. (C) Results of the Cell Counting Kit-8 assay for the cells at passages 4 and 7. The viability of cells derived from both locations of the synovium was high at passage 4 (ATFL vs talus: 100% vs 94%, respectively). Approximately half of both types of cells were dead at passage 7 (ATFL vs talus: 49% vs 46%, respectively). Cells from 4 donors were examined. p, passage.
Adipogenesis
Based on adipogenesis differentiation assays, all samples generated adipocyte colonies, which stained positively for oil red O (Figure 4A). However, the proportion of oil red O–positive colonies was significantly higher in the cells derived from the anterior fat pad than in cells from the surrounding ATFL synovium (Figure 4B). In addition, quantitative real-time PCR to determine the expression of adipogenesis-related genes was performed (n = 6). The expression of CFD in the anterior fat pad group was significantly higher than that in the ATFL group; however, no significant differences were observed in PPARG or LPL mRNA levels between the 2 groups (Figure 4C).

Adipogenic potential of synovial mesenchymal stem cells (MSCs). (A) Colonies that stained positively with oil red O or crystal violet for each cell type. (B) The proportion of oil red O–positive colonies with respect to the total number of colonies for each MSC source. Values represent the mean ± SD of 6 donors. The proportion of oil red O–positive colonies of the anterior fat pad was significantly higher than that of the surrounding anterior talofibular ligament (ATFL) synovium (P = .028; Wilcoxon signed rank test). (C) Results of quantitative real-time polymerase chain reaction from 6 donors. The expression of adipsin (gene) in the anterior fat pad group was significantly higher than that in the ATFL group; however, there were no significant differences in the expression of LPL and PPARG between the groups (Wilcoxon signed rank test). Error bars indicate the SD. *P < .05. LPL, lipoprotein lipase; PPARG, peroxisome proliferator-activated receptor γ.
Osteogenesis
Based on osteogenesis differentiation assays, the cells derived from the synovium-generated colonies of osteogenic cells stained positively with both von Kossa and ALP (Figure 5A). There was no significant difference in the rates of von Kossa– and ALP-positive colonies, representing the total number of colonies, between the 2 groups (Figure 5B). Moreover, according to quantitative real-time PCR analysis, there were no significant differences in the expression of COL1A1, ALPP, RUNX2, and BGLAP between the 2 groups (Figure 5C). These findings suggest that the osteogenic potential of MSCs from the anterior fat pad is similar to that of MSCs from the surrounding ATFL synovium.

Osteogenic potential of mesenchymal stem cells (MSCs). (A) Colonies stained positively with von Kossa and alkaline phosphatase (ALP) as well as with crystal violet. (B) Proportion of von Kossa– and ALP-positive colonies with respect to the total number of colonies. Values represent the mean ± SD of 6 donors. There was no significant difference in the proportion of von Kossa– and ALP-positive colonies between the groups (P = .866; Wilcoxon signed rank test). (C) Quantitative real-time polymerase chain reaction data from 6 donors. There were no significant differences in the gene expression of COL1A1, ALPP, RUNX2, and BGLAP between the 2 types of synovium (COL1A1: P = .250; ALPP: P = .625; RUNX2: P = .375; BGLAP: P = .250 [Wilcoxon signed rank test]). Error bars indicate the SD. ALPP, alkaline phosphatase placental type; BGLAP, bone γ-carboxyglutamate protein; COL1A1, collagen type I alpha 1; RUNX2, Runt-related transcription factor 2.
Chondrogenesis
Chondrogenesis differentiation assays revealed that most pellets from each tissue were >0.5 mm in diameter. The pellets of both samples had a positive toluidine blue–stained cartilage matrix and were stained with safranin O. Immunoreactivity for collagen type II was also observed in both pellets (Figure 6A). According to quantitative real-time PCR analysis, there were no significant differences in the mRNA expression of COL2A1, COL10A1, and SOX9 between the 2 types of pellets (Figure 6B). These findings suggest that the chondrogenic potential of MSCs from the anterior fat pad is similar to that of MSCs from the surrounding ATFL synovium.
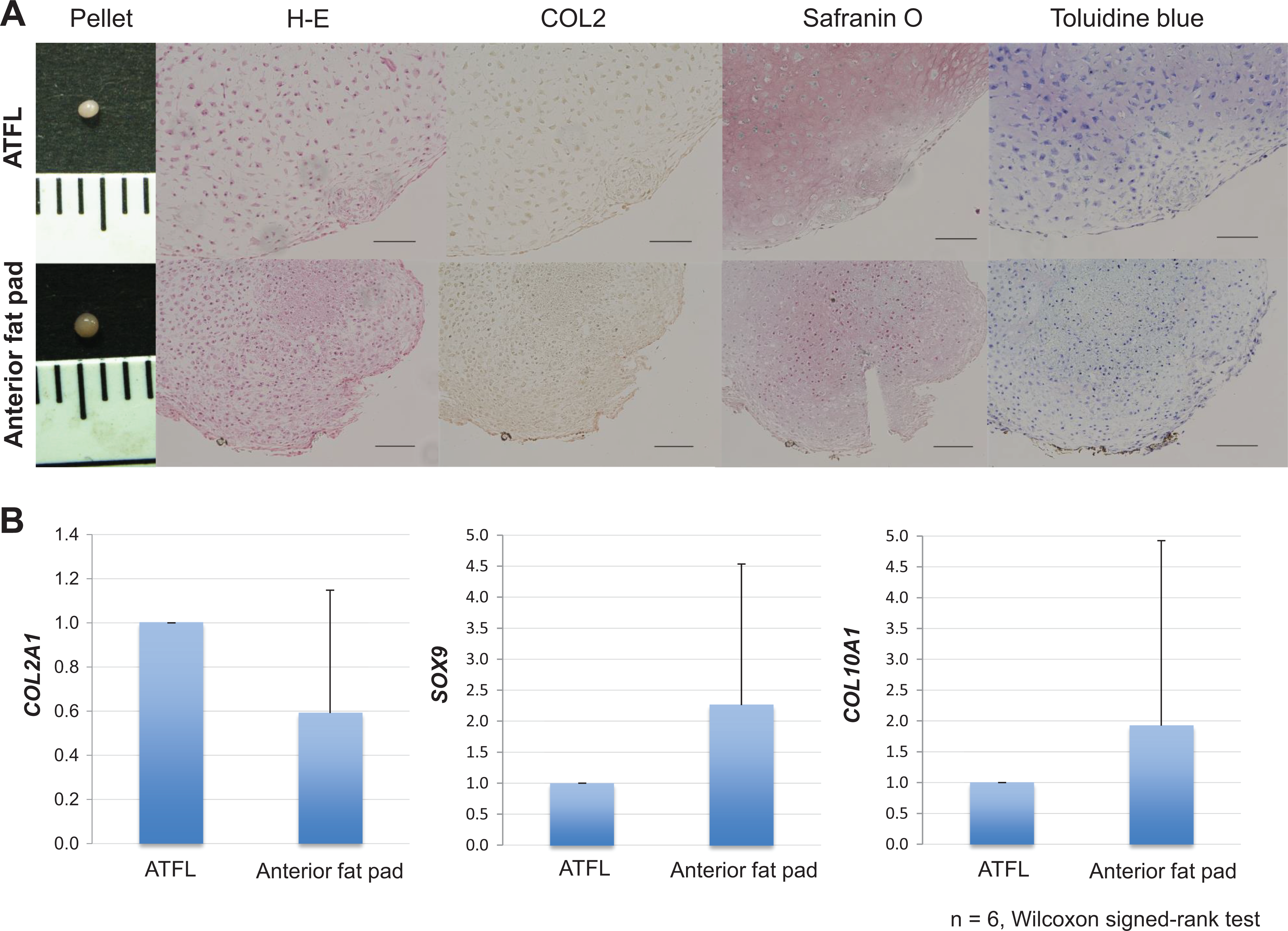
Chondrogenic potential of the 2 types of synovial mesenchymal stem cells (MSCs). A chondrogenic assay was performed using MSCs derived from 2 locations of synovial tissues, that is, the surrounding anterior talofibular ligament (ATFL) synovium and the anterior fat pad. (A) Pellets from each sample are shown. Histological analysis was performed on tissues stained with safranin O and toluidine blue, and immunocytochemical analysis was performed to assess collagen type II expression. (B) Quantitative real-time polymerase chain reaction data from 6 donors. Error bars indicate the SD. There were no significant differences in the mRNA expression of COL2A1 (P = .188), COL10A1 (P = .438), or SOX9 (P = .313) between both types of pellets (Wilcoxon signed rank test). Scale = 100 μm. COL2A1, collagen type II alpha 1; COL10A1, collagen type X alpha 1; SOX9, sex-determining region Y-box 9. COL2, collagen type Ⅱ; H-E, hematoxylin and eosin.
Epitope Profile
The frequency of cells that stained positive with CD45 (hematopoietic cell marker), CD31 (endothelial cell marker), CD117 (c-Kit; stem cell factor receptor), CD34 (hematopoietic progenitor cell antigen), VEGFR-2 (Flk-1), and nerve growth factor receptor was <2% in samples derived from both synovium types. The frequency of cells expressing CD44 (hyaluronan receptor) in samples from the anterior fat pad and the surrounding ATFL synovium was 41.0% and 49.4%, respectively, and the frequency of cells expressing CD90 (Thy1) was 89.2% and 89.8%, respectively. Similarly, the proportion of cells positive for CD105 (SH-2) in samples from the anterior fat pad and the surrounding ATFL synovium was 6.3% and 8.5%, respectively, and that for CD147 (neuregulin) was 12.5% and 10.2%, respectively (Figure 7). No significant differences were observed in the relative frequency of various cell types between the 2 groups. These findings demonstrate that the surface markers were similarly expressed in the 2 cell types.

Flow cytometry analysis of cell surface markers related to mesenchymal stem cells (MSCs), hematopoietic stem cells, and endothelial cells in samples derived from 2 synovial locations. Passage 2 cells from 2 synovial tissue samples were analyzed via flow cytometry for the relative frequency of cells expressing various cell surface markers. No significant differences existed in the rate of positivity between the 2 groups (CD31: P = .655; CD34: P = .655; CD44: P = .068; CD45: P > .999; CD90: P = .715; CD105: P = .593; CD117: P = .655; CD147: P = .465; VEGFR-2: P = .285; NGFR: P > .999 [Wilcoxon signed rank test]). Samples from 4 donors were examined with similar results. NGFR, nerve growth factor receptor; VEGFR-2, vascular endothelial growth factor receptor–2.
Discussion
The key findings of this study are as follows: (1) Adherent cells derived from the surrounding ATFL synovium and the anterior fat pad of ankle joints from patients with CLAI, but without OLTs, could be isolated; they exhibited consistent proliferation and multilineage differentiation potential. (2) No significant differences were evident in the number of colonies between MSCs derived from the 2 synovial tissues, despite a larger number of nucleated cells per colony and a higher yield obtained in samples from the anterior fat pad than from the surrounding ATFL synovium. (3) MSCs derived from the anterior fat pad exhibited greater adipogenic potential than did those from the surrounding ATFL synovium, with no significant differences in osteogenic or chondrogenic differentiation potential between the 2 sources. (4) Colonies were not observed in any samples obtained from synovial fluid of patients with CLAI.
The International Society for Cell & Gene Therapy position statement defines MSCs as cells that adhere to plastic, have specific surface marker expression, and exhibit multipotent differentiation potential. 34 In this study, we isolated adherent cells derived from the anterior fat pad and the surrounding ATFL synovium, which were negative for the expression of CD34, CD45, CD31, CD117, nerve growth factor receptor, and VEGFR-2 and positive for the expression of CD44, CD90, CD105, and CD147 (Figure 7). Cells derived from the human synovium and bone marrow are negative for CD34 and CD45 and positive for CD90, CD105, and CD44. 25,27 These findings indicate that both ankle synovial-derived MSC types expressed similar cell surface markers compared to the expression in cells from other sources, including bone marrow and adipose tissues, and thus they were defined as MSCs.
As part of the assessment for proliferation potential, we investigated the yield obtained and expandability of MSCs derived from synovial fluid and tissue of the ankle joint. However, we did not observe colony formation in any of the samples derived from synovial fluid of patients with CLAI without OLTs. This result indicates that synovial fluid harvested from patients with CLAI without OLTs is not suitable as an MSC source. In addition, the expandability of MSCs from both sources of the ankle synovium was lost at the same time during in vitro culture (passage 8), thus revealing similar proliferation potentials between the 2 groups. Several other studies have also compared MSCs harvested from the fibrous synovium and fatty synovium. For instance, Mochizuki et al 22 reported that MSCs derived from the fibrous synovium and from the adipose synovium in the knee joints demonstrate equivalent chondrogenic potentials. Hatakeyama et al 9 demonstrated no significant differences in the number of colonies per 104 nucleated cells or in the number of nucleated cells per colony between samples from the hip and knee joints. In contrast, Murata et al 23 investigated synovium-derived MSCs from the hip joints of patients undergoing hip arthroscopic surgery and found the number of colony-forming units per nucleated cell and cell yield from the cotyloid fossa fatty synovium to be significantly greater than those from the paralabral fibrous synovium. Our findings indicate that MSCs derived from the anterior fat pad had greater adipogenic differentiation potential than the MSCs from the surrounding ATFL synovium, although no significant difference in the yield obtained was observed between the anterior fat pad and the surrounding ATFL synovium.
We also investigated the multilineage differentiation potential of synovium-derived MSCs and found that the adipogenic differentiation potential of the synovium anterior to the talus was significantly greater than that of the surrounding ATFL synovium. Interestingly, our histological findings revealed more fatty tissue in the synovium anterior to the talus than in the surrounding ATFL synovium. These findings may provide an explanation for why MSCs of the synovium anterior to the talus had greater adipogenic differentiation potential than those of the surrounding ATFL synovium had. Hatakeyama et al 9 also reported significantly greater adipogenic differentiation potential for cells derived from the fatty synovium of the knee joint than for those derived from the fibrous synovium of the hip joint. Similarly, Murata et al 23 reported that the adipogenic differentiation potential of cells from the cotyloid fossa fatty synovium was significantly greater than that of cells from the paralabral fibrous synovium. These previous studies used similar methods to those used in our study, and their results agree with our observations. Moreover, we determined that MSCs derived from the fatty synovium have greater adipogenic potential than MSCs from the fibrous synovium have.
Limitations
Our study has some limitations. First, it is difficult to extrapolate the potential of MSCs in vivo based on this in vitro study. Second, this study comprised a relatively small sample size; thus, sample bias may have occurred in the proliferation assay. However, to minimize sample bias, we performed a power analysis. Third, we did not compare synovial MSCs to MSCs from other sources, such as bone marrow or adipose tissue, because our institutional review board did not approve the harvesting of MSCs from other sources, as this would have required a more invasive procedure and prolonged postoperative recovery. Nevertheless, this study utilized synovial tissue harvested from humans, which is one of the strengths of this study. Fourth, as the number of colonies was counted by one author (H.N.), we could not calculate the intraclass correlation coefficient values for this method. Finally, we did not compare MSCs from the ankle joint with MSCs from other joint mesenchymal tissue, including the knee or hip joint, because the prevalence of simultaneous ankle and other joint arthroscopic procedures is very low in the clinic; it is extremely rare to have an opportunity to harvest synovial tissue from the ankle as well as from the knee or other joints on the same day. Although we observed that the adipose synovium had superior adipogenesis potential to the fibrous synovium, no investigation was performed to compare ankle joints with other joints. Future large cohort studies on the synovium from other joints may be necessary to clarify the specific findings of the synovium from the ankle joint.
Conclusion
Synovial MSCs obtained from the ankle joint had self-renewal and multilineage differentiation potential. The osteogenic and chondrogenic differentiation potentials of MSCs derived from the synovium of 2 locations, that is, the anterior fat pad and the surrounding ATFL synovium, were comparable, although the adipogenesis potential of MSCs from the anterior fat pad was superior to the potential of MSCs isolated from the surrounding ATFL synovium. Finally, the synovium in the ankle joints of patients with CLAI might be a good source of MSCs for stem cell therapy applications, whereas synovial fluid appeared unsuitable.
Footnotes
Acknowledgment
Final revision submitted December 24, 2021; accepted February 17, 2022.
One or more of the authors has declared the following potential conflict of interest or source of funding: S.U. has received research support from Smith & Nephew and consulting fees from ConMed and Smith & Nephew. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the University of Occupational and Environmental Health (No. H27-137).
